Research Article, Int J Ophthalmic Pathol Vol: 7 Issue: 2
Ocular Surface Evaluation in Patients Treated with Pharmacological Treatment for Presbyopia
Sonia Facal1,2, Juliana Leiro1, Ariel Gualtieri3, Cristian Perez1, Giovanna Benozzi1,2 and Betina Orman2*
1Centro de Investigación Avanzada para la Presbicia, Buenos Aires, Argentina
2Pharmacology Unit, Facultad de Odontología. Universidad de Buenos Aires, Buenos Aires, Argentina
3Biophysics Unit, Facultad de Odontología. Universidad de Buenos Aires, Buenos Aires, Argentina
*Corresponding Author : Betina Orman
Pharmacology Unit, School of Dentistry, Universidad de Buenos Aires, M. T. de Alvear 2142 – 4to. Piso “B” 1122AAH Buenos Aires; Argentina
Tel: +54 11 52 87 62 35
E-mail: betina.orman@odontologia.uba.ar
Received: July 17, 2018 Accepted: August 02, 2018 Published: August 08, 2018
Citation: Facal S, Leiro J, Gualtieri A, Perez C, Benozzi G, et al. (2018) Ocular Surface Evaluation in Patients Treated with Pharmacological Treatment for Presbyopia. Int J Ophthalmic Pathol 7:2. doi: 10.4172/2324-8599.1000218
Abstract
Background: Presbyopia is the progressive deterioration of accommodation and can be treated with a non-invasive pharmacological treatment using eye drops.
Objective: To evaluate the ocular surface integrity and the tear production in patients under pharmacological treatment for presbyopia using eye drops with the combination of pilocarpine and diclofenac during one year. Methods: A total of thirty eyes were included in this study. The evaluation of the influence of the pharmacological treatment of the ocular surface, Schirmer test and tear film break-up time, ocular surface staining and conjunctival impression cytology were performed at baseline (T0) and after one year (T1).
Results: For Schirmer test measurements there were no significant differences in patients under the pharmacological treatment (T=-0.56; P=0.58; n=30). Values of tear film break-up time increased significantly on T1, (T=-3.53; P=0.0047; n=30) indicating amelioration of the lachrymal film. For all stains the values on T1 were diminished, pointing out the amelioration of the corneo conjunctival surface. For conjunctival impression cytology, on T1, 33.3% of the samples suffered no changes, 23.3% of the samples got worst and 43.3% ameliorated their condition.
Conclusion: Our results suggest that the pharmacological treatment for presbyopia produced a corneo conjunctival surface amelioration and no changes in tear production after one year of chronically use.
Keywords: Dry eye disease; Ocular surface; Presbyopia; Pilocarpine; NSAIDs; Pharmacological treatment; Conjunctival impression cytology
Abbreviations
DED: Dry Eye Disease; TFBUT: Tear Film Break-Up Time; ST: Schirmer Test; NSAIDs: Nonsteroidal Anti- Inflammatory Drugs.
Introduction
Presbyopia is the progressive deterioration of accommodation resulting in loss of the visual ability to focus on objects located at different distances. Accommodation in humans is performed by ciliary muscle and iris sphincter contractions, convergence and changes in the shape and position of the lens [1-3]. The muscarinic receptors on iris and ciliary muscle are stimulated by the cholinergic neurotransmitter, acetylcholine [4]. In the plethora of presbyopia treatment available, there is a patented pharmacological treatment for presbyopia [5] (US 8.524.758 B2- EP1.938.839 B1) that consists in daily eye drops with the combination of pilocarpine and diclofenac. Pilocarpine, as a cholinergic agonist, produces an spasmodic contraction of the ciliary muscle and miosis [6,7]. The combination of NSAIDs and parasympathetic agonists allows that the intensity of the contraction of the pupil [8] and the ciliary muscle decrease, changing the shape and position of the lens for good visual acuity at all distances restoring accommodation. Furthermore, the association with NSAIDs eliminates local inflammation that would appear as secondary’s to the chronic stimulation of pilocarpine [8].
Pilocarpine is used as an agonist of the muscarinic receptor for the treatment of the oral and ocular dryness [9] and by its binding, to the muscarinic receptors M3 and M1, stimulates the watery secretions of lachrymal and salivary glands. Pilocarpine increases tear and saliva production in patients with dry eye and dry mouth consequent to Sjögren Syndrome or gland irradiation for the treatment of cranial tumors.
The tear film is a key element for the ocular surface health and the quality of the optical image. It may be altered by environment that disrupts the homeostasis inducing inflammation and increase of evaporation. Dry eye disease and ocular surface damage are the primary concerns related to chronic use of eye drops as glaucoma patients.
Dry eye disease is a multifactorial condition involving changes in tear composition, volume and ocular surface integrity [10] as well. This condition is characterized by discomfort, visual disturbances and tear-film instability with potential damage to the ocular surface. DED symptoms include: dryness, discomfort, foreign body sensation, red eye, pain and blurring or vision fluctuation. Ocular surface staining score by the Oxford system [11], TFBUT, and Schirmer test score are used as objective signs for the diagnosis of DED. Patients should be diagnosed with DED when they have at least one symptom and one objective sign. If several objective signs are present at different levels, the severity level of the DED could be determined following ocular surface staining [12].
The aim of this study was to evaluate the ocular surface integrity and tear production in patients under pharmacological treatment for presbyopia using eye drops chronically with the combination of pilocarpine and diclofenac.
Materials and Methods
Study design
All of the patients of this prospective study were informed of the nature of the study, and the research adhered to the tenets of the Declaration of Helsinki. The ethics committees of the Argentinian Society of Clinical Investigation approved this study and written informed consent was obtained from all participants before enrollment.
Patients
A total of 253 patients with presbyopia diagnosed at the “Centro de Investigación Avanzada de la Presbicia” underwent a complete ophthalmologic examination, including slit lamp biomicroscopy and fundoscopy. A total of thirty eyes of fifteen patients complimented the inclusion criteria and were included in this study (Figure 1).
Inclusion criteria were: (a) male between 45 to 55 years old; (b) with no ocular and systemic pathology; (c) normal McMonnies questionnaire; (d) no ocular or systemic medication; (e) no smoking habit; (f) indication for pharmacological treatment for presbyopia Benozzi Method (US 8.524.758 B2- EP1.938.839 B1).
Exclusion criteria were: (a) active ocular infection; (b) ocular allergy; (c) history of refractive surgery or use of contact lens; (d) DED or conditions with clinical overlapping of the aforementioned diseases; (e) patients who abandoned pharmacological treatment for presbyopia.
All measurements on the presbyopia patients were performed previously to start and one year after the beginning of the pharmacological treatment for presbyopia, by the same calibrated investigators, under similar testing conditions, and at room temperature. Uncorrected distance visual acuity (UDVA) and uncorrected near visual acuity (UNVA) were measured before pharmacological treatment (PT=previous treatment), at the beginning of pharmacological treatment (T0) and one year after treatment (T1).
Evaluation consisted in a protocol encompassing: (1) McMonnies questionnaire as described below and according to the following sequence waiting 30 min between each test; (2) Schirmer test; (3) TFBUT; (4) corneal staining with fluorescein; and conjunctival staining with (5) lissamine green and (6) rose Bengal; and (7) conjunctival impression cytology.
McMonnies questionnaire: The McMonnies questionnaire is among the earliest and most widely used screening instruments for dry eye syndromes, and it is composed by questions focus on clinical risk factors for DED with a sensitivity 98% and specificity 97% [13]. The test is used to screen patients for the possibility of DED to discriminate marginal dry eye from normal and more severe dry eye. Values less than 10 are qualified like normal, values from 10 to 20 are marginal dry eye and values over 20 are considered as a pathological dry eye [14].
Schirmer test: The Schirmer test measures tear secretion; tear flow as a result of <10 mm indicating impaired secretion [15,16]. Schirmer test without anesthesia was performed and tears production was measured in both eyes simultaneously with Schirmer test strip for 5-min [17].
Tear film break-up time: The patient waited for 30 min, and TFBUT was measured 10-30 s after instillation of 50 μl of a 0.25% sodium fluorescein (Poen, Buenos Aires, Argentina) and determined manually using a stopwatch (in seconds) [17]. The TFBUT is an indirect measure of tear film stability indicated by a break-up time of >10 s [18]. Two measurements were taken in succession and averaged [19].
Corneal fluorescein staining: Corneal fluorescein staining was evaluated using cobalt blue illumination following the 15-point NEI/ Industry scale (grades of 0-3 for five regions of the ocular surface), after TFBUT measurements. The maximum staining score is 15 for the cornea with values above 3 was considered abnormal [17,20].
Lissamine green conjunctival staining: Lissamine green conjunctival staining was evaluated after instilling 50 μl of a 1% sodium lissamine green dye with previous anesthesia. Conjunctival staining assessment used the Oxford grading scheme [11]. For the conjunctiva, the maximum score is 18 for each eye with values above 3 being abnormal.
Rose bengal conjunctival staining: After waiting for another 30 min, the Rose bengal test was performed after instilling 50 μl of a 0.5% Rose bengal dye over the inferior bulbar conjunctiva with previous anesthesia. Conjunctival staining assessment used the Oxford grading scheme [11]. For the conjunctiva, the maximum score is 18 for each eye with values above 3 being abnormal.
Conjunctival impression cytology: Semicircular filters approximately 15 mm diameter in size (Polyvinylidene Fluoride – PVDF filter, 22-um pore; Millipore Corp., Bedford, MA, USA) were applied to the inferior tarsal and bulbar conjunctiva after instillation of one drop of topical anesthetic proparacaine (Poen, Buenos Aires, Argentina) in each eye, and the excess fluid was wiped away. The paper fragments were applied for approximately 10 s, and after gentle pressure with the blunt end of the forceps; the fragments were peeled off and immediately immersed in tubes containing absolute ethanol. After fixation, specimens were rehydrated in 70% ethyl alcohol, and then placed successively in periodic acid–Schiff reagent, sodium metabisulfite, Gill’s hematoxylin and Scott’s tap water. Slides were examined under a conventional light microscope, under a 400 X magnification. Morphometric analysis was carried out using a point-counting technique, were PAS positive areas were counted across 15 random microscopic high-power fields on a 100-point and 50 lines grid on a video system coupled to the microscope. The result for each subject was the average of the 15 fields [21]. The CIC was graded by the scale of Nelson, a four grades scale, based on the number of globet cells and the appearance of epithelial cells. Grade 0, is considerate as normal; Grade 1, minimal alteration; Grade 2, moderate alteration and Grade 3, severely altered [22]. The same investigator performed all the observations and was blinded to the origin of the samples [22].
Statistical analysis
Data were statically evaluated. To compare values between baseline and after one year of a pharmacological presbyopia treatment the paired test was performed; t paired or Wilcoxon for paired dates when not met the normality assumption. The normality assumption was tested by Shapiro-Wilk test. In all cases a significance level of 5% (α=0.05) was fixed.
Results
A total of 30 eyes of 15 patients were included in this study. All the patients had presbyopia and a McMonnies questionnaire score less than 10. All the subjects followed the pharmacological treatment prescribed with eyedrops consisting of pilocarpine and diclofenac. To evaluate the influence of the pharmacological treatment on the ocular surface, Schirmer test and TFBUT, ocular surface staining and conjunctival impression cytology were performed.
All patients had UDVA 20/20 before beginning the pharmacological treatment of presbyopia (PT). The UDVA was 20/20 after starting treatment (T0) and remained constant after 1 year of treatment (T1). All patients needed correction for near vision, UNVA values ranged between J2 and J5 (PT). Once they started the pharmacological treatment of presbyopia (T0), the mean UNVA changed to J1 and remained constant after 1 year of treatment (T1).
The evaluation of the ocular surface by the Schirmer test and TFBUT
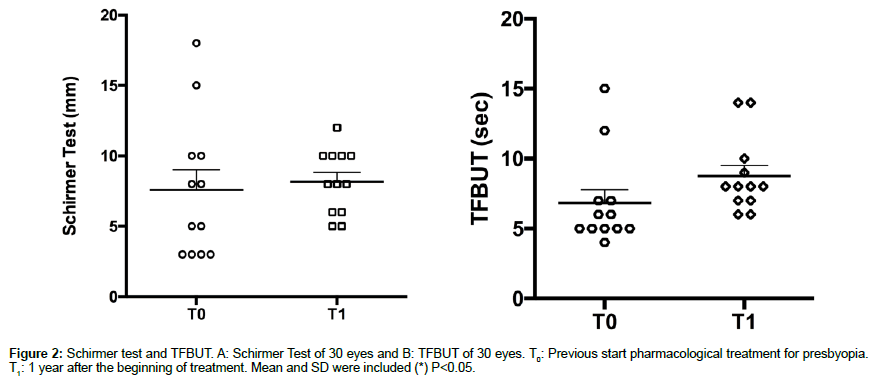
Schirmer test measurements in patients under the pharmacological treatment showed no significant differences between baseline (T0) and one year later (T1) (T=-0.56; P=0.58; n=30). Whereas, the resulting values of TFBUT increased significantly on T1, (T=-3.53; P=0.0047; n=30) indicating amelioration of the lachrymal film (Figure 2).
Corneal and conjunctival staining
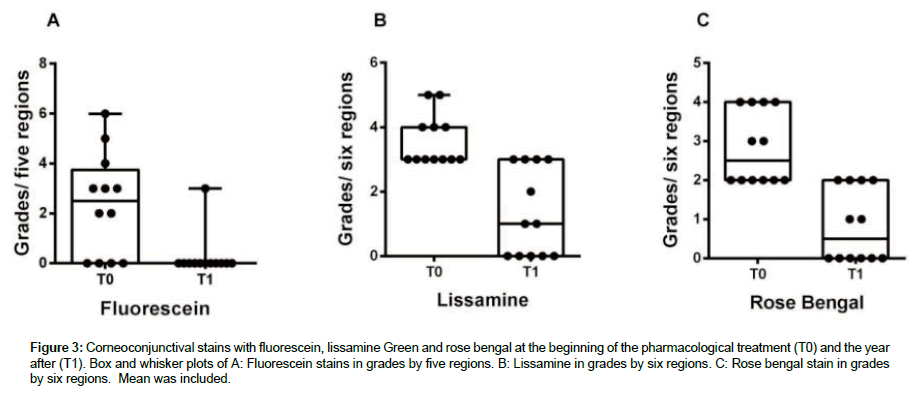
The box-and-whisker plots in (Figure 3) illustrate corneoconjunctival stains for T0 and T1. The average of fluorescein stain for T0 was 3.3 grades and 0.2 grades for T1, for lissamine stain for T0 were 4 grades and 0.9 grades for T1 and for rose bengal 2.2 grades for T0 and 1.0 grade for T1. The comparison of the values obtained at T0 y T1 for lissamine green (t paired) and for fluorescein and rose bengal (Wilcoxon for paired dates) showed significant differences for the three stains (P<0.05). These results pointed out the amelioration of the corneo conjuctival surface of the patients under pharmacological treatment for presbyopia during one year.
Figure 3: Corneoconjunctival stains with fluorescein, lissamine Green and rose bengal at the beginning of the pharmacological treatment (T0) and the year after (T1). Box and whisker plots of A: Fluorescein stains in grades by five regions. B: Lissamine in grades by six regions. C: Rose bengal stain in grades by six regions. Mean was included.
Conjunctival impression cytology
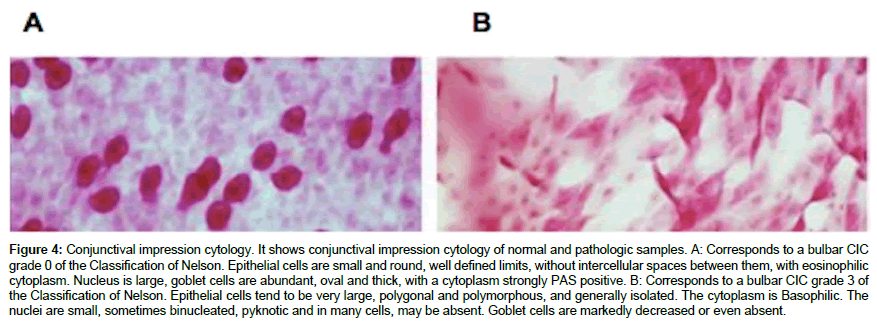
To evaluate if the chronic use of eye drops employed for the pharmacological treatment for presbyopia influence the apparition of metaplasia or changes on the eyelid or bulbar conjunctiva, conjunctival cytology was performed (Figure 4).
Figure 4: Conjunctival impression cytology. It shows conjunctival impression cytology of normal and pathologic samples. A: Corresponds to a bulbar CIC grade 0 of the Classification of Nelson. Epithelial cells are small and round, well defined limits, without intercellular spaces between them, with eosinophilic cytoplasm. Nucleus is large, goblet cells are abundant, oval and thick, with a cytoplasm strongly PAS positive. B: Corresponds to a bulbar CIC grade 3 of the Classification of Nelson. Epithelial cells tend to be very large, polygonal and polymorphous, and generally isolated. The cytoplasm is Basophilic. The nuclei are small, sometimes binucleated, pyknotic and in many cells, may be absent. Goblet cells are markedly decreased or even absent.
Table 1 shows the eyelid and bulbar CIC obtained from all the patients. For eyelid CIC all the samples were grade 0 except one sample at baseline (T0) and one year later (T1). For the bulbar CIC, at baseline, 50% of the samples were normal, and 50% of the samples were anomalous. After one year of presbyopia treatment, 33.3% of the samples, corresponding to normal samples, suffered no changes; 23.3% got worst and, 43.3% ameliorated their condition. However, there were not significant differences for eyelid CIC and bulbar CIC at T0 and T1 (Wilcoxon test for paired samples: P=0.75).
| Sample | Eyelid CIC | Bulbar CIC | ||
|---|---|---|---|---|
| T0 | T1 | T0 | T1 | |
| 1 | 0 | 0 | 0 | 0 |
| 2 | 1 | 1 | 0 | 0 |
| 3 | 0 | 0 | 0 | 1 |
| 4 | 0 | 0 | 0 | 2 |
| 5 | 0 | 0 | 1 | 2 |
| 6 | 0 | 0 | 2 | 1 |
| 7 | 0 | 0 | 0 | 0 |
| 8 | 0 | 0 | 1 | 0 |
| 9 | 0 | 0 | 3 | 0 |
| 10 | 0 | 0 | 2 | 0 |
| 11 | 0 | 0 | 1 | 0 |
| 12 | 0 | 0 | 0 | 0 |
| 13 | 1 | 1 | 0 | 0 |
| 14 | 0 | 0 | 1 | 0 |
| 15 | 0 | 0 | 3 | 1 |
| 16 | 0 | 0 | 2 | 0 |
| 17 | 0 | 0 | 0 | 1 |
| 18 | 0 | 0 | 0 | 0 |
| 19 | 1 | 1 | 0 | 0 |
| 20 | 0 | 0 | 0 | 1 |
| 21 | 0 | 0 | 0 | 2 |
| 22 | 0 | 0 | 1 | 2 |
| 23 | 0 | 0 | 2 | 1 |
| 24 | 0 | 0 | 0 | 0 |
| 25 | 0 | 0 | 1 | 0 |
| 26 | 0 | 0 | 3 | 0 |
| 27 | 0 | 0 | 2 | 0 |
| 28 | 0 | 0 | 1 | 0 |
| 29 | 0 | 0 | 0 | 0 |
| 30 | 0 | 0 | 0 | 0 |
Table 1: Eyelid and bulbar Conjunctival impression cytology. T0 correspond to baseline and T1 one year after the pharmacological treatment for presbyopia.
Discussion
The results of this study showed a statistically significant amelioration or no changes in the condition of the ocular surface integrity and the tear production in patients during a year under the pharmacological treatment for presbyopia. The pharmacological treatment consists of topical eye drops with a combination of a parasympathetic agonist and a non-steroidal anti-inflammatory, pilocarpine, and diclofenac, respectively. Our results showed that the patients under this treatment achieve J1 UNVA and it is maintained during 1 year of treatment.
Pilocarpine was proposed to increase tear production in patients with dry eye or gland irradiation for the treatment of cranial tumors. Lachrymal gland fluid production is largely under muscarinic receptor control, and in fact, M3 and M1 receptor subtypes were identified in the lachrymal gland tissue [23] and on goblet cells in the human conjunctiva [24]. The stimulation of these receptors showed increased watery secretions of lachrymal and salivary glands and to prevent acinar apoptosis [25]. Goblet cells can release their secretory granules as a reflex response mediated by the activation of either parasympathetic or sympathetic nerves that surround them. Goblet cells synthesize and secrete mucin and are essential for maintaining the health of the ocular surface [26,27].
NSAIDs are inhibitors of the cyclooxygenases, which mediate the breakdown of arachidonic acid to produce prostaglandins and other metabolic products. Prostaglandins are known to be mediators of inflammation. Although, superficial punctuate keratitis, corneal infiltrates and the higher incidence of persistent epithelial defects have been reported following the use of these anti-inflammatory agents [28,29], most of these patients had recently undergone cataract or refractive surgery [30]. Furthermore, it is felt that corneal melting requires a compromised ocular surface to occur and other comorbid conditions as trichiasis and glaucoma.
Schirmer test is one of the most commonly used measurements of tear production including reflex and basal tears. TFBUT is used to assess tear film stability. The patients after one year under pharmacological treatment for presbyopia showed no significant differences for Schirmer test; in contrast, TFBUT values increased significantly. The inclusion criteria for this study were healthy patients with normal McMonnies questionnaire. Nevertheless, according to the International Dry Eye Workshop, dry eye syndrome is a multifactorial disease. Dry eye etiology includes allergies and environmental factors as computer use, concentrated near work and fatigue, may result in inhibition of blinking and the presence of dry eye symptoms [31,32]. Thus, many dry eye states are likely to be intermittent and variable in severity, and may not be present or recalled at the time of the subject’s examination, or when participating in a study [33]. Furthermore, modern industrialized cities resulted in high levels of air pollution being the ocular surface vulnerable to it. Due to the contact of the preocular tear film, cornea and conjunctiva with the surrounding air, toxins have direct access to the ocular structures and can abnormally change them. A previous study suggested that the quality of environmental air could result in a premature break-up of the preocular tear film and corneal epithelial damage [34]. Studies have shown that chronic exposure to ambient air pollution is associated with a significantly decreased TFBUT [35] and significantly higher levels of abnormal Schirmer test [36]. All subjects of this study live in a polluted area and, this explains why our results showed that the patients had abnormally low values for Schirmer test and TFBUT, as normal citizens. The pharmacological treatment did not improve tear production quantify by Schirmer test. The volume of tears in the eye depends on the balance of the rate of production and elimination by drainage and evaporation. Mathers et al. [37] revealed that the rate of tear secretion decreased with aging. The prevalence of DED was increased with aging and was significantly higher in subjects older than 40 years old [38]. Samarkos and Moutsopoulos [39] showed that the treatment with pilocarpine augmented tear production and is depending on aging.
Staining with dyes allows the measurement of the extent of damage of the ocular surface. Typically, corneal epithelial cells are impermeable to the tear film as the lipid membranes at the ocular surface are effective barriers against polar and water-soluble substances. When the barrier is disrupted, the tear film can gain access to deeper tissues. Rose bengal stains healthy, dead and degenerate cells; lissamine green only dead and degenerate cells and; fluorescein none. Staining with fluorescein, Rose bengal and lissamine green showed significant differences in the number of sites, at baseline and one year later. The diminution of the number of sites calls attention to the amelioration of the corneoconjuctival cells condition. Dryness of the eye is often accompanied by an alteration in the morphology of epithelial cells of conjunctiva and reduction in conjunctival goblet cell density since the non-keratinized stratified conjunctival epithelium progressively loses goblet cells and differentiates into a non-secretory keratinized epithelium [40].
CIC showed no significant differences during the year of treatment. It’s interesting to note that the inclusion criteria were healthy patients with a normal McMonnies questionnaire, which is interpreted as a diagnosis of no DED. While, bulbar CIC of 50% of the patients revealed like no normal at baseline and after one year of presbyopia treatment, 43.3% of them improved, 23.3% deteriorated, and 33.3% had no changes in their condition.
The purpose of the present study was to establish if there was an association between the use of pharmacological treatment for presbyopia and modifications at the ocular surface. We evaluated the ocular surface by Schirmer test, TFBUT, stains and conjunctival cytology impression. Our results suggest that the pharmacological treatment for presbyopia produced a corneoconjunctival surface amelioration put forward by ocular surface staining as lissamine green, fluorescein and rose bengal and CIC or at least no changes in tear production as demonstrated by the consistent results of Schirmer test and the increased values of TFBUT. Patients under Benozzi Method pharmacological treatment for presbyopia ameliorate or do not modify their ocular surface. However, further and more complete studies are needed to confirm the effect of the pharmacological treatment for presbyopia on the ocular surface and tear production.
Acknowledgements
The authors thank Isabel Marquez for her professional collaboration.
Conflict of Interest Statement
Betina Orman, Sonia Facal, Juliana Leiro, Cristian Perez and Ariel Gualtieri declare no conflict of interest. Giovanna Benozzi is the owner of the patents of the pharmacological treatment eye drops mentioned in this paper.
Financial Support
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
References
- Atchison DA (1995) Accommodation and presbyopia. Ophthalmic Physiol Opt 15: 255-272.
- Glasser A (2006) Accommodation: Mechanism and measurement. Ophthalmol Clin North Am 19: 1-12.
- Benozzi G, Leiro J, Facal S, Perez C, Benozzi J, et al. (2013) Developmental changes in accommodation evidenced by an ultrabiomicroscopy procedure in patients of different ages. Med Hypothesis Discov Innov Ophthalmol 2: 8-13.
- Koeppl C, Findl O, Kriechbaum K, Drexler W (2005) Comparison of pilocarpine-induced and stimulus-driven accommodation in phakic eyes. Exp Eye Res 80: 795-800.
- Benozzi J, Benozzi G, Orman B (2012) Presbyopia: a new potential pharmacological treatment. Med Hypothesis Discov Innov Ophthalmol 1: 3-5.
- Pardue MT, Sivak JG (2000) Age-related changes in human ciliary muscle. Optom Vis Sci Off Publ Am Acad Optom 77: 204-210.
- Ostrin L, Glasser A (2005) Comparisons between pharmacologically and Edinger-Westphal-stimulated accommodation in rhesus monkeys. Invest Ophthalmol Vis Sci 46: 609-617.
- Schalnus R (2003) Topical nonsteroidal anti-inflammatory therapy in ophthalmology. Ophthalmologica 217: 89-98.
- Aragona P, Pietro R, Spinella R, Mobrici M (2006) Conjunctival epithelium improvement after systemic pilocarpine in patients with Sjogren’s syndrome. Br J Ophthalmol 90: 166-170.
- Foulks GN (2007) 2007 Report of the International Dry Eye Workshop (DEWS). Ocul Surf 5: 75-89.
- Bron AJ, Evans VE, Smith JA (2003) Grading of corneal and conjunctival staining in the context of other dry eye tests. Cornea 22: 640-650.
- Hyon JY, Kim H-M, Lee D, Chung ES, Song JS, et al. (2014) Korean guidelines for the diagnosis and management of dry eye: development and validation of clinical efficacy. Korean J Ophthalmol 28: 197-206
- Gothwal VK, Pesudovs K, Wright TA, McMonnies CW (2010) McMonnies questionnaire: enhancing screening for dry eye syndromes with Rasch analysis. Invest Ophthalmol Vis Sci 51: 1401-1407.
- McMonnies C, Ho A, Wakefield D (1998) Optimum dry eye classification using questionnaire responses. Adv Exp Med Biol 438: 835-838.
- Lemp MA (1995) Report of the National Eye Institute/Industry workshop on Clinical Trials in Dry Eyes. CLAO J 21: 221-232.
- Bijsterveld OP (1969) Diagnostic tests in the Sicca syndrome. Arch Ophthalmol 82: 10-14.
- Alves M, Reinach PS, Paula JS, Vellasco e Cruz AA, Bachette L, et al. (2014) Comparison of diagnostic tests in distinct well-defined conditions related to dry eye disease. PLoS One 9: e97921.
- Lemp MA, Hamill JR (1973) Factors affecting tear film breakup in normal eyes. Arch Ophthalmol 89: 103-105.
- Kumar P, Bhargava R, Kumar M, Ranjan S, Kumar M, et al. (2014) The correlation of routine tear function tests and conjunctival impression cytology in dry eye syndrome. Korean J Ophthalmol 28: 122-129.
- Dundas M, Walker A, Woods RL (2001) Clinical grading of corneal staining of non-contact lens wearers. Ophthalmic Physiol Opt 21: 30-35.
- Aguilar AJ, Marquez MI, Albera PA, Tredicce JL, Berra A (2014) Effects of systane(®) balance on noninvasive tear film break-up time in patients with lipid-deficient dry eye. Clin Ophthalmol 8: 2365-2372
- Nelson JD (1988) Impression cytology. Cornea 7: 71-81.
- Mauduit P, Jammes H, Rossignol B (1993) M3 muscarinic acetylcholine receptor coupling to PLC in rat exorbital lacrimal acinar cells. Am J Physiol 264: C1550-1560.
- Fox RI, Konttinen Y, Fisher A (2001) Use of muscarinic agonists in the treatment of Sjögren’s syndrome. Clin Immunol 101: 249-263.
- Diebold Y, Ríos JD, Hodges RR, Rawe I, Dartt DA (2001) Presence of nerves and their receptors in mouse and human conjunctival goblet cells. Invest Ophthalmol Vis Sci 42: 2270-2282.
- Argüeso P, Gipson IK (2001) Epithelial mucins of the ocular surface: structure, biosynthesis and function. Exp Eye Res 73: 281-289.
- Inatomi T, Spurr-Michaud S, Tisdale AS, Zhan Q, Feldman ST, et al. (1996) Expression of secretory mucin genes by human conjunctival epithelia. Invest Ophthalmol Vis Sci 37: 1684-1692.
- Radtke N, Meyers S, Kaufman HE (1978) Sterile corneal ulcers after cataract surgery in keratoconjunctivitis sicca. Arch Ophthalmol 96: 51-52.
- Flach AJ (2001) Corneal melts associated with topically applied nonsteroidal anti-inflammatory drugs. Trans Am Ophthalmol Soc 99: 205-10-2.
- Holly FJ, Lemp MA (1977) Tear physiology and dry eyes. Surv Ophthalmol 22: 69-87.
- Dainoff MJ, Happ A, Crane P (1981) Visual fatigue and occupational stress in VDT operators. Hum Factors 23: 421-437.
- Nakamori K, Odawara M, Nakajima T, Mizutani T, Tsubota K (1997) Blinking is controlled primarily by ocular surface conditions. Am J Ophthalmol 124: 24-30.
- Isreb MA, Greiner JV, Korb DR, Glonek T, Mody SS, et al. (2003) Correlation of lipid layer thickness measurements with fluorescein tear film break-up time and Schirmer’s test. Eye (Lond) 17: 79-83.
- Saxena R, Srivastava S, Trivedi D, Anand E, Joshi S, et al. (2003) Impact of environmental pollution on the eye. Acta Ophthalmol Scand 81: 491-494.
- Pan Z, Mølhave L, Kjaergaard SK (2000) Effects on eyes and nose in humans after experimental exposure to airborne office dust. Indoor Air 10: 237-245.
- Gupta SK, Gupta SC, Agarwal R, Sushma S, Agrawal SS, et al. (2007) A multicentric case-control study on the impact of air pollution on eyes in a metropolitan city of India. Indian J Occup Environ Med 11: 37-40.
- Mathers WD, Lane JA, Zimmerman MB (1996) Tear film changes associated with normal aging. Cornea 15: 229-234.
- Albietz JM (2000) Prevalence of dry eye subtypes in clinical optometry practice. Optom Vis Sci 77: 357-363.
- Samarkos M, Moutsopoulos HM (2005) Recent advances in the management of ocular complications of Sjögren's syndrome. Curr Allergy Asthma Rep 5: 327-332.
- Rolando M, Terragna F, Giordano G, Calabria G (1990) Conjunctival surface damage distribution in keratoconjunctivitis sicca. An impression cytology study. Ophthalmologica 200: 170-176.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi