Case Report, J Vet Sci Med Diagn Vol: 8 Issue: 3
New Zealand White Rabbits Fed High Cholesterol Diets Develop Morbid Systemic Diseases before Intracranial Atherosclerosis is Detected
- *Corresponding Authors:
- Matthew Alexander
Departments of Radiology and Imaging Sciences
University of Utah, Salt Lake City, Utah, USA,
Tel: 8015877996
Received: August 05, 2019 Accepted:August 28, 2019 Published: September 03, 2019
Abstract
Intracranial atherosclerotic disease (ICAD) lacks sufficient human data; a suitable animal model is needed. Watanabe heritable hyperlipidemic (WHHL) rabbits have limitations for ICAD. New Zealand white (NZW) rabbits fed high cholesterol diets and WHHL rabbits were evaluated. 14 rabbits underwent evaluation with MRI and pathology. No NZW rabbits (8 fed high cholesterol diet, 2 fed regular diets) developed ICAD. 6 (75%) high-cholesterol rabbits did not survive for the planned timeframe. All 4 WHHL rabbits developed ICAD. Methods other than induction with a high cholesterol diet should be pursued for an ICAD model.
Keywords: Atherosclerosis; Hyperlipidemia; Rabbit; New Zealand white
Introduction
Intracranial atherosclerotic disease (ICAD) is a significant cause of ischemic stroke that leads to long-term morbidity and mortality in millions of patients worldwide each year, placing substantial economic and emotional costs on society [1]. There are differences in treatment algorithms among practitioners, medical centers, and geographic regions. Conflicting algorithms result from conflicting research findings, and pathophysiology data in particular are lacking. In the quest for improved pathophysiological understanding, sufficient human data are impossible to acquire, so animal model data must suffice. The Watanabe heritable hyperlipidemic (WHHL) rabbit model is the most extensively utilized atherosclerotic animal known to develop ICAD [2-5]. However, the mechanism by which it develops this disease does not replicate the way by which ICAD develops in the vast majority of humans [3-7]. Furthermore, supply of these animals has recently proven unreliable, so the need for a more sufficient model is acute [8]. Diet-based models exist for atherosclerosis, producing disease in wild type rabbits fed high cholesterol diets, yet these models have not been evaluated for development of intracranial disease [4,5,9-12].
Case Study
This investigation examines the intracranial arteries of animals fed a high cholesterol diet, utilizing vessel wall magnetic resonance imaging (VWI) and histopathological stains to evaluate for the development of ICAD.
Materials and Methods
IACUC protocol
All rabbits were evaluated under a protocol approved by the Institutional Animal Care and Use Committee at our institution.
MRI protocol
All rabbits underwent baseline VWI using a standardized protocol at 3T. General anesthesia was induced with intramuscular injection of buprenorphine (0.03 mg/kg) followed approximately 30 minutes later by a ketamine (25-35 mg/kg) and xylazine (3 mg/kg) mixture; anesthesia was then maintained as needed with isoflurane in oxygen via endotracheal tube. Following induction of general anesthesia, diffusion-weighted imaging (DWI) and fluid attenuated inversion recovery (FLAIR), 3D T1-weighted black-blood vessel wall imaging (T1BB), susceptibility weighted imaging (SWI) sequences were obtained. After the intravenous injection of 0.1 mmol/kg of gadolinium contrast, T1BB was repeated. The same protocol was employed in all subsequent VWI studies performed on a monthly basis for the remainder of the rabbits’ lives.
Diet protocol
WHHL and New Zealand white (NZW) rabbits were continued on regular diets to serve as diseased and normal controls, respectively. Additional NZW rabbits were transitioned from regular diets to custom 2% or 4% cholesterol pellets (Envigo, Madison, WI) supplemented with hay, alfalfa, and greens. Monthly VWI studies were scheduled with the intent of performing euthanasia after three months to evaluate for signs of ICAD. Different cholesterol amounts were utilized to assess for a dose effect. If animals demonstrated failure to thrive prior to three months, euthanasia was performed.
Euthanasia and pathology
Euthanasia was performed with perfusion fixation. Maintaining deep general endotracheal anesthesia, rabbits were moved to a fume hood. A midline incision was made over the neck and blunt dissection performed to expose one of the carotid arteries. Using salinger technique, a 4 French vascular sheath was introduced into the exposed carotid artery and secured with sutures. The internal jugular veins, having been exposed during surgical access for catheterization of the carotid artery, were transected. A steady flow of 2% paraformaldehyde and 5% glutaraldehyde solution was then infused into the indwelling sheath to achieve fixation and allow ex vivo histological analysis. The animal was decapitated and the brain and intracranial arteries harvested and placed in a container with formalin. After two weeks, the brain specimens were sliced to prepare slides. Slices were oriented in such a way to maximize the cross-sectional orientation of the proximal intracranial arteries. Analysis was performed with light microscopy after hematoxylin and eosin (H&E) staining to evaluate the vessel walls for presence of ICAD. Qualitative comparisons were made between control and high cholesterol diet rabbits, specifically evaluating vessels for presence of intracranial atherosclerosis and brain parenchyma for presence of infarct.
Statistical analysis
Descriptive statistics were performed to characterize the animals, diets implemented, and survival to the targeted three month duration of exposure. Independent sample T-tests was performed to assess development of ICAD and survival among the animal groups.
Results
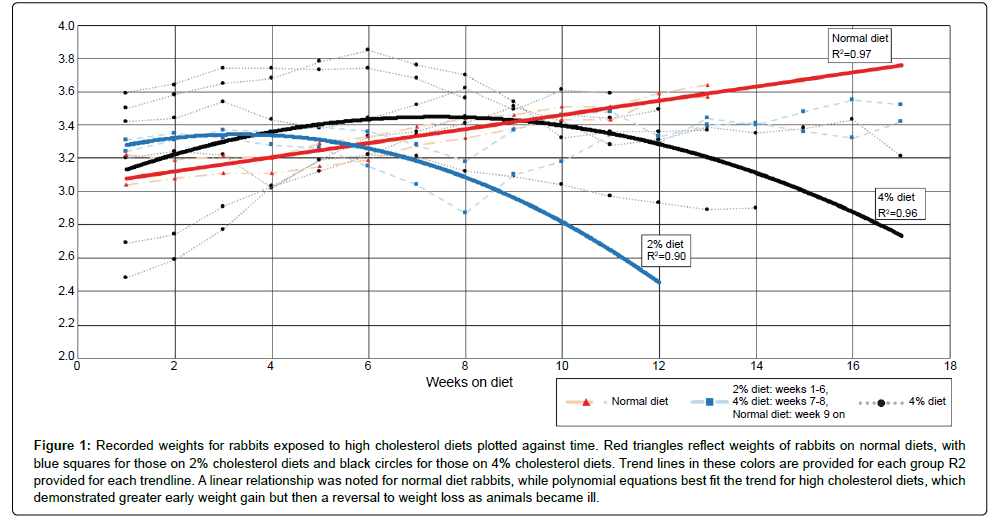
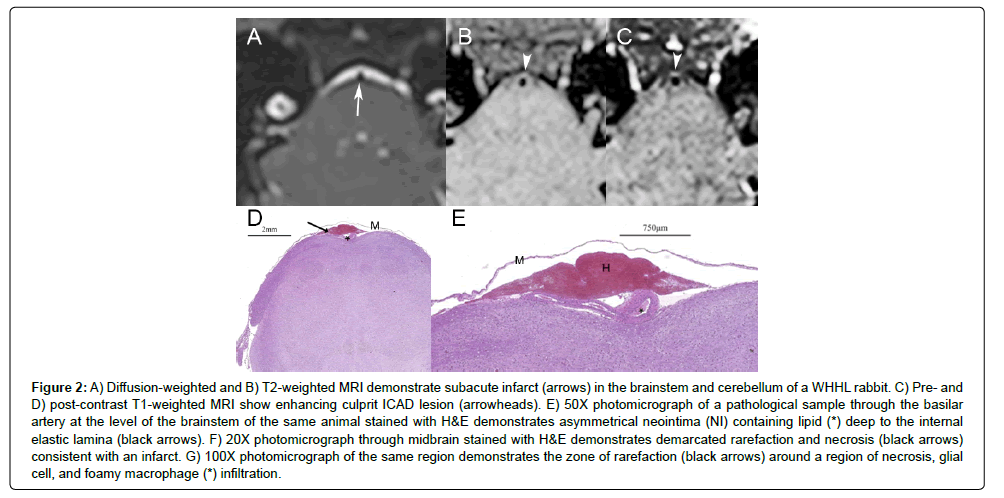
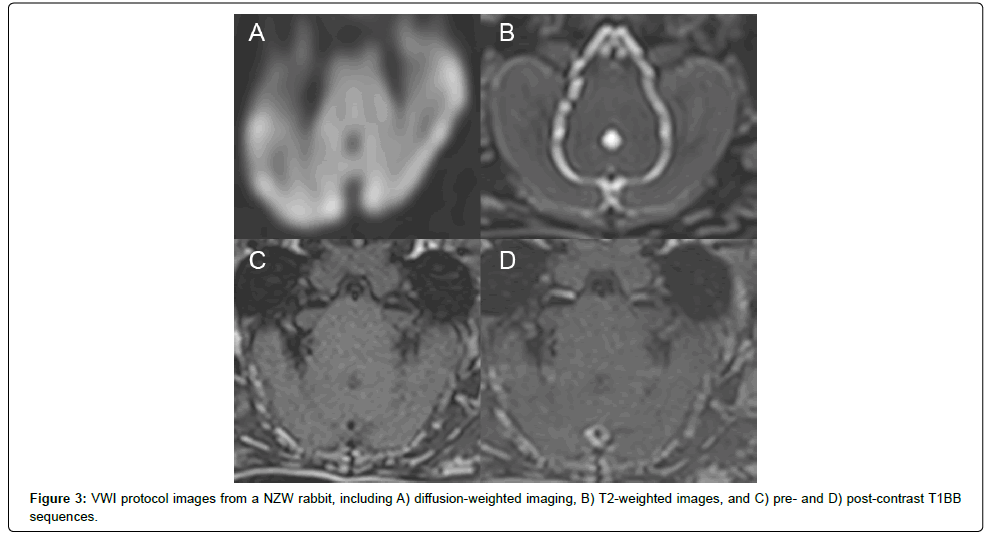
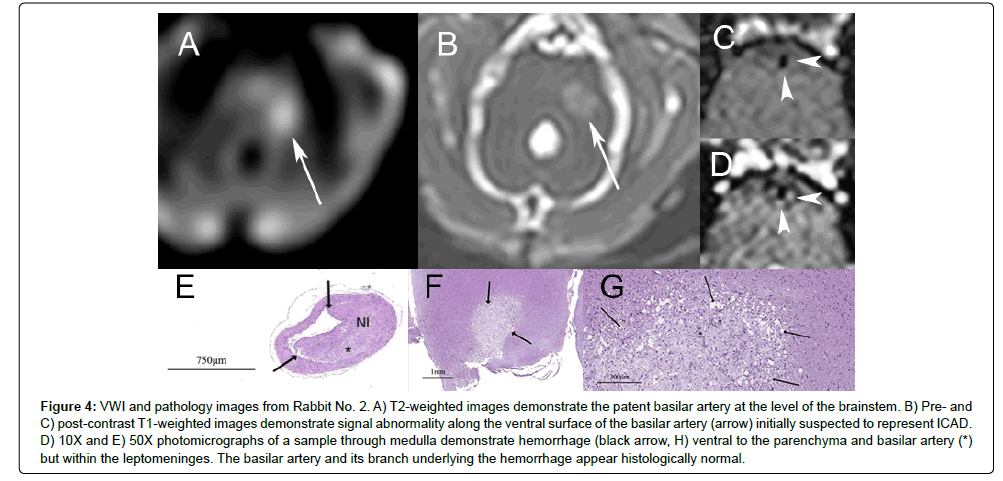
14 total rabbits underwent investigation. 8 animals were exposed to high cholesterol diet. Two NZW rabbits received 2% cholesterol diet, while 6 received 4% cholesterol diet. Table 1 summarizes the animals fed high cholesterol diets. Two NZW and 4 WHHL rabbit received normal diets. Changes in animal weights with respect to time are graphed in Figure 1. ICAD was demonstrated in all WHHL rabbits on both VWI and histopathology (Figure 2); no ICAD was visualized on VWI or histopathology in any of the NZW rabbits, regardless of diet (Figure 3). One NZW rabbit on 4% cholesterol diet demonstrated abnormality in the basilar artery on VWI that was initially suspected to represent early ICAD (Figure 4). However, pathological analysis following perfusion fixation demonstrated subleptomeningeal blood but no ICAD (Figure 3).
Figure 1:Recorded weights for rabbits exposed to high cholesterol diets plotted against time. Red triangles reflect weights of rabbits on normal diets, with blue squares for those on 2% cholesterol diets and black circles for those on 4% cholesterol diets. Trend lines in these colors are provided for each group R2 provided for each trendline. A linear relationship was noted for normal diet rabbits, while polynomial equations best fit the trend for high cholesterol diets, which demonstrated greater early weight gain but then a reversal to weight loss as animals became ill.
Figure 2:A) Diffusion-weighted and B) T2-weighted MRI demonstrate subacute infarct (arrows) in the brainstem and cerebellum of a WHHL rabbit. C) Pre- and D) post-contrast T1-weighted MRI show enhancing culprit ICAD lesion (arrowheads). E) 50X photomicrograph of a pathological sample through the basilar artery at the level of the brainstem of the same animal stained with H&E demonstrates asymmetrical neointima (NI) containing lipid (*) deep to the internal elastic lamina (black arrows). F) 20X photomicrograph through midbrain stained with H&E demonstrates demarcated rarefaction and necrosis (black arrows) consistent with an infarct. G) 100X photomicrograph of the same region demonstrates the zone of rarefaction (black arrows) around a region of necrosis, glial cell, and foamy macrophage (*) infiltration.
Table 1: Summary of NZW rabbits fed high cholesterol diet and outcomes.
| Rabbit No. | Diet | Days of Diet | Euthanasia Indication | Intracranial Artery Pathology Results |
|---|---|---|---|---|
| 1 | 4% | 94 | N/A (Died Under Anesthesia) | Normal |
| 2 | 4% | 108 | Failure to Thrive | Subleptomeningeal Hemorrhage |
| 3 | 4% | 63 | Failure to Thrive | Normal |
| 4 | 4% | 63 | Failure to Thrive | Normal |
| 5 | 4% | 49 | Failure to Thrive | Normal |
| 6 | 4% | 91 | Scheduled | Normal |
| 7 | 4% | 98 | N/A(Died in Cage) | Normal |
| 8 | 4% | 114 | Scheduled | Normal |
All 4 WHHL and the 2 normal diet NZW rabbits survived to the three month target for euthanasia. Two (25.0%) high cholesterol diet rabbits survived to scheduled euthanasia at three months. Four (50.0%) required earlier euthanasia due to failure to thrive but were able to undergo euthanasia on an elective basis. Two (25.0%) high cholesterol diet animals died outside protocol. One died under anesthesia for VWI on diet day 94. Necroscopy demonstrated pale appearance of the liver, a pale spleen with multifocal white spots, pale brown kidneys with multifocal tan spots, pale lungs, and fatty deposits on the heart. The remaining high cholesterol diet rabbit was found dead in its cage on diet day 98. Necroscopy demonstrated yellow and firm liver, an enlarged spleen with mottled appearance, pale lungs, and free fluid in the abdominal and thoracic cavities.
WHHL rabbits were more likely to have ICAD confirmed on pathology compared to NZW rabbits (100% vs. 0.0%, p<0.001). NZW rabbits fed a high cholesterol diet were less likely to survive to the planned three month timeframe (62.5% vs. 100%, p=0.003). High cholesterol diet rabbits were also less likely to survive to scheduled euthanasia (25% vs. 100%, p=0.001).
Discussion
ICAD is the most common cause of ischemic stroke worldwide [1,13]. Current conceptual understanding of atherosclerosis invokes systemic processes that cause lesions at focal points of vulnerability, but this understanding is based on atherosclerosis research that has been performed outside the intracranial circulation [14,15]. Such extrapolation can be misleading because intracranial and extracranial arterial systems are derived from different germ cell layers, so vascular diseases in these separate systems should be considered distinct processes [16,17]. Differences between ICAD and extracranial atherosclerotic disease have undergone little investigation, particularly when considering recent insights into the role of inflammation in atherosclerosis, yet many approaches to ICAD diagnosis and treatment are based on extrapolation from outmoded extracranial research.
To perform investigation of ICAD that adequately characterizes its pathophysiology and natural history, human investigations alone will be insufficient for several reasons. ICAD often begins with an asymptomatic interval that can last for years and is typically diagnosed only at later, advanced stages. Those with the disease often have concomitant diseases, many of which are they risk factors for vascular disease. These additional diseases, or treatments for them, can confound analysis of ICAD.
Additionally, the above-described discrepancies in surveillance and treatment schemes employed among managing providers further hinder analysis in humans. Indeed, approaches to management are often directly contradictory to one another. Finally, histopathologic confirmation, widely considered the gold standard in assessment of human disease, is prohibitively morbid to obtain, so current management relies on potentially faulty extrapolation of atherosclerosis research performed elsewhere in the body. Longitudinal evaluation completed with histopathological confirmation will require analysis in an animal model.
Rabbit models of atherosclerosis have a long history for the investigation of peripheral and coronary artery atherosclerosis [5]. However, little investigation has occurred for ICAD. The Watanabe heritable hyperlipidemic (WHHL) rabbit model lacks low density lipoprotein (LDL) receptors and reliably develops systemic atherosclerosis [2,3,5,18,19].This model provided the first characterization of underlying abnormality in individuals with familial hypercholesterolemia, the human condition WHHL rabbits most closely model [3]. While indispensable for the understanding of coronary atherosclerosis, this model has limited applicability for intracranial disease. Humans with familial hypercholesterolemia often demonstrate sparing of the intracranial vasculature, and the vast majority of ICAD occurs in individuals without this rare disease [20-22]. WHHL rabbits only reliably develop intracranial disease after induction of systemic hypertension, which can be problematic considering their already general poor health [6,7]. Additionally, these rabbits breed poorly, and all but one breeding colony has been depopulated [8]. Considering its poor fidelity to the pathophysiology of human ICAD and uncertain availability in the future, and in light of the pressing need for an animal model of ICAD as described above, we aimed assess a possible rabbit model that replicates a high cholesterol diet described elsewhere that has not yet been evaluated for intracranial disease[11].
In an effort to preliminarily evaluate development of ICAD in an expedited fashion, we began by assessing six NZW rabbits, two each fed regular, 2% cholesterol, and 4% cholesterol diets. Among these initial six animals, one developed abnormality on VWI within weeks. Weeks later, pathological analysis the abnormality on VWI was shown to actually represent subleptomeningeal blood, but initially the finding was erroneously interpreted as early ICAD (Figure 4). After initial observation of the VWI abnormality, the rabbit was continued on the 4% diet to allow further progression of the presumed lesion. This animal did uncharacteristically well on what would typically be considered a morbidly unhealthy diet until approximately 100 days after diet initiation, when it began demonstrating signs of failure to thrive. In light of these results, four additional NZW rabbits were placed on 4% cholesterol diets. No ICAD developed in any of these animals, and poor health was observed in keeping with previously described reports of high cholesterol diets [5]. It is possible that rabbits may eventually develop ICAD when fed high cholesterol diets, but the morbid systemic effects that result from such a diet precede these findings and make this an unsatisfactory model of ICAD.
Figure 4:VWI and pathology images from Rabbit No. 2. A) T2-weighted images demonstrate the patent basilar artery at the level of the brainstem. B) Pre- and C) post-contrast T1-weighted images demonstrate signal abnormality along the ventral surface of the basilar artery (arrow) initially suspected to represent ICAD. D) 10X and E) 50X photomicrographs of a sample through medulla demonstrate hemorrhage (black arrow, H) ventral to the parenchyma and basilar artery (*) but within the leptomeninges. The basilar artery and its branch underlying the hemorrhage appear histologically normal.
Since creation of the WHHL model decades ago, several other rabbit models have been created [5]. While the diet-based approach described in this report did not achieve desired results, additional models should be investigated. These include transgenic or knockout rabbits [23-25]. While active investigations continue to examine this multitude of rabbit models of atherosclerosis, to date none of them have been applied to investigations of ICAD in particular. Investigation of intracranial disease is warranted and should be pursued in the future.
Conclusion
Given difficulties in acquiring high quality data in humans, a robust animal model of ICAD is needed. In this study we evaluated a high-cholesterol NZW rabbit model in an attempt to induce ICAD. ICAD was not observed in any NZW rabbits on the high cholesterol diet, and substantial morbidity from systemic illnesses ensued in animals on the altered diet. This argues that other rabbit models should be pursued.
Acknowledgements
The authors wish to thank Dr. Jessica Durrant of HistoTox Labs, Inc. for pathology analysis. Contributions: MSZ and MDA designed the study, analyzed data and edited the manuscript. SW, SEK, JSM, AHD, and DLP participated in study design, analyzed data, and edited the manuscript. NIH S10OD018482, NIH R01HL127582, NIH/NINDS K23NS105924, American Heart Association Scientist Development Grant 17SDG33460420, and American Heart Association Transformational Project Award 19TPA34910194.
Conflict of Interest
This research was funded in part by the Joe Niekro Research Grant from the Joe Niekro Foundation and Society of NeuroInterventional Surgery Foundation.
References
- Chimowitz MI, Lynn MJ, Howlett-Smith H, Stern BJ, Hertzberg, VS, et al. (2005) Comparison of warfarin and aspirin for symptomatic intracranial arterial stenosis. N Engl J Med 352: 1305-1316.
- Aliev G, Burnstock G (1998) Watanabe rabbits with heritable hypercholesterolaemia: A model of atherosclerosis. Histol Histopathol 13: 797-817.
- Goldstein JL, Kita T, Brown MS (1983) Defective lipoprotein receptors and atherosclerosis. Lessons from an animal counterpart of familial hypercholesterolemia. N Engl J Med 309: 288-296.
- Wang Z, Zhang J, Li H, Li J, Niimi M, et al. (2016) Hyperlipidemia-associated gene variations and expression patterns revealed by whole-genome and transcriptome sequencing of rabbit models. Sci Rep 6: 26942.
- Yanni AE (2004) The laboratory rabbit: an animal model of atherosclerosis research. Lab Anim 38: 246-256.
- Ito T, Shiomi M (2001) Cerebral atherosclerosis occurs spontaneously in homozygous whhl rabbits. Atherosclerosis 156: 57-66.
- Kong J, Tamaki N, Asada M (2000) Early lesions of cerebral atherosclerosis from induced hypertension in watanabe heritable hyperlipidemic rabbits. Kobe journal of medical sciences 46: 87-101.
- Shiomi M (2017) Whhl rabbits at kobe and elsewhere. In: Alexander MD, editor. Email describing plans to close the WHHL/WHHLMI rabbit colony at Kobe University in March 2018. Kobe University, Kobe, Japan.
- Fan J, Watanabe T (2000) Cholesterol-fed and transgenic rabbit models for the study of atherosclerosis. J Atheroscler Thromb 7: 26-32.
- Niimi M, Yang D, Kitajima S, Ning B, Wang C, et al. (2016) Apoe knockout rabbits: A novel model for the study of human hyperlipidemia. Atherosclerosis 245: 187-193.
- Kolodgie FD, Katocs AS, Largis EE, Wrenn SM, Cornhill JF, et al. (1996) Hypercholesterolemia in the rabbit induced by feeding graded amounts of low-level cholesterol. Methodological considerations regarding individual variability in response to dietary cholesterol and development of lesion type. Arterioscler Thromb Vasc Biol 16: 1454-1464.
- Bocan TM, Mueller SB, Mazur MJ, Uhlendorf PD, Brown EQ, et al. (1993) The relationship between the degree of dietary-induced hypercholesterolemia in the rabbit and atherosclerotic lesion formation. Atherosclerosis102: 9-22.
- Wong KS, Li H, Chan YL, Ahuja A, Lam WW, et al. (2000) Use of transcranial doppler ultrasound to predict outcome in patients with intracranial large-artery occlusive disease. Stroke; A J Cerebral Circulation 31: 2641-2647.
- Stoll G, Bendszus M (2006) Inflammation and atherosclerosis: Novel insights into plaque formation and destabilization. Stroke 37: 1923-1932.
- Libby P, Ridker PM, Hansson GK (2009) Inflammation in atherosclerosis: from pathophysiology to practice. J Am Coll Cardiol 54: 2129-2138.
- Lasjaunias PL, Berenstein A, Brugge KGT (2001) Surgical neuroangiography. Springer,New York, USA.
- Postiglione A, Nappi A, Brunetti A, Soricelli A, Rubba P, et al. (1991) Relative protection from cerebral atherosclerosis of young patients with homozygous familial hypercholesterolemia. Atherosclerosis 90: 23-30.
- Rosenfeld ME, Tsukada T, Gown AM, Ross R (1987) Fatty streak initiation in watanabe heritable hyperlipemic and comparably hypercholesterolemic fat-fed rabbits. Arteriosclerosis 7: 9-23.
- Shiomi M, Ito T, Shiraishi M, Watanabe Y (1992) Inheritability of atherosclerosis and the role of lipoproteins as risk factors in the development of atherosclerosis in whhl rabbits: Risk factors related to coronary atherosclerosis are different from those related to aortic atherosclerosis. Atherosclerosis. 96: 43-52.
- Ding X, Li C, Yu K, Gao A, Xiao L, et al. (2014) Different risk factors between intracranial and extracranial atherosclerotic stenosis in asian population: A systematic review and meta-analysis. Int J Neurosci 124: 834-840.
- Lindenholz A, Kolk AGVD, Schaaf IVD, Worp BVD, Harteveld A, et al. (2018) Vascular risk factors and intracranial atherosclerosis at 7t vessel wall mri in caucasian ischemic stroke and tia patients. International Society for Magnetic Resonance in Medicine, Paris, France.
- Qureshi AI, Caplan LR (2014) Intracranial atherosclerosis. Lancet 383: 984-998.
- Brousseau ME, Hoeg JM (1999) Transgenic rabbits as models for atherosclerosis research. J Lipid Res 40: 365-375.
- Ji D, Zhao G, Songstad A, Cui X, Weinstein EJ (2015) Efficient creation of an apoe knockout rabbit. Transgenic Res 24: 227-235.
- Taylor JM, Fan J (1997) Transgenic rabbit models for the study of atherosclerosis. Front Biosci 2: d298-308.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi