Research Article, J Clin Exp Oncol Vol: 7 Issue: 2
Prognostic Markers in Osteosarcoma Patients in the Najaf Province of Iraq
Hamdallah H Al-Baseesee1, Imad AlSabri2,4, Amina B Aldujele3 and Zuhair Allebban4*
1Department of Surgery, College of Medicine, Kufa University, Najaf, Iraq
2Middle Euphrates Cancer Therapy Center, Najaf, Iraq
3Department of Physiology, College of Medicine, Kufa University, Najaf, Iraq
4Middle Euphrates Unit for Cancer Research, College of Medicine, University of Kufa, Najaf, Iraq
*Corresponding Author : Zuhair Allebban
Middle Euphrates Unit for Cancer Research, College of Medicine/University of Kufa, P.O. Box 21 Najaf, Iraq
Tel: 009647804272223
E-mail: zuhair.allabban@uokufa.edu.iq
Received: February 10, 2018 Accepted: March 07, 2018 Published: March 14, 2018
Citation: Al-Baseesee HH, AlSabri I, Aldujele AB, Allebban Z (2018) Prognostic Markers in Osteosarcoma Patients in the Najaf Province of Iraq. J Clin Exp Oncol 7:2. doi: 10.4172/2324-9110.1000215
Abstract
Background: Among the solid malignancies of bone, osteosarcomas are the most common, but unfortunately there has not been solid new therapy for this disease. In this study, we emphasized on the prognostic markers of the disease and our goal was to present the clinical and demographic outcomes of osteosarcoma patients.
Materials and Methods: A total of 30 patients (17 male, 13 female) osteosarcoma patients treated and followed up in Middle Euphrates Cancer Center Therapy in Najaf, Iraq from 2016 to 2017 were reviewed prospectively and retrospectively. Serum alkaline phosphatase (ALP), lactate dehydrogenase (LDH) and bone-specific ALP (bsALP) were analyzed both before and after chemotherapy. Patients were also examined by magnetic resonance imaging (MRI).
Results: Seventeen male and thirteen female patients were diagnosed with metastatic (except one male and one female) osteosarcoma located at extremities. The patients showed an increase in the level of serum alkaline phosphatase (ALP), lactate dehydrogenase (LDH), bone specific ALP. A strong correlation was demonstrated between LDH and MRI results.
Conclusions: In this study, LDH and MRI were found to be the most important prognostic factors in osteosarcoma patients. An increase in serum level of ALP, LDH, and bone specific ALP was observed. A Significant correlation was indicated between LDH and MRI results. An increase in sample size and the use of more effective and active agents of chemotherapy, especially for metastatic cases are needed.
Keywords: Osteosarcoma; Prognostic marker; Alkaline phosphatase; Lactate dehydrogenase; Bone-specific ALP; MRI
Introduction
The origin of the osteosarcoma is the primitive mesenchymal cells of bone; it is rarely derived from soft tissue. It is considered the most common primary bone tumor in childhood and adolescence and the most common malignancy [1]. It often leads to fatal metastasis, usually in the lungs causing the highest rate of death in these age groups [2]. Its incidence is lower among older people (2-3 per 106) if compared to its incidence in age group 15-19 years where it is 8-11 per 106. In addition, the incidence in females is higher than in males with a ratio of 4:1 [3,4]. Distal femur, proximal tibia and proximal humerus are the origin of osteosarcomas in many patients and 10% of osteosarcoma developed in the axial skeleton [5]. Swelling and local pain with limitation of joint movement are common symptoms in osteosarcoma patients with some cases where pathological fracture is the first sign of disease.
MRI is a better indicator to determine soft tissue involvement in the whole bone, it is used in combination with plain radiography, and the latter assess the changes at the bone. In addition, a thorax computed tomography is also done to determine metastasis to the lungs. Lungs and to a lesser extent bones are the most common sites involved in metastasis and approximately 15% of patients end up with metastatic osteosarcoma [1]. Osteosarcoma patients are treated with chemotherapy and the most used chemotherapeutic agent are doxorubicin, cisplatin, methotrexate and ifosfamid [6]. Survival rates with these agents are 10-50%. Surgery is used in addition to chemotherapy in patients with limited lung metastasis [5].
Before the advent of chemotherapy, 80-90% of osteosarcoma patients ended up with metastatic cancer that led to their death [7,8]. Chance of survival is improved with the addition of surgical resection of tumors in metastatic patients although the prognosis in metastatic osteosarcoma remains poor. Biomarkers can be helpful in predicting osteosarcoma which in turn can improve patient’s survival. In this study, we used some of these markers such as LDH, ALP, bsALP and MRI to determine their value in predicting metastasis of the disease at the time of diagnosis.
Although the use of surgery along with chemotherapy have drastically improved the treatment outcomes for osteosarcoma patients; however, metastatic lesions and poor response to chemotherapy have been reported as important prognostic factors as well [9,10]. Histological analysis is used to assess the response to chemotherapy despite the difficulty of assessment before tumor excision. Although histological analysis after chemotherapy has a significant prognostic value in osteosarcoma patients, prognostic value of radiological examinations in the treatment of osteosarcoma has been reported in scant cases [11,12].
ALP belongs to a group of metalloenzymes with low substrate specificity that requires alkaline environment [13]. Tissue-nonspecific ALP (TNALP) gene expression takes place, among other places, in tissues as osteoblast, kidney, hepatocytes, and early placenta. Serum ALP originates mostly from hepatic tissues, bone, or kidney [14]. Serum ALP level increases in patients with osteosarcoma. There exist a relationship between serum ALP and clinical outcome in osteosarcoma patients [15], although that relationship has been shown to be different in different studies, so the role of serum ALP as a prognostic value in osteosarcoma patients has been shown to be inconsistent in these studies. In addition to that, normal values of serum ALP in children are usually higher in children [16] which make it difficult to regard serum ALP as a prognostic biomarker in patients with osteosarcoma.
Another variable factor to be considered is the stage of cancer. One study, using 816 osteosarcoma patients, has shown significant relationship between elevated serum ALP and metastasis [17-19]. Elevated serum ALP in 372 non-metastatic osteosarcoma patients showed no significant correlation with the spread of the disease [20-22]. To improve the prognosis of the cancer, there is still a need for better biomarkers to be implemented in aggressive treatment in patients with poor prognosis [23].
Materials and Methods
Thirty patients with osteosarcoma between the age of 24 to 60 years (average 41.7 years) for male, and 15-68 years of age (average 38 years) for female were treated at the Middle Euphrates Cancer Therapy Center in Najaf, Iraq.
The values of LDH, ALP, and bsALP were determined using from blood sample obtained from patients. Along with MRI, values were used to determine diagnosis and assess stage of cancer. Pathological assessment was done on samples obtained from biopsy and surgery. Cancer stage was determined and only patients treated with chemotherapy and surgery were included in the study while those patients that had low grade osteosarcoma or who did not undergo surgery were excluded from the this study.
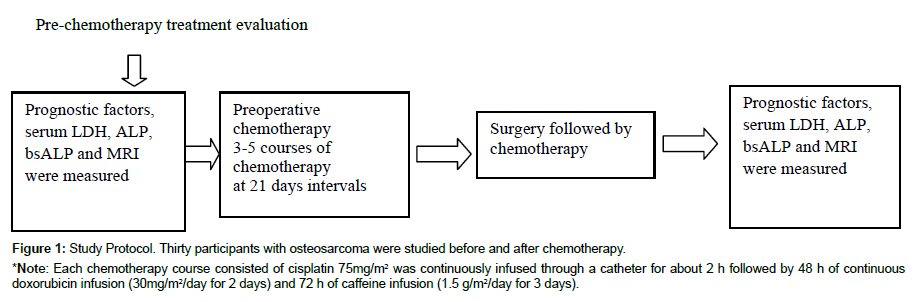
Pre-chemotherapy and post-chemotherapy values of serum ALP, LDH, bsALP, and MRI were performed on all patients. Patients received 3-5 courses of chemotherapy at interval of 21 days; surgery was performed after the culmination of chemotherapy. In first line of chemotherapy course, cisplatin 75 mg/m2 was infused for 1-2 h followed by doxorubicin infusion (30 mg/m2/day) for 2 days and 72 h of caffeine infusion (1.5 g/m2/day for 3 days). Non-metastatic patients received only first line of treatment while metastatic patients were followed with second line of treatment with ifosfamide 3 g/m2/5day, etoposide (100 mg/m2 for 3 days and mesna 3 gm/m2). To detect local recurrence and metastasis, a follow-up evaluation was performed consisted of MRI, serum LDH, ALP and bone-specific ALP.
Compliance with ethical standards
Consent was obtained from all patients and/or their families and the study was approved by the Institutional Review Board of college of medicine/Kufa university, Najaf, Iraq.
Magnetic resonance imaging
MRI sequences included a standard (spin-echo) T1-weighted sequence (repetition time [ms]/echo time [ms], 400-900/10-20), with or without gadolinium enhancement, and an intermediate weighted/ T2-weighted sequence (1,500-2,500/70-100), without fat suppression (Philips Ingenia 1.5T, Philips Healthcare, Best, Netherlands).
Intramedullary tumor lengths were measured in coronal sections of unenhanced T1-weighted sequences, and tumor widths and depths were measured in axial enhanced T1- and T2-weighted sequences without fat suppression [24]. Final decisions of MRI evaluation were made by the consensus of three reviewers whenever there was a 10% discrepancy in image readings.
Laboratory analysis
In all patients a chemistry panel and complete blood count tests were performed before the start of chemotherapy including ALP, LDH and bsALP. The normal ranges for ALP at our institution are defined according to gender and age: in males aged <12, 14-18 and >17 years the upper limit of the normal ranges are 304, 385 and 128 U/l, and in females are 310, 188 and 101 U/l, respectively.
Histological examination
Fresh tumor tissue samples were processed for histological reading according to standard procedure of fixation in 10% formalin, embedding in paraffin, sectioning at 5 μm thickness, xylene deparaffinization, and final rehydrating in an ethanol series. Hematoxylin and eosin (H &E) staining was performed according to standard protocols. Histological examination was performed with a BHS System Microscope (Olympus Corporation, Tokyo, Japan). Images were acquired with INFINITY ANALYZE software (Lumenera Corporation, Ottawa, Canada) [25].
Statistical analysis
Statistical analyses were performed using SPSS software version 21.0 (SPSS, Inc., Chicago, Illinois, USA). Correlation between the prognostic factors was assessed by Pearson’s correlation coefficient. The correlation between the prognostic factors, LDH, ALP, bonespecific ALP serum level and MRI was done using Pearson’s rank correlation coefficient. A P value of ≤ 0.05 was considered statistically significant.
Results
Regarding the study participants, the mean age of patients was 24-60 years for males and 15-68 years for female. Most patients (57%) were female, and 43% were male. The most frequent tumor location was the femur (53%), followed by the humerus (30%) and tibia (17%). Twenty-eight out of the thirty patients included in this study were found to be metastatic at the time of diagnosis. The majority of pathological type was osteoblastic followed by chondroblastic and 50% of the tumor was stage IV as shown in Table 1. The normal values of ALP and LDH, were established according to the reference values of our laboratory in our center (LDH range 26-440 U/L, ALP range 32-131 U/L). Analyses were performed by a Roche/Hitachi COBAS modular machine. All patients with ALP and LDH, bone-specific ALP values were above the normal range as shown in Table 2.
| Characteristic | No. of patients | % | Average Years |
Range Years |
Metastatic site |
|---|---|---|---|---|---|
| Age at diagnosis (y) Male Female |
30 13 17 |
100 | 41.7 38 |
24-60 15-68 |
Lung* Lung |
| Gender Male Female |
13 17 |
43 57 |
|||
| Primary tumor location Femur Tibia Humerus |
16 5 9 |
53 17 30 |
|||
| Pathologic subtype Osteoblastic Chondroblastic Fibroblastic |
18 10 2 |
60 33 7 |
|||
| Stage IIA IIB IIIB IV |
2 7 6 15 |
7 23 20 50 |
Table 1: Characteristics of study participants.
| Marker | N | Mean | SD |
|---|---|---|---|
| LDH (U/L) | 30 | 510.5 | 503.9 |
| ALP (U/L) | 30 | 144.3 | 103.8 |
| Bone specific ALP (mcg/L) | 30 | 0.4 | 0.2 |
| MRI | 30 | 9.8 | 8.9 |
Table 2: Descriptive Statistics, Measurement of the prognostic markers.
Pearson correlation analysis was used to any correlative changes between any two of the following prognostic factors: ALP, LDH, bsALP and MRI. The results are shown in Table 3.
| LDH | ALP | MRI | Bone specific ALP | ||
|---|---|---|---|---|---|
| LDH (U/L) | Pearson Correlation | 1 | -0.121 | -0.376(*) | -0.117 |
| Sig.(2-tailed) | 0.523 | 0.041 | 0.537 | ||
| N | 30 | 30 | 30 | 30 | |
| ALP (U/L) |
Pearson Correlation | -0.121 | 1 | -0.060 | -0.054 |
| Sig.(2-tailed) | 0.523 | 0.751 | 0.777 | ||
| N | 30 | 30 | 30 | 30 | |
| MRI | Pearson Correlation | - 0.376(*) | -0.060 | 1 | -0.125 |
| Sig.(2-tailed) | 0.041 | 0.751 | 0.511 | ||
| N | 30 | 30 | 30 | 30 | |
| Bone specific ALP (mcg/L) | Pearson Correlation | -0.117 | -0.054 | -0.125 | 1 |
| Sig.(2-tailed) | 0.537 | 0.777 | 0.511 | ||
| N | 30 | 30 | 30 | 30 |
Table 3: Pearson Correlation coefficient among four of the prognostic factors of osteosarcoma.
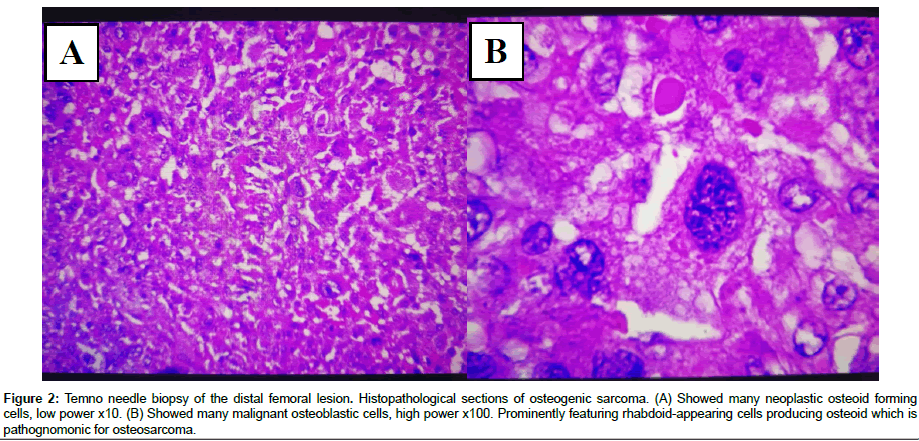
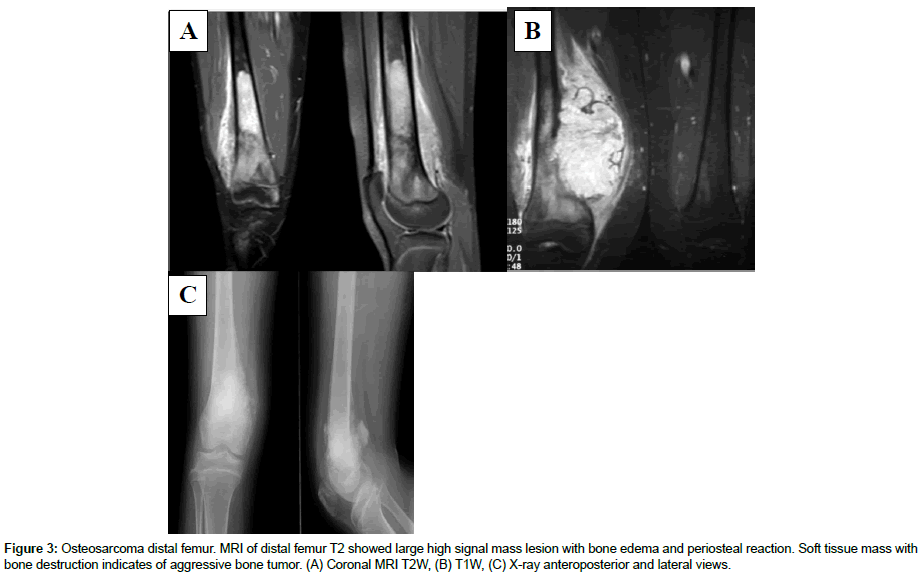
Histopathological examination was conducted on study participants. The results are shown in Figures 1 and 2. Additionally, MRI results are demonstrated in Figure 3.
Figure 1: Study Protocol. Thirty participants with osteosarcoma were studied before and after chemotherapy.
*Note: Each chemotherapy course consisted of cisplatin 75mg/m2 was continuously infused through a catheter for about 2 h followed by 48 h of continuous doxorubicin infusion (30mg/m2/day for 2 days) and 72 h of caffeine infusion (1.5 g/m2/day for 3 days).
Figure 2: Temno needle biopsy of the distal femoral lesion. Histopathological sections of osteogenic sarcoma. (A) Showed many neoplastic osteoid forming cells, low power x10. (B) Showed many malignant osteoblastic cells, high power x100. Prominently featuring rhabdoid-appearing cells producing osteoid which is pathognomonic for osteosarcoma.
Discussion
Lack of homogeneity in analysis and methods in previous studies is to blame for the contradictory conclusions reached by different studies. Histological subtype, gender, age, genetic variations, elevated values of serum AP or LDH, bsALP, and MRI are all markers that have been used to assess the prognosis of osteosarcoma patient but with variable results.
Among these potential markers, histological response, presence of metastasis, was found to be important prognostic factors [26]. In this study, we found the rate of clinically detectable metastatic disease at initial presentation is significantly higher compared to expected rates in developed countriesa (10-20%) [27-29], or when the results of our study compared to studies done in Brazil 20.8% [30-32] and in South Africa 49-66% [33,34]. The differences in these studies probably due to delay in diagnosis and advanced disease at the moment of diagnosis, and possibly the first cause of mortality among these patients, and considered one of the significant prognostic factors of this disease [30,35].
Also, age was found to be another significant factor in outcome of disease. Prognosis was found to be poor in patients with age above 40 years [36], our study showed the same result. European and American studies have shown the gender not to be a significant prognostic factor [37,38] although Brazilian [31] and Scandinavian [39] studies concluded that females having a better prognosis, the later findings were not observed in our study.
In addition to gender not being significant factor in survival rate, we found the histological subtype also to be as such, as previously reported [40].
Our result confirmed the previous result in terms of ALP having a prognostic value. Our current study represents the first in its kind about this cancer in our country, although its limitation is in the small number of osteosarcoma patients. More expanded studies are needed in the future [41,42].
Previous studies that showed metastasis to bones indicates a worse prognosis than metastasis to the lungs [43]. In this study, we aimed to evaluate if the pretreatment value of serum ALP, LDH and bsALP could be used as specific markers in high grade metastatic osteosarcoma of the extremities to determine the presence of skeletal and pulmonary metastases. Our results is similar to previously reported results [43] but disagrees with other reported results that showed no value in these parameters in predicting metastasis to the lungs [43].
Previous study showed that LDH can be used to reflect systemic malignancies and its significance has been illustrated in various types of cancers including Ewing’s sarcoma [43]. BsALP has been suggested to be a reliable prognostic tumor marker of osteosarcoma [44].
Although elevation of LDH predicted metastasis with high specificity, but it did not differentiate between metastasis to bones or to lungs [43], and this is similar to our result in which all the skeletal osteosarcoma patients had pulmonary metastasis.
ALP, LDH, and bsALP did not show distinction between skeletal and pulmonary metastasis in previous study. To the best of our knowledge, our study is the first of its kind in Iraq to use the elevation of ALP and LDH as specific predictive markers for metastatic skeletal osteosarcoma. Han et al found that serum ALP elevation to more than twice the upper limit of normal value in patients that had pulmonary metastases [45]. Rizolli et al found similar results [46].
The original goal of this study was to determine the predictive value of ALP, LDH and bsALP as markers in determining the disease outcome and survival, but few shortcomings such as small sample size inhibits us from accomplishing the goal of the study. Several patients were lost to the follow up and therefore the long term survival could not be determined.
The incidence of metastases was higher than those reported by Marais et al (80%) and much higher than those reported by Bacci et al. [46]. These outcomes could be due to either more advanced disease stage, both locally and systemically or its aggressiveness at the time of diagnosis.
MRI should be used to detect skeletal osteosarcoma at the initial stage before treatment and during the follow-up. All imaging findings were reviewed by three experienced musculoskeletal radiologists who had image diagnostic experience and high knowledge about this tumor.
In this study, we found serum ALP provides important information in determining prognosis in osteosarcoma patients and associated with the presence of metastasis. Since this test is simple and cost-effective, it is one of the routine fast diagnostic tests in clinical laboratories.
Following treatment, serum LDH levels almost uniformly returned to normal and no correlation between postoperative levels and relapse of disease could be identified. As another outcome of this study, we believe serum LDH level is a reliable marker to determine prognosis in patients with osteosarcoma of the extremities and that it can be used to determine the efficacy of different therapeutic protocols.
Conclusion
In our study, we find that LDH and MRI are the most important prognostic markers for osteosarcoma. An increase in serum level of ALP, LDH, bsALP were observed in all patients compared to normal reference values in our institution. A significant correlation was observed between LDH and MRI results. The uses of more effective and active agents of chemotherapy, especially for metastatic cases are needed. Sample size should be increased. More specific clinical trials are needed for further exploration of these serum biomarkers for use in clinical monitoring of osteosarcoma patients.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Author Contributions
HHA, IA, and ABA actively contributed to collect data and statistical process.HHA, ABA, and ZA have reviewed and made corrections to the final version of the manuscript. All authors made important contributions to the conception, design and analysis of the work, critical revision, and a final approval for publication.
References
- Ritter J, Bielack SS (2010) Osteosarcoma. Ann Oncol 21 Suppl 7: vii320-325.
- Ta HT, Dass CR, Choong PF, Dunstan DE (2009) Osteosarcoma treatment: state of the art. Cancer Metastasis Rev 28: 247-263.
- Stiller CA, Passmore SJ, Kroll ME, Brownbill PA, Wallis JC, et al. (2006) Patterns of care and survival for patients aged under 40 years with bone sarcoma in Britain, 1980-1994. Br J Cancer 94: 22-29.
- Qureshi A, Ahmad Z, Azam M, Idrees R (2010) Epidemiological data for common bone sarcomas. Asian Pac J Cancer Prev 11: 393-395.
- Bielack SS, Kempf-Bielack B, Delling G, Exner GU, Flege S, et al. (2002) Prognostic factors in high-grade osteosarcoma of the extremities or trunk: an analysis of 1,702 patients treated on neoadjuvant cooperative osteosarcoma study group protocols. J Clin Oncol 20(3):776-790.
- Kager L, Zoubek A, Pötschger U, Kastner U, Flege S, et al. (2003) Primary metastatic osteosarcoma: presentation and outcome of patients treated on neoadjuvant Cooperative Osteosarcoma Study Group protocols. J Clin Oncol 21: 2011-2018.
- Anninga JK, Gelderblom H, Fiocco M, Kroep JR, Taminiau AH, et al. (2011) Chemotherapeutic adjuvant treatment for osteosarcoma: where do we stand? Eur J Cancer 47: 2431-2445.
- Dahlin DC, Unni KK (1977) Osteosarcoma of bone and its important recognizable varieties. Am J Surg Pathol 1: 61-72.
- Davis AM, Bell RS, Goodwin PJ (1994) Prognostic factors in osteosarcoma: a critical review. J Clin Oncol 12: 423-431.
- Bielack SS, Kempf-Bielack B, Delling G, Exner GU, Flege S, et al. (2002) Prognostic factors in high-grade osteosarcoma of the extremities or trunk: an analysis of 1,702 patients treated on neoadjuvant cooperative osteosarcoma study group protocols. J Clin Oncol 20: 776-790.
- Miwa S, Shirai T, Taki J, Sumiya H, Nishida H, et al. (2011) Use of 99mTc-MIBI scintigraphy in the evaluation of the response to chemotherapy for osteosarcoma: comparison with 201Tl scintigraphy and angiography. Int J Clin Oncol 16: 373-378.
- Miwa S, Shirai T, Taki J, Sumiya H, Nishida H, et al. (2011) Use of 99mTc-MIBI scintigraphy in the evaluation of the response to chemotherapy for osteosarcoma: comparison with 201Tl scintigraphy and angiography. Int J Clin Oncol 16: 373-378.
- Kaplan MM (1972) Alkaline phosphatase. N Engl J Med 286: 200-202.
- Cho SR1, Lim YA, Lee WG (2005) Unusually high alkaline phosphatase due to intestinal isoenzyme in a healthy adult. Clin Chem Lab Med 43: 1274-1275.
- Mckenna Rj, Schwinn Cp, Soong Ky, Higinbotham N (1964) Osteogenic Sarcoma Arising In Paget's Disease. Cancer 17: 42-66.
- Rauchenzauner M, Schmid A, Heinz-Erian P, Kapelari K, Falkensammer G, et al. (2007) Sex- and age-specific reference curves for serum markers of bone turnover in healthy children from 2 months to 18 years. J Clin Endocrinol Metab 92: 443-449.
- Wu PK, Chen WM, Chen CF, Lee OK, Haung CK, et al. (2009) Primary osteogenic sarcoma with pulmonary metastasis: clinical results and prognostic factors in 91 patients. Jpn J Clin Oncol 39: 514-522.
- Han J, Yong B, Luo C, Tan P, Peng T, et al. (2012) High serum alkaline phosphatase cooperating with MMP-9 predicts metastasis and poor prognosis in patients with primary osteosarcoma in Southern China. World J Surg Oncol 10: 37.
- Hung GY, Yen HJ, Yen CC, Chen WM, Chen PC, et al. (2015) Experience of pediatric osteosarcoma of the extremity at a single institution in Taiwan: prognostic factors and impact on survival. Ann Surg Oncol 22: 1080-1087.
- Bacci G, Picci P, Ferrari S, Orlandi M, Ruggieri P, et al. (1993) Prognostic significance of serum alkaline phosphatase measurements in patients with osteosarcoma treated with adjuvant or neoadjuvant chemotherapy. Cancer 71: 1224-1230.
- Smeland S, Müller C, Alvegard TA, Wiklund T, Wiebe T, et al. (2003) Scandinavian Sarcoma Group Osteosarcoma Study SSG VIII: prognostic factors for outcome and the role of replacement salvage chemotherapy for poor histological responders. Eur J Cancer 39: 488-494.
- Kim SH, Shin KH, Kim HY, Cho YJ, Noh JK, et al. (2014) Postoperative nomogram to predict the probability of metastasis in Enneking stage IIB extremity osteosarcoma. BMC Cancer 14: 666.
- Bielack S, Carrle D, Casali PG; ESMO Guidelines Working Group (2009) Osteosarcoma: ESMO clinical recommendations for diagnosis, treatment and follow-up. Ann Oncol 20 Suppl 4:137-139.
- Kim MS, Lee SY, Cho WH, Song WS, Koh JS, et al. (2008) Tumor necrosis rate adjusted by tumor volume change is a better predictor of survival of localized osteosarcoma patients. Ann Surg Oncol 15: 906-914.
- Murakami T, DeLong J, Eilber FC, et al. (2016) Tumor-targeting Salmonella typhimurium A1-R in combination with doxorubicin eradicate soft tissue sarcoma in a patient-derived orthotopic xenograft (PDOX) model. Oncotarget 7: 12783-12790.
- Bramer JA, van Linge JH, Grimer RJ, Scholten RJ (2009) Prognostic factors in localized extremity osteosarcoma: a systematic review. Eur J Surg Oncol 35: 1030-1036.
- Ozger H, Eralp L, Atalar AC, Toker B, Ayan I, et al. (2007) Survival analysis and the effects of prognostic factors in patients treated for osteosarcoma. Acta Orthop Traumatol Turc 41: 211-219.
- Philip T, Iliescu C, Demaille MC, Pacquement H, Gentet JC, et al. (1999) High-dose methotrexate and HELP [Holoxan (ifosfamide), eldesine (vindesine), platinum]--doxorubicin in non-metastatic osteosarcoma of the extremity: a French multicentre pilot study. Fédération Nationale des Centres de Lutte contre le Cancer and Société Française d'Oncologie Pédiatrique. Ann Oncol 10: 1065-1071.
- Provisor AJ, Ettinger LJ, Nachman JB, Krailo MD, Makley JT, et al. (1997) Treatment of nonmetastatic osteosarcoma of the extremity with preoperative and postoperative chemotherapy: a report from the Children's Cancer Group. J Clin Oncol 15: 76-84.
- Petrilli AS, de Camargo B, Filho VO, Bruniera P, Brunetto AL, et al. (2006) Results of the Brazilian Osteosarcoma Treatment Group Studies III and IV: prognostic factors and impact on survival. J Clin Oncol 24: 1161-1168.
- Petrilli AS, Gentil FC, Epelman S, Lopes LF, Bianchi A, et al. (1991) Increased survival, limb preservation, and prognostic factors for osteosarcoma. Cancer 68: 733-737.
- Petrilli S, Penna V, Lopes A, Figueiredo MT, Gentil FC (1991) IIB osteosarcoma.Current management, local control, and survival statistics – Sao Paulo, Brazil. Clin Orthop Relat Res 270: 60-66.
- Ferreira N, Marais LC (2012) Osteosarcoma presentation stages at a tumour unit in South Africa. S Afr Med J 102: 673-676.
- Shipley JA, Beukes CA (2012) Outcomes of osteosarcoma in a tertiary hospital. S Afr J Orthop 11:18-22.
- Meyers PA, Gorlick R, Heller G, Casper E, Lane J, et al. (1998) Intensification of preoperative chemotherapy for osteogenic sarcoma: results of the Memorial Sloan-Kettering (T12) protocol. J Clin Oncol 16: 2452-2458.
- Berner K, Hall KS, Monge OR, Weedon-Fekjær H, Zaikova O, et al. (2015) Prognostic factors and treatment results of high-grade osteosarcoma in norway: a scope beyond the "classical" patient. Sarcoma 2015: 516843.
- Fuchs N, Bielack SS, Epler D, Bieling P, Delling G, et al. (1998) Long-term results of the co-operative German-Austrian-Swiss osteosarcoma study group's protocol COSS-86 of intensive multidrug chemotherapy and surgery for osteosarcoma of the limbs. Ann Oncol 9: 893-899.
- Meyers PA, Heller G, Healey J, Huvos A, Lane J, et al. (1992) Chemotherapy for nonmetastatic osteogenic sarcoma: the Memorial Sloan-Kettering experience. J Clin Oncol 10: 5-15.
- Saeter G, Wiebe T, Wiklund T, Monge O, Wahlqvist Y, et al. (1999) Chemotherapy in osteosarcoma. The Scandinavian Sarcoma Group experience. Acta Orthop Scand Suppl 285: 74-82.
- Taylor WF, Ivins JC, Unni KK, Beabout JW, Golenzer HJ, et al. (1989) Prognostic variables in osteosarcoma: a multi-institutional study. J Natl Cancer Inst 81: 21-30.
- Mavrogenis AF, Abati CN, Romagnoli C, Ruggieri P (2012) Similar survival but better function for patients after limb salvage versus amputation for distal tibia osteosarcoma. Clin Orthop Relat Res 470: 1735-1748.
- Bacci G, Longhi A, Bertoni F, Briccoli A, Versari M, et al. (2006) Bone metastases in osteosarcoma patients treated with neoadjuvant or adjuvant chemotherapy: the Rizzoli experience in 52 patients. Acta Orthop 77: 938-943.
- Marais LC, Bertie J, Rodseth R, Sartorius B, Farreira B (2014) Pre-treatmentserum lactate dehydrogenase and alkaline phosphatase as predictors of metastases in extremities osteosarcoma. J Bone Oncol 4: 80-84.
- Lui PPI, Leung KS, Kumta SM (1996) Bone specific alkaline phosphatase in plasma a tumour marker for osteosarcoma. Oncology 53: 275-283.
- Han J, Yong B, Lou C, Tan P, Teng B, et al. (2012) High serum Alkaline Phosphatase cooperating with MMP-9 predicts metastases and poor prognosis in patients with primary osteosarcoma in Southern China. World J Surg Oncol 10: 37-46.
- Bacci G, Picci P, Ferrari S, Orlandi M, Ruggieri P, et al. (1993) Patients with osteosarcoma treated with adjuvant or neoadjuvant chemotherapy. Cancer 71: 1224-1230.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi