Research Article, Arch Med Biotechnol Vol: 1 Issue: 2
Isolation and Characterization of Starter Culture Bacteria for Ensete Ventricosum Fermentation
Meskerem Elfu*
College of Natural Sciences, School of Graduate Studies, Arba Minch University, Ethiopia
*Corresponding Author : Meskerem Elfu
College Of Natural Sciences, School of Graduate Studies Arba Minch University, Ethiopia
Tel: 0913645540; 0911021609
E-mail: meskielfu@gmail.com; tabula.nassa@gmail.com
Received: March 27, 2018 Accepted: June 05, 2018 Published: June 12, 2018
Citation: Elfu M (2018) Isolation and Characterization of Starter Culture Bacteria for Ensete Ventricosum Fermentation. Arch Med Biotechnol 1:2.
Abstract
In Ethiopia different kinds of traditionally fermented foods and beverages are processed and consumed. Traditional fermentation takes place by microorganisms present in the substrate without addition of functional starter culture. This usually leads to long fermentation time, extensive work load on workers and inconsistent product quality. Kocho is one of the widely used Ethiopian fermented foods that suffer this problem. In developed countries production of fermented foods is based on the use of starter cultures, for instance lactic acid bacteria that initiate rapid acidification of the raw material. In Ethiopia the use of starter culture is not a common practice. In this study eight lactic acid producing bacteria were isolated as potential starter culture organisms for enset fermentation. They were characterized to genus level and screened by their desirable characteristics for fermentation. Isolates k15 (Lactobacillus spp.) and C (Streptococcus spp.) were found to be promising starter culture organisms. Both isolates were able to grow best at pH 4-8, 5�??-10�?? 2-6%NaCl and fermented enset in 12 days. The fermented enset (Kocho) was processed for nutritional value and sensory evaluations. The starter culture fermented enset had superior quality than traditionally fermented one in terms of nutritional content, color, odor, texture, test and overall acceptability. These isolates can be used for preparation of starter culture for enset fermentation that would reduce fermentation time, enhance the quality and market value of kocho there by contributing for ensuring food security in enset growing regions of Ethiopia.
Keywords: Lactic acid bacteria; Starter culture; Kocho; Enset; Traditional fermentation
Introduction
Background
In Ethiopia traditionally fermented foods and beverages are consumed in different occasions such as marriages, at festivals, social gatherings and ceremonies (Figure 1). A variety of fermented, nonfermented foods and beverages are processed and consumed from a wide range of raw materials using traditional techniques. In Ethiopia fermentation is usually natural, with no defined starter cultures used to initiate it [1]. Using functional starter cultures offer an additional functionality compared to traditional starter cultures and represent a way of improving and optimizing the substrate fermentation process and achieving tastier, safer and healthier products [2].
In Ethiopia preparation of many indigenous or traditional fermented foods and beverages is still a household art. In addition, traditional foods and traditional food processing techniques form part of the culture of the people. Traditional food processing activities constitute a vital body of indigenous knowledge handed down from parent to child over several generations. Unfortunately, this vital body of indigenous knowledge is often undervalued.
According to Voula [3] the food processing sector in industrialized countries is well organized. Large-scale enterprises generally have sufficient resources at their disposal, both for supporting Research and Development (R&D) and for securing sustainable use of modern technologies, including the controlled application of microbial strains in food processing.
Lactic acid bacteria (LAB) are the primary microorganisms used to ferment maize, sorghum or millet based foods that are processed in West Africa. The use of LAB starter cultures during cereal dough fermentation is a subject of increasing interest in efforts to standardize this step and guaranty product uniformity [4].
Ethiopian traditional fermentation includes foods like injera, bread, kocho and ambasha. Beverages include alcoholic and nonalcoholic. Alcoholic beverage includes tella, areki and tej, and nonalcoholic include borde, shamita, bukre, cheka, merissa and korefe. Kocho is one of traditionally fermented foods consumed in southwestern part of Ethiopia. It is obtained from Enset [5]. The plant has different parts such as pseudo stem, pulverized corm and stalk of inflorescence. Kocho is the main food product obtained by fermenting the mixture of the scraped pulp of the pseudo stem, pulverized corm and stalk of inflorescence [6].
Traditional methods of enset production are time consuming and labor intensive. It takes, for example, up to 3 month to obtain a good quality kocho from enset. Most of this time is for fermentation. In addition, traditional fermentation methods leave offensive odor in kocho. In addition to its nutritional deficiency in protein and vitamin Yewelsew [7] kocho has get reduced market price compared to other cereals and crops consumed in the country (Mogesse). However it has economic and social advantages it is popular and used as staple food for 20 millions of people in southern and southwestern parts of the country [8]. It is drought resistant, (improved food security in Enset growing areas of Ethiopia)can be stored for longer period, can be harvested at any time, and provides huge product harvested from a single plant than other cereals consumed in the country [9].
To reduce its problems of offensive smell on consumes on, traditionally washing is used as technique to remove its odor. However, this technique may reduce its nutritional level and result in resource wastage. Some people consume it unwashed after cutting the non-fermented fiber. As fermentation lowers the pH of the medium, complaints about stomach disorder are common. Some people also consume it along with other sources of protein and vitamin foods to increase its nutritional level [7]. Using fermentation technology is preferred to improve its fermentation problems. One of these is by using starter culture technology, which reduces fermentation time, increase sensory acceptance in different regions, reduces work load of women and children, increase its nutritional value, increase its lower market price, improves food security problem from the country, benefits farmers and inset growing areas and to produce quality product. There is no research on functional starter culture technology for Ethiopian traditional fermented foods. To the knowledge of the investigator of this research, much effort has not been made to produce potential starter culture from bacteria using easily available fermented foods as a source of bacteria. This research is designed to develop functional starter culture for enset fermentation, by using different fermented foods as a source of organisms to be isolated. The significance of this study is thus production of quality kocho by using starter culture technology from bacteria using locally available fermented foods at laboratory level which could be scaled up to pilot and industrial levels. There for this study was designed to develop potential starter culture by the following objectives.
Objectives
General objective
To improve the value of enset by using defined starter culture for fermentation.
Specific objectives
• To determine the contents of the supernatant of traditionally fermented and washed enset.
• To isolate and select potential bacterial starter culture for enset fermentation.
• To characterize the selected isolates in terms of their suitability for the desired fermentation.
• To optimize enset fermentation with the selected bacterial consortia.
• To compare the starter culture fermented enset with traditionally fermented one in terms of sensory and nutritional values.
Methodology
Sample collection
Enset and traditionally fermented enset samples were collected from Chencha (woreda), Gamu-Gofa Zone, SNNP and Ethiopia. The “Genna” Enset variety was selected for kocho preparation, based on the consumption frequencies and local preference of Enset varieties for Kocho and Bulla preparation. Kidney beans, maize and pumpkin were collected from Arba Minch market by using sterile plastic bags. Traditionally fermented milk was collected from Arba Minch University Omo lounge using sterile flask. Banana and Mango fruits were from Arba Minch University banana trip in sterile plastic bags. The samples were kept in Microbial Biotechnology laboratory in refrigerator at 4°C until use.
Measurement of nutritional parameters of samples Nutritional parameters: pH, Titrable acidity (TA), Total suspended solid (TSS), Total dissolved solid (TDS), Total fat content (TFC), Total ash content (TAC), Total protein content (TPC), Total moisture content (TMC), Total fiber content (TFbC) and Total carbohydrate content (TCC) were measured after the samples were prepared as supernatant and sediment. The same parameter were measured for crude samples of traditionally fermented and starter culture fermented kocho samples. Traditionally fermented enset (500 g) was washed by using 1L tap water and filtered by using 3 mm size sieve. Then the residue which contains the fiber part at the top of sieve was removed. The remaining part was allowed to settle for ten minutes. The supernatant was carefully decanted and stored in small plastic bottles at 4°C. The sediment was dried at 70°C for 2 h and processed for the measurements of nutritional parameters.
pH: The pH of crude samples of both traditionally and starter culture fermented enset was measured by mixing 1 g of sample with 9 ml of distilled water vortexes for 5 min. The pH was determined by using digital pH meter.The pH of the traditionally fermented enset supernatant was measured directly. The pH of starter culture containing samples was determined at the 3rd, 6th, 9th , 12th, 14th and 16th days of fermentation time.
Titrable acidity (TA): The TA of crude samples of both traditionally and starter culture fermented enset was determined by titrating according to the method described by [10]. One gram of each of the starter containing and traditionally fermented enset sample was mixed in 9 ml of distilled water independently. The supernatant TA was measured directly by titrating 10 ml. Then it was determined by titrating with 0.1 M NaOH, using 1% phenolphthalein as indicator.
Total suspended solids (TSS): TSS of 100 volumes was dried at 105°C for 30 min. Their weights were measured and recorded as w1. The Supernatant of 100 ml was poured in to the crucibles and placed at 105°C for 12 h. The final weight of the crucible and the dry matter was recorded as w2.The total suspended solids percent of the supernatant was calculated as:
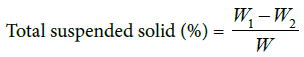
Total dissolved solids (TDS): The TDS content of the supernatant was measured by using High performance multi meter. The supernatant of 100 ml was placed on 100 ml flask. The TDS probe sterilized by using distilled water and inserted in to 100ml flask containing supernatant of kocho then TDS recorded from probe reading.
Total fat content (TFC): Traditionally fermented and starter containing Kocho 2 g dried crude samples and 2 ml of kocho supernatant was extracted with 100 ml petroleum ether independently, for a minimum period of 4 h in the soxhlet extractor. The solvent was then evaporated by heating on a steam bath. The flask containing the extracted fat was dried on steam bath to a constant mass [10]. The total crude fat was calculated as percentage by weight:
![]()
Where, W1=weight of the extraction flask
W2=weight of the extraction flask plus the dried crude fat (g)
W=weight of the sample
Total ash content (TAC): Clean drying dish were dried at 120°C in hot air oven and ignited at 550°C for about 3 h in a muffle furnace. Then, cooled in desiccators and weighed using analytical balance (M1). Then, 2 g of starter culture containing and traditionally fermented dried kocho powder and 2 ml supernatant was placed on crucible glass (M2). The samples was dried at 105°C for 1 h and carbonized by blue flame of Bunsen burner, until the contents turn black. The dish with its contents was transferred to a muffle furnace and ignited at about 550°C. The residue was weighed (M3) [10]. The total ash was expressed as percentage on dry basis as follow:
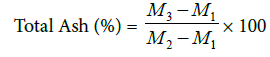
Where, M1=weight of dish
M2=weight of samples
M3=weight of sample and dish after drying
Total protein content (TPC): Crude protein was analyzed by micro Kjeldahl method as presented on AOAC [11]. The general procedure included the following steps of digestion: 2 g traditionally fermented and starter used kocho and 2 ml of supernatant was digested by adding 5 ml of concentrated sulfuric acid, in the presence of potassium sulfate catalyst in a Kjeldahl flask, then neutralization and distillation as the digest was diluted with 30 ml distilled water. 25 ml of NaOH (40%) was added to neutralize the sulfuric acid. Upon addition of NaOH, the ammonium was distilled off and trapped into a boric acid and solution containing methyl blue and methyl red indicators. Finally, titration of the ammonium attached to borate anion was titrated with standardized HCl and total crude protein of kocho was calculated as total nitrogen [10].
Total moisture content (TMC): Moisture content of traditionally fermented and starter culture containing sample was analyzed by oven drying method. A drying dish was dried in an oven at 105°C for 1 h and placed in desiccators to cool. The weight of the drying dish (W1) was determined. 2 g of kocho samples were weighed in the dry dish (W2) oven dried at 105°C for 2 h and after cooling in a desiccators to room temperature, it was again weighed (W3).
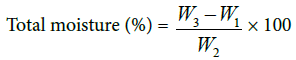
Where, W1=weight of dish
W2=weight of sample
W3=weight of sample and dish after drying
Total fiber content (TFC): The fiber contents of traditionally fermented and starter used kocho were dried at 90ºC 2 g sample was transferred to 400 ml beaker. After digestion with 1.25% sulfuric acid and washed with distilled water and then digested by 1.25% NaOH, the sample was filtered. The residue left after refluxing was washed again with 1.25% sulfuric acid near the boiling point. The residue was then dried at 95°C overnight, cooled in desiccators and weighed (M1). After mashing for 2 h. at 500°C, it was cooled in desiccators and weighed again (M2). The total crude fiber was expressed in percentage as:

Total carbohydrate content (TCC): Total carbohydrate was determined by difference.
Total Carbohydrate (%) =100-(%TFC+%TAC+%TPC+%TMC+%TFbC+%TCC)
Where,
%TPC=Crude protein content in percent
%TFC=Crude Fat content in percent
%TAC=Ash content in percent
%TFbC=Total fiber content in percent
%TCC=Total carbohydrate content in percent
%TMC=Total moisture content in percent
Isolation of LAB for enset fermentation
Isolation of Lab from 15 days and 4 month traditionally fermented enset and yogurt: Traditionally fermented enset (15 days and 4 month) 1 g and 24 h traditionally fermented yogurt 1 ml placed in to 9 ml of distilled water independently. Six fold serial dilutions were made from the mixed solution and 1ml from the last 3 dilutions (10-4, 10-5 and 10-6) were pipetted and plated on sterilized de Man Rogosa and Sharpe (MRS) agar which consisted of peptone (10 g) yeast extract (10 g) meat extract (5 g) glucose (2 g), dipotasium sulfate (0.4 g), tri sodium acetate (1 g), ammonium citrate (0.4 g), Magnesium Sulfate (0.1 g) Manganese Sulfate (0.05 g) and agar (15 g) in 1 liter water was done by autoclaving at 12°C for 20 min. (100) μl of suspension of were spread onto a plate containing MRS agar by pour plate method. The plates were then incubated at 30°C for 48 h anaerobically. Morphologically distinct and well isolated colonies were picked and transferred to new MRS agar plates by streaking to obtain pure colonies [12].
Isolation of LAB from kidney beans and maize grains: The samples dried at 105°C for 2 h dried samples was grounded and sieved by 2 mm sieve. The 5 g flour was mixed with 5 ml tap water then stored for 48 h at room temperature anaerobically. The bacteria were isolated by using isolation method used in isolation of LAB for enset fermentation.
Isolation of LAB from mango, banana and pumpkin: Samples were pilled Edible part grounded and stored for 48 h anaerobically at room temperature. The bacteria were isolated by using isolation method used in isolation of LAB for enset fermentation.
Characterization of starter culture bacteria isolates
The positive lactic acid producing bacterial isolates were subjected to asset of morphological physiological and biochemical tests for identification. Biochemical characterization was done by Bergey’s Manual of determinative bacteriology [13]. Colonies differ in morphology; pigmentation, shape and size were subculture in MRS agar. Each individual sub cultured sample was examined by gram staining and catalase production activity. Only gram positive and catalase negative samples were then purified by streaking and were stored in slant MRS agar and with 20% glycerol at -4°C for further experiment [14].
Catalase test: Clean microscopic slide was taken. A drop of 3% H2O2 was placed on the microscopic slide aseptically. A loop full of 48 h cultured bacteria was taken and mixed with 3% H2O2 solution on the slide and the presence of the bubble production observed [15].
Gram staining: A drop of distilled water was placed on clean slide. 48 h incubated culture was added on the slide and smeared with distilled water by using sterile loop. Heat fixed by passing 3-4 times on Bunsen burner. The fixed bacterial smear is subjected to Crystal Violet, Iodine Solution, Alcohol (decolorizing agent) and safranin respectively. Gram positive bacteria retain crystal violet and hence appear deep violet in color, while Gram negative bacteria lose the crystal violet and are counterstained by the Safranin. Hence they appear red in color [16].
Motility test: A loop full of 48 h incubated culture was placed on depressed slide. The slide was covered by using slide cover. Motility of isolate was observed by at 100x magnification power by using microscope. Motile bacteria move one part to another then non motile one stay one place.
Spore former: To determine spore former from non-spore former isolated strains was heat treated for 15 minute at 80°C then inoculated in MRS medium incubated for 48 h at 30°C. If isolates are spore former growth obtained on the medium there is no growth observed isolates are non-spore former.
Screening for potential starter culture bacteria
The isolated strains were individually inoculated by single streaking on selective media such as Starch agar (2% starch), milk agar and nutrient agar plates to isolate amylase, protease, cellulose and lipase producers respectively. Then inoculated plates incubated overnight at 30°C and checked for a zone of clearing around each bacterial isolate. For starch agar, the zone of clearing observes after flooding the plates with iodine.
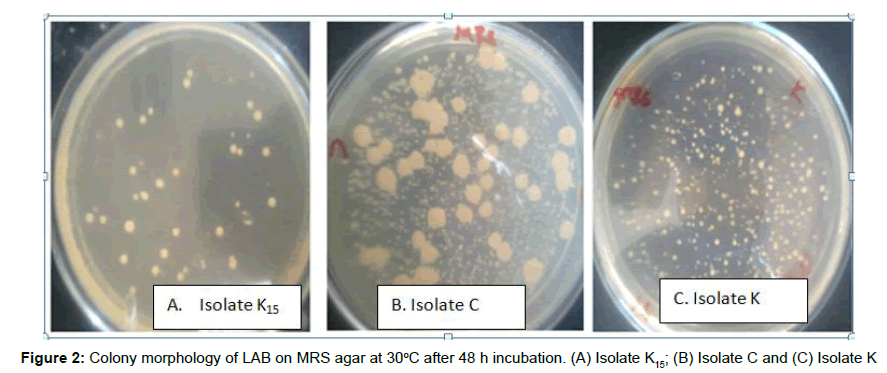
NaCl tolerance: To determine NaCl tolerance, all the isolates were grown in MRS medium supplemented with different concentrations of NaCl (2, 4 and 6%). The medium were inoculated with loop full overnight culture of the isolates and incubated in anaerobic condition at 30°C for 48 h. After 48 h incubation, growth was determined by the formation of colony on the medium (Figure 2) [17].
Determination of optimal growth at different pH: To determine the optimal growth of isolated strain at different pH, 1% (v/v) fresh overnight cultures of isolated strain were inoculated into MRS broth with varying pH ranging from 4 to 8. The pH were adjusted with concentrated acetic acid (99%) and 5 N NaOH. The inoculated broths were incubated in anaerobic condition 24 h at 37°C. Over growth, of the bacteria was measured using a spectrophotometer, reading the optical density at 600 nm (OD600) against the un-inoculated broth [17]. Un-inoculated broth was used as control.
Determination of optimal growth temperature: The optimal growth of isolated strain was streak plated on MRS agar for 24 h at 5°C, 10°C, 15°C, 30°C and 45°C for 24 h.
Carbohydrate utilization and gas production of isolates: Carbohydrate fermentation was determined by inoculating 24 hrs growth culture on MRS broth in the presence of Durham tube. Bubbles trapped within the Durham tube indicate the production of gas. Even a single bubble is significant and denotes evidence of gas production. No bubbles within the Durham tube indicate a non-gas producing or anaerogenic organism.
Selection of potential starter culture bacteria for enset fermentation
Potential isolates was selected based on their ability to degrade cellulosic and hemicellulose materials. Isolates was selected as potential isolate based on clear zone formation, carbohydrate source fermentation and gas production, temperature, NaCl and pH tolerance.
Optimization of fermentation conditions by selected isolates: Chopped enset pulp 50 g was measured and placed in sterilized beaker and aluminum foil covered. Heated at 80ºC for 5 min and cooled for 30 min inside laminar air flow. Inoculants introduced by using sterile micropipette.
Inoculum size: 50 g of chopped enset pulp was introduced with 2 ml, 4 ml and 6 ml of 24 h incubated anaerobically covered with Para film.
Temperature tolerance: The fermenting temperature of selected isolate inoculated non fermented enset sample was stored at different temperature at 5°C, 10°C, 15°C, 30°C and 45°C.
Fermentation time: Fermentation time of selected isolate introduced enset was determined at 3, 6, 9, 12, 14 and 16 days.
Comparison with traditionally fermented enset
Traditionally fermented and starter culture fermented kocho samples were compared in terms of their nutritional parameters: pH, TA, TFC, TAC, TPC, TMC TFbC and TCC as indicated at Titrable acidity.
Sensory evaluation of starter culture containing and traditionally fermented enset: Traditionally fermented and starter culture containing enset was prepared by using traditional method cutting. Fermented samples squeezed by using fiber to remove its moisture content. Squeezed enset were cut and mixed with tap water until it became smooth and baked. 5 cm sized used in the screening of enset bread, based on their sensory quality characteristics. A total of 15 panelists were involved in the sensory evaluation. Panelists selected randomly from local people. Starter fermented and traditionally fermented enset bread 15 cuts were presented for panelists to evaluate the taste, color, flavor and overall acceptability of the samples. All panelists were instructed to make their own individual assessments, according to the evaluation criteria provided for each samples on the basis of taste, color, flavor and overall acceptability. The sensory evaluation format listed at Appendix 1
Result and Discussion
Measurement of nutritional parameters of samples
pH: The acidity obtained from traditional method (5.5) and supernatant (5) than sediment (3.2) kocho as presented on (Table 1). The difference in pH is due to removal of acid during washing. Due to acidity traditionally fermented enset non-washed is very sour test than washed kocho. There is also difference in test color, smell and texture of washed and non-washed kocho. Non washed kocho was small black and sour test than washed kocho was white in color and tasteless (water test). The difference in color and test is due to the removal of color and test producing water soluble substances. Concerning processing methods, the lower pH (high acidity) (5.5) was recorded for kocho samples from traditionally fermented and the lower pH (less acidity) (6.5) was obtained from kocho samples from starter culture fermented enset. The effect of fermentation organisms was significant on pH value of kocho samples. The highest acidity shows the production became very sour test than lower pH [18] (Table 1).
| Parameters | Traditionally fermented | Starter culture containing | |||
|---|---|---|---|---|---|
| Crude | Supernatant | Sediment | Fiber | Crude | |
| pH | 5.5 | 5 | 3.2 | 5 | 6.5 |
| TA (%) | 0.39 | 0.37 | 0.12 | 0.32 | 0.58 |
| TFC (%) | 1.93 | 1.22 | 0.31 | 0.41 | 3.36 |
| TAC (%) | 2.24 | 0.14 | 1.31 | 1.02 | 1.06 |
| TPC (%) | 2.81 | 2.01 | 0.53 | 0.36 | 3.02 |
| TFbC (%) | 2.75 | 0.00 | 0.02 | 2.62 | 0.67 |
| TMC (%) | 49.30 | ND | 26.25 | 22.41 | 51.20 |
| TCC (%) | 40.97 | ND | ND | ND | 40.14 |
| TSS (mg/l) | ND | 1.4 mg/l | ND | ND | ND |
| TDS (mg/l) | ND | 178 mg/l | ND | ND | ND |
Table 1: Nutritional parameters of samples.
Titrable acidity (TA): The maximum TA percent among the two processing method (cutting and washing) was found in kocho prepared by cutting (0.39) and supernatant decanted during washing (0.37). The sediment kocho TA was lower than supernatant and traditional method (0.12). This is due to removal of contents during washing.
The increase in pH and increase in TA in starter culture fermented enset during the entire enset fermentation could be attributed to the activities of acid producing microorganism’s mainly lactic acid bacteria (Table 1). Enset fermented with starter culture had initial pH range of (9.0 to 6.2) (Appendix 3 and 4) with TA (0.21 to 0.58) in 16 day fermentation. This pH value is slightly lower than the pH value (5.5) with reduced TA (0.39) recorded in traditional fermentation. The difference in TA was due to the presence high number of microorganisms in traditional fermentation and single strain in starter culture used enset [5].
Total suspended solids (TSS): The Supernatant decanted during washing were contains 1.4 mg/l suspended solids which are removed from fermented enset during washing. The result shows that people who consume kocho by washing they lose 1.4 mg/l amount of suspended solids. This removed solid may contain nutritional part of soluble substances.
Total dissolved solids (TDS): The TDS of supernatant were contains 178 mg/1 dissolved solids removed during washing. As mentioned in TSS this removed TDS also remove nutritional substances. This substance was lost during washing (Table 1).
Total ash content (TAC): Traditionally fermented enset contains higher ash content than supernatant decanted one and sediment kocho. The difference in ash content is due to the edible part in traditional fermented enset is higher than supernatant decanted.
Concerning the two processing methods (Starter used and traditionally fermented enset) starter culture containing enset contains lower amount of ash than traditionally fermented one. The decrease on ash content is due to fermenting organisms. According to Melese 2013 study the reduction in ash content is due to fermentation time. Starter culture containing kocho sample contained lower amount of ash than traditionally fermented kocho sample. The difference in ash content is due to the degradation of enset pulp by fermenting microorganisms.
Total fat content (TFC): The traditional method contains highest fat (1.9%) content than the supernatant (1.7%). The sediment kocho (0.7) was lower fat content this is due to the removal of water soluble fat from traditionally fermented enset during washing. The difference in fat content is due to removal of soluble fat substances during washing. Washed kocho became dry exposed to air than traditionally fermented enset this is due to loss of fatty substances during washing. Starter culture used enset contains the highest crude fat content (3.31%) than traditionally fermented one (1.9%) (Table 1). The difference in crude fat content among traditionally fermented and starter culture used is due to strains used as starter they not degrade fat. In traditionally fermented one different microbe are available they may degrade the content of fat [10]. The reduction in fat content is due to the degradation of fats by lepolytic enzymatic activities and microbial proliferation causing the consumption of nutrients.
Total protein content (TPC): The TPCs of the traditionally fermented enset was highest protein content (2.81%) than the sediment and supernatant 1.43% and 2.01% respectively. The difference is due to the removal of soluble protein substances during washing. Due to removal of protein substances washed kocho is very protein deficiency than traditional method. The TPC of starter used enset contains highest protein content than traditionally fermented one as presented on (Table 1). The crude protein content of kocho increased due to the action of fermenting microorganisms in the synthesis of some amino acids and improved the quality of the protein, as determined by amino acid profiles. It is also known that fermentation had the general effect of increasing the essential amino acid content of kocho. Heating the Enset pulp increases the solubility of protein in to their smaller units [19]. Mogesse [1] report Protein reduction is due to leaching of the more soluble proteins and amino acids. Fermentation had the general effect of increasing the essential amino acid content of kocho.
Total fiber content (TFC): The TFbC Traditionally fermented enset contains highest fiber than starter used one it can be seen from (Table 1) the crude fiber content of Enset samples showed significant difference due to the degradation of enset pulp by starter organism. The highest value, 2.75% in crude fiber content was recorded for traditionally fermented enset and lowest 0.97% recorded from starter used kocho.
The decrease in fiber content during fermentation could be attributed to the partial solubilization or degradation of cellulosic and hemi-cellulosic structural materials in the plant (pulp) by microbial enzymes. In the current work, fermentation increases the breakdown of dietary fiber to soluble and digestible form, and which decrease the total indigestible crude fiber content [20]. In addition to this the fiber content decreases due to increasing pH (Appendix 3 and 4).
Total moisture content (TMC): Traditional method used enset and sediment kocho was lowest moisture content than supernatant decanted. The supernatant was the highest moisture content. Starter culture fermented enset was highest moisture content than traditionally fermented one. The difference in moisture content is due to degradation of pulp by organisms involved in fermentation process. In [10] study the moisture content reduced due to increasing fermentation time and fermentation.
Total carbohydrate content (TCC): Among the two methods washed kocho was highest carbohydrate content than used by washing. People who consume kocho by washing have highest carbohydrate content. The difference in washed and non-washed kocho was water soluble carbohydrate substances settle during washing and the highest value of non-degraded carbohydrate content removed with fiber. In this study, there was significant difference in carbohydrate content among the microorganisms involved in fermentation. Traditionally fermented enset was highest carbohydrate content than starter culture fermented one. The reduction of carbohydrate content during the fermentation process was possibly due to the breakdown of more complex components by enzymes produced by fermenting microorganisms in starter culture containing kocho sample. And this might be also associated with the reduction of crude fiber content, also advanced the decrease in carbohydrate content. Therefore, decrease in carbohydrate content is related with the addition of fermentation time, resulting to enzymatic degradation and depolimeraization of carbohydrate to their smaller forms [10]. The general trend observed in this study as presented from (Table 1).was decreasing carbohydrate content due to fermenting organisms. During enset fermentation there is also production of gas by inoculated fermenting organisms (Appendix 6). This shows that strain lactic acid producing ability [21].
Isolation of starter culture bacteria
A total of 155 colonies were taken from serial dilution 10-4 (Appendix 5). The number, shape and color of isolates are shown in (Table 1). Representative colonies were obtained and purified by repeated streaking on MRS agar. Each colony of pure culture was individually picked based on distinct morphological characteristics (Table 2). In this study starter culture bacteria were isolated from different fermented food sources and the efficient strain was selected for further studies as presented on [12].
| Isolate code | Colony texture | Edge | Colony color | Colony shape |
|---|---|---|---|---|
| K | Shinny mucoid surface | Rough | Creamy white | Cocci in chain |
| C | Shinny surface | Smooth | White Creamy | Cocci in chain |
| B | Shinny surface | Rough | White | Short rod |
| M | Shinny surface | Rough | White | Cocci in chain |
| Kb | Shinny mucoid surface | Smooth | Creamy white | Cocci in chain |
| Pu | Shinny mucoid surface | Smooth | Creamy white | cocci |
| K15 | Shinny mucoid surface | Smooth | Creamy white | cocci |
| Y | Shinny mucoid surface | Smooth | Creamy white | Short rod |
Table 2: Colony morphology of isolates from sampling areas.
Biochemical characterization of isolated strains
The morphological and biochemical characterization of isolates strains are presented in Table 3. Out of 155 isolates 103 catalase positive, 52 catalase negative, 17 non motile, 35 motile, 6 spore former, 10 non spore former, 8 gram positive and 2 gram negative isolates was characterized. Most isolates were cocci and few species were rod. Isolated strains are similar characteristics with LAB. Many investigators reported production of starter culture by various species of LAB. According to [22-26] study LAB are utilized as starter culture organisms in different developed countries. They are isolated from different fermented food sources. This result can be compared with the study of [27]. Eight gram positive strains were selected for starter culture preparation. Based on Bergeys manuals of bacteriology the selected strain was identified as LAB. In addition to this isolated strains were similar with Ekundayo [28] study.
| Isolates code | Catalase test | Motility | Spore former | Gram staining | Closest Genera | ||||
|---|---|---|---|---|---|---|---|---|---|
| -ve | +ve | Motile | Non motile | Spore former | Non spore former | -ve | +ve | ||
| K | 1 | 1 | 0 | 1 | 0 | 1 | 0 | 1 | streptococcus |
| C | 11 | 9 | 8 | 3 | 1 | 2 | 1 | 1 | streptococcus |
| B | 7 | 16 | 5 | 2 | 2 | 1 | 0 | 1 | streptococcus |
| M | 4 | 2 | 3 | 1 | 0 | 1 | 0 | 1 | streptococcus |
| Kb | 12 | 18 | 8 | 4 | 0 | 2 | 1 | 1 | lactobacillus |
| Pu | 10 | 30 | 8 | 2 | 1 | 1 | 0 | 1 | Leuconostoc |
| K15 | 4 | 2 | 1 | 3 | 2 | 1 | 0 | 1 | lactobacillus |
| Y | 3 | 25 | 2 | 1 | 0 | 1 | 0 | 1 | lactobacillus |
| Total | 52 | 103 | 35 | 17 | 6 | 10 | 2 | 8 | |
Table 3: Biochemical characterization of isolated strains.
Screening of starter culture bacteria
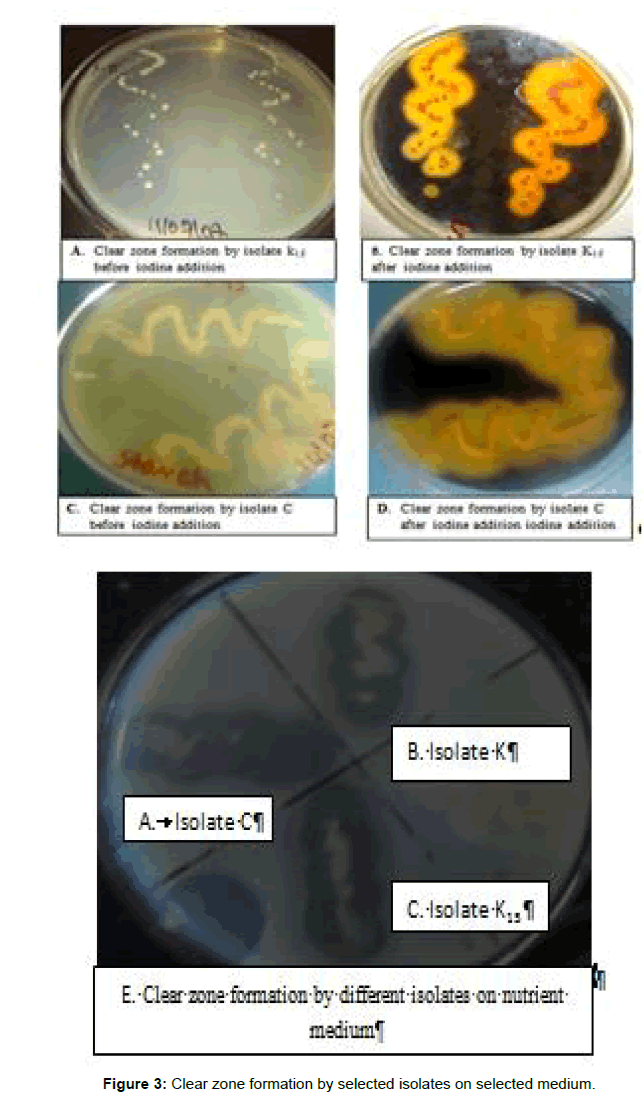
Out of the eight bacterial isolates obtained from various samples, were screened based on tolerance of NaCl, clear zone formation, temperature, pH and fermenting ability of carbohydrate sources. Two isolates (K15 and K) tolerate 2% and 4% NaCl media, one isolate (C) grow at higher concentration of NaCl containing medium (Table 4). Among eight isolates 1 isolate not grow in NaCl medium. Addition of NaCl in the food inhibits the growth of pathogenic and food spoilage bacteria but not LAB as mentioned by [22,29]. The experimental result represented in (shows that the isolated bacterial strains from different samples were able to survive in extreme acidic pH (pH 4) to basic pH (pH 8). Maximum growth of isolated strain k (OD=1.22) was observed at pH 5 and maximum growth of isolated strain K15 (OD=1.20) and C (OD=0.86) at pH 6. The reason for choosing this pH range was to determine whether LAB spp. can grow in acidic and alkaline conditions and also to predict the optimum pH value for good growth [17]. Isolate C and K15 were growing best at 5°C to 45°C. As described by [1], enset fermentation in cooler environment product stored up to years until three years anaerobically but in higher temperature up to 6 month. Isolated strains grow best at lower temperature except isolate C grows at higher temperature. Clear zone formation is another criterion for selection of potential starter culture bacteria (Figure 3). Clear zone formation is due to the production of lactic acid by inoculated strain. Isolate K15 form more clear zone 1.5 cm than isolate C forms 1 cm. All selected potential strains ferment carbohydrate sources but isolate C and K15 ferment glucose with gas production isolate K ferment without gas production. Growth determined by using UV spectrophotometer isolate C optical density (OD) measured at 600 nm 0.514 at 24 h and 1.511 at 48 h. Increase in OD is due to the growth of bacteria. Isolate k15 0.833 at 24 h and 1.485 at 48 hrs, isolate k 0.912 at 24 h and 1.868 at 48 h control 0.207 at 24 h and 48 h. Increasing in OD shows the isolated strain produce lactic acid. The highest gas productions by inoculated strain are due to the production of lactic acid. According to [25] study Organisms which produced acid readily are considered to be those for which acid production was more formed on the glucose containing medium. Isolates strains are similar to Merrill study in carbohydrate fermentation. The highest acid production observed by isolate K15 and C (Figure 4).
| Isolates | NaCl (%) | pH (OD) | Temperature | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 2 | 4 | 6 | 4 | 5 | 6 | 8 | 5 | 10 | 15 | 30 | 45 | |
| K15 | + | + | - | 0.987 | 1.021 | 1.201 | 0.941 | + | + | + | + | - |
| K | + | + | - | 1.122 | 1.220 | 1.110 | 0.972 | + | + | + | + | - |
| C | + | + | + | 0.682 | 0.821 | 0.861 | 0.917 | + | + | + | + | + |
| control | ND | ND | ND | 0.341 | 0.341 | 0.341 | 0.341 | ND | ND | ND | ND | ND |
Table 4: Screening parameters of selected isolates.
Selection of potential lactic acid producing bacteria
Potential starter culture bacteria selected based on their properties: fermentation of carbohydrate sources, NaCl, pH and temperature tolerance and clear zone formation, isolate K15 and C was selected for further work as presented on (Table 5).
| Isolates | Carbohydrate source utilization (OD) | Gas production (cm) | Durham tube height (cm) | ||||||
|---|---|---|---|---|---|---|---|---|---|
| glucose | sucrose | fructose | starch | glucose | sucrose | fructose | starch | ||
| K15 | 1.86 | 0.95 | 1.43 | 1.69 | 3.0 | 2.5 | 2.8 | 3.0 | 7 |
| K | 1.48 | 1.21 | 1.44 | 1.21 | 1.0 | 1.0 | 1.1 | 1.2 | 7 |
| C | 1.51 | 0.95 | 1.32 | 1.21 | 2.0 | 1.5 | 1.5 | 1.0 | 7 |
| control | 0.20 | 0.20 | 0.20 | 0.20 | 0.0 | 0.0 | 0.0 | 0.0 | 7 |
Table 5: Carbohydrate utilization and gas production by selected isolates.
Optimization of fermentation conditions by selected isolates
Isolate K15 and C was selected due to its highest ability to produce lactic acid (Figure 5). The bacterial fermentation conditions and acid production were optimized under various environmental and culture conditions (Table 6).
| Isolate | Inoculum size (ml) | Temperatures (oC) | Incubation (days) | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 2 | 4 | 6 | 5 | 10 | 15 | 30 | 45 | 3 | 6 | 9 | 12 | 14 | 16 | |
| K15 | +++ | + | + | +++ | +++ | +++ | + | + | + | + | ++ | +++ | +++ | ++ |
| C | +++ | + | + | +++ | +++ | +++ | + | + | + | + | ++ | +++ | +++ | ++ |
Table 6: Optimization of fermentation conditions.
Effect on inoculums size: The optimum inoculums size for enset fermentation was 2 ml of 24 h growth culture. The maximum acid production happened at 6 ml at 12 days incubation time. Increase in inoculums size affects the end product in terms of test, color, smell and flavor by changing the product sour test, black in color and forming offensive odor. The product became very sour test and black in color.
At lower and higher inoculums sizes the lactic acid production was decreased. This may be accounted for the limited number of cells to perform the fermentation at low inoculums and the resulting competition at high inoculums levels.
Effect on temperature: Temperature is an important factor for good enset fermentation. Mogesse [1] reported that enset fermentation product stored for longer periods in cooler environment than higher temperature. In this study good fermentation obtained from optimum temperature 5ºC. Both Isolate k15 and C strains have ability to give good product in cooler environment similar to the Mogesse [1]. Enset fermentation in higher temperature inhibits the pathogenic microorganisms than lower temperature fermentation.
Effect on fermentation time: From an economic point of view, fermentation time is a very important factor in enset fermentation. The lesser the incubation period the better is the isolate. Different incubation period was reported for enset fermentation. According to Tiruha [5] study reported that enset fermentation takes minimum one month to obtain good product. Similarly Mogesse [1] reported enset fermentation takes minimum weeks to several months. However, a much lesser optimum incubation time was recorded from using starter culture bacteria.
Comparison of starter used and traditionally fermented enset
The starter culture fermented and traditionally fermented enset was compared in terms of nutritional values using similar parameters mentioned in Titrable acidity. The highest nutritional content obtained from starter fermented enset than traditionally fermented enset in terms of fat, protein, ash, fiber, moisture content and carbohydrate content as presented on the (Table 1). The highest TA and PH were obtained from starter fermented than traditionally fermented enset. This difference was due to the production of lactic acid by starter culture organisms during fermentation periods (Figure 6).
Sensory evaluation of starter used and traditionally fermented enset
Sensory properties were adopted as the main criterion of the quality of fermented products, being the most important attribute for the consumers. The addition of starters that have improved sensory characteristics of the enset fermented in single strain compared to the traditionally fermented one. Isolate C and K15 inoculated samples found to be better than traditionally fermented enset in terms of taste, texture, flavor, color and nutritional value. During evaluation panelists were surprised by starter culture containing sample by its color and test. They asked “how to ferment this kind of kocho? What you used to ferment? If the process reduces the fermentation time we need it.” Starter culture used kocho accepted in color, test and smell than traditionally fermented enset. Evaluators tell that “starter used kocho is better for consumption and health compared to the traditionally fermented.” Starter used kocho has preferred smells by consumers than traditionally fermented enset and its test is also accepted by panelists. Isolate C and K15 are better acceptance during evaluation in terms of color, test and texture. The control was worst in quality due to less sour (taste), smell (flavor), and black (color), wherever the samples treated with single culture were acceptable for sensory quality (Figure 7).
However, no significant differences for texture and test appearance were found among batches with single starters except color difference in isolate C. The samples K15 and C had significantly higher scores for flavor than the traditionally fermented one; the sample K15 and C had significantly higher scores for taste than the control. Therefore, K15 and C was the most preferred by panelists. The better flavor score was obtained from K15 and C better color score was obtained from isolate C. Mixed strain (C and K15) containing sample similar with isolate K15 with a little color difference but the degradation is the same as K15. Isolate C and K15 single strains fermented enset positively affected by white color development and stability. Enset fermented with multiple strain isolate K15 mixed with isolate C there is a small color difference only. Control heat treated without inoculums and traditionally fermented enset turned to black when exposed to air. Bacteria contribute for color development and stability. This is due to the production of antioxidant by bacteria. Anti-oxidants are the most frequently used functional food compounds. The main source of these substances is plant material. Antioxidants play a fundamental role in the prevention of cancer and cardiovascular diseases. They act as scavengers of reactive oxygen species and metal chelators that protect human cells and reduce oxidative damages [30]. In this study isolated strain used as starter culture for enset fermentation have multiple benefits such as reduce incubation time by 36.7%. This means traditional fermentation is time consuming process and lobar intensive [1]. But in this study prepared starter reduces by 12 days [5]. It also increases nutritional value this is because in traditional fermentation has nutritional deficiency in protein and vitamin A so this starter increases its nutritional value. This may be due to the presence of organisms which degrade amino acids. In traditional fermentation different kinds of organisms involved but due to interaction for food they do not function properly. In addition to this isolates are acceptable by panelists in terms of color, test, flavor and smell. Using this starter culture add value for the farmers by reducing fermentation time, increase nutritional value and provide quality product.
Conclusion and Recommendations
Among 8 lactic acid bacteria isolated 2 potential strains were able to degrade selected substrate than other strains. Based on their higher degrading ability and production of color and smell isolate K15 and C were selected for further study. The isolate was identified as Lactic acid bacteria (lactobacillus spp. and streptococcus spp.) by using Bergaye’s manual of bacteriology. It grew best and ferment enset at 5ºC temperature in MRS medium. Glucose and peptone were best carbon and nitrogen sources for lactic acid production respectively. The type of lactic acid produced was affected by the type of substrate used for the fermentation. The best source for strong lactic acid production applicable for enset fermentation was 15 day pre-fermented enset and maize. Enset fermented by lactobacillus spp. was benefit for consumers as selected by panelists. This isolates can be regarded as potential strain for the fermentation of enset. This would have health, economic and environmental benefits for the country in particular and the world in general.
• Based on the results of this study, the following recommendations are forwarded.
• Further characterization of the isolated bacteria by using metagenomics or 16s rRNA to confirm the isolate as LAB.
• Scale up of the starter culture production to pilot and industrial levels using easily available substrates.
• Commercialize the technology as it would improve kocho quality, fermentation time and create employment opportunity to list some for the people in enset growing regions.
• Reduce work load of women and children involved in enset processing by introducing the technology.
References
- Mogessie A (2006) A review on the microbiology of indigenous fermented foods and beverages of Ethiopia. Ethiop J Biol Sci 5: 89-245.
- Leroy F, Verluyten J, De Vuyst L (2006) Functional meat starter cultures for improved sausage fermentation. Int J Food Microbiol 106: 270-285
- Voula A, Effie T, Konstantinos P, Wilhelm H (2013) Status and trends of the conservation and sustainable use of microorganisms in food processes, commission on genetic resources for food and agriculture. background study paper No. 65
- Soro Y, Kouakou B, Georges A, Philippe T, Koffi M (2014) The Use of Lactic Acid Bacteria Starter Cultures during the Processing of Fermented Cereal-Based Foods in West Africa: A Review. Trop Life Sci Res 25: 81-100
- Tiruha H, Kebede A, Edessa N (2014) The microbiology of kocho: An Ethiopian traditional fermented food from Enset. Int J Life Sci 8: 7-13.
- Admasu T, Struik P (2001) Enset (Ensete ventricosum(Welw) Cheesman) kocho yield under different crop establishment methods as compared to yields of other carbohydrate-rich food crops. Netherlands J Agri Sci 49: 81-94
- Yewelsew A, Barbara J, Margaret J, Gail E (2006) Nutritive value and sensory acceptability of corn and kocho based foods supplemented with legumes for infant feeding in southern Ethiopia. Afr J Food Agric Nutr Dev 6: 1684-5376.
- Temesgen M, Bizuayehu T, Catellani M, Enrico M (2014) Indigenous knowledge, use and on farm management of Enset (Ensete ventricosum (Welw) Cheesman) diversity in Wolaita, Southern Ethiopia. J Ethnobiol Ethnomed 10: 41.
- Ayalew D (2006) Studies of Enset /Enseteventricosum/ for major, minor and trace elements MSc thesis, chemistry department Addis Ababa University, Ethiopia.
- Melese T (2013) Improving the Indigenous Processing of Kocho, an Ethiopian Traditional Fermented Food. J Nutr Food Sci 3:1
- AOAC, 1995, Official methods of analysis. (16th edtn), Gaithers burg, MD.
- Tefera T, Ameha K, Biruhtesfa A (2014) Cassava based foods: microbial fermentation by single starter culture towards cyanide reduction, protein enhancement and palatability. Int Food Res J 21: 1751-1756.
- Buchanan R, Gibbonns N (1994) Bergeys Manual of Determinative Bacteriology, 8th edtn. Afr J Biotechnol 2: 3151-3157.
- Patra A, Sil J, Das B (2011) Isolation and characterization of dominant lactic acid bacteria from Dahi at Medinipur and evaluation of their antibacterial activity. Int J of Food Safety 13: 157-163.
- Goyal R, Dhingra H, Bajpai P, Joshi N (2012) Characterization of the Lactobacillus isolated from different curde samples. Afr J Biotechnol 11: 14448-14452
- Narasimhulu K, Rao P, Vinod A (2010) Isolation and Identification of Bacterial Strains and Study of their Resistance to Heavy Metals and Antibiotics. J Microb Biochem Technol 2: 074-076.
- Sharma K, Roy R, Sathiavelu M, Arunachalam S (2013) Study of Probiotic and Antioxidant activity of Lactobacillus spp. Res J Pharm Biol and Chem S 4: 0975-8585.
- Anjum R, Zahoor T, Akhtar S (2007) Comparative study of yoghurt prepared by using local isolated and commercial imported starter culture. J Res Sci 18: 35-41.
- Adebowale O, Maliki K (2011) Effect of fermentation period on the chemical composition and functional properties of Pigeon pea (Cajanuscajan) seed flour. Int Food Res J 18: 1329-1333.
- Wizna, Abbas H, Rizal Y, Dharma A, Kompiang I (2009) Improving the quality of tapioca by-products (onggok) as poultry feed through fermentation by Bacillus amyloliquefaciens. Pak J Nutr 8: 1636-1640.
- Merrill N, Max S, Dunn S, Salle A (2015) The effect of carbohydrates on acid production by twenty four lactic acid bacteria. J Biol Chem 168: 33-42.
- Chang HY, Shim Y, Cha K Chee M (2009) Probiotic characteristics of lactic acid bacteria isolated from kimchi, Original article. J Appl Microbiol 109: 220-230.
- Teshome G (2015) Review on lactic acid bacteria function in milk fermentation and preservation. Afr J Food Sci 9: 170-175.
- Kucerova K, Svobodova H, Tuma S, Ondrackova I, Ova P (2009) Production of Biogenic Amines by Enterococci. Czech J Food Sci 27: 50-55.
- Kockova M, Gerekova P, Petrulakova Z, Hybenova E, Sturdik E, et al. (2011) Evaluation of fermentation properties of lactic acid bacteria isolated from sourdough. Acta Chimica Slovaca 4: 7-87
- Goyal R, Dhingra H, Bajpai P, Joshi N (2012) Characterization of the Lactobacillus isolated from different curde samples. Afr J Biotechnol 11: 14448-14452
- Patil M, Pal A, Anand T, Ramana V (2010) Isolation and characterization of lactic acid bacteria from crud and cucumber. Indian J Biotechnol 9: 166-172.
- Ekundayo O (2014) Isolation and identification of lactic acid bacteria from rhizosphere soils of three fruit trees, fish and ogi Original Research. ArticleInt J Curr Microbiol App Sci 3: 991-998.
- Cai Y, Ohomom S, Ogawa M, Kumai S (1997) Effect of NaCl tolerant lactic acid bacteria and NaCl on the fermentation characteristics and aerobic stability of silage. J Appl Microbiol 83: 307-313.
- Grajek W, Olejnik A, Sip A (2005) Probiotics, prebiotics and antioxidants as functional foods. Acta Biochim Pol 52: 665-671.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi