Research Article, J Clin Immunol Res Vol: 1 Issue: 1
Phenotypic IgA Deficit (SIgAD) or Probably (PIgAD) and Related Diseases Associations during 4 years of Research in Latin-American Kids
Fernández C1*, Roxo-Junior P2, Marina SC2, Juan CAB3, Felipe MB4, Deysi LAF5 and Lorena B6
1Board member EAACI IDP WORK GROUP, Paraguay
2Ribeirão Preto Medical School, University of São Paulo, Brazil
3Allergy and Immunology Division Hospital Nacional Edgardo Rebagliati Martins, Lima Peru
4Department of Diagnostic Processes and Evaluation, Faculty of Health Sciences, Universidad Católica de Temuco, Chile
5Allergy and Immunology Division INMUNE Medical Center CDE-Paraguay
6Hospital Central IPS Neurology Division, Asunción - Paraguay
*Corresponding Author : Fernández Carmen
Board member EAACI IDP WORK GROUP, Paraguay
Tel: +595982634013
E-mail: yoajere@yahoo.com
Received: March 5, 2018 Accepted: June 13, 2018 Published: June 18, 2018
Citation: Fernández C, Roxo-Junior P, Marina SC, Juan CAB, Felipe MB, et al. (2018) Phenotypic IgA Deficit (SIgAD) or Probably (PIgAD) and Related Diseases Associations during 4 years of Research in Latin-American Kids. J Clin Immunol Res 1:1.
Abstract
Abstract Introduction: The early diagnosis of IgA Deficit confirmed (SIgAD) or probably (PIgAD) is very important, the correct diagnosis and clinical follow-up improves the quality of life of these patients. We know, within the years, they can development other pathologies, actually, the guides only follow up in patients with confirmed deficit, but we see many kids with normal levels of IgA with symptoms and we consider it very important to evaluate of both group. Objective: Describe the phenotype characteristic and associated pathologies of SIgAD and PIgAD during 4 years of research in Latin-American kids. Materials and methods: Descriptive observational design, of boys and girls from 8 months to 18 years old, diagnosed with low levels of IgA, registered from 1998 until June 2012, who had completed 4 years of follow up since diagnosis, clinical and laboratory registration. For the study were divide in 2 groups. The first group (A) patients older than 4 years and with IgA level less than or equal to 7 mg/dl (called group SIgAD), and the second one (B) patients with of varied ages and IgA levels higher than 7 mg/dl and lower than 50 mg/dl (called group PIgAD). Results: 43 patients filled the selection criteria and were register, from 6-referent center, average age 27, 6 months (2.3 years old), were included 18 in the GROUP A and 25 in the GROUP B. Both groups presented associate pathology, 10 patients in the GROUP A such as Vitíligo (2), Protein allergy to cow's milk (4) and allergy Rhinitis (4); on the other hand, 8 kids in the GROUP B, Allergic Rhinitis (6) and Asthma (2). No one patient of any group has presented oncology diseases and autoimmune diseases until the end of research. Conclusions: It is very important the follow up of both groups, because of the both group presented complications with the years. In this study, we can observe that there is more complication with allergic pathologies in the group B and they are not having food allergy. On the other hand, in the group A, the absence of Asthma, like the pathology associated, is strange, but we need more study like these, with a bigger population, to get conclusions more accurate and avoid statistics interpretation mistakes.
Keywords: Allergy; Asthma; Cow’s milk; Selective IgA déficit; VitÃligo
Introduction
In 1960 Thomas Tomasi discovered this antibody secreted (IgA), made up of two basic units linked by a secretory piece synthesized by epithelial cells in tears, milk, saliva and intestinal tracts mucous and digestive system. This secretory piece is a polypeptide responsible for the transport of IgA across the epithelium
They not fix complement and not act like opsonin; however, it is very important to avoid the entry of microorganisms and macromolecules into the organism [1].
When we talk about primary immunodeficiency, comes us to mind, more than 300 probably alterations of the immune system [2].
The Selective Deficiency of IgA (SIgAD) is consider the most frequently primary immunodeficiency [3] and defined as undetectable serum immunoglobulin A (IgA) with normal levels of immunoglobulin G (IgG) and Immunoglobulin M (IgM), in patients older than 4 years of age [4].
The provabily deficiency IgA (PIgAD) defined as low level of IgA in two standard deviations with normal levels of IgG and IgM. The actually guides only give follow up patients with confirmed deficit (SIgAD), but we see many kids with values below 50 mg/dl, with symptoms like SIGAD, and no one consider evaluate this group, even if they can have pathology association over the years [5,6].
In the review, made from Inamine, publicized in the Journal of Gastroenterology, June 2018, describe IgA functions on the gut microbiome and discuss the role of IgA levels for liver diseases especially alcoholic liver disease and non-alcoholic fatty liver disease/non-alcoholic steatohepatitis [7]. The some conditions we can see in the recently review about Celiac Allergy Disease, made for Oliveira et al. [8], this group revered low levels of IgA before development Celiac Allergy, but this levels are not low than 7 mg/dl, the levels around 10 mg/dl.
Also in the review about Autoimmunity and Primary Immunodeficiency [9-11] the levels was around 4 to 20 mg/dl. All this previously publications and more [12,13], give as the idea that maybe we need do the follow up in all the patients with low levels of IgA to defined if this populations really have pathologies associations with the years.
The early diagnosis of SIGAD or PIGAD is very important, even if more than 80% are asymptomatic [14,15], the lack of association with a specific gene and the variety phenotypic presentations, make necessary to investigate them, in many infectious and non-infectious diseases, mainly in the pediatric age [16-18].
Objective
Describe the phenotypic characteristics and pathological associations during 4 years of follow-up, in the children populations with SIgAD or PIgAD.
Materials
Population
All children with a diagnosis of Selective IgA Deficit (SIgAD) or Probably (PIgAD), under 18 years old, from the 6 participating centers, who had complete records of clinical and laboratory follow-up during the 4 years after the diagnosis, from June 1999 to June 2012.
SIGAD case definition
Serum IgA level of <7 mg/dl in individuals older than four years in the presence of normal levels of both immunoglobulin G (IgG) and immunoglobulin M (IgM) and exclusion of other causes of Hipogammaglobulinemia and T cell defect as well as normal IgG antibody response to all vaccinations [19].
PIGAD case definition
Serum IgA levels less than or equal to 50 mg/dl, in individuals less than 18 years, in the presence of normal levels of both immunoglobulin G (IgG) and immunoglobulin M (IgM) and exclusion of other causes of Hipogammaglobulinemia and T cell defect as well as normal IgG antibody response to all vaccinations [20-22]. For the patients follow-up, was used the protocol proposed by Domínguez et al. (Table 1) [23].
| Exámenes Complementarios | Periodicidad |
|---|---|
| Examen clínico en cada control | Every month |
| hemograma | Every month |
| vsg | Every month |
| Immunoglobulin g, a, m, e | Every 1-2 years old under 10 years old |
| subclases iGg | Every 1-2 years old uo to 10 years old |
| ac. antidifterico | In the diagnosis time |
| ac. antitetanico | In the diagnosis time |
| ac. antineumococcico | In the diagnosis time |
| CH50 | Every 2 years |
| aNTICUERPO ANTINUCLEARES | Every 2 years |
Table 1: Laboratory control of patients with sigad.
The diagnoses was confirm with three different laboratory result, a difference between one and another was 1 to 3 months.
The value elected for the record was the better of the first three, and for follow-up only one laboratory result.
For the screening of diabetes used: Fasting glycemic, determination of blood glucose at any time and oral glucose overload test (World Health Organization guidelines) and Glycosylated Hemoglobin.
For the screening to autoimmunity and oncology diseases, was decide to recommend every 2 years, Rheumatologist, Endocrinologist and Onco-hematological evaluation.
The pulmonary evaluation performed by pulmonologist, every 2 years and Spirometer every 1 year compared with NHANES III according to the international standards.
For the diagnosis of allergic pathology associations, was evaluating for allergist, with the corresponding studies according to the clinical criteria for the diagnosis confirmation.
Methods
For the data analysis, used the Statistical Package for the Social Sciences SPSS 14.0 for windows. The population was dividing in two groups.
GROUP A patients older than 4 years with IgA less than or equal to 7 mg/dl (IgA deficiency confirmed) SIgAD.
GROUP B patients of all ages with IgA greater than 7 and less than 50 mg/dl (probable IgA deficiency) PIgAD.
Results
43 Patients met the selection criteria, with complete records during 4 years of follow-up, the average age at diagnosis was 33 months (2.75 years), 24 male and 19 female.
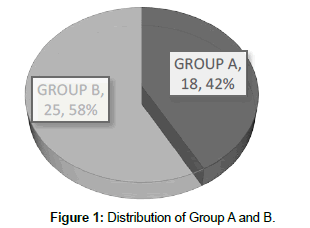
Of the 43 patients, 18 patients were included in the GRUPO A and 25 in the GROUP B (Figure 1).
The time from the onset of symptoms to the diagnosis was for GROUP A on average 48 months, and for GROUP B 18 months (12- 48 months).
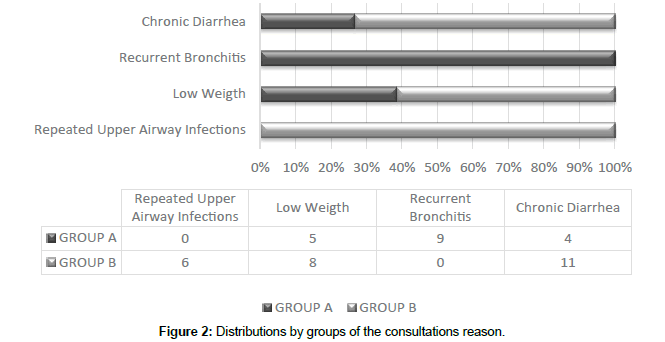
The reason of the consultation in GROUP A was Chronic Diarrhea 4 (22.2%), Recurrent Bronchitis 9 (50%), and Low Weight 5 (27.8%), in the GROUP B was Chronic Diarrhea 11 (44%), Low Weight 8 (32%) and Repeated Upper Airway Infections 6 (24%) (Figure 2).
In all of them IgG immunoglobulin values and subtypes 1, 2, 3 and 4 were measured. In the GROUP A no patient, until to the date of the manuscript elaboration, presented alterations in the controls, in GROUP B, 2 patients presented low levels of IgG2 in only one time of the 3 control, and had recurrent upper Airway Infections.
The other patients until the completion of this work did not present alteration in the values of the other immunoglobulin.
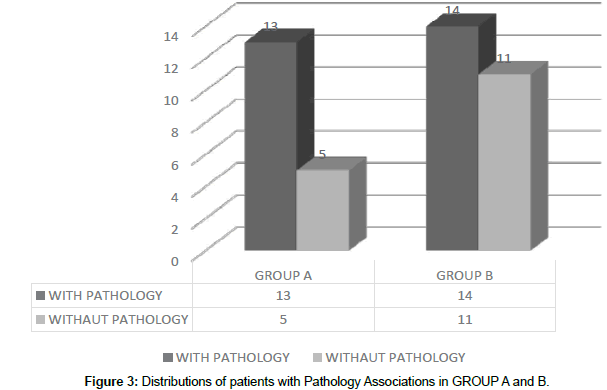
In relation to the associated pathologies during the 4 years of evolution, in Group A 13 of 18 patients (72.2%), in Group B 14 of 25 patients (56%) (Figure 3).
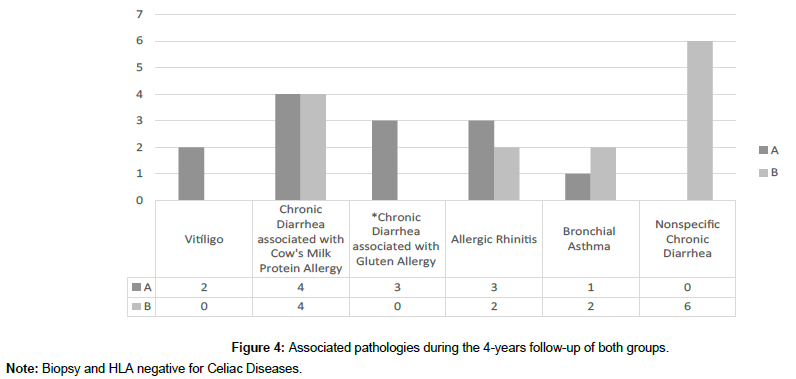
In the GROUP A, Vitíligo in 2 patients, chronic diarrhea associated with Cow's Milk Protein Allergy 4 patients, Chronic Diarrhea associated with non-Celiac Gluten Allergy 3 patients (HLA and Biopsy Negative), Allergic Rhinitis 3 patients, and 1 patient with Bronchial Asthma.
In the GROUP B, 4 patients presented Cow's Milk Protein Allergy, 2 patients with Allergic Rhinitis, 2 patients presented Bronchial Asthma and 6 patients nonspecific Chronic Diarrhea (Figure 4).
Discussion
In our study, SIgAD was more prevalence in male, coincident with de others publications, like the recent study was estimated, in over 12.000 Swedish twins by Viktorin et al. [22].
In the Alexander study, found a higher prevalence of selective IgA deficiency and subnormal IgA levels in men, demonstrated that the importance of inherited genes is 50% for the level of IgA and that there are important shared environmental influences [22].
In relation to time from start the symptoms and diagnosis, the median diagnosis delay, was around to 5-18 months, in relations to other studies [23,24]. We do the diagnosis very early, maybe because the centers works only with immunology diseases, this point make easier and early diagnosis.
In one of the six center, used PRIDE SCALE in all patients, when they suspect primary Immunodeficiency disease, it was valid in Mexicans people, with 98.3% sensibility and 98.3 specificity, 86.6%, positive valor predictive (VPP) 96.7%, negative valor predictive (NVP) 92.8% [25].
However, in reference to PIGAD the average that we found was 18 months (12-48 months), we not found refers papers to the average age for the diagnosis, probably because in these groups the follow-up studies are null.
Just Çipe et al. [20] in the Journal of Investig Allergol Clin Immunol, wrote a little about Partial IgA Deficiency, but they not refer average age for the diagnosis.
The reason of the consultations was Recurrent Bronchitis 50%, in GROUP A, the same percentage we find in others studies 40%-90%, but in the other hand, we not find sinopulmonar infections, like other groups, associated with IgG subclass deficiency, especially IgG2 [26-28].
In the GROUP B, Chronic Diarrhea 44% with Low Weight 32% was the more frequently reason of the consultations, the findings are very high, in comparison to other findings, and we assume it is due to the small number of individuals studied.
The recently report of the International Union of Immunological Sciences, approximately one third of the 354 primary immunodeficiency diseases have gastrointestinal component [29,16].
Also we can read in the Pallav et al. article, about PIGAD and Celiac Disease in the United States, in the universe of the 1000 patients with gastrointestinal diseases, they are not find relations with PIGAD an Celiac Diseases, but do reference to 4.4% Chronic Diarrhea in from to 5.9% in healthy patients, differences were not statistically significant [16,30].
We find associate pathology in Group A 72.2% and in Group B 56%, in the same way, even if the different was not significant, let see that both group have associate pathology with the years.
We know that previous studies talk about SIgAD and the predisposition to complications [31-33], but what about PIgAD group, only one study has mention infectious diseases in patients with low IgA levels performed by Hauge et al. [34] published in the Journal of Pathology, Microbiology and Immunology.
In this work, they find IgA deficiency in blood donors with mental diseases, enhanced inflammation and possibly an increased risk of infection [34].
In our study we not find statistical difference in referent to allergic pathologies associated, GROUP A 61.5% and GROUP B 57%, Kim et al. in the work about Relationship between serum IgA level and allergy/ asthma show that low IgA level is associated with atopic disease, but in relationship to Allergy/asthma this relationship is inversely [35].
We know that asthma have too many agents involved in it development [36,37] so, it is not logical to associate only with the presence or absence of IgA and/or blood levels [38].
In reference to Cow's Milk Protein Allergy, we find the same result in of both group, this finding coincides with recent studies that highlight the importance of the affinity of IgA for the bacteria of the Microbiota [39,40], like as increased binding of epitope-specific IgA to maternal cow's milk (CM) proteins [41], minimizing serum IgA levels as a determining factor.
In the recent studies mentioned the predisposition in the SIgAD to develop autoimmune disease [42-45], in our casuistry during 4 years of follow-up we not observed the appearance until the date of this publication, maybe with more big populations, this topic change.
About the associations Vitiligo and SIgAD or PIgAD, we find only in SIgAD group, it could be a casual finding, considering that studies show that there is a greater relationship between Vitiligo and increase or decrease of IgG subtypes, and not precisely related to IgA and its values [46-48].
Although the Hereditary pattern was not the objective in this study, it is important to highlight that genetic susceptibility is not very clear, and apparently, the most important risk factor is a family history of SIgAD [49].
Conclusion
The study concludes of both group A and B, have pathological associations with the years, although these findings interpreted in the context of a small population, we consider it important to follow both groups.
In the future, we need to increase the study population to obtain accurate results and conclusions about the incidence of complications in both groups.
References
- Bonilla FA, Bernstein, IL, Khan DA, Ballas ZK, Chinen J, et al. (2015) Practice parameter for the diagnosis and management of primary immunodeficiency. J Investig Allergol Clin Immunol 94: S1-S63.
- Bousfiha A, Jeddane L, Al-Herz W, Ailal L, Casanova JL, et al. (2015) The 2015 iuis phenotypic classification for primary immunodeficiencies. J Clin Immunol 132: 78-92.
- Quinn J (2015) Primary immunodeficiency is worldwide: updated overview from the jeffrey modell centers global network. J Immunol Res 22: M122-M133.
- Yazdani R, Latif A, Tabassomi F, Abolhassani H, Azizi G, et al. (2015) Clinical phenotype classification for selective immunoglobulin a deficiency. Expert Rev Clin Immunol 11: 1245-1254.
- Picard C, Al-Herz W, Bousfiha A, Casanova JL, Chatila T (2015) Primary immunodeficiency diseases: an update on the classification from the international union of immunological societies expert committee for primary immunodeficiency 2015. J Clin Immunol 35: 696-726.
- Zelazko M, Sampaio MC, de Luigi MC, de Olarte DG, Madrigal OP, et al. (1998) primary immunodeficiency diseases in latin america: first report from eight countries participating in the lagid. J Cin Immunol 18: 161-166.
- Inamine T, Schnabl B (2018) Immunoglobulin A and liver diseases. J Gastroenterol 53: 691-700.
- Oliveira GN, Mohan R, Fagbemi A (2018) review of celiac disease presentation in a pediatric tertiary centre. Arq Gastroenterol 55: 86-93.
- Azizi G, Bagheri Y, Tavakol M, Askarimoghaddam F, Poorrostami K, et al. (2018) The clinical and immunological features of patients with primary antibody deficiencies. Endocr Metab Immune Disord Drug Targets.
- Slade C, Bosco JJ, Giang TB, Kruse E, Stirling RG, et al. (2018) Delayed diagnosis and complications of predominantly antibody deficiencies in a cohort of australian adults. Front Immunol 9: 694.
- Gutierrez MJ, Sullivan KE, Fuleihan R; USIDNET Consortium, Bingham CO (2018) Phenotypic characterization of patients with rheumatologic manifestations of common variable immunodeficiency. Science direct 30654-30656.
- Ludvigsson JF, Neovius M, Ye W, Hammarström L (2015) IgA deficiency and risk of cancer: a population-based matched cohort study. J Clin Immunol 3: 182-188.
- Le Leiva M, Zelazco M, Oleastro M, Sampaio AC, Neto BTC, et al. (2007) Primary immunodeficiency diseases in latin america: the second report of the lagid registry. J Clin Immunol 27: 101-108.
- Sanabria D, Giménez V, Carpinelli MM, de Cuellar CM (2017) estallido respiratorio de neutrófilos por las técnicas del nitroazul tetrazolio (nbt) y dihidrorodamina (dhr) en niños con sospecha clínica de enfermedad granulomatosa crónica (EGC). Revista Paraguaya de Pediatria 44: 49-55.
- Yazdani R, Azizi G, Abolhassani H, Aghamohammadi A (2017) selective IgA deficiency: epidemiology, pathogenesis, clinical phenotype, diagnosis, prognosis and management. Scand J Immunol 85: 3-12.
- Pallav K, Xu H, Leffler D, Kabbani T, Kelly CP (2015) Immunoglobulin a deficiency in celiac disease in the united states. J Gastroenterol Hepatol 31: 133-138.
- Picard C, Al-Herz W, Bousfiha A, Casanova JL, Chatila T, et al. (2015) Primary Immunodeficiency Diseases: an Update on the Classification from the International Union of Immunological Societies Expert Committee for Primary Immunodeficiency 2015. J ClinImmunol 35: 696-726.
- Yazdani R, Latif A, Tabassomi F, Azizi G, Rezaei N, et al. (2015) Clinical phenotype classication for selective immunoglobulin a deiciency. Expert Rev Clin Immunol 11: 1245-1254.
- Domínguez O, Giner MT, Alsina L, Martín MA, Lozano J, et al. (2012) Fenotipos clínicos asociados a la deficiencia selectiva de IgA: revisión de 330 casosy propuesta de un protocolo de seguimiento. Anales de Pediatria 76: 261-267.
- Çipe FE, Dou F, Güloùlu D, Aytekin C, Polat M, et al. (2013) B-Cell Subsets in Patients with Transient Hipogammaglobulinemia of Infancy, Partial IgA Deficiency, and Selective IgM Deficiency. J Investig Allergol Clin Immunol 23: 94-100.
- Giorgio F, Principi M, Losurdo G, Piscitelli D, Iannone A, et al. (2015) Seronegative celiac disease and immunoglobulin deficiency: where to look in the submerged iceberg? Nutrients 7: 7486-7504.
- Viktorin A, Frankowiack M, Padyukov L, Chang Z, Melén E, et al. (2014) IgA measurements in over 12.000 swedish twins reveal sex differential heritability and regulatory locus near cd30l. Hum Mol Genet.
- Domínguez O, Giner MT, Alsina L, Martín MA, Lozano J, et al. (2012) Clinical phenotypes associated with selective IgA deficiency: a review of 330 cases and a proposed follow-up protocol. An Pediatr (Barc) 76: 261-267.
- Graziano V, Pecoraro A, Mormile I, Quaremba G, Genovese A, et al. (2017) Delay in diagnosis affects the clinical outcome in a cohort of CVID patients with marked reduction of IgA serum levels. Clin Immunol 180: 1-4.
- Dolores MMM, Arturo VL, Antonio OMJ, Carlos GA, Raimundo ROA, et al. (2012) Validación de la escala PRIDE (Primary Immunodeficiency Disease Evaluation) para determinar nivel alto de sospecha de inmunodeficiencia primaria en población mexicana. Rev Alergia Mex 59: 192-198.
- Maródi L (2018) Interdisciplinary immunodeficiency: a novel approach to understanding primary immunodeficiency disorders. Orv Hetil 159: 895-897.
- Reust CE (2013) Evaluation of primary immunodeficiency disease in children. Am Fam Physician 87: 773-778.
- Jorgensen GH, Gardulf A, Sigurdsson MI, Gudmundsson S, Ludviksson BR, et al. Clinical symptoms in adults with selective IgA deiciency: a case-control study. J Clin Immunolgy 33: 742-747.
- Hartono S, Ippoliti MR, Mastroianni M, Torres R, Rider NL (2018) Gastrointestinal Disorders Associated with Primary Immunodeficiency Diseases. Clin Rev Allergy Immunol 1-21.
- Picard C, Gaspar HB, Al-Herz W, Bousfiha A, Casanova JL, et al. (2018) International union of immunological societies: 2017 primary immunodeficiency diseases committee report on inborn errors of immunity. J Clin Immunol 38: 96-128.
- Azizi G, Kiaee F, Hedayat E, Yazdani R, Dolatshahi E (2018) Rheumatologic complications in a cohort of 227 patients with common variable immunodeficiency. Scand J Immunol 87: e12663.
- Mertin S, Thomson I (2013) What you need to know about IgA deiciency: a case study. J Am Assoc Nurse Pract 26: 268-272.
- Modell V, Knaus M, Modell F, Roifman C, Orange J, et al. (2014) Global overview of primary immunodeiciencies: a report from jeffrey modell centers worldwide focused on diagnosis, treatment and discovery. Immunol Res 60: 132-144.
- Hauge SC, Jensen CK, Nielsen LK, Pedersen OB, Sørensen E, et al. (2018) The association of IgA deiciency on infection rate,self-perceived health, and levels of c-reactive protein in healthy blood donors. Apmis 126: 248-256.
- Kim WJ, Choi IS, Kim CS, Lee JH, Kang HW (2017) Relationship between serun IgA level and allergy/asthma. Kjim 2: 137-145.
- Urm SH, Yun HD, Fenta YA, Yoo KH, Abraham RS, et al. (2013) Asthma and risk of selective IgA deficiency or common variable immunodeficiency: a population-based case-control study. Mayo Clin Proc 88: 813-821.
- Reisi M, Azizi G, Kiaee F, Masiha F, Shirzadi R, et al. (2017) Evaluation of pulmonary complications in patients with primary immunodeficiency disorders. Eur Ann Allergy Clin Immunol 49: 122-128.
- Orange JS1, Akhter J, Seeborg FO, Boyle M, Scalchunes C, et al. (2016) Pulmonologist perspectives regarding diagnosis and management of primary immunodeficiency diseases. Allergy Asthma Proc 37: 162-168.
- Dzidic M, Abrahamsson TR, Artacho A, Björkstén B, Collado MC (2016) Aberrant IgA responses to the gut microbiota during infancy precede asthma and allergy development. J Allergy Clin Immunol 139: 1017-1025.
- Kansu A, Yüce A, Dalgıç B, Şekerel BE, Çullu-Çokuğraş F, et al. (2016) Consensus statement on diagnosis, treatment and follow-up of cow's milk protein allergy among infants and children in Turkey. Turk J Pediatr 58: 1-11.
- Seppo AE, Savilahti EM, Berin M, Sampson HA, Järvinen KM (2017) Breast milk IgA to foods has different epitope specificity than serum IgA-Evidence for entero-mammary link for food-specific IgA?.Clin Exp Allergy 47: 1275-1284.
- Guffroy A, Gies V, Martin M, Korganow AS (2017) Primary immunodeficiency and autoimmunity. Rev Med Interne 38: 383-392.
- Schmidt RE, Grimbacher B, Witte T (2017) Autoimmunity and primary immunodeficiency: two sides of the same coin? Nat Rev Rheumatol 14: 7-18.
- Azizi G, Tavakol M, Rafiemanesh H, Kiaee F, Yazdani R, et al. (2017) Autoimmunity in a cohort of 471 patients with primary antibody deficiencies Expert Rev Clin Immunol 13: 1099-1106.
- Abolhassani H, Gharib B, Shahinpour S, Masoom SN, Havaei A, et al. (2015) Autoimmunity in patients with selective IgA deficiency. J Investig Allergol Clin Immunol 25: 112-119.
- Li Y, Yang M, Zhang R, Liu W, Zhang K, et al. (2016) Evaluation of serum immunoglobulins concentrations and distributions in vitiligo patients. Immunol Res 64: 1150-1156.
- Gualdi G, Lougaris V, Baronio M, Vitali M, Tampella G, et al. (2015) Burden of skin disease in selective IgA deficiency and common variable immunodeficiency. J Investig Allergol Clin Immunol 25: 369-371.
- Mohammadinejad P, Pourhamdi S, Abolhassani H, Mirminachi B, Havaei A, et al. (2015) Primary antibody deficiency in a tertiary referral hospital: a 30-year experiment. J Investig Allergol Clin Immunol 25: 416-425.
- Toker O, Aggmon-Levin N, Somech R (2016) How to approach a patient with suspected immunodeficiency. Harefuah 155: 170-176.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi