Research Article, J Clin Exp Oncol Vol: 7 Issue: 4
Post-Prostatectomy Radiotherapy in a Single Tertiary Institution: Outcomes Relating to Pre-Radiotherapy Prostate Specific Antigen
Janice Yeh1*, Ivan Iankov2, Myo Min3,4,5, Mohamad Rukasri Abdul Rahim4,6 and Daniel Roos2,7
1Alfred Health Radiation Oncology, The Alfred, Melbourne, Australia
2Department of Radiation Oncology, Royal Adelaide Hospital, Adelaide, Australia
3Department of Radiation Oncology, Sunshine Coast University Hospital, Birtinya, Queensland, Australia
4Faculty of Medicine, University of Queensland, Queensland, Australia
5School of Medicine, Griffith University, Queensland, Australia
6Genesis Cancer Care Australia, South West Campus, Bunbury, Australia
7School of Medicine, University of Adelaide, Adelaide, Australia
*Corresponding Author : Janice Yeh
Alfred Health Radiation Oncology, The Alfred, Melbourne, VIC 3004, Australia
Tel: +613-9076-2337
Fax: +613-9076-2916
E-mail: jsyyeh@gmail.com
Received: May 11, 2018 Accepted: May 30, 2018 Published: June 07, 2018
Citation: Yeh J, Iankov I, Min M, Abdul Rahim MR, Roos D (2018) Post-Prostatectomy Radiotherapy in a Single Tertiary Institution: Outcomes Relating to Pre- Radiotherapy Prostate Specific Antigen. J Clin Exp Oncol 7:4. doi: 10.4172/2324-9110.1000220
Abstract
Objective: Timing of post-radical prostatectomy (RP) radiotherapy (RT) in patients with high risk prostate cancer continues to be debated. This is a retrospective review aiming to evaluate the influence of pre-RT prostate specific antigen (PSA) values on postprostatectomy RT outcomes in one Australian center.
Method: Eligible patients were treated at the Royal Adelaide Hospital between January 2004 and December 2013, excluding those with nodal or distant metastatic disease pre-RT, or those who received neoadjuvant androgen deprivation therapy pre-RT. The primary endpoint of biochemical failure-free survival (bFFS) was defined as time from RP to date of biochemical failure (bF). Covariates of Gleason score, post-RP PSA, and pre-RT PSA were further analysed in relation to bFFS.
Results: 103 of 122 patients underwent final analysis (8 were excluded for the above reasons; 11 had missing data). Median follow-up from RP was 60 months. Kaplan-Meier (KM) estimates of 1, 2, 3, 4 and 5-year survival probabilities were 93.5%, 83.4%, 82.4%, 76.6% and 71% respectively. There was no statistically significant correlation between bFFS and pathological T-stage (p=0.1), surgical margin involvement (p=0.7), or RT total dose (p=0.8). Analysis based on KM survival distributions and log-rank tests suggest that pathological Gleason score may have some influence on bFFS (p=0.04). Doubling the pre-RT PSA whilst holding all other factors and covariates constant, increases the hazard of bF at a particular time-point by approximately 19% on average.
Conclusion: This single-institution retrospective study provides reasonable evidence for influence of pre-RT PSA on post-RP RT outcomes, arguing for earlier referral for RT.
Keywords: Prostatectomy; Prostate-specific antigen; Prostatic neoplasms; Radiotherapy
Introduction
In Australia, prostate cancer is the second most common cause of cancer deaths in men, with 19,233 new cases diagnosed in 2013 [1]. Of those who undergo radical prostatectomy (RP) to treat their prostate cancer, up to 50% will have histopathology showing at least one adverse feature [2]. These include surgical margin involvement, extracapsular extension, or seminal vesicle involvement. These patients have an approximately 40% risk of developing biochemical recurrence in the future, with median time to recurrence of 2.4 years [3].
There exists extensive literature to support consideration of offering patients radical radiotherapy (RT) in the post-RP setting [4], with the goal of lengthening biochemical failure free survival (bFFS). Radiotherapy in this setting can be initiated in multiple different clinical scenarios. These include shortly post-RP in patients who have adverse pathological features but with undetectable serum Prostate Specific Antigen (PSA), so-called “adjuvant RT” (ART); shortly post- RP in patients who have adverse pathological features, and inadequate biochemical response; and delayed post-RP in patients who later develop biochemical failure (“salvage RT”) (SRT).
In terms of predictive factors of disease relapse in patients who have received RT in either of these settings, recurring factors in the literature include pathological Gleason Score, pathological T stage, involvement of surgical margin, PSA velocity prior to RT, PSA prior to RP, and most prominently, PSA prior to RT [5-14].
As a large public tertiary referral centre, we were interested to review the outcome of our patients who have undergone RT after RP, focusing particularly on their pre-RT PSA value.
Methods
Relevant institution Human Research Ethics Committee approval was obtained to collect and analyse data of patients referred to and treated at our centre with ART or SRT, between January 2004 and December 2013.
Patient data was retrieved from ARIA Oncology-Information System (Varian Medical Systems), medical records (both paper and digital), OACIS Clinical Information System (TELUS Health), and local private pathology laboratories. Follow-up details were collected up to at least the censor date of 31st of March 2016. Data were analysed using Microsoft Access and R statistical software for Windows. Exclusion criteria included patients with nodal or distant metastatic disease prior to RT, and patients who received neoadjuvant androgen deprivation therapy (ADT) prior to RT. The definition of biochemical failure used in this study was the same as that of the RAVES study protocol page 39 (“first occasion following radiotherapy that the serum PSA is ≥ 0.40 ng/mL and rising (from the previous value). The date of biochemical failure will be the date of the first PSA level ≥ 0.40 ng/mL”) [15]. Patients in this review were not enrolled in RAVES.
The primary endpoint of bFFS was defined as time from RP surgery to date of biochemical failure.
Note that we did not attempt to analyse outcomes by ART vs SRT referral intent. Such retrospective analyses are inherently flawed due to intrinsic differences between the two groups and the fact that an indeterminate number of the adjuvant group will never relapse.
Radiotherapy
Post-RP RT was delivered by 3D-conformal technique or intensity-modulated RT technique, to a dose range of 60-66 Gy, delivered in 1.8 to 2 Gy fractions.
Follow-up
Patients were typically seen initially 4 to 6 weeks after completing RT, and then at 3 to 6 month intervals for the first two years after treatment. Afterward, patients were followed at least once annually. Post-RT serum PSAs were requested by the treating Radiation Oncologist, Urologist, and/or General Practitioner. Patients with a rising serum PSA level had further evaluation for recurrent disease, including history, exam, and diagnostic imaging.
Statistical analysis
The primary endpoint of bFFS was estimated utilizing the Kaplan-Meier (KM) method. Log-rank test was utilized to find suitable candidates for regressors. Small sample size ensured only limited statistical power of the performed tests for effect of factors on biochemical relapse. Cox regression was employed to investigate the influence of the analysed covariates and factors on bFFS.
As the primary covariate of interest (pre-RT PSA) has a positively skewed distribution, it was transformed by using logarithm with base 2 function, which resulted in a relatively symmetric distribution with sufficient number of cases in every sub-region of interest. This was utilized in the Cox regression analysis. The increase of log2 (pre-RT PSA) by one unit is equivalent to doubling absolute pre-RT PSA. Undetectable PSAs were assumed to be equal to 0.01.
After careful examination of results from the exploratory data analysis (EDA) and initial Cox proportional hazard modeling, the Penalized Cox proportional hazard regression was chosen as the most appropriate strategy for quantifying the effect of pre-RT PSA on bFFS. The coefficients of log2 (pre-RT PSA) and post-operative PSA were penalized in the final model by using Ridge methodology to prevent collinearity.
Given that there is potential for patients who received RT shortly post-RP with undetectable PSA to never develop biochemical failure even without RT, a selection bias would be introduced if all patients were included in our analysis. We therefore employed methodology to eliminate selection bias by analysing “samples” of patients. Each “sample” contains all patients who had PSA failure during their respective follow-up period and a fraction of the patients who did not have PSA failure during their respective follow-up period. We utilized data from historical Randomized Controlled Trials (RCT) on survival probability of patients who do not receive RT in this setting to determine if a patient is included or excluded in a sample.
Results
Between January 2004 and December 2013, 122 patients received post-RP RT at the Royal Adelaide Hospital. Eight of these patients were excluded (3 patients had N1 disease, and 5 patients had received neoadjuvant ADT). Of the remaining 114 eligible patients, 11 had missing critical data, resulting in 103 patients undergoing analysis.
Median follow-up period was 60 months. This is considered sufficient to minimize bias introduced by patients lost to followup.
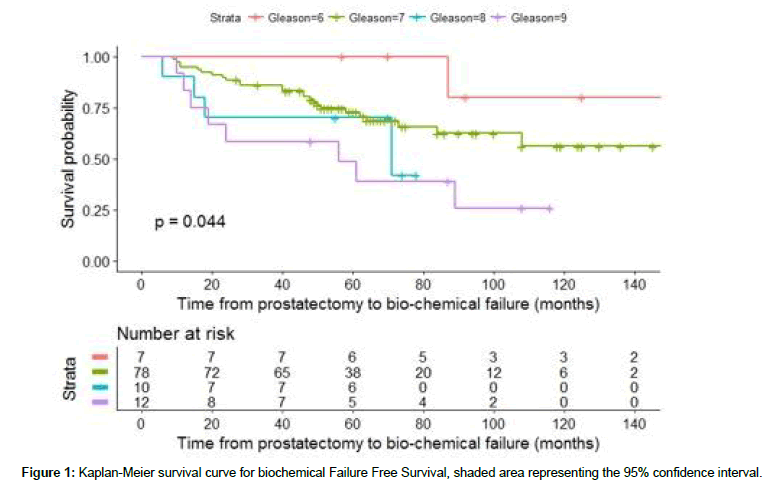
Table 1 shows descriptive statistics for the two groups of patients: eligible patients and patients considered in our analysis. Pathological T3 (pT3) and Gleason 7 disease dominated, whereas surgical margin status, as well as the two subgroups of RT doses were reasonably well balanced. The descriptive statistics are similar for the two groups; hence our results are not expected to be affected significantly by selection bias.
| Variable | Statistics name/Factor level/Category name | Statistic value/Frequency | |
|---|---|---|---|
| Eligible patients (n=114) | Included in the analysis (n=103) | ||
| Age at surgery (years) | Median | 62.5 | 62 |
| Range | (47-75) | (47-75) | |
| Pathological T stage | T2 | 25 | 21 |
| T3a | 58 | 53 | |
| T3b | 30 | 28 | |
| Not available | 1 | 1 | |
| Gleason score (prostatectomy) | 5 | 1 | 1 |
| 6 | 9 | 5 | |
| 7 | 82 | 75 | |
| - 7 (3+4) | 39 | 34 | |
| - 7 (4+3) | 43 | 41 | |
| 8 | 10 | 10 | |
| 9 | 12 | 12 | |
| Surgical margins | Positive | 63 | 58 |
| Negative | 50 | 44 | |
| Not available | 1 | 1 | |
| Post-op PSA (ug/L) | Median | 0.045 | 0.04 |
| Range | (Undetectable-0.77) | (Undetectable-0.77) | |
| Pre-radiotherapy PSA (ug/L) | Median | 0.16 | 0.15 |
| Range | (Undetectable-3.0) | (Undetectable-3.0) | |
| Total dose (Gy) | 60-64 | 67 | 61 |
| 64.8-66 | 47 | 42 | |
| Time from surgery to start of radiotherapy (months) | Median | 14.45 | 13.6 |
| Follow up - months from surgery to: | |||
| - PSA failure | Median | 40 | 40 |
| - Censoring or lost to follow-up | Median | 70 | 70 |
| - Overall | Median | 60 | 60 |
Table 1: Clinical and pathological patient data.
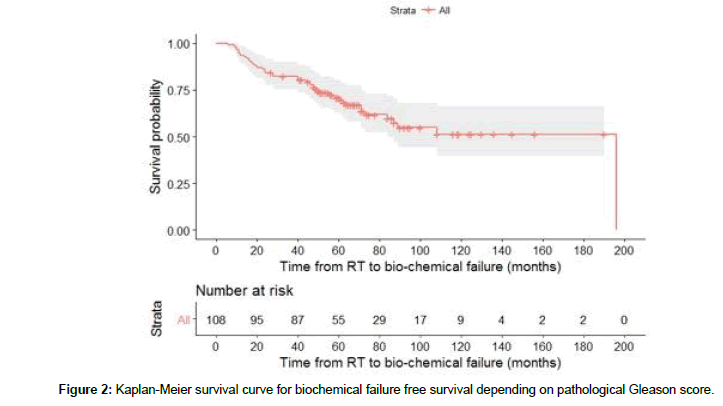
Figure 1 displays KM overall biochemical failure free survival (bFFS) probabilities based on measuring time to failure as duration from prostatectomy to PSA failure or censoring. The estimates of 1, 2, 3, 4 and 5-year survival probabilities were 93.5%, 83.4%, 82.4%, 76.6% and 71% respectively. The standard errors of the obtained survival probabilities vary from 2.4% to 4.5%, providing opportunities for determining practical confidence intervals.
Analysis based on KM survival distributions and log-rank tests suggest that pathological Gleason score may have some influence on bFFS. The test for Gleason score was significant (p=0.04, Figure 2), however due to the small sample size the tests for several known risk factors returned statistically insignificant p-values at 5% significant level, namely pT-stage (p=0.1), surgical margin involvement (p=0.7) and RT total dose (p=0.8). Cox regression analysis was utilized to obtain the hazard ratio (HR) associated with one unit increase of log2 (pre-RT PSA). The result was an average hazard ratio (HR) of 1.19 (95% confidence interval of 0.98-1.45). In other words, doubling the pre-RT PSA whilst holding all other factors and covariates constant increases the hazard of biochemical failure at a particular time-point by approximately 19% on an average.
Discussion
This retrospective review evaluating the influence of pre-RT PSA values on post-RP RT outcomes in one Australian centre, within its limitations, further confirms bFFS advantages with initiation of RT at a lower PSA value.
Thompson et al. in their systematic review [16] recommended that radiotherapy should be administered at the earliest sign of PSA recurrence as lower levels of pre-RT PSA have been shown in multiple studies to be associated with improved bFFS.
Gandaglia et al. [17] published an excellent literature review on adjuvant and salvage radiotherapy after RP, which included a table on contemporary retrospective studies evaluating outcomes and predictors of response in prostate cancer patients undergoing salvage radiotherapy. Pre-RT PSA levels feature on almost all studies. The overall median Pre-RT PSA level was 0.2-0.8 ng/ml. Other published articles [8,9,11] reviewing indications and optimal timing of initiating post-RT RT have also identified the impact of pre-RT PSA value on progression free probability, as a continuous spectrum, with inversely proportional relationship.
A recent tumour control probability model predicting five-year risk of biochemical relapse in hormone-naïve patients with greater than pT2, pN0 prostate cancer [18] also found that the detrimental effect of higher PSA levels can never be fully compensated by increasing the radiation dose.
The rate at which serum PSA is rising prior to RT is also a factor that should be taken into account. In ASCO’s endorsement of the 2013 AUA/ASTRO guideline on adjuvant and salvage radiotherapy after prostatectomy [19], the authors acknowledged the many technical issues relating to the measurement of the rate of PSA rise. They also recommended still offering SRT to men with short PSA doubling time (PSADT), as they are at highest risk of clinically meaningful progression and stand the most to gain from SRT, even though Stephenson et al. [5] reported a poorer biochemical response to SRT in those men with short PSADT. In other words, although knowing the patients’ PSADT will unlikely alter recommendations to proceed with SRT; however it may be considered as a predictive factor relating to outcome of treatment.
Despite having congruent evidence to support initiation of post-RP RT at “low” PSA levels, the challenge of identifying which patients and at what post-RP PSA level to initiate RT still remains. The Australian eviQ guidelines on SRT [20] included “persistently elevated PSA >6 weeks post radical prostatectomy” and “detectable and rising PSA” as indications for treatment. However, it did not provide actual values of PSA levels, further emphasizing the clinical conundrum.
The role of ADT in conjunction with post-RP RT is also evolving. The GETUG-AFU 16 randomized, multicenter, open-label phase 3 trial supports the use of concurrent short-term androgen suppression [21], and the Shipley et al. randomized, placebo controlled phase 3 trial likewise demonstrates benefit from two years of antiandrogen therapy [22] with salvage RT.
Furthermore, the emerging use of 68Ga-PSMA-11 PET/CT scans in the detection of recurrent prostate cancer must also be taken into account, as clinicians are beginning to utilize them to guide initiation of RT. One significant advantage would be avoiding offering locoregional treatment to patients who have recurred distantly. A large retrospective analysis [23] demonstrated strong correlation of serum PSA level and positive scan, however, unfortunately, there is also a small risk of false negatives (some patients with PSA >10 ng/ml did not have a positive scan). Future studies to evaluate 68Ga- PSMA-11 PET/CT imaging findings in the setting of post-RP RT, and its impact on bFFS, metastasis-free survival, disease-specific survival etc. are also warranted.
Limitations to this study must be considered when interpreting its results. This is a single centre retrospective study, which lacks additional data regarding PSA velocity, treatment toxicity, and clinical progression. Due to the dominance of pT3 and Gleason 7 disease, our findings are most relevant to those subsets of patients. Similarly, results should be considered most applicable to cases where the PSA is within the interval 0.1-0.3, which is the inter-quartile range of the pre-RT PSA values in our analysis. This conservative approach ensures that our recommended range is not influenced by outliers and observations not following the underlying data pattern.
Conclusion
This single institution retrospective study, within its limitations, provides reasonable evidence for influence of pre-RT PSA on post-RP RT bFFS outcomes, in particular in patients with pT3 and Gleason 7 prostate cancer. This argues for consideration of earlier referral for RT.
Conflict of Interest
No conflict of interest to disclose.
Acknowledgements
We thank Radiation Oncologists at the Royal Adelaide Hospital for support and advice.
References
- Prostate cancer in Australia (2017) Australian Government. Cancer Australia. Commonwealth of Australia; Prostate cancer statistics.
- Karakiewicz PI, Eastham JA, Graefen M, Cagiannos I, Stricker PD, et al. (2005) Prognostic impact of positive surgical margins in surgically treated prostate cancer: multi-institutional assessment of 5831 patients. Urology 66: 1245-1250.
- Swanson GP, Riggs M, Hermans M (2007) Pathologic findings at radical prostatectomy: risk factors for failure and death. Urol Oncol: Seminars and Original Investigations 25: 110-114.
- Shaikh MP, Alite F, Wu MJ, Solank AA, Harkenrider MM (2017) Adjuvant radiotherapy versus wait-and-see strategy for pathologic T3 or margin-positive prostate cancer: A meta-analysis. Am J Clin Oncol 33: 19-19.
- Stephenson AJ, Shariat SF, Zelefsky MJ, Kattan MW, Butler EB, et al. (2004) Salvage radiotherapy for recurrent prostate cancer after radical prostatectomy. JAMA 291: 1325–1332.
- Tendulkar RD, Agrawal S, Gao T, Efstathiou JA, Pisansky TM, et al. (2016) Contemporary update of a multi-institutional predictive nomogram for salvage radiotherapy after radical prostatectomy. J Clin Oncol 34: 3648-3454.
- Mir MC, Li J, Klink JC, Kattan MW, Klein EA, et al. (2014) Optimal definition of biochemical recurrence after radical prostatectomy depends on pathologic risk factors: identifying candidates for early salvage therapy. Eur Urol 66: 204-210.
- Briganti A, Joniau S, Gandaglia G, Cozzarini C, Sun M, et al. (2013) Patterns and predictors of early biochemical recurrence after radical prostatectomy and adjuvant radiation therapy in men with pT3N0 prostate cancer: Implications for multimodal therapies. Int J Radiat Oncol Biol Phys 87: 960-967.
- King CR (2012) The timing of salvage radiotherapy after radical prostatectomy: A systematic review. Int J Radiat Oncol Biol Phys 84: 204-211.
- Neuhof D, Hentschel T, Bischof M, Sroka-Perez G, Hohenfellner M, et al. (2007) Long-term results and predictive factors of three-dimensional conformal salvage radiotherapy for biochemical relapse after prostatectomy. Int J Radiat Oncol Biol Phys 67: 1411-1417.
- Hagan M, Zlotecki R, Medina C, Tercilla O, Rivera I, et al. (2004) Comparison of adjuvant versus salvage radiotherapy policies for postprostatectomy radiotherapy. Int J Radiat Oncol Biol Phys 59: 329-340.
- Pisansky TM, Kozelsky TF, Myers RP, Hillman DW, Blute ML, et al. (2000) Radiotherapy for isolated serum prostate specific antigen elevation after prostatectomy for prostate cancer. J Urol 163: 845-850.
- Grossfeld GD, Chang JJ, Broering JM, Miller DP, Yu J, et al. (2000) Impact of positive surgical margins on prostate cancer recurrence and the use of secondary cancer treatment: Data from the CaPSURE database. J Urol 163: 1171-1177.
- Lowe BA, Lieberman SF (1997) Disease recurrence and progression in untreated pathologic stage T3 prostate cancer: Selecting the patient for adjuvant therapy. J Urol 158: 1452-1456.
- Trans Tasman Radiation Oncology Group Ltd-TROG (2011) RAVES Radiotherapy Adjuvant Versus Early Salvage Study Protocol.
- Thompson IM, Valicenti RK, Albertsen P, Davis BJ, Goldenberg SL, et al. (2013) Adjuvant and salvage radiotherapy after prostatectomy: AUA/ASTRO guideline. J Urol 190: 441-449.
- Gandaglia G, Briganti A, Clarke N, Karnes RJ, Graefen M, et al. (2017) Adjuvant and salvage radiotherapy after radical prostatectomy in prostate cancer patients. Eur Urol 72: 689-709.
- Fiorino C, Broggi S, Fossati N, Cozzarini C, Goldner G, et al. (2016) Predicting the 5-year risk of biochemical relapse after postprostatectomy radiation therapy in >pT2, pN0 patients with a comprehensive tumour control probability model. Int J Radiat Oncol Biol Phys 96: 333-340.
- Freedland SJ, Rumble RB, Finelli A, Chen RC, Slovin S, et al. (2014) Adjuvant and salvage radiotherapy after prostatectomy: American Society of Clinical Oncology Clinical Practice Guideline Endorsement. J Clin Oncol 32: 3892-3898.
- EviQ Cancer Treatment Online Protocols (2017) Cancer institute NSW, Prostate Adenocarcinoma Salvage EBRT Post Radical Prostatectomy.
- Carrie C, Hasbini A, de Laroche G, Richaud P, Guerif S, et al. (2016) Salvage radiotherapy with or without short-term hormone therapy for rising prostate-specific antigen concentration after radical prostatectomy (GETUG-AFU 16): a randomized, multicenter, open-label phase 3 trial. Lancet Oncol 17: 747-756.
- Shipley WU, Seiferheld W, Lukka HR, Major PP, Heney NM, et al. (2017) Radiation with or without antiandrogen therapy in recurrent prostate cancer. N Engl J Med 376: 417-428.
- Afshar-Oromieh A, Holland-Letz T, Giesel FL, Kratochwil C, Mier W, et al. (2017) Diagnostic performance of 68Ga-PSMA-11 (HBED-CC) PET/CT in patients with recurrent prostate cancer: evaluation in 1007 patients. Eur J Nucl Med Mol Imaging 44: 1258-1268.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi