Research Article, J Regen Med Vol: 7 Issue: 1
Progenitor Subpopulations of Valvular Cells Resembling Hematopoietic and Mesenchymal Stem Cells and their Role in Myofibroblastic Activation
Mir S Ali, Xinmei Wang and Carla MR Lacerda*
Department of Chemical Engineering, Texas Tech University, Lubbock, TX, USA
*Corresponding Author : Carla MR Lacerda
Department of Chemical Engineering, Texas Tech University, USA
Tel: 806-834-4089
E-mail: carla.lacerda@ttu.edu
Received: March 13, 2018 Accepted: April 11, 2018 Published: April 17, 2018
Citation: Ali MS, Wang X, Lacerda CMR (2018) Progenitor Subpopulations of Valvular Cells Resembling Hematopoietic and Mesenchymal Stem Cells and their Role in Myofibroblastic Activation. J Regen Med 7:1. doi: 10.4172/2325-9620.1000141
Abstract
valvular interstitial cells (qVICs) resembling fibroblasts. In diseased valves, a myofibroblastic phenotype, activated VICs (aVICs), is highly proliferative, synthesize extracellular matrix and repair/remodel the valve. Adult valves also have an understudied small population of progenitor cells (pVIC), which can differentiate into other VIC phenotypes. A better understanding is needed for the role of pVIC in valvular pathophysiology. We hypothesize that pVICs mediate deactivation of VICs, to control or prevent pathological development. Methods: In this study, we isolated two subpopulations of pVICs, Mesenchymal Stem Cells (MSC) and Hematopoietic Stem Cells (HSC), and evaluated their role in myofibroblastic deactivation of VICs. Porcine pVIC subpopulations were magnetically isolated with CD90 and CD34 respectively serving as markers of MSC and HSC. MSC and HSC subpopulations were validated using secondary MSC and HSC markers CD105 and CD117 respectively. Three culture types were designed. Results: A pVIC-supplemented culture was created by increasing pVIC concentration in VIC population by 50% (positive response) Native culture maintained pVIC concentration identical to native valves postharvest (physiologic response). Negative culture had pVICs removed (negative response). Supplemented culture with MSC subpopulation did not have any effect on VIC activation. Supplemented culture with HSC subpopulation induced deactivation in VICs. Conclusion: To our knowledge, this is one of the first observations of pVIC subpopulations mediating myofibroblastic deactivation in VICs and further studies are needed for a more detailed understanding of pVIC function in valvular biology.
Keywords: Valvular cell progenitors; Hematopoietic stem cells; Mesenchymal stem cells; Magnetic separation; Valvular interstitial cells; Regenerative potential; Tissue engineering
Introduction
About 5 million people in the USA are burdened with heart valve diseases [1] and from them about 50,000 people die every year [2]. Degenerative diseases are the major valvular disease in modern industrial world and affect mitral and aortic valves mostly [3,4]. Non-surgical treatment for valvular diseases is lacking and current knowledge of valvular pathophysiology is not sufficient. Heart valves are populated by two cell types. Valvular endothelial cells (VECs) line the outer surface of the valves and are in contact with blood [5]. Valvular interstitial cells (VIC) reside throughout the valve and maintain valvular pathophysiology [6,7]. VICs are a heterogeneous population and five distinct phenotypes have been identified [7]. They are embryonic endothelial/mesenchymal, quiescent (qVIC), activated (aVIC), osteoblastic (obVIC) and progenitor (pVIC). Embryonic endothelial/mesenchymal VIC undergo endothelial to mesenchymal transition (EndoMT) during valvulogenesis and form heart valves through proliferation and extracellular matrix (ECM) synthesis [8]. All other phenotypes are present only in adult valves. qVICs have fibroblast-like characteristics and reside in adult healthy valves [9], qVICs remain quiescent until injury triggers myofibroblastic activation to aVIC [9,10]. aVICs play a leading role in valvular degenerative pathogenesis. aVICs are contractile and proliferative [7]. They repair and remodel the valve leaflet by synthesizing ECM [7]. aVICs are commonly characterized by high expression of cytoskeletal protein α smooth muscle actin (α-SMA) [10,11]. Approximately 90% of VIC population from adult healthy valves are qVIC and approximately 2.5% are aVIC [9]. In myxomatous valves, approximately 60% of VIC population are aVIC [9]. obVICs are only seen in calcific valves and have osteoblastic properties [12].
The least understood among VIC phenotypes is pVIC [7]. pVICs have progenitor or stem cell properties and can differentiate into other VIC phenotypes [7]. pVICs have been shown to consist of several subpopulations. Using mouse models, a hematopoietic stem cell (HSC) subpopulation has been shown to reside in heart valves [13,14]. Heterogeneity in pVIC population is visible when tested with different stem cell markers [15-17]. A subpopulation of pVIC, resembling mesenchymal stem cells (MSC), was identified using stage-specific embryonic antigen 4 [15], CD68 [18], CD117 [16] and colony forming unit assays [19]. These two stem cell populations are well-known bone marrow residents [13,14,18-20], potentially traveling to the valves via the bloodstream. Besides finding an MSC subpopulation, Wang et al. [15] identified two other pVIC subpopulations resembling pericytes and side population progenitors. For pericytes, they employed chondroitin sulfate proteoglycan 4 as marker. For side population progenitors, they employed ATPbinding cassette sub-family G member 2 as marker. Side population progenitors are an adult tissue derived population of multipotent progenitor cells [21]. The marker used for side population progenitor has also been shown to be an HSC marker [22,23] suggesting identification of an HSC progenitor subpopulation in VICs. VICs, positive for this marker, also deposited more calcified matrix in vitro [15]. An osteoprogenitor subpopulation of pVIC contributing to osteogenic calcification has also been identified [19]. pVICs also contain subpopulations of circulating endothelial progenitor cells [17,24-26] and dendritic cells [17,27].
HSC and MSC are two major adult stem cells in bone marrow. HSCs are responsible for production of all blood cell lineages to maintain hematopoiesis [28,29]. HSCs have several positive and negative markers. HSCs are generally marked as CD34+ [29-34] but contradictory viewpoints also exist in the current literature [35]. MSCs, also referred to as stromal cells, are found in many tissues [36] and can differentiate into osteoblasts, chondroblasts, adipocytes etc [31,37]. MSCs are generally characterized by in vitro plastic adherence, specific marker expression (STRO-1+, CD90+, CD105+ and CD34-) and specific differentiation abilities [31,37,38]. Contradiction for CD34 as negative marker for MSC also exists [35,39].
Deactivation of aVIC to qVIC is essential in maintaining normal valve physiology and preventing degenerative pathology. aVICs have been deactivated in vitro using substrate stiffness [40] and cocultures with VEC [41]. Although both HSC and MSC subpopulations of pVIC have been identified in VIC population before [13-16,18,19], their function in valvular pathophysiology is unknown.
Here, we tested the function of MSC and HSC subpopulations of pVIC in VIC deactivation. pVICs were isolated from porcine VIC populations from aortic valves using MSC marker CD90 [30] and HSC marker CD34 [30,31]. MSC and HSC subpopulations were validated using secondary MSC and HSC markers CD105 and CD117 respectively. Three culture types were prepared to study pVIC subpopulation functions, particularly in regards to their role mediating VIC deactivation. The experimental groups are as follows:
Native: This group represents the native population of VICs as extracted from aortic valve leaflets. It serves as the environmental control maintaining the concentration of pVICs in VIC population identical to that in native valves postharvest.
Supplemented: The supplemented group is a 1:1 mixture of native and positive pVICs. Positive pVICs are MSC or HSC subpopulations of pVICs magnetically isolated from native VIC populations using CD90 or CD34 respectively. The supplemented culture is hypothesized to amplify the role of pVICs by increasing their relative ratio in a VIC population.
Negative: The negative group includes VICs that are negative for MSC or HSC subpopulation markers CD90 or CD34 respectively. This group corresponds to a VIC population completely devoid of pVIC of MSC or HSC origin. Negative cultures were used to explore how VICs would behave in the absence of pVIC.
Methods
Collection of cells for magnetic separation: Aortic valve leaflets were excised from porcine hearts within 4 h of slaughter. Leaflets were washed with phosphate buffer saline (137 mM sodium chloride, 2.7 mM potassium chloride, 4.3 mM sodium phosphate dibasic and 1.46 mM potassium phosphate monobasic (all Fisher Scientific, Waltham, MA)). VECs were scrubbed from leaflets after 10 min digestion in 600 U/mL collagenase (Sigma-Aldrich, St. Louis, MO) in cell culture medium at 37°C. Leaflets were then incubated in used collagenase medium overnight at 37°C. All 37°C incubations were performed in a humidified chamber with 5% carbon dioxide. Cell culture medium contains Dulbecco’s modified eagle medium (Mediatech, Corning, Manassas, VA) supplemented with 10% bovine growth serum (Atlanta Biologicals Inc., Flowery Branch, GA) and 1% antibiotics/antimycotics (10,000 U/mL penicillin G, 10 mg/ mL streptomycin sulfate and 25 μg/mL amphotericin B (Quality Biologicals, Gaithersburg, MD)). Digested VIC from leaflets were seeded on plastic culture plates at 105 cells/cm2 and cultured for 3 days. VICs were collected using 0.125% (w/v) trypsin (Sigma-Aldrich) in phosphate buffer saline at 37°C for 15 min. Collected VICs were used for magnetic separation.
Magnetic separation: 3 × 107 VICs were used for magnetic separation. Cells were centrifuged at 100xg and resuspended in 500 μL serum replacement medium (Dulbecco’s modified eagle medium supplemented with 10% serum replacement (Sigma-Aldrich)). 10 μg/ mL of anti CD34 antibody (Abcam, Cambridge, MA) or 20 μg/mL of anti CD90 antibody (Fisher Scientific) were added to serum replacement supplemented medium. Serum replacement was used to avoid the interaction of antibodies with bovine growth serum. VICs were incubated with antibodies at 37°C for 3 h with gentle rocking. In parallel, another batch of VICs was incubated in serum replacement medium for 3 h without antibodies to be used as native VICs. After incubation with primary antibodies, cells were centrifuged at 100xg and resuspended in serum replacement medium (80 μL/107 cells) and secondary IgG conjugated with magnetically capturable microbeads (Miltenyi Biotech, Auburn, CA) 20 μL/107 cells at 37°C for 30 min with gentle rocking. Cells were centrifuged again and resuspended in phosphate buffer saline with 2 mM ethylene diamine tetraacetic acid and 0.5% bovine serum albumin for maintaining cell viability (both Fisher Scientific). Cell suspension was then flown through magnetic columns (Miltenyi Biotech) along with OctoMACS magnet (Miltenyi Biotech). Magnetically labelled cells were retained by magnet and remained in the column. Unlabeled cells, not captured by magnet, were collected as negative cells. Magnetically labeled cells were then eluted from the column and collected as positive cells. Negative, positive and native cells were centrifuged at 100xg and resuspended in culture medium.
To validate the magnetic separation process, a 1:1 mixture of adipose derived stem cells (ADSCs) and VICs were separated following same protocol using 10 μg/mL CD34 antibody as ADSC marker. Approximately 56% positive cells after separation from this ADSC-VIC mixture was expected as ADSCs are positive for CD34 [35,42,43] and 6% of VICs have been found positive for CD34 [17]. Cell counting was performed using a hemocytometer (Fisher Scientific) after diluting cell solution with 0.4% trypan blue (Fisher Scientific). Concentration of pVIC in VIC population was counted as ratio of number of positive VICs to the sum of numbers of positive and negative VICs.
A parallel batch of VIC incubated without antibodies were used for native VIC culture. Positive VICs collected using magnet and native VICs were mixed in a 1:1 ratio for supplemented culture. Negative VICs, not caught by magnet, were used for negative culture. Native, supplemented and negative cultures were started on cover glass or plastic culture plates at 105 cells/cm2 seeding density, continued for 2 days and then used for immunofluorescence and immunoblot.
Immunofluorescence: Cultures on cover glass were fixed with 2% formaldehyde (Fisher Scientific) for 10 min and permeabilized with 0.1% IGEPAL CA630 (Sigma-Aldrich) for 5 min. Nonspecific binding was blocked with 1% goat serum (MP Biomedicals, Solon, OH). Incubation with primary and secondary antibodies were performed for 2 h in 0.1% bovine growth serum in MES buffer (10 mM 2-(N-morpholino) ethane sulfonic acid, 10 mM sodium chloride, 1.5 mM magnesium chloride, 10% glycerol and 100 KIU aprotinin at pH 6.2 (all Fisher Scientific)). Primary antibodies were anti-CD105 (0.5 μg/mL) as MSC marker and anti-α-SMA (0.5 μg/mL) as aVIC marker (all Fisher Scientific). Secondary antibodies used are goat anti-mouse and anti-rabbit IgG DyLight 488 0.2 μg/mL and DyLight 550 0.5 μg/mL (all Fisher Scientific). 1 μg/mL DAPI stain (Fisher Scientific) was performed for 1 min. 3 x 1 min MES buffer washes were performed after every step. Fluorescent microscopy was performed using Leica DMI6000 B system (Leica Microsystems, Buffalo Grove, IL).
Immunoblot: Cells on plastic culture plates were washed with phosphate buffer saline three times. Complete extraction buffer (2mM dithiothreitol, 25 μg/mL digitonin, 150 mM sodium chloride, 50 mM HEPES (all Fisher Scientific) and 1% IGEPAL CA630) was added at 50 μL/cm2 of plate area and incubated at 4°C for 1 h with gentle rocking. Samples were centrifuged at 1100xg for the removal of insoluble debris. 1% Halt protease inhibitor cocktail (Active Motif, Carlsbad, CA) was added to samples. Bicinchoninic acid assay (Fisher Scientific) was used following manufacturer’s protocol to determine protein concentration in samples. 40 μg protein samples were precipitated with 9X volume ethanol (Fisher Scientific) at -80°C for 4 h to overnight. Gel electrophoresis was performed at 300 V for 15 min followed by membrane transfer. Membranes were incubated overnight with primary antibodies diluted in 3% nonfat milk in tris buffer saline (10 mM tris base (Fisher Scientific), 150 mM sodium chloride at pH 8.0). Primary antibodies were anti-α-SMA (1:2000), anti-CD105 (1:750), anti-CD117 (1:500) (Novus Biologicals, Littleton, CO) and anti-glyceraldehyde 3-phosphate dehydrogenase (GAPDH) (1:750) (EMD Millipore, Temecula, CA). Membranes were incubated with goat anti-mouse and anti-rabbit IgG (H+L) conjugated with horseradish peroxidase (AnaSpec, Fremont, CA) for 1 h. Protein bands were developed using luminol chemiluminescence (Bio-Rad Laboratories, Hercules, and CA) and were imaged in Chemidoc XRS imager (Bio-Rad Laboratories). Band intensities were quantified using ImageJ (Image processing and analysis in Java by NIH Image, National Institute of Health, Bethesda, MD) and normalized according to GAPDH intensities.
Results
Concentration of pVIC in VIC population: VICs have distinct phenotypes. Of them pVIC is the least studied and understood phenotype. Although subpopulations of pVIC have been identified before, their function in VIC pathophysiology remains largely unknown. Here, we focused on isolating two pVIC subpopulations resembling MSC and HSC and testing their role in VIC myofibroblastic activation. We isolated pVIC subpopulations from porcine aortic valve VICs through magnetic separation technique using molecular markers for MSC and HSC. We used CD90 as MSC marker and CD34 as HSC marker. Magnetic separation of VICs yielded on average 4% of VICs positive for both CD34 and CD90 individually. This suggests that concentrations of either MSC or HSC subpopulation in native VIC population are roughly 4%.
To validate the magnetic separation protocol used in this experiment, we separated a 1:1 mixture of ADSC and VIC using CD34 as marker. For this validation experiment, we considered ADSC as CD34+ and VIC as partially CD34+. Although ADSCs have been generally considered CD34- [44,45], recent evidence suggests that ADSC are CD34+ [35,42,43]. 6% of VIC population have also been found to be CD34+ before [17]. From this ADSC-VIC mixture, 61% of cells were separated using magnetic separation as CD34+. A positive separation of close to 56% (50% for ADSC + 6% for VIC) indicates that the protocol used here includes considerable technical and biological variability with an error of 9%.
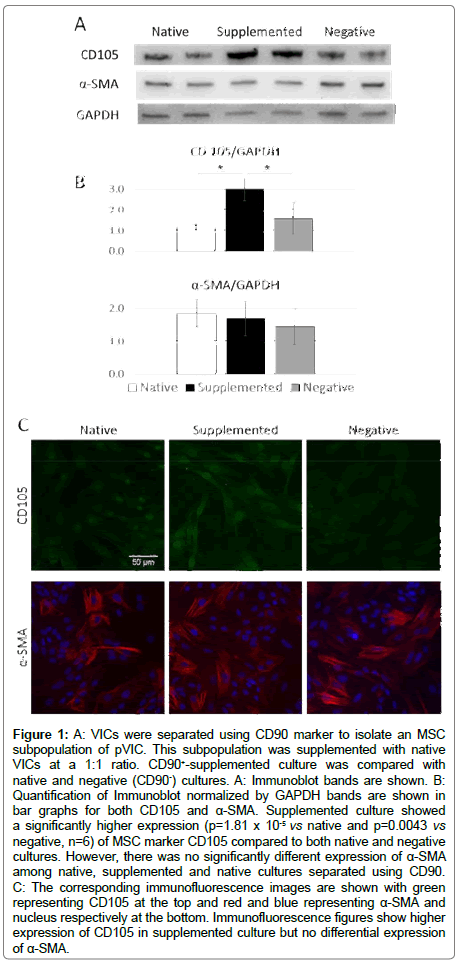
MSC subpopulation is positive for another MSC marker: To test the validity of our MSC subpopulation separation process, we tested them with another MSC marker CD105 [31,46]. We tested CD105 expression in native, supplemented and negative cultures prepared using VICs separated using CD90. Supplemented culture showed higher expression of CD105 compared to both native and negative cultures (p=1.81 x 10-5 vs native and p=0.0043 vs negative, n=6) (Figure 1). Immunofluorescence images also showed higher expression of CD105 (green) in supplemented culture compared to both native and negative cultures (Figure 1). These results validate the effectiveness in isolating a pVIC subpopulation with true MSC characteristics.
Figure 1: A: VICs were separated using CD90 marker to isolate an MSC subpopulation of pVIC. This subpopulation was supplemented with native VICs at a 1:1 ratio. CD90+-supplemented culture was compared with native and negative (CD90-) cultures. A: Immunoblot bands are shown. B: Quantification of Immunoblot normalized by GAPDH bands are shown in bar graphs for both CD105 and α-SMA. Supplemented culture showed a significantly higher expression (p=1.81 x 10-5 vs native and p=0.0043 vs negative, n=6) of MSC marker CD105 compared to both native and negative cultures. However, there was no significantly different expression of α-SMA among native, supplemented and native cultures separated using CD90. C: The corresponding immunofluorescence images are shown with green representing CD105 at the top and red and blue representing α-SMA and nucleus respectively at the bottom. Immunofluorescence figures show higher expression of CD105 in supplemented culture but no differential expression of α-SMA.
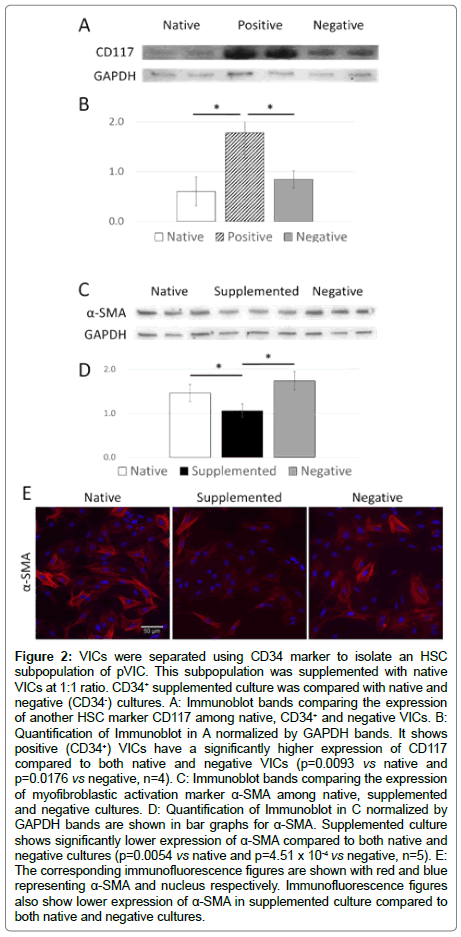
HSC subpopulation is positive for another HSC marker: To test the validity of our CD34+ HSC subpopulation separation process, we tested them with another HSC marker CD117 [32,34]. CD34+ VICs showed higher expression of CD117 compared to both native and negative VICs (p=0.0093 vs native and p=0.0176 vs negative, n=4) (Figure 2). This validates the effectiveness in isolating a pVIC subpopulation with true HSC characteristics.
Figure 2: VICs were separated using CD34 marker to isolate an HSC subpopulation of pVIC. This subpopulation was supplemented with native VICs at 1:1 ratio. CD34+ supplemented culture was compared with native and negative (CD34-) cultures. A: Immunoblot bands comparing the expression of another HSC marker CD117 among native, CD34+ and negative VICs. B: Quantification of Immunoblot in A normalized by GAPDH bands. It shows positive (CD34+) VICs have a significantly higher expression of CD117 compared to both native and negative VICs (p=0.0093 vs native and p=0.0176 vs negative, n=4). C: Immunoblot bands comparing the expression of myofibroblastic activation marker α-SMA among native, supplemented and negative cultures. D: Quantification of Immunoblot in C normalized by GAPDH bands are shown in bar graphs for α-SMA. Supplemented culture shows significantly lower expression of α-SMA compared to both native and negative cultures (p=0.0054 vs native and p=4.51 x 10-4 vs negative, n=5). E: The corresponding immunofluorescence figures are shown with red and blue representing α-SMA and nucleus respectively. Immunofluorescence figures also show lower expression of α-SMA in supplemented culture compared to both native and negative cultures.
MSC subpopulation does not affect VIC activation: pVICs resembling MSC subpopulations have been identified in previous work [15,16,18,19]. But not many experiments have been performed to research their role in valvular pathophysiology. There are some studies showing a role of MSC subpopulation in inducing calcification in VICs [19,47]. But none has explored the role of MSC subpopulation in a key phenomenon in valvular pathophysiology, VIC myofibroblastic activation. Here we explore the role of MSC subpopulation in VIC activation by amplifying their function using a supplemented culture. MSC subpopulation was separated from a VIC population using CD90 marker. This subpopulation was supplemented with native VICs at 1:1 ratio. This increased the concentration of MSC in a VIC population from 4% to >50%. CD90+ supplemented culture was compared with native and negative (CD90-) cultures. There was no significantly different expression of α-SMA among native, supplemented and native cultures (Figure 1). The immunofluorescence images also do not show differential expression of α-SMA (red) among these native, supplemented and native cultures. These results suggest, MSC subpopulation does not play a direct role in VIC activation even if their concentration is amplified in a VIC culture. To further investigate pVIC function, we assessed the HSC subpopulation.
HSC subpopulation induces deactivation in VICs: Similarly to the MSC subpopulation, HSC subpopulation has also been identified in valves [13-15]. Knowledge of HSC subpopulation function in valvular pathophysiology is also lacking. To test the function of HSC subpopulation in VIC myofibroblastic activation, supplemented cultures with amplified HSC concentration were created. The supplemented culture has HSC concentration increased from 4% to >50%. VICs were separated using CD34 marker to isolate the HSC subpopulation. Supplemented culture was created by mixing CD34+ VICs with native VICs at 1:1 ratio. CD34+ supplemented culture was compared with native and negative (CD34-) cultures. Supplemented cultures showed significantly lower expression of aVIC marker α-SMA compared to native and negative cultures (p=0.0054 vs native and p=4.51 x 10-4 vs negative, n=5) (Figure 2). The corresponding immunofluorescence images also show a lower expression of α-SMA (red) in supplemented culture compared to both native and negative cultures (Figure 2). These results suggest that pVIC subpopulation with HSC characteristics play a role in deactivation of VIC when their concentration is amplified.
Discussion
Distinct subpopulations of pVIC have been identified and isolated before. Among the identified pVIC subpopulations are HSC [13-15], MSC [15,16,18,19], pericytes [15], endothelial progenitor cell [17,24- 26], dendritic cell [17,27] etc. There is minimal knowledge about their function in valvular pathophysiology, specially related to function in VIC activation. Here, we identified and separated MSC and HSC subpopulations of pVIC and explored their role in VIC activation. We found that HSC subpopulation induces deactivation in VICs and MSC subpopulation showed no such effect.
The concentrations of either MSC and or HSC subpopulation in native VIC population resulted to be 4%. Previous studies of pVIC subpopulation resulted in 7% [15] and 10% [46] of VICs positive as MSC and 5% [15] and 6% [17] of VICs positive as HSC using other markers. These numbers suggest that pVICs reside in VIC population in small concentrations.
MSCs are adult stem cell found in different tissues [36] and show several positive and negative markers (STRO-1+, CD90+, CD105+ and CD34-) [31,37,38]. CD90 was used as a marker for isolation of MSC subpopulation in this experiment. Lack of CD34 expression has been generally used to distinguish MSC from hematopoietic cells [1,37,38]. But several studies have questioned CD34 as negative marker for MSC [35,39]. Because of this uncertainty, we used a second MSC marker, CD105, to verify that the isolated MSC subpopulation shows MSC characteristics. Significantly higher expression of CD105 in supplemented MSC subpopulation culture verifies our effectiveness in MSC subpopulation isolation (Figure 1). There is also evidence that MSCs become CD34- with increasing subcultures [35]. So, the MSC subpopulation in this experiment should also be CD34- due to subculturing of VICs.
In this study, MSC subpopulation did not show any function in VIC activation, even when their concentration in VIC population is amplified (Figure 1). Two studies regarding MSC subpopulation function have shown their ability to induce osteogenic calcification in valvular cell and tissue models [19,47]. MSCs from bone marrow also have the ability to differentiate into osteoblasts [37]. This suggests MSC subpopulation may be involved in osteoblastic transformation of VIC, which was not the focus of our study. A combined study looking at both myofibroblastic and osteoblastic VIC transformation may provide us with better insight into MSC subpopulation function.
Although, in our study, MSC subpopulation did not show any effect on VIC activation, MSCs have been shown to affect myofibroblastic activation on other cell types. In liver fibrosis, hepatic stellate cells become myofibroblastic similarly to a VIC. Adult stem cells from bone marrow [48,49], specifically MSCs [50], have been shown to be the source of these fibroblastic and myofibroblastic cell types. MSCs also modulate stellate cell activation via paracrine signaling [50]. MSCs play both positive and negative roles in pathophysiology of liver fibrosis [50-53]. Activated carcinoma associated fibroblasts contribute to tumor growth and cancer progression. Bone marrow derived MSCs have also been shown to be a source of this cell type [54,55]. MSCs also activate this cell type in vitro via paracrine signaling [56]. MSCs contribute to cutaneous wound repair [57,58] and regeneration of ischemic myocardium [59,60] using paracrine signaling.
The HSC subpopulation of pVIC was isolated using CD34 as marker. CD34 is universally considered an HSC marker in current literature and has been widely used for HSC detection and isolation [29-34]. However, new studies are emerging arguing against CD34 as an HSC marker [35]. That is why, to make sure the CD34+ VICs isolated in this experiment represent a true HSC population, we tested it with another HSC marker CD117. CD117, also known as c-Kit, is a receptor for hematopoietic growth factors responsible for HSC survival and proliferation [32-34]. Results show that CD34+ VICs have a significantly higher expression of CD117 compared to both native and CD34- VICs (Figure 2). This suggests that the CD34+ subpopulation of VIC represent a true HSC population as it is positive for a second HSC marker.
In this experiment we tested the role of HSC subpopulation in myofibroblastic activation of VICs. The HSC subpopulation of pVIC did show a role in VIC deactivation. When the effect of HSC subpopulation on VIC population was amplified using a supplemented culture, activation in VICs decreased significantly (Figure 2). Previous studies show that an HSC subpopulation of pVIC travel from bone marrow to heart valves and become valvular residents [13,14]. The same study showed that healthy valves have a higher concentration of this bone marrow derived HSC subpopulation compared to transplanted valves [14]. This suggests HSCs play a role in valvular homeostasis. Our results also suggest that HSC subpopulation maintains valvular physiology by keeping VIC activation and subsequently degeneration in check.
HSC paracrine signaling has not been fully elucidated but several developmentally conserved signaling pathways have been shown to be involved in HSC renewal and differentiation [29]. These pathways include Notch, Wnt and TGF-β/Smad. Notch and Wnt signaling have been shown to induce HSC proliferation and selfrenewal in vitro [29,61,62]. TGF-β works as an inhibitor of HSC growth [29]. These three pathways are heavily involved in valvular development and pathophysiology as well [63]. In adult valves, Notch1 plays an inhibitory role to calcification [64]. Both Wnt and TGF-β are involved in myofibroblastic activation in VICs and in degenerative diseased valves [65-67]. Functions of these pathways in both HSC and valvular biology suggest that, they may be involved in HSC subpopulation mediated deactivation of VICs seen in this study. A low concentration (4%) of HSC subpopulation in a VIC population also suggests that HSC subpopulation interacts with VICs using paracrine signaling mechanism to induce deactivation. This myofibroblastic deactivating function of pVIC could be used in valvular tissue engineering approaches. Valvular tissue engineering aims to maintain physiologically quiescent phenotype of VICs [68,69]. This HSC subpopulation could be used as a cellular approach to induce deactivation in tissue engineered valves.
Despite the significant finding of pVIC function in VIC deactivation, there exist some limitations to this study. Although subpopulations resembling MSC and HSC were separated using molecular markers, we did not test their differentiation abilities. Differentiation abilities are often necessary to more accurately validate isolation of particular stem cell populations. The magnetic cell separation technique used in this experiment has some technical limitations due to non-specific binding of antibodies to negative cells producing false positives. Also, not all the cells containing antigens are always captured by antibodies resulting in false negatives. Despite these possible limitations, the concentrations of positive cells found after the separation process is correct as detected by our validation study.
Conclusions
Here we isolated two subpopulations of VICs, MSC and HSC, and tested their role in mediating myofibroblastic activation in VICs. Magnetic separation was used to separate pVIC subpopulations form porcine VICs. CD90 and CD34 were used for MSC and HSC markers respectively in the magnetic separation process. MSC and HSC subpopulations were validated using secondary MSC and HSC markers CD105 and CD117 respectively. Supplemented cultures were created by amplifying pVIC concentration in VIC population to >50%. They were compared with native cultures containing native pVIC concentration and negative cultures containing no pVIC. Cultures were tested with stem cell markers for validation of pVIC separation process and with α-SMA for VIC phenotype determination. Supplemented culture with MSC subpopulation did not have any effect on VIC activation. Supplemented culture with HSC subpopulation induced deactivation in VICs. This is a very important finding, which could lead to important changes in how valvular diseases are treated in the future. If progenitor valvular cells are indeed capable of reversing disease, this will tremendously impact clinical practice and how we design and engineer devices. This is the first study showing function of pVIC subpopulation in VIC activation. pVIC research is in its infancy and this study could open the door to new pVIC function based studies and valvular tissue engineering techniques using pVICs.
Acknowledgements
We would like to thank Jackson Brothers Meat Locker, Post, TX for providing us with porcine hearts.
References
- Nkomo VT, Gardin JM, Skelton TN, Gottdiener JS, Scott CG (2006) Burden of valvular heart diseases: a population-based study, Lancet 368:1005-1011.
- Mozaffarian D, Benjamin EJ, Go AS, Donna KA, Michael JB, et.al. (2016) Heart disease and stroke statistics-2016 update. Circulation 133:38-360.
- Roberts WC, Ko JM (2008) Some observations on mitral and aortic valve disease. 21:282-299.
- Soler Soler J, Galve E (2000) Worldwide perspective of valve disease, Heart 83:721-725
- Butcher JT, Nerem RM (2007) Valvular endothelial cells and the mechanoregulation of valvular pathology. Philos Trans R Soc L B Biol Sci 362:1445-1457.
- Chester AH, Taylor PM (2007) Molecular and functional characteristics of heart-valve interstitial cells. Philos Trans R Soc B Biol Sci 362:1437-1443.
- Liu AC, Joag VR, Gotlieb AI (2007) The emerging role of valve interstitial cell phenotypes in regulating heart valve pathobiology. Am J Pathol 171:1407-1418.
- Hinton RB, Yutzey KE (2011) Heart valve structure and function in development and disease, Annu Rev Physiol 73:29-46.
- Rabkin-Aikawa E, Farber M, Aikawa M, Schoen FJ (2004) Dynamic and reversible changes of interstitial cell phenotype during remodeling of cardiac valves. J Heart Valve Dis 13:841-847
- Wang H, Leinwand LA, Anseth KS (2014) Cardiac valve cells and their microenvironment - insights from in vitro studies, Nat Rev Cardiol 11:715-727.
- Rabkin E, Aikawa M, Stone JR, Fukumoto Y, Libby P, et.al. (2001) Activated interstitial myofibroblasts express catabolic enzymes and mediate matrix remodeling in myxomatous heart valves. Circulation. 2001;104:2525-2532. doi:10.1161/hc4601.099489.
- Rajamannan NM, Subramaniam M, Rickard D, Stuart RS, Janis D, et.al. (2003) Human aortic valve calcification is associated with an osteoblast phenotype, Circulation 107:2181-2184.
- Visconti RP, Ebihara Y, LaRue AC, Paul A. Fleming, Tim C. McQuinn, et.al. (2006) An in vivo analysis of hematopoietic stem cell potential: hematopoietic origin of cardiac valve interstitial cells, Circ Res 98:690-696.
- Hajdu Z, Romeo SJ, Fleming PA, Markwald RR, Visconti RP, et.al. (2011) Recruitment of bone marrow-derived valve interstitial cells is a normal homeostatic process, J Mol Cell Cardiol 51:955-965.
- Wang H, Sridhar B, Leinwand LA, Anseth KS (2013) Characterization of cell subpopulations expressing progenitor cell markers in porcine cardiac valves, PLoS One 8:1-11.
- Veinot JP, Prichett-Pejic W, Song J, Waghray G, Parks W, et.al. (2006) CD117-positive cells and mast cells in adult human cardiac valves - Observations and implications for the creation of bioengineered grafts. Cardiovasc Pathol;15:36-40.
- Skowasch D, Schrempf S, Wernert N, Steinmetz M, Jabs A, et.al. (2005) Cells of primarily extravalvular origin in degenerative aortic valves and bioprostheses. Eur Heart J, 26:2576-2580.
- Deb A, Wang SH, Skelding K, Miller D, Simper D, et.al. (2005) Bone marrow-derived myofibroblasts are present in adult human heart valves, J Heart Valve Dis 14:674-678.
- Chen JH, Yip CYY, Sone ED, Simmons C (2009) Identification and characterization of aortic valve mesenchymal progenitor cells with robust osteogenic calcification potential, Am J Pathol 174:1109-1119.
- Tanaka K, Sata M, Fukuda D (2005) Age-associated aortic stenosis in apolipoprotein E-deficient mice, J AmColl Cardiol 46:134-141.
- Martin CM, Meeson AP, Robertson SM (2004) Persistent expression of the ATP-binding cassette transporter, Abcg2, identifies cardiac SP cells in the developing and adult heart, Dev Biol 265:262-275.
- Zhou S, Schuetz JD, Bunting KD, Anne-Marie Colapietro, Janardhan Sampath (2001) The ABC transporter Bcrp1/ABCG2 is expressed in a wide variety of stem cells and is a molecular determinant of the side-population phenotype. Nat Med 7:1028-1034.
- Kim M, Turnquist H, Jackson J, Magda S, Ying Y, et al. (2002) The multidrug resistance transporter ABCG2 (Breast Cancer Resistance Protein 1) Effluxes Hoechst 33342 and is overexpressed in hematopoietic stem cells, Clin Cancer Res.
- Matsumoto Y, Adams V, Walther C, Caroline K, Peter B, et al. (2009) Reduced number and function of endothelial progenitor cells in patients with aortic valve stenosis: A novel concept for valvular endothelial cell repair, Eur Heart J. 30:346-355.
- Vaturi M, Perl L, Leshem-Lev D, Dadush O, Bental T, et al. (2011) Circulating endothelial progenitor cells in patients with dysfunctional versus normally functioning congenitally bicuspid aortic valves. Am J Cardiol, 108:272-276.
- Gossl M, Khosla S, Zhang X, Nara H, Kyra LJ et.al. (2012) Role of circulating osteogenic progenitor cells in calcific aortic stenosis, J Am Coll Cardiol 60:1945-1953.
- Choi J-H, Do Y, Cheong C, et al. Identification of antigen-presenting dendritic cells in mouse aorta and cardiac valves, J Exp Med 206:497-505.
- Ogawa M (1993) Differentiation and proliferation of hematopoietic stem cells, Blood 81:2844-2853
- Blank U, Karlsson G, Karlsson S (2008) Signaling pathways governing stem-cell fate, Blood 111:492-503.
- Colter DC, Sekiya I, Prockop DJ (2001) Identification of a subpopulation of rapidly self-renewing and multipotential adult stem cells in colonies of human marrow stromal cells, Proc Natl Acad Sci U S A 98:7841-7845.
- Pittenger MF, Mackay AM, Beck SC, Jaiswal RK, Douglas R (1999) Multilineage potential of adult human mesenchymal stem cells, Science 284:143-147.
- Wognum AW, Eaves AC, Thomas TE (2003) Identification and isolation of hematopoietic stem cells. Arch Med Res 34:461-475.
- Wilson A, Trumpp A (2006) Bone-marrow hematopoietic-stem-cell niches, Nat Rev Immunol 6:93-106.
- Bryder D, Rossi DJ, Weissman IL (2006) Hematopoietic stem cells, Am J Pathol 169:338-346.
- Lin CS, Ning H, Lin G, Lue TF (2012) Is CD34 truly a negative marker for mesenchymal stromal cells? Cytotherapy 14:1159-1163.
- Kassem M, Abdallah BM (2008) Human bone-marrow-derived mesenchymal stem cells: Biological characteristics and potential role in therapy of degenerative diseases. Cell Tissue Res 331:157-163.
- Dominici M, Le Blanc K, Mueller I, Slaper Cortenbach I, Marini F (2006) Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy 8:315-317.
- Chamberlain G, Fox J, Ashton B, Middleton J (2007) Concise review: mesenchymal stem cells: their phenotype, differentiation capacity, immunological features, and potential for homing, Stem Cells 25:2739-2749.
- Deans RJ, Moseley AB (2000) Mesenchymal stem cells: biology and potential clinical uses, Exp Hematol 28:875-884.
- Kloxin AM, Benton JA, Anseth KS (2010) In situ elasticity modulation with dynamic substrates to direct cell phenotype, Biomaterials 31:1-8.
- Shapero K, Wylie-sears J, Levine RA, Mayer JE, Bischoff J (2015) Reciprocal interactions between mitral valve endothelial and interstitial cells reduce endothelial-to-mesenchymal transition and myofibroblastic activation, J Mol Cell Cardiol 80:175-185.
- Baer PC (2014) Adipose-derived mesenchymal stromal/stem cells: An update on their phenotype in vivo and in vitro. World J Stem Cells 6:256-265.
- Lin G, Xin Z, Zhang H, Lia B, Guifang W (2012) Identification of active and quiescent adipose vascular stromal cells. Cytotherapy 14:240-246.
- Zhu Y, Liu T, Song K, Fan X, Ma X, et al. (2008) Adipose-derived stem cell: a better stem cell than BMSC, Cell Biochem Funct 26:664-675.
- Lei L, Liao W, Sheng P, Fu M, He A, et al. (2007) Biological character of human adipose-derived adult stem cells and influence of donor age on cell replication in culture, Sci China Ser C Life Sci 50:320-328.
- Latif N, Sarathchandra P, Thomas PS, Antoniw J, Batten P (2007) Characterization of structural and signaling molecules by human valve interstitial cells and comparison to human mesenchymal stem cells, J Heart Valve Dis 16:56-66.
- Leskelä HV, Satta J, Oiva J, Eriksen H , Juha R (2006) Calcification and cellularity in human aortic heart valve tissue determine the differentiation of bone-marrow-derived cells, J Mol Cell Cardiol 41:642-649.
- Baba S, Fujii H, Hirose T, Kentaro Y, Hisaya A (2004) Commitment of bone marrow cells to hepatic stellate cells in mouse, J Hepatol 40:255-260.
- Forbes SJ, Russo FP, Rey V (2004) A Significant Proportion of Myofibroblasts Are of Bone Marrow Origin in Human Liver Fibrosis. Gastroenterology 126:955-963.
- Parekkadan B , van Poll D , Megeed Z , Kobayashi N , Tilles AW, et al. (2007) Immunomodulation of activated hepatic stellate cells by mesenchymal stem cells. Biochem Biophys Res Commun 363:247-252.
- Higashiyama R, Moro T, Nakao S, KenichiroM, Hiroshi F, et al. (2009) Negligible contribution of bone marrow-derived cells to collagen production during hepatic fibrogenesis in mice, Gastroenterology 137:1459-1466.
- Kallis YN, Forbes SJ (2009) The Bone Marrow and Liver Fibrosis: Friend or Foe? Gastroenterology 137:1218-1221.
- Kisseleva T, Uchinami H, Feirt N, Oscar Q, Jose C, et al. (2006) Bone marrow-derived fibrocytes participate in pathogenesis of liver fibrosis, J Hepatol 45:429-438.
- Mishra PJ1, Mishra PJ, Humeniuk R, Medina DJ, Alexe G, et al. (2008) Carcinoma-associated fibroblast-like differentiation of human mesenchymal stem cells. Cancer Res 68:4331-4339.
- Quante M, Tu SP, Tomita H, Gonda T, Wang SS, et al. (2011) Bone Marrow-Derived Myofibroblasts Contribute to the Mesenchymal Stem Cell Niche and Promote Tumor Growth, Cancer Cell 19:257-272.
- Spaeth EL, Dembinski JL, Sasser AK, Keri W, Ann K, et al. (2009) Mesenchymal stem cell transition to tumor-associated fibroblasts contributes to fibrovascular network expansion and tumor progression. PLoS One
- Hocking AM, Gibran NS (2010) Mesenchymal stem cells: Paracrine signaling and differentiation during cutaneous wound repair, Exp Cell Res 316:2213-2219.
- Wu Y, Zhao RCH, Tredget EE (2010) Concise review: Bone marrow-derived stem/progenitor cells in cutaneous repair and regeneration, Stem Cells 28:905-915.
- Mirotsou M, Jayawardena TM, Schmeckpeper J, Gnecchi M, Dzau VJ (2011) Paracrine mechanisms of stem cell reparative and regenerative actions in the heart, J Mol Cell Cardiol 50:280-289.
- Gnecchi M, Zhang Z, Ni A, Dzau VJ (2008) Paracrine mechanisms in adult stem cell signaling and therapy, Circ Res 103:1204-1219.
- Reya T, Duncan AW, Ailles L, Jos D, David CS, et al. (2003) A role for Wnt signalling in self-renewal of haematopoietic stem cells. Nature 423:409-414.
- Scheller M, Huelsken J, Rosenbauer F, Makoto M, Walter B et al. (2006) Hematopoietic stem cell and multilineage defects generated by constitutive beta-catenin activation, Nat Immunol 7:1037-1047.
- Combs MD, Yutzey KE (2009) Heart valve development: Regulatory networks in development and disease, Circ Res 105:408-421.
- Nigam V, Srivastava D (2009) Notch1 represses osteogenic pathways in aortic valve cells, J Mol Cell Cardiol 47:828-834.
- Chen JH, Chen WLK, Sider KL, Yip CYY, Simmons CA (2011) β-catenin mediates mechanically regulated, transforming growth factor-β1-induced myofibroblast differentiation of aortic valve interstitial cells, Arterioscler Thromb Vasc Biol 31:590-597.
- Cushing MC, Liao JT, Anseth KS (2005) Activation of valvular interstitial cells is mediated by transforming growth factor-?? Interactions with matrix molecules, Matrix Biol 24:428-437
- Walker G, Masters KS, Shah DN, Anseth KS, Leinwand L (2004) Valvular myofibroblast activation by transforming growth factor-β: Implications for pathological extracellular matrix remodeling in heart valve disease, Circ Res 95:253-260.
- Kheradvar A, Groves EM, Dasi LP, Alavi S, Tranquillo R, et al. (2015) Emerging Trends in Heart Valve Engineering: Part I. Solutions for Future, Ann Biomed Eng 43: 833-843.
- Cheung DY, Duan B, Butcher JT (2015) Current progress in tissue engineering of heart valves: multiscale problems, multiscale solutions, Expert Opin Biol Ther 15:1155-1172.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi