Research Article, J Spine Neurosurg Vol: 8 Issue: 1
Spontaneous Spinal Epidural Haematomas in Adults: A Systematic Review
Soltani S1, Nogaro MC1, Haleem S1, Rupani N1, Pyrovolou N1 and Kieser DC2*
1Oxford University NHS Foundation Trust, Division of Spinal Surgery, Oxford, England
2Department of Orthopaedics and Musculoskeletal Medicine, University of Otago, Christchurch School of Medicine, Christchurch, New Zealand
*Corresponding Author : David Kieser
Department of Orthopaedics and Musculoskeletal Medicine, University of Otago, Christchurch School of Medicine, Christchurch, New Zealand
Tel: +64 211499829
E-mail: kieserdavid@gmail.com
Received: December 26, 2018 Accepted: January 03, 2019 Published: January 10, 2019
Citation: Soltani S, Nogaro MC, Haleem S, Rupani N, Pyrovolou N (2019) Spontaneous Spinal Epidural Haematomas in Adults: A Systematic Review. J Spine Neurosurg 8:1. doi: 10.4172/2325-9701.1000313
Abstract
Study design: Systematic review Objectives: Understand the typical presentation, risk factors, location, size, treatment and outcome of spontaneous spinal epidural haematomas (SSEH). Methods: Systematic review of the English literature from 1 January 1960 to 1 March 2018. Individual patient data was extracted and collated. Outcome measures were mode of presentation, risk factors, initial neurological findings, presumed diagnosis, diagnostic investigations, site and size of the SSEH, treatment, neurological recovery and survival. Results: 160 publications (254 patients) were reviewed. Most patients (98%) presented with back pain and radicular pain. Pregnancy was common in those aged under 40years (24%), whereas anticoagulation therapy (30%) and hypertension (30%) was common in those aged 50-80years. Most patients present with neurological dysfunction. 15% were initially suspected of having an alternative diagnosis, including 7.5% who were suspected of ischaemia for which anti-coagulation therapy was either planned or given. MRI and/or CT myelogram was performed in 98% of cases. The mid-cervical spine and cervico-thoracic junction were most commonly affected and the average haematoma size extended across 5.4 vertebral levels. Most patients underwent surgical decompression (76%) and most improved their neurological function (85%). Conclusions: SSEH affects all age groups and presents with variable neurological signs sometimes mimicking cerebral or cardiac ischaemia. Cross-sectional spinal imaging with MRI or CT is diagnostic and avoids inappropriate anticoagulation therapy. Most SSEHs are located in the mid-cervical spine and cervico-thoracic junction and affect multiple spinal levels. Surgical intervention is not always indicated, but with appropriate management most patients will improve or resolve their neurological deficit.
Keywords: Spinal epidural haematomas; Neurological dysfunction; Anticoagulation therapy; Midcervical spine
Introduction
Spinal epidural haematomas (SEH) occur from extra-dural bleeding, most commonly after trauma, epidural anaesthesia or operative intervention [1,2]. These haematomas are rare but carry a significant risk of spinal cord or nerve root compression potentially resulting in permanent neurological dysfunction [3,4]. Although the management of epidural haematomas is debated, most centres would treat a compressive spinal epidural haematoma causing neurological dysfunction with surgical decompression and evacuation of the haematoma.
Spontaneous spinal epidural haematomas (SSEH) occur spontaneously, without an obvious inciting event, and often mimic other conditions such as cerebro-vascular accidents [5,6]. These are rare with a quoted incidence of approximately 0.1 cases per 100 000 population annually [7-9]. This makes them a difficult condition to diagnose and may result in delayed diagnosis and potentially mismanagement with anticoagulation therapy [6,9,10].
The consequence of a SSEH can be devastating with progressive neurological decline resulting in permanent spinal cord injury and death [3,11,12]. Thus, early and accurate diagnosis and management is paramount in order to preserve neurological function and optimise the chance of recovery [13].
This study reviews the literature on the presentation, diagnosis and treatment of SSEH to provide an overview for clinicians treating these patients and to ensure early and accurate treatment for this time-dependent condition.
Methods
We performed a comprehensive search of all publications between 1 January 1960 and 1 March 2018 using PubMed, Medline, Embase, Scopus, QxMD, Google Scholar and the Cochrane Library. The MeSH term ‘epidural haematoma’ and keywords (spine, spinal and spontaneous) connected by the Boolean operators AND and OR were used to identify all possible studies. This was followed by an analysis of the text words contained in the title and abstract, and of the index terms used to describe articles. Then a manual search was conducted of the reference lists of identified articles and relevant reviews for additional studies.
Our inclusion criteria were: All studies written in English reporting on SSEH in humans aged 18 years or older. Our exclusion criteria were: Review articles, animal studies and cases with recognised causes (traumatic or iatrogenic) for epidural haematoma. We did not exclude articles based on cohort size or length of follow-up. We defined a ‘recognised causes of epidural haematoma’ as preceding trauma, spinal procedures and injections.
The outcomes of interest were: mode of presentation, risk factors (specifically bleeding disorders, hypertension, cardio-vascular disease, diabetes and anti-coagulation), initial neurological findings, and initial presumed diagnosis, diagnostic investigations, site and size of the SSEH, treatment, neurological recovery and survival.
The key papers identified were reviewed independently by two of the authors (SS and MN) with the data from each paper collated to answer our selected outcome measures. Variance between the authors was checked and corrected by DK.
Because of the paucity of comparative trials on the management of SSEH and most studies (126) being case reports, our data entry involved each case being individually entered on an excel sheet. Thus, this was a study of individual patients rather than grouped studies. The outcomes of interest are therefore presented as patient means with associated ranges.
Patient age, sex and diagnostic investigation were described in all cases, but some studies failed to report the degree of neurological dysfunction, the location and size of the haematoma, the time to surgical intervention and the follow-up. In these situations, we excluded their results for the specific outcome of interest. Equally, some articles failed to accurately define the change in neurological status preventing the determination of an American Spinal Injury Association (ASIA) score, however they did state whether neurological improvement occurred. In these cases, we independently present an ASIA score, for those in which this was discernable, and a neurological outcome defined as worsened, unchanged, partial or complete recovery.
Statistical analysis
The outcomes of interest are presented as patient means with associated ranges or standard deviations. Comparison between the average pre- and post-treatment neurological score was performed with a two-tailed homoscedastic t test. To compare patients with neurological recovery (complete or partial) to those without neurological recovery (unchanged or worsened) we used an ANOVA for age, number of levels and time to operation, and Chi-square tests for gender, location and operative versus non-operative treatment.
Statistical significance was defined as a p value less than 0.05.
Results
The initial literature search identified a total of 936 articles. On reviewing the abstracts, 306 publications were initially identified that fulfilled our search criteria. No further relevant studies were identified from the references of the included studies (Figure 1).
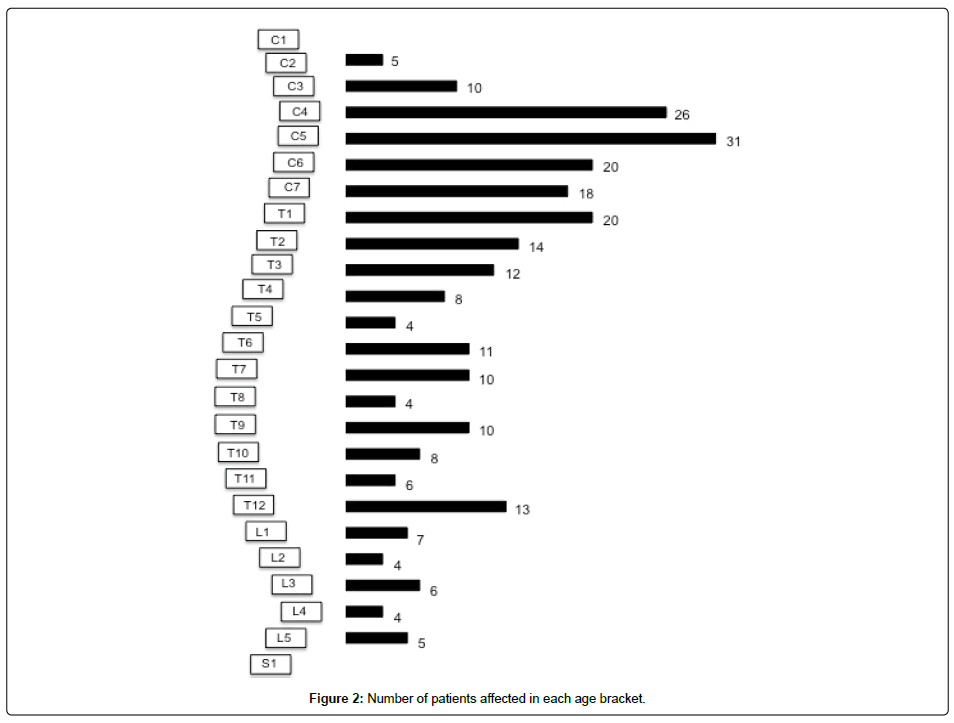
After exclusion criteria, a total of 160 publications and 254 patients were included in this review. The level of evidence ranged from IV to V. The average number of patients per publication was 3.8 (range 1-15 patients) and age at operation was 56.4 years (range 18-93 years) (Figure 2).
Male patients numbered 133 and female patients 121. The average follow-up was 158 days (range 2 days to 4 years). The large variance related to most publications ceasing follow-up after full neurological recovery was obtained.
Mode of presentation
Not all studies accurately described the patients’ initial presentation, but 98.4% (124/126) of patients complained of back pain and 97.5% (39/40) experienced radicular pain.
Risk factors
Although not all articles described the individual patient risk factors, seven patients had bleeding tendencies including Haemophilia, Von Willibrands disease and factor X deficiency. Forty-nine patients had hypertension, eight had diabetes, six were obese, 13 had atrial fibrillation or flutter and 22 had cardiovascular disease. Fifty-seven patients were on anticoagulation therapy, most frequently warfarin (43.9%), aspirin (36.8%) or clopidogrel (10.5%). Three patients had recently received thrombolysis therapy for vascular ischaemia, one patient had recently had surgery (coronary artery bypass grafting) and one had an endovascular stenting procedure. Fifteen patients were either pregnant or within two weeks of delivery.
Pregnancy accounted for a large proportion of those aged below 40 years (24.2%). In contrast, of those aged between 50 and 80 years, 30.0% were on anticoagulation and 30.0% were hypertensive. Bleeding disorders were rarely reported causes of SSEH and did not appear to be age specific (1.7% of those aged less than 40 years and 2.0% of those aged 50-80 years).
Initial neurological findings
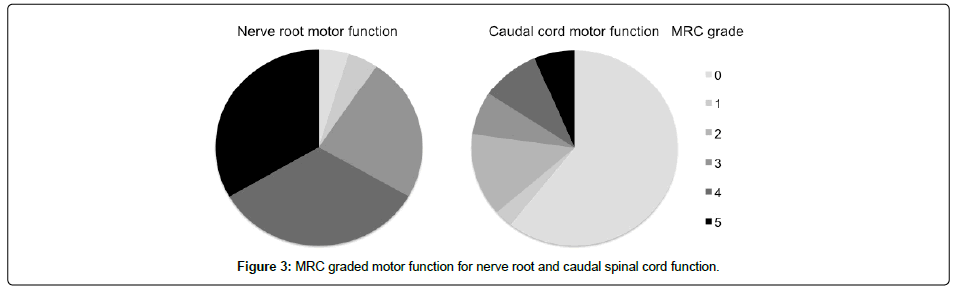
Twenty-two patients with nerve root compression and 148 with spinal cord compression had their motor function defined according to the medical research council (MRC) grading system. In contrast 148 patients with spinal cord compression had their distal motor function defined (Figure 3).
In those with nerve root compression, 62.5% (10/16) were described as having normal nerve root sensation, 25.0% (4/16) had reduced sensation and 12.5% (2/16) had abscent sensation. The caudal sensation was poorly described in those with spinal cord compression. However, 55.8% (29/52) had reduced distal reflexes, 7.7% (4/52) had normal reflexes and 36.5% (19/52) had increased reflexes.
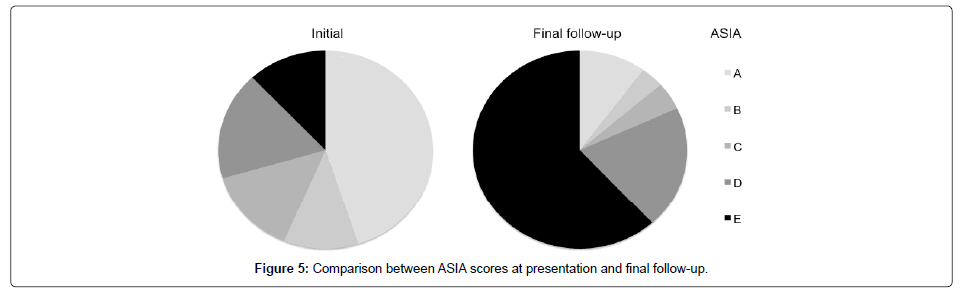
In those with spinal cord dysfunction, 45.1% (64/142) were ASIA A, 11.3% (16/142) were ASIA B, 14.1% (20/142) were ASIA C, 17.6% (25/142) were ASIA D and 12.0% (17/142) were ASIA E (Figure 5).
Initial presumed diagnosis
Thirty-nine patients (15.4%) were initially suspected of having an alternative diagnosis, including 16 suspected of having an ischaemic cerebral infarction, three intracranial haemorrhage, three coronary ischaemia, three spinal tumours and two disc prolapses. In total, 19 patients (7.5%) were diagnosed with an ischaemic incident for whom anticoagulation was either considered or administered.
Investigations
An MRI was conducted on 81.1% (202/249) of patients, 12.0% (30/249) had a CT myelogram, 2.0% (5/249) had a non-contrast CT scan and 4.8% (12/249) had a CT and MRI scan.
Location and size of SSEH
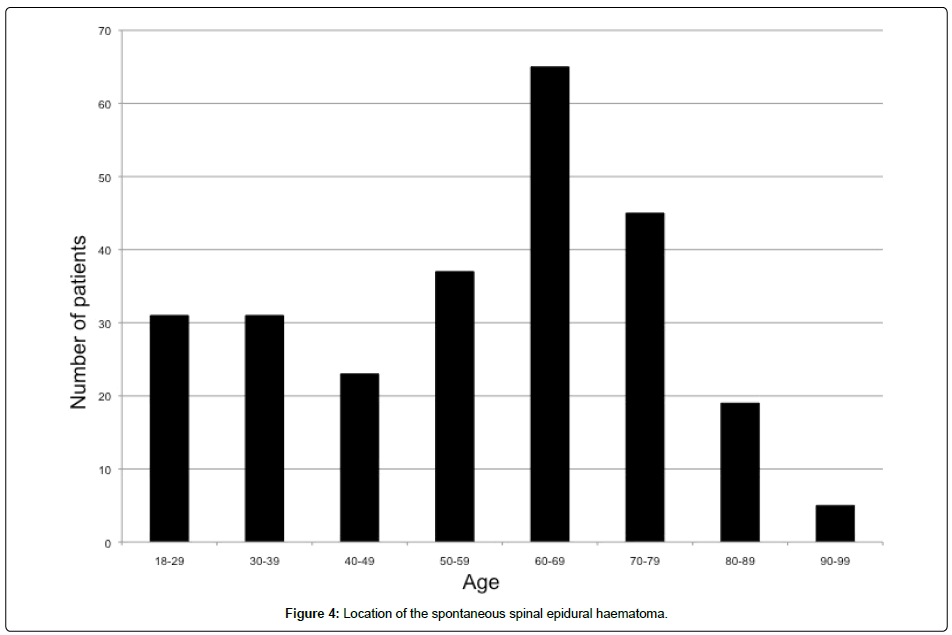
Two patients had two separate haematomas; the remainder had a single haematoma. The location of the centre of the SSEH is shown in figure 4.
The average size of the haematoma extended across 5.4 levels (range 1-28 levels).
Treatment
A total of 76.2% (192/252) of patients were treated with surgical decompression and 23.8% (60/252) were treated without surgical decompression. The average time from the beginning of symptoms until surgical decompression was 73.8 hours (range 3 hours - 6 months).
Neurological recovery
A total of 47.1% (113/240) of patients were described as having a complete neurological recovery, 37.9% (91/240) had a partial recovery, 10.0% (24/240) remained unchanged and 5.0% (12/240) worsened. In those patients obtaining a full neurological recovery, the average time until recovery was 52.4 days (range 1 day - 2 years).
At final follow-up 10.1% (20/199) of patients were ASIA A, 3.5% (7/199) ASIA B, 4.5% (9/199) ASIA C, 20.1% (40/199) ASIA D, 61.8% (123/199) ASIA E (Figure 5).
Table 1 compares the patient demographics and treatment between patients on outcome. There were no significant differences between those with and without neurological recovery according to age (p=0.251), sex (p=0.366), haematoma size (p=0.392) or location (p=0.264). There was also no significant difference between those that underwent surgical decompression and those that did not (p=0.395). Furthermore, there was no significant difference related to the time to decompression in those undergoing operative intervention (p=0.686).
| Number of patients | % of patients | Age | % male | % decompressed | Mortality rate | Location | Size | Time to operation | |
|---|---|---|---|---|---|---|---|---|---|
| Complete | 113 | 47.1 | 53.5 (18.3) | 55.8 | 66.1 | 0 | 55C, 44T, 10L | 5.1 (4.1) | 109.3 (616.9) |
| Partial | 91 | 37.9 | 58.5 (18.6) | 52 | 86.8 | 3.3 | 34C, 46T, 7L | 5.7 (3.9) | 59.7 (194.6) |
| Unchanged | 24 | 10.0 | 58.6 (19.5) | 42 | 83.3 | 8.3 | 5C, 13T, 3L | 5.4 (2.6) | 45.3 (41.7) |
| Worse | 12 | 5.0 | 61.4 (15.3) | 50 | 83.3 | 33.3 | 5C, 6T, 1L | 7.2 (4.7) | 45 (66.0) |
| Total | 240 | 56.5 | 52.0 | 76.4 | 3.7 | 99C, 109T, 21L | 5.5 | 74.2 |
Table 1: Comparison of patient demographics and treatment between outcome groups.
The effect on location is shown in Table 2. Overall the rate of neurological recovery (complete or partial) was greater in the cervical spine (89.9%) than the thoracic (81.5%) or lumbar spine (80.9%), but this was not statistically significant (p=0.264).
| Cervical | Thoracic | Lumbar | |
|---|---|---|---|
| Complete | 55 (55.6%) | 44 (42.7%) | 10 (47.6%) |
| Partial | 34 (34.3%) | 40 (38.8%) | 7 (33.3%) |
| Unchanged | 5 (5.1%) | 13 (12.6%) | 3 (14.3%) |
| Worsened | 5 (5.1%) | 6 (5.8%) | 1 (4.8%) |
Table 2: The rate of neurological recovery related to the haematoma site.
Survival
Nine patients were reported to have died within three months of their presentation. This included three patients who suffered a myocardial infarction, one with a cardiac arrest, one with extensive pneumonia and one with a large bowel haemorrhage. The cause of death in the remainder was not defined. Eight of these patients were either ASIA A or B on presentation. One patient was ASIA D. All affected the cervical or thoracic spine and ranged in size from 2-20 vertebral levels.
Discussion
SSEHs are rare but serious conditions that may result in permanent neurological dysfunction and/or death [3,4]. This condition affects all adult age groups, but predominantly those aged between 50 and 80 years. Pregnancy attributes a significant risk in those aged below 40 years (24% of patients), whereas anti-coagulation therapy and hypertension are more commonly associated with those aged between 50 and 80 years (30% respectively). Interestingly, bleeding disorders are rarely associated with SSEH and are not more frequent in a younger patient population (1.6% of those aged less than 40 years and 1.9% of those aged 50-80 years).
While most patients present with back pain and radiculopathy, the initial diagnosis can be difficult to determine with an estimated 15% being suspected of an alternative diagnosis including 7.5% with ischaemia and the potential for anti-coagulation therapy. However, cross-sectional imaging is diagnostic and predominantly performed with an MRI or CT myelogram. The expected MRI features of this condition are an intracanal, epidural lesion with variable T1 signal intensity, heterogenous hyperintense T2 signal with hypo-intense foci, compression of epidural fat, subarachnoid sac and spinal cord [14]. In contrast, the CT features are of a hyperdense (50-70 HU) extradural mass.
Most lesions affect the mid-cervical spine or cervico-thoracic junction and on average affect more than five spinal segments. The higher prevalence of cervical and cervico-thoracic lesions may be due to this area being the only area of the spinal canal with a compact continuous venous network in the epidural space, although this remains unproven [15].
SSEHs can affect a local nerve root and/or the spinal cord. In those with nerve root compression, most (70%) retain MRC grade 4 or 5 power and normal sensation (63%). In contrast, in those with spinal cord compression, most present with profound spinal cord dysfunction (56% ASIA A/B), caudal motor deficits (58% MRC grade 0) and reduced distal reflexes (56%).
In this review surgical decompression and the time to surgical decompression was not associated with an improved outcome. However, this must be interpreted with caution because most articles describing non-operative treatment were in patients with improving neurology. The authors feel that expeditious surgical decompression should therefore remain as the treatment of choice in patients with severe or worsening neurology. Further research is necessary to confirm this opinion.
In this review, the outcome of SSEHs appeared favourable with 85% of patients improving their neurological dysfunction. The likely reason why some patients fail to improve is permanent spinal cord damage caused by direct compression or vascular compression causing spinal cord ischaemia. The reason for worsened neurology despite treatment is likely further spinal cord compression, progressive spinal cord oedema or ischaemic penumdra [16]. Younger patients and those affecting the cervical spine were more likely to have neurological recovery, but this was not statistically significant. Of interest, the size of the SSEH does not predict the degree of neurological recovery.
This study has multiple limitations that should be recognised. Firstly, this is a review of the literature and is therefore subject to the limitations of the articles included. This includes reporting bias and variable completeness of the data provided. Secondly, with no randomised controlled trials comparing operative and non-operative care, a conclusion on the optimal treatment cannot be provided by this study. Lastly, SSEHs can affect different anatomical locations, with variable effects on patient’s symptoms and signs. Thus, a conclusive treatment algorithm for each presentation cannot be determined on the data provided.
In conclusion, SSEHs affect all age groups and present with variable neurological signs sometimes mimicking cerebral or cardiac ischaemia. Cross-sectional spinal imaging is diagnostic and avoids inappropriate anticoagulation therapy. Most SSEHs are located in the mid-cervical spine and cervico-thoracic junction and affect multiple spinal levels. Surgical intervention is not always indicated, but with appropriate management most patients will improve or resolve their neurological deficit.
Data archiving
An appendix is supplied of the reviewed articles
Acknowledgments
Glynny Kieser for her editorial input
References
- Awad JN, Kebaish KM, Donigan J, Cohen DB, Kostuik JP (2005) Analysis of the risk factors for the development of post-operative spinal epidural haematoma. J Bone Joint Surg Br 87: 1248-1252.
- Wulf H (1996) Epidural anaesthesia and spinal haematoma. Can J Anaesth 43: 1260-1271.
- Hejazi N, Thaper PY, Hassler W (1998) Nine cases of nontraumatic spinal epidural hematoma. Neurol Med Chir (Tokyo) 38: 718-723.
- Endo T, Suzuki S, Inoue T, Utsunomiya A, Uenohara H, et al. (2014) Prediction of neurological recovery in spontaneous spinal epidural hematoma using apparent diffusion coefficient values. Spinal Cord 52: 729-733.
- Deger SM, Emmez H, Bahadirli K, Kale A, Ebinc FA, et al. (2009) A spontaneous spinal epidural hematoma in a hemodialysis patient: a rare entity. Intern Med 48: 2115-2118.
- Liou KC, Chen LA, Lin YJ (2012) Cervical spinal epidural hematoma mimics acute ischemic stroke. Am J Emerg Med 30: 1322 e1321-1323.
- Aksay E, Kiyan S, Kitis O, Yuruktumen A (2008) A rare diagnosis in emergency department: spontaneous spinal epidural hematoma. Am J Emerg Med 26: 835 e833-835.
- Huh J, Kwak HY, Chung YN, Park SK, Choi YS (2016) Acute Cervical Spontaneous Spinal Epidural Hematoma Presenting with Minimal Neurological Deficits: A Case Report. Anesth Pain Med 6: e40067.
- Shima H, Yasuda M, Nomura M, Mori K, Miyashita K, et al (2012) A spinal epidural hematoma with symptoms mimicking cerebral stroke. Nagoya J Med Sci 74: 207-210.
- Hsieh C, Tung C, Shih P, Lin W (2005) Cerebral hemosiderosis as a causative factor of vascular parkinsonism. Acta Neurol Taiwan 14: 201-207.
- Aristedis R, Dimitrios B (2017) Spinal epidural hematoma mimicking subarachnoid hemorrhage: A case study. Surg Neurol Int 1: 182-182.
- Duffill J, Sparrow OC, Millar J, Barker CS (2000) Can spontaneous spinal epidural haematoma be managed safely without operation? A report of four cases. J Neurol Neurosurg Psychiatry 69: 816-819.
- Xian H, Xu LW, Li CH, Hao JM, Wan WX, et al. (2017) Spontaneous spinal epidural hematomas: One case report and rehabilitation outcome. Medicine (Baltimore) 96: e8473.
- Sklar EM, Post JM, Falcone S (1999) MRI of acute spinal epidural hematomas. J Comput Assist Tomogr 23: 238-243.
- Groen RJ, Begeer JH, Wilmink JT, Le Coultre R (1991) Acute cerebellar ataxia in a child with transient pontine lesions demonstrated by MRI. Neuropediatrics 22: 225-227.
- Park J, Lee JB, Park JY, Lim DJ, Kim SD, et al (2007) Spinal cord infarction after decompressive laminectomy for spontaneous spinal epidural hematoma: case report. Neurol Med Chir (Tokyo) 47: 325-327.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi