Case Report, J Clin Exp Oncol Vol: 7 Issue: 2
Primary Intramedullary Diffuse Large B cell Lymphoma Mimicking a Spinal Meningioma: A Case Report and Review of the Literature
Homajoun Maslehaty1*, Roman Frantsev2, Jan van de Nes2 and Ulrich Sure2
1Department of Neurosurgery, Nordstadt Hospital Hannover, Germany
2Department of Neurosurgery, University Hospital Essen, Germany
*Corresponding Author : Homajoun Maslehaty
Department of Neurosurgery, Nordstadt Hospital Hannover Haltenhoffstrasse 41, 30167 Hannover, Germany
Tel: +49511-9700
E-mail: h.maslehaty@gmx.de
Received: February 15, 2018 Accepted: March 05, 2018 Published: March 12, 2018
Citation: Maslehaty H, Frantsev R, Nes J, Sure U (2018) Primary Intramedullary Diffuse Large B cell Lymphoma Mimicking a Spinal Meningioma: A Case Report and Review of the Literature. J Clin Exp Oncol 7:2. doi: 10.4172/2324-9110.1000216
Abstract
Non-Hodgkin lymphomas account for only 0.3 to 1.5% of all central nervous neoplasms and are mainly subdivided in B-cell and T-cell lymphomas. Primary spinal involvement of PNCLS is a very rare disease, most being metastatic. We report a rare case of a 62-yearold male with a short history of progressive paraparesis. MRI of the spine showed a homogeneous contrast enhanced tumor mass with dural attachment, assuming a spinal meningioma. During surgery the tumor appeared diffusely grown into the spinal cord without detectable margins. Histological work-up brought up the diagnosis of a diffuse large B-cell-lymphoma (DLBCL). We illustrate the diagnostic steps and pitfalls and highlight the multimodal therapy and oncological features of this rare disease.
Keywords: Diffuse large B cell lymphoma; Intramedullary; Primary CNS lymphoma; Surgery
Introduction
Primary central nervous lymphomas (PCNSL) amount up to 6% of intracranial neoplasms and are divided in Hodgkin and non- Hodgkin lymphomas [1,2]. Most of these lesions are localized in the supratentorial space, including the periventricular area and corpus callosum in 86%. Infratentorial localization is determined with about 13% and spinal cord involvement in about 1% [3,4]. CNS affection of Hodgkin lymphomas occur more frequently in the setting of grade III or IV systemic diseases [5].
Non-Hodgkin lymphomas account for only 0.3 to 1.5% of all central nervous neoplasms and are mainly subdivided in B-cell and T-cell lymphomas [6].
Primary spinal involvement of PNCLS is a very rare disease, most being metastatic [7]. Current literature reports only a few cases, however, predominantly without involvement of the spinal cord [3,8-10].
In the presented article, we present a rare case of a primary intramedullary spinal cord B-cell lymphoma mimicking a spinal meningioma in a patient with progressive paraparesis. We illustrate the diagnostic steps, stepwise therapy and highlight the oncological features.
Case Presentation
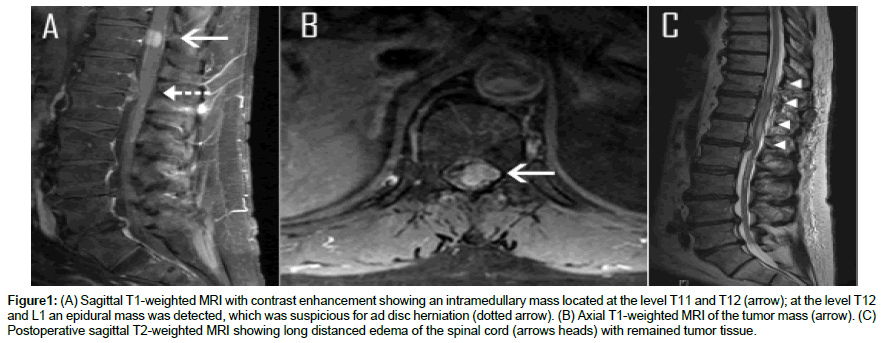
A 62-years-old male presented with a three days history of progressive weakness and numbness of the left leg in the referring hospital. Medical history was uneventful, without tumor anamnesis. Spinal MRI showed a contrast enhanced tumor adjacent to the dura with compression of the spinal cord at the level of Th11 and Th12, suggestive of a spinal meningioma (Figure 1A and B).
Figure 1: (A) Sagittal T1-weighted MRI with contrast enhancement showing an intramedullary mass located at the level T11 and T12 (arrow); at the level T12 and L1 an epidural mass was detected, which was suspicious for ad disc herniation (dotted arrow). (B) Axial T1-weighted MRI of the tumor mass (arrow). (C) Postoperative sagittal T2-weighted MRI showing long distanced edema of the spinal cord (arrows heads) with remained tumor tissue.
At admission the patient was alert, without any cranial nerve complaints. He was unable to walk due to a paresis of the left leg (iliopsoas paresis grade 1 and foot flexor paresis grade 3) with accompanied left leg hypoesthesia (Frankel grade C). There was no evidence of bladder dysfunction.
Written informed consent was obtained and surgery was performed at the following day with intraoperative electrophysiological monitoring. The dura was opened via laminectomy of Th11 and 12. Surprisingly, the tumor showed diffuse growth patterns into the spinal cord, without detectable margins. Hence, total tumor resection was not done and only extended biopsy was performed. Tumor specimens were sent for histo-pathological work-up. During surgery evoked muscle potentials of the left leg diminished and were not reproducible at the end of surgery. Postoperatively, the patient presented a complete plegia of the left leg. CT scan of the spine ruled out any hemorrhagic complications.
A few days later the patient developed a complete paraplegia with bladder dysfunction. MRI of the spine showed an enormous edema of the spinal cord. There was no evidence for postoperative bleeding (Figure 1C).
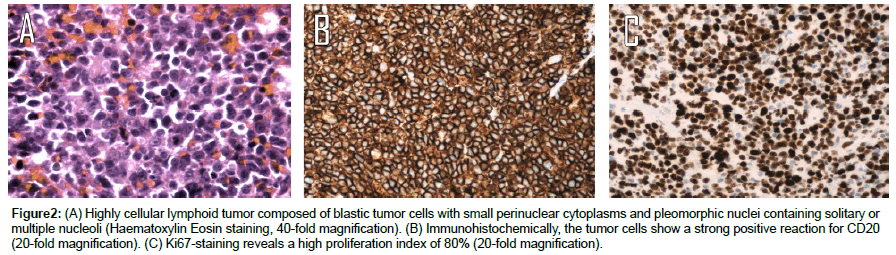
Histopathological examination showed scanty fragments of connective tissue with diffuse infiltration of a highly cellular lymphoid neoplasm. The blastic lymphoid tumor cells displayed small perinuclear cytoplasms and relatively large pleomorphic nuclei with solitary or multiple nucleoli (corresponding to immunoblasts or centroblasts). In addition, numerous mitoses und foci of necrosis were seen. Immunohistochemically, the tumor cells showed a strong positive reaction for the B-cell differentiation marker CD20. Additionally, a Ki67-labelling index of 80% was seen. According to the Revised European-American Lymphoma- (REAL-) and the WHO-classification, these findings lead to the diagnosis of diffuse large B-cell-lymphoma (DLBCL) Ann-Arbor-Stage IV (Figure 2). The patient was referred to the department of haemato-oncology for further treatment.
Figure 2: (A) Highly cellular lymphoid tumor composed of blastic tumor cells with small perinuclear cytoplasms and pleomorphic nuclei containing solitary or multiple nucleoli (Haematoxylin Eosin staining, 40-fold magnification). (B) Immunohistochemically, the tumor cells show a strong positive reaction for CD20 (20-fold magnification). (C) Ki67-staining reveals a high proliferation index of 80% (20-fold magnification).
The patient was started on a chemotherapy protocol GMALLB- ALL for patients > 55 years of life with methotrexate (8600 mg), rituximab (800 mg) and prednisolone 16 mg (i.v) for 5 days. Additionally to chemotherapy the patient received treatment of external beam radiation at 3 Gy fractions for a total of 7 Gy of the spinal cord at level Th11-L2 for 2 weeks. The radiotherapy was stopped due to lack of neurological improvement of the patient.
Additional PET-CT scan did not reveal any evidence of a nodal disease, despite the residual intramedullary tumor mass, assuming a PCNSL.
Despite decreased swelling in the spinal cord and a diminished contrast-enhancement of the tumor on the three months followup MRI the patient remained paraplegic with presence of bladder dysfunction.
Discussion
PCNSL are a form of extra-nodal lymphoma and account for 6% of all intracranial neoplasms. PCNSL are in many cases (90%) diffuse large B-cell-lymphomas and amount up to 1%-2% of all non- Hodgkin Lymphomas [1]. Diffuse large B-cell-lymphomas involve the spinal cord in approximately 1% [4].
Only few cases of primary intramedullary spinal cord non- Hodgkin’s diffuse large B-cell lymphoma have been reported in the literature [6,11-13].
In our case spinal MRI showed a contrast-enhanced tumor adjacent to the dura with compression of the spinal cord at the level T11 and T12. Since the patient was in good health, without preexisting neoplasm, the MRI findings appeared to us typically for a spinal meningioma. Other possible entities of this mass like metastasis, infections, (toxoplasmosis, tuberculosis, and granulomata’s) and other malignant tumors were considered unlikely. Hence, we performed surgery with the intention of complete tumor resection. The operative strategy was changed immediately after recognition of the infiltrative growth of the tumor into the spinal cord and tumor tissue was collected for histopathological work-up.
Nakamizo et al. report 2 cases of primary spinal intramedullary lymphomas and review all previously reported magnetic resonance imaging findings of this disease. The most common finding was a solid and homogeneously enhanced mass that was hyper intense on T2- weighted images, without associated syringomyelia and hypointense on T1-weighted images [14]. However, these signal characteristics are similar to those of other spinal tumors, such as meningiomas or schwannomas, like in our case.
Diagnosis of DLBCL can only be done by histopathological workup, which requires presence of tumor specimen collected surgically via lymph node or extra-nodal tissue biopsy. According to the European Society for medical Oncology, immunohistochemistry using CD45 and CD20 is mandatory for setting the diagnosis. Moreover, complete blood count and routine blood chemistry including lactate dehydrogenase (LDH) are required, as well as protein electrophoresis [15].
Some authors suggest the collection of cerebrospinal fluid for diagnosis. Flanagan et al. described the characteristic CSF findings of elevated protein levels (>100 mg/dl), lack of oligo-clonal bands and elevated cell count were also proposed to discriminate lymphoma and from multiple sclerosis [11].
In our case the patient developed a delayed complete paraplegia with bladder dysfunction. MRI of the spine showed long distanced edema of the spinal cord. After assessment of all findings, we assumed fast tumor growth with compression of the spinal cord vessels with interruption of blood supply causing an anterior spinal artery syndrome with neurological deterioration and presence of a spinal cord edema as the possible pathogenesis behind it. Unfortunately, the tumor showed aggressive and rapid growth and caused neurological deterioration prior to the results of the histopathological examination before any treatment could be administered.
The standard systemic treatment for DLBCL is the CHOPtherapy (cyclophosphamide, doxorubicin, vincristine, and prednisone), or with rituximab (R-CHOP), a chimeric monoclonal antibody against the CD20 antigen. Both of the treatment modalities can lead to lower relapse and improved survival. However, radiation remains an integral part of the treatment of localized disease, achieving high complete response rates and long term recurrence free survival after chemotherapy [16]. Various alternative regimens were explored in an attempt to improve on R-CHOP, such as R-ACVBP (rituximab, doxorubicin, cyclophosphamide, vindesin, bleomycin and prednisone), which significantly improves the survival of patients aged 18-59 years compared with R-CHOP [17].
The clinical and radiological features of this disease can mimic other medical conditions leading to different treatment strategies. In inconclusive situations infrequent tumor entities like intramedullary PCNS lymphomas should be kept in mind.
Conflict of Interest
We declare that we have no conflict of interest.
References
- Iwamoto FM, DeAngelis LM (2006) An update on primary central nervous system lymphoma. Hematol Oncol Clin North Am 20: 1267-1285.
- Miller DC, Hochberg FH, Harris NL, Gruber ML, Louis DN, et al. (1994) Pathology with clinical correlations of primary central nervous system non-Hodgkin's lymphoma. The Massachusetts General Hospital experience 1958-1989. Cancer 74: 1383-1397.
- Cugati G, Singh M, Pande A, Ramamurthi R, Balasubramanyam M, et al. (2011) Primary spinal epidural lymphomas. J Craniovertebr Junction Spine 2: 3-11.
- Hochberg FH, Miller DC (1988) Primary central nervous system lymphoma. J Neurosurg 68: 835-853.
- Sapozink MD, Kaplan HS (1983) Intracranial Hodgkin's disease. A report of 12 cases and review of the literature. Cancer 52: 1301-1307.
- Bekar A, Cordan T, Evrensel T, Tolunay S (2001) A case of primary spinal intramedullary lymphoma. Surg Neurol 55: 261-264.
- Alves OL (1998) Current Management of Primary Cerebral Lymphomas. Neurosurg Quart 8: 71-140.
- Cağavi F, Kalayci M, Tekin IO, Numanoğlu G, Cağavi Z, et al. (2006) Primary spinal extranodal Hodgkin's disease at two levels. Clin Neurol Neurosurg 108: 168-173.
- Higgins SA, Peschel RE (1995) Hodgkin's disease with spinal cord compression. A case report and a review of the literature. Cancer 75: 94-98.
- Love JG, Miller RH, Kernohan JW (1954) Lymphomas of spinal epidural space. AMA Arch Surg 69: 66-76.
- Flanagan EP, O'Neill BP, Porter AB, Lanzino G, Haberman TM, et al. (2011) Primary intramedullary spinal cord lymphoma. Neurology 77: 784-791
- McDonald AC, Nicoll JA, Rampling R (1995) Intramedullary non-Hodgkin's lymphoma of the spinal cord: a case report and literature review. J Neurooncol 23: 257-263.
- Sivri M, Erdoğan H, Allahverdiyev I, Koplay M, Temizöz O (2015) A rare cause of spinal mass: primary intramedullary spinal cord lymphoma. Spine J 15: e43-4
- Nakamizo T, Inoue H, Udaka F, Oda M, Kawai M, et al. (2002) Magnetic resonance imaging of primary spinal intramedullary lymphoma. J Neuroimaging 12: 183-186.
- Tilly H, Vitolo U, Walewski J, da Silva MG, Shpilberg O, et al. (2012) Diffuse large B-cell lymphoma (DLBCL): ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol 26: v116-125.
- Persky DO, Unger JM, Spier CM, Stea B, LeBlanc M, et al. (2008) Phase II study of rituximab plus three cycles of CHOP and involved-field radiotherapy for patients with limited-stage aggressive B-cell lymphoma: Southwest Oncology Group study 0014. J Clin Oncol 26: 2258-2263
- Récher C, Coiffier B, Haioun C, Molina TJ, Fermé C, et al. (2011) Intensified chemotherapy with ACVBP plus rituximab versus standard CHOP plus rituximab for the treatment of diffuse large B-cell lymphoma (LNH03-2B): an open-label randomised phase 3 trial. Lancet 378: 1858-1867.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi