Research Article, J Spine Neurosurg Vol: 6 Issue: 3
The Etiology And Prognosis Of C5 Palsy After Anterior Decompression With Spinal Fusion
Imajo Y* Kanchiku T, Suzuki H, Funaba M, Nishida N and Taguchi T
Department of Orthopaedic Surgery, Yamaguchi University Graduate School of Medicine 1-1 Minami-kogushi, Ube, Yamaguchi, 755-8505, Japan
*Corresponding Author : Dr. Yasuaki Imajo
Department of Orthopaedic Surgery, Yamaguchi University Graduate School of Medicine 1-1 Minami-kogushi, Ube, Yamaguchi, 755-8505, Japan
Tel: 81-836-22-2268
Fax: 81-836-22-2267
E-mail: i-yasuak@yamaguchi-u.ac.jp
Received: March 25, 2017 Accepted: June 06, 2017 Published: June 14, 2017
Citation: Imajo Y, Kanchiku T, Suzuki H, Funaba M, Nishida N, et al. (2017) The Etiology and Prognosis of C5 Palsy after Anterior Decompression with Spinal Fusion. J Spine Neurosurg 6:3. doi: 10.4172/2325-9701.1000270
Abstract
Background: There are currently few reports on the etiology and the prognosis of C5 palsy using electrophysiological examination. The aim of this study was to discuss the etiology and prognosis of C5 palsy after anterior decompression with spinal fusion (ASF) using radiological findings and electrophysiological examination.
Methods: 219 patients underwent ASF for cervical degenerative disease. We assessed the cervical sagittal alignment, the local angle at the fused level, and the height of the fused vertebral body on lateral radiographs in a neutral position preoperatively and at final follow-up. We performed intraoperative motor evoked potentials (MEPs) from deltoid and biceps, and measured compound muscle action potentials (CMAPs) in deltoid and central motor conduction time (CMCT) preoperatively and approximately 1 month after onset of the C5 palsy. C5 palsy was defined as a paresis of the deltoid (manual muscle testing (MMT) score of 1 or 2) with involvement of the biceps brachii muscle and supination. To compare the radiological and neurological findings of patients with C5 palsy and those without C5 palsy, 40 patients (designated as group C) were randomly selected from 209 patients without C5 palsy.
Results: The incidence of C5 palsy was 4.6% (10 patients). We calculated the CMCT pre- and postoperatively in 8 patients. Compared with preoperatively, the CMCT shortened in 7 patients (87.5%) postoperatively, except for case 6. Intraoperatively, there were no patients with a decrease in MEPs from deltoid and biceps. Patients had a CMAP amplitude that exceeded 1.5 mV for the deltoid on the C5 palsy side recovered deltoid function. There were no significance differences in radiological findings between the group with C5 palsy and group C.
Conclusions: We considered the prognosis to be good in patients with a CMAP amplitude of the deltoid muscle on the C5 palsy side that exceeded 1.5 mV.
Keywords: C5 palsy; Etiology; Prognosis; Anterior decompression with spinal fusion; Compound muscle action potentials
Abbreviations
ASF: Anterior Decompression With Spinal Fusion; Meps: Motor Evoked Potentials (Meps); Cmaps: Compound Muscle Action Potentials; CMCT: Central Motor Conduction Time; MMT: Manual Muscle Testing
Introduction
Postoperative C5 palsy are more frequently described following posterior rather than anterior cervical surgery [1-3]. There are fewer reports of C5 palsy after the anterior approach than after the posterior approach.
The suggested etiologies of C5 palsy include iatrogenic nerve injury during surgery, reperfusion injury of the spinal cord, a tethering effect to the nerve root due to the posterior shift of the spinal cord, spinal cord ischemia, and a segmental spinal cord disorder [1,4-9]. No tethering effect has been shown to play a role in the etiology of C5 palsy after the anterior approach [8,10].
In this study, C5 palsy was defined as a paresis of the deltoid (manual muscle testing (MMT) score of 1 or 2) with involvement of the biceps brachii muscle and supination, but without the loss of intrinsic muscle strength.
There are currently few reports on the prognosis of C5 palsy [9]. The aim of this study was to discuss the etiology and prognosis of C5 palsy after anterior decompression with spinal fusion (ASF) using radiological findings and electrophysiological examination.
Materials and Methods
Between 1997 and 2012, 219 patients underwent ASF for cervical degenerative disease (Table 1). There were 131 men and 88 women, with a mean age of 57.4 (range, 29–85) years at the time of surgery. There were 75 patients with cervical spondylotic myelopathy (CSM), 71 with cervical disc herniation (CDH), 36 with ossification of the posterior longitudinal ligament (OPLL), 28 with cervical spondylotic amyotrophy (CSA) (proximal-type: 19 and distal-type: 9), 6 with cervical spondylotic radiculopathy (CSR), and 3 with other diseases. There were 74 patients who underwent 1 level of fusion, 139 who underwent 2 levels of fusion, 5 who underwent 3 levels of fusion and 1 who underwent 4 levels on fusion. Patients were confined to bed for 3 days after surgery. Patients were braced in a soft collar for 2 months postoperatively, and were followed for a minimum of the one postoperatively year. We excluded patients with injuries, tumors, multifocal motor neuropathy, and amyotrophic lateral sclerosis.
| Mean age | 57.4 (29-85) | |
|---|---|---|
| Sex | ||
| Men | 131 | |
| Women | 88 | |
| Disease | ||
| CSM | 75 | |
| CDH | 71 | |
| OPLL | 36 | |
| CSA | ||
| Proximal-type | 19 | |
| Distal-type | 9 | |
| CSR | 6 | |
| Others | 3 | |
| No. of levels fused | Fused level | No. of cases |
| 1 | C3-4 | 16 |
| C4-5 | 11 | |
| C5-6 | 35 | |
| C6-7 | 10 | |
| C7-T1 | 2 | |
| subtotal | 74 | |
| 2 | C2-4 C3-5 C4-6 C5-7 |
1 31 64 41 |
| C6-T1 | 2 | |
| subtotal | 139 | |
| 3 | C3-6 | 2 |
| C4-C7 | 3 | |
| subtotal | 5 | |
| 4 | C4-T1 | 1 |
Table 1: The characteristic of 219 patients who underwent anterior decompression with spinal fusion.
Radiological findings
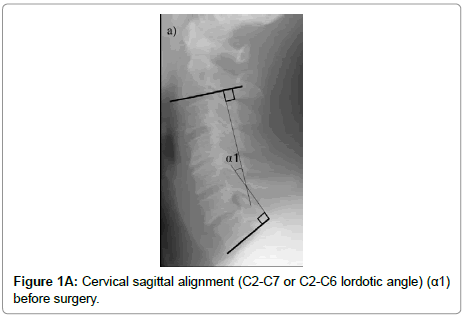
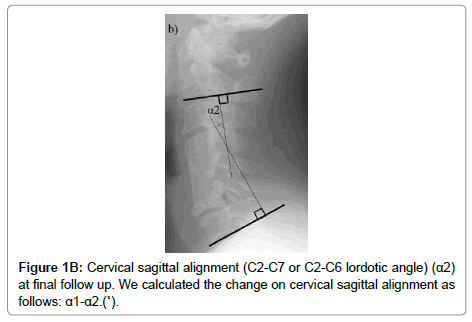
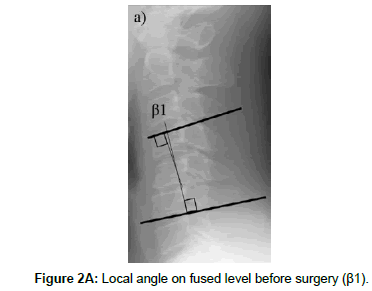
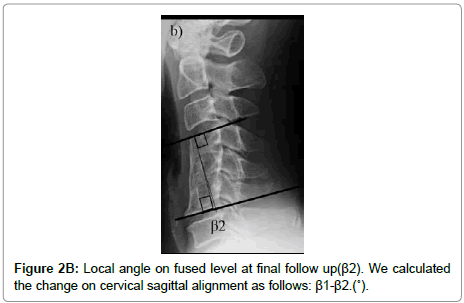
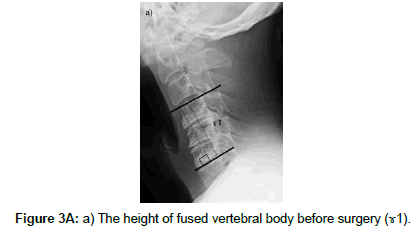
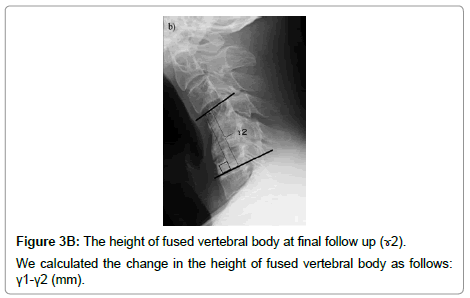
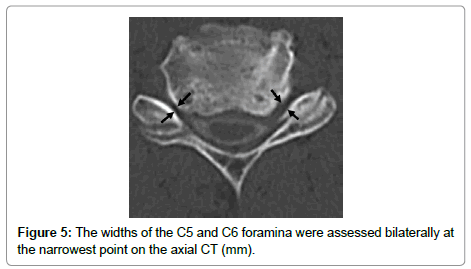
The cervical sagittal alignment (C2-C7 or C2-C6 lordotic angle), the local angle at the fused level, and vertebral body height on standard lateral radiographs in a neutral position were evaluated preoperatively and at final follow-up (Figures 1-3). Lateral tilting angle of graft bone was assessed on postoperative anterior-posterior radiographs (Figure 4). Foraminal size was assessed utilizing CT studies at the C5 and C6 levels. The widths of these foramina were assessed bilaterally at the narrowest point on the axial CT (Figure 5). MRI were assessed the presence of high intensity areas (HIAs) in the spinal cord on the sagittal T2-weighted views preoperatively and the extent of HIAs postoperatively.
Neurological findings
We examined muscle strength of the deltoid, biceps brachii muscle, supination, the biceps tendon reflex (BTR), the triceps tendon reflex (TTR), the area of sensory disturbance, the onset of weakness, and the postoperative course of motor function.
To compare the radiological and neurological findings of patients with C5 palsy and those without C5 palsy at the fused C4-C5 or C3-C5 or C4-C6 levels, 40 patients (designated as group C) were randomly selected from 209 patients without C5 palsy. Group C were matched by age at the time of surgery, disease, and fused levels for patients with C5 palsy.
Electrophysiological examinations
We performed all electrophysiological examinations using a Nicolet Viking IV instrument and Viking Select (Nicolet Biomedical Madison, Wisconsin, USA).
Measurement of central motor conduction time (CMCT)
We placed self-adhesive surface recording electrodes on bilateral abductor digiti minimi (ADM) using a standard belly-tendon method and recorded motor evoked potentials (MEPs). Transcranial magnetic stimulation (TMS) was delivered by Magstim 200 (Magstim, Machida City, Tokyo) using a circular coil with an outer diameter of 140 mm. We applied TMS while the patients exerted isometric voluntary contraction of the ADM. The coil was held with its center on the Cz position of a 10–20 system for recording MEPs from the ADM. The TMS intensity was set at 20% above the MEP’s threshold and at least 4 consecutive trials were recorded. We recorded the shortest onset latency of the MEPs. Compound muscle action potentials (CMAPs) and F-waves were recorded following supramaximal electric stimulation (square wave 0.2 ms) of the ulnar nerve at the wrist. We obtained 16 serial responses and the shortest latency of the F-waves was measured. We calculated the CMCT as follows: MEPs latency - (CMAPs latency + F wave latency − 1)/2 (ms). The ADM responses were amplified and filtered with a bandpass of 5–5,000 Hz.
Recording intraoperative MEPs
We performed intraoperative spinal cord monitoring using transcranial electrical stimulation. The transcranial stimulus conditions comprised 5 train stimuli, a stimulus interval of 2 ms, a stimulus of 300–600 V, and a filter of 50–1000 Hz. The MEPs were recorded from the deltoid, biceps brachii muscle, ADM, and abductor hallucis.
Measurement of CMAPs in the deltoid
We measured CMAPs for 5 patients (cases 6, 7, 8, 9, and 10) preoperatively and for 8 patients (cases 1, 2, 3, 6, 7, 8, 9, and 10) approximately 1 month after the onset of the C5 palsy.
We recorded Erb-point stimulated CMAPs in the deltoid. One 11-mm disc electrode was placed over the middle of the deltoid as an active electrode and 1 was placed on the acromion as a reference electrode. The bipolar stimulator consisted of a pair of bare-metal contact surfaces, approximately 3 mm in diameter, with an adjustable interelectrode distance. We gradually increased the stimulus intensity until it no longer altered the size of the recorded response. The CMAPs measurements included the negative-peak amplitude from baseline to peak.
Statistical analysis
Statistical analyses were performed using Statcel 2. Mann- Whitney’s U test and Student’s t-test was applied for patients with C5 palsy and Group C. A P value of <0.05 was considered statistically significant in all the analyses.
This study received approval from the institutional review board of our hospital.
Results
Table 2 showed degenerative diseases and fusion levels of patients with C5 palsy and group C.
| Patients with C5 palsy | Group C | p-value | ||
|---|---|---|---|---|
| No. of cases (%) | 10 (4.6%) | 40 | ||
| Mean age | 63.8 (37-79) | 62.8 (31-80) | NS | |
| Sex | ||||
| Men | 9 | 20 | NS | |
| Women | 1 | 20 | ||
| Disease CSM |
3(4.0%) | 12 | NS | |
| CDH | 1(1.4%) | 4 | NS | |
| OPLL | 2(5.6%) | 8 | NS | |
| CSA Proximal type | 3(15.8%) | 12 | NS | |
| CSR | 1(16.7%) | 4 | NS | |
| No. of levels fused | Fused level | |||
| 1 | C4-5 | 1(9.1%) | 7 | NS |
| subtotal | 1(1.4%) | 7 | NS | |
| 2 | C3-5 | 5(16.1%) | 16 | NS |
| C4-6 | 4(6.3%) | 17 | NS | |
| subtotal | 9(6.5%) | 33 | NS | |
Table 2: The relationship between patients with C5 palsy and group C.
The incidence of C5 palsy was 4.6% (10 out of 219 patients) in patients who underwent ASF. There were 9 men and 1 woman with a mean age of 63.8 (range, 37–79) years at the time of surgery. The incidence of C5 palsy was 0.0% (0 out of 8 patients) in patients who underwent ASF with plate. There was no significant difference between patients underwent ASF without plate and with plate.
The highest incidence of C5 palsy was 16.7% in patients with CSR, followed by 15.8% for proximal-type CSA, 5.6% for OPLL, 4.0% for CSM, and 1.4% for CDH.
ASF at C4-C5 intervertebral level was indicated by all patients with C5 palsy. We identified C5 palsy in 1 of 74 (1.4%) patients who underwent 1 level of fusion and 9 of 139 (6.5%) patients who underwent 2 levels of fusion. There were no patients with C5 palsy who underwent more than 3 levels of fusion. There was no correlation between 1 fusion level and 2 fusion levels (P = 0.09). The highest incidence of C5 palsy was 16.1% in patients with C3-C5 fusion levels, followed by 9.1% at the C4-5 fusion level, and 6.3% at the C4-6 fusion levels.
In group C, there were 20 men and 20 women with a mean age of 62.8 (range, 31–80) years at the time of surgery. There were 12 patients with CSM, 4 with CDH, 8 with OPLL, 12 with proximal-type CSA, and 4 with CSR. There were 7 patients fused at the C4-5 level, 16 fused at the C3-5 level, and 17 fused at the C4-6 level.
The preoperative diagnosis and neurological findings of C5 palsy (Table 3).
| Case | Diagnosis | BTR (Rt/Lt) |
TTR (Rt/Lt) |
MMT(pre-op) (Rt/Lt) |
S-D area | ||
|---|---|---|---|---|---|---|---|
| Deltoid | Biceps | Supination | |||||
| 1 | CDH | ↓/↓ | ↓/↓ | 5/5 | 5/5 | NR/NR | C6 |
| 2 | CSM | ↓/→ | ↑/↑ | 5/5 | 5/5 | NR/NR | C6 |
| 3 | CSR | ↓/→ | →/→ | 4/5 | 4/5 | NR/NR | C6 |
| 4 | CSM | ↑/↑ | ↑/↑ | 5/4 | 5/5 | NR/NR | C6 |
| 5 | OPLL | ↓/↑ | ↓/↑ | 5/3 | 5/4 | NR/NR | C5 |
| 6 | OPLL | ↓/↓ | ↑/↑ | 5/5 | 5/5 | NR/NR | C5 |
| 7 | CSA | ↓/→ | ↓/→ | 2/5 | 4/5 | 2/5 | C6 |
| 8 | CSA | ↓/↓ | ↓/↓ | 5/4 | 5/3 | 5/3 | C6 |
| 9 | CSM | →/↓ | ↓/↓ | 5/5 | 5/5 | 5/5 | C6 |
| 10 | CSA | ↓/→ | →/→ | 4/5 | 4/5 | 4/5 | None |
S-D; sensory disturbance, NR; not reported,↓:diminished,→:normal,↑:exaggerated
Table 3: Preoperative diagnosis and neurological findings of C5 palsy.
With regard to BTR and TTR, there were 2 patients (cases 3 and 10) with radiculopathy, and other patients with myelopathy and/or radiculomyelopathy. Six of 10 (60.0%) patients had motor weakness and 9 (90%) had areas of sensory disturbance.
Postoperative course of C5 palsy
The C5 palsy occurred on the right side in 5 patients and the left side in 5. In 5 of 6 patients with preoperative motor weakness, the side of the C5 palsy did not correlate with the side affected with the motor weakness (it did correlate in case 10). The mean follow-up period was 60.2 (range, 20–199) months. The mean onset of muscle weakness was 12 (range, 1–26) days. Weakness was present in the deltoid and biceps brachii muscles in all 10 patients and in the supinator muscle in all 5 patients that had complete medical records (Table 4).
| Case | Palsy side | FU period (months) |
Onset of C5 palsy (days) | MMT of palsy side (At onset / At FU) |
CMCT of palsy side (ms) |
CMAPs amplitude of deltoid (mV) (palsy side/non-palsy side) |
Period of recovery for deltoid (months) |
Degree of recovery for deltoid | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Deltoid | Biceps | Supinator | Before surgery | After surgery* | Before surgery | After surgery* | ||||||
| 1 | Lt | 199 | 16 | 2/5 | 2/5 | NR/4 | NR | NR | NR | 2.3/11.0 | 6 | complete |
| 2 | Lt | 15 | 10 | 2/5 | 2/4 | NR/NR | 6.6 | 6.0 | NR | 1.5/7.5 | 15 | complete |
| 3 | Lt | 128 | 26 | 2/5 | 2/5 | NR/NR | NR | NR | NR | 1.9/9.0 | 4 | complete |
| 4 | Rt | 68 | 23 | 2/5 | 2/5 | NR/NR | 6.5 | 6.3 | NR | NR | 10 | complete |
| 5 | Rt | 53 | 5 | 1/4 | 1/4 | NR/NR | 7.9 | 6.6 | NR | NR | - | incomplete |
| 6 | Rt | 45 | 7 | 2/5 | 2/5 | 2/4 | 11.6 | 10.3 | 7.5/7.3 | 2.1/6.1 | 20 | complete |
| 7 | Lt | 13 | 10 | 2/4 | 2/4 | 2/3 | 6.1 | 6.3 | 10.2/1.6 | 1.7/0.3 | - | incomplete |
| 8 | Rt | 14 | 19 | 1/3 | 1/3 | 1/2 | 7.2 | 5.5 | 11.0/3.3 | 4.1/2.9 | - | incomplete |
| 9 | Lt | 16 | 3 | 1/3 | 1/2 | 1/2 | 6.9 | 6.8 | 8.9/10.2 | 0.3/10.1 | - | incomplete |
| 10 | Rt | 24 | 1 | 1/2 | 1/2 | 1/2 | 5.8 | 5.1 | 4.0/11.6 | 0.3/10.7 | - | incomplete |
Table 4: Postoperative course.
We calculated the CMCT pre- and postoperatively in 8 patients. In comparison with preoperatively, the CMCT was shortened postoperatively in 7 of these patients.
With regard to spinal cord monitoring, there were no patients with a decrease in MEPs from the deltoid, biceps brachii muscle, ADM, and abductor hallucis intraoperatively.
We measured CMAPs of the deltoid bilaterally in 5 patients (except for cases 1, 2, 3, 4, and 5) before surgery and in 8 patients (except for cases 4 and 5) after surgery. The surgical outcome was poor for all 3 patients with CSA (cases 7, 8, and 10). Patients that did not have CSA that had CMAP amplitude that exceeded 1.5 mV for the deltoid on the C5 palsy side recovered deltoid function. However, case 8 had CSA and, even though the amplitude of the CMAP exceeded 4.1 mV for the deltoid on the C5 palsy side, the patient has not recovered deltoid function. The CMAP amplitude of the deltoid on the nonpalsy side was decreased for all patients (cases 6-10) postoperatively in comparison with preoperatively.
Radiological findings
There were no correlations between the group with C5 palsy and group C with regard to the antero-posterior diameter of the C5 canal, the change in the cervical sagittal alignment, the change in the local angle, the change in the height of the fused vertebral body, and the width of the C5 and C6 foramina. In the group with C5 palsy, there was a progression in the cervical sagittal alignment and local angle with kyphosis in all patients (100%) and shortening in the height of the fused vertebral body that exceeded 2 mm in 7 patients (70%). In group C, there was a progression in the cervical sagittal alignment and local angle with kyphosis in 32 patients (80%) and shortening in the height of the fused vertebral body that exceeded 2 mm in 14 patients (35%). There was no significant difference between patients with C5 palsy and Group C with regard to lateral tilting angle of grafted bone. But in patients with C5 palsy, there were tilting of grafted bone that exceeded 2Ëš in 5 patients (50%). Tilting side of grafted bone corresponded with palsy side in all 5 patients (Table 5).
| Patients with C5 palsy (n=10) |
Group C (n=40) | p-value | ||
|---|---|---|---|---|
| Plain radiograph | AP diameter of C5camnal (mm) | 14.2 ± 1.1 | 14.7 ± 1.2 | 0.30 |
| The change in cervical sagittal alignment (C2-7 or C2-6 lordotic angleËš) (α1-α2) | -11.9 ± 12.5 | -5.0 ± 8.1 | 0.06 | |
| The change in local angleËš (β1-β2) | -10.1 ± 8.0 | -5.9 ± 8.0 | 0.21 | |
| The change in the height of fused vertebral body (mm) (γ1-γ2) | 3.3 ± 2.8 | 1.5 ± 2.2 | 0.06 | |
| Lateral tilting angle of grafted bone | 2.5 ± 4.0 | 2.0 ± 3.5 | 0.67 | |
| CT | Width of the C5 foramen (mm) Palsy side(C5palsy)/small side(control) |
3.0 ± 1.3 | 2.9 ± 1.3 | 0.74 |
| Normal side(C5palsy)/large side(control) | 3.4 ± 1.6 | 3.7 ± 1.0 | 0.57 | |
| Width of the C6 foramen (mm) Palsy side(C5palsy)/small side(control) |
3.2 ± 1.2 | 2.8 ± 1.0 | 0.21 | |
| Normal side(C5palsy)/large side(control) | 3.1 ± 1.1 | 3.7 ± 1.3 | 0.23 |
Table 5: Radiological findings.
In the group with C5 palsy, the C5 palsy side corresponded with the narrow side at a rate of 50% and 40% with regard to width of the C5 and C6 foramina, respectively. There was no significant difference between the C5 palsy side and non-palsy side with regard to width of the C5 and C6 foramina, respectively.
The preoperative T2-weighted MRI demonstrated spinal cord compression in all 10 patients with C5 palsy and a HIA in 6 of the 10 patients (60%). The spinal cord compression was at the C4-C5 intervertebral level in 9 patients (90%), at C5-C6 in 6, and at C3-C4 in 2. HIAs were present at the C3-C4 intervertebral level in 1 patient, at C4-C5 in 4, and at C5-C6 in 1. There were no patients who extended HIAs postoperatively.
Discussion
The average reported incidence of C5 palsy following surgery for cervical compression myelopathy for the anterior approach is 6.7% (range, 1.6-9.1%) [5,8,10-14].
Most investigators have reported the etiology of C5 palsy using radiological findings. There are currently few reports on the etiology and the prognosis of C5 palsy using electrophysiological examination. The aim of this study was to discuss the etiology and prognosis of C5 palsy after anterior decompression with spinal fusion (ASF) using radiological findings and electrophysiological examination. We performed intraoperative motor evoked potentials (MEPs) from deltoid and biceps, and measured compound muscle action potentials (CMAPs) in deltoid and central motor conduction time (CMCT) preoperatively and approximately 1 month after onset of the C5 palsy. This is the first paper on the etiology and prognosis of C5 palsy using intraoperative MEPs, CMCT, and CMAPs.
The etiology of C5 palsy
According to spinal cord monitoring using MEPs, there were no patients with either intraoperative nerve root or spinal cord injuries. Postoperative MRI did not show any novel or expanded T2-HIAs in the spinal cord. Therefore, reperfusion injury of the spinal cord may not be an applicable etiology. Some researchers have reported that the tethering phenomenon may not be applicable to ASF because the spinal cord tends to move anteriorly after decompression [8,10]. It is very difficult to diagnose spinal cord ischemia and segmental spinal cord disorders.
Compared with preoperatively, the CMCT shortened in 7 patients (87.5%) postoperatively, except for case 6. There were no patients with prolonged CMCTs that exceeded 1 ms. The CMCT can be used to electrophysiologically evaluate corticospinal function [15-17]. It has been found to be prolonged in patients with cervical spinal cord compression [15-17]. Kaneko et al. reported the normal CMCT value as 5.2 ± 1.1 ms [16]. The preoperative CMCT of case 6 was 6.1 ms, and it increased by 0.2 ms postoperatively. As a result, we could not assess any functional deterioration of the corticospinal tracts. There were no patients with functional deterioration of the corticospinal tracts in this study.
The postoperative amplitude of the CMAPs of the deltoid contralateral to the palsy side was slightly decreased in all patients (cases 6-10) compared with the preoperatively CMAPs. The CMAPs of the deltoid reflect the number of axons that innervate it. The number of axons decreased on the non-palsy side postoperatively. We previously reported that the deterioration of the amplitudes of the CMAPs on bilateral sides meant the involvement of the anterior horns [18]. However, the deterioration of the amplitude of the CMAPs was too low on the non-palsy side. Thus, we considered nerve root involvement to play a role in the etiology of the C5 palsy.
There were no significance differences in radiological findings between the group with C5 palsy and group C. However, the P value was 0.06 for the change in both cervical sagittal alignment and the height of the fused vertebral body. Tilting side of grafted bone corresponded with palsy side in all 5 patients who exceeded 2Ëš in lateral tilting angle of grafted bone. The height of the fused vertebral body of C5 palsy side was shorter than that of non-palsy side in these patients. Therefore, we considered that the height of foramen of C5 palsy was shorter than that of non-palsy side. The incidence of C5 palsy may reflect the progression of kyphosis and the reduced height of the foramen. The mean onset of C5 palsy was 12 days, which meant that the progression of kyphosis and the reduced height of the foramen gradually occurred after ASF. These findings imply that the etiology of the C5 palsy may include involvement of the nerve roots.
There were no patients with C5 palsy who underwent ASF with plate in this study. ASF with plate may prevent the progression of kyphosis and the reduced height of the foramen
Prognosis of C5 palsy
Although C5 palsy has a good prognosis for functional recovery, some patients experience persistent muscle weakness [10]. The prognosis of C5 palsy in relation to the severity of motor weakness has been described [9]. However, there are no reports on the prognosis of C5 palsy using the postoperative amplitude of CMAPs of the deltoid. Patients (except for those with CSA) with a CMAP amplitude of the deltoid on the C5 palsy side that exceeded 1.5 mV recovered deltoid function in severe C5 palsy patients with a muscle power of 2 or less on MMT. There were no patients who fully recovered their supination function. We considered the prognosis to be good in patients (except for those with CSA) with CMAP amplitude of the deltoid on the C5 palsy side that exceeded 1.5 mV.
Conclusions
According to intraoperative spinal cord monitoring using MEPs, there were no patients with either intraoperative nerve root or spinal cord injuries. There were no patients with functional deterioration of the corticospinal tracts postoperatively. We believe that C5 palsy after ASF may be due to the involvement of the nerve roots with progression of kyphosis and a reduced height of the foramina. We considered the prognosis to be good in patients (except for those with CSA) with CMAP amplitude of the deltoid on the C5 palsy side that exceeded 1.5 mV.
References
- Imagama S, Matsuyama Y, Yukawa Y, Kawakami N, Kamiya M, et al. (2010) C5 palsy after cervical laminoplasty: a multicenter study. J Bone Joint Surg 92: 393-400.
- Kaneko K, Hashiguchi A, Kato Y, Kojima T, Imajo Y, at al. (2006) Investigation of motor dominant C5 paralysis after laminoplasty from the results of evoked spinal cord responses. J Spinal Disord Tech 19: 358-361.
- Katsumi K, Yamazaki A, Watanabe K, Ohashi M, Shoji H (2012) Can Prophylactic Bilateral C4/C5 Foraminotomy Prevent Postoperative C5 Palsy After Open-Door Laminoplasty? A Prospective Study. Spine 37: 748754.
- Chiba K, Toyama Y, Matsumoto M, Maruiwa H, Watanabe M, et al. (2002) Segmental motor paralysis after expansive open-door laminoplasty. Spine 27: 2108-2115.
- Hasegawa K, Homma T, Chiba Y (2007) Upper extremity palsy following cervical decompression surgery results from a transient spinal cord lesion. Spine 32: E197-E1202.
- Tuzuki N, Abe R, Saiki K, Li Z (1996) Extradural tethering effect as one mechanism of radiculopathy complicating posterior decompression of cervical spinal cord. Spine 21: 203-211.
- Shinomiya K, Okawa A, Nakao K, Mochida K, Haro H, et al. (1994) Morphology of C5 ventral nerve rootlets as part of dissociated motor loss of deltoid muscle. Spine 19: 2501-2504.
- Hashimoto M, Mochizuki M, Aiba A, Okawa A, Hayashi K, et al. (2010) C5 palsy following anterior decompression and spinal fusion for cervical degenerative diseases. Eur Spine J 19: 1702-1710.
- Sakaura H, Hosono N, Mukai Y, Ishii T, Yoshikawa H (2003) C5 palsy after decompression surgery for cervical myelopathy. Spine 28: 2447-2451.
- Odate S, Shikata J, Yamamura S, Soeda T (2013) Extremely wide and asymmetric anterior decompression causes postoperative C5 palsy. Spine 38: 2184-2189.
- Bydon M, Macki M, Kaloostian P, Sciubba DM, Wolinsky JP, et al. (2014) Incidence and Prognostic Factors of C5 Palsy: A Clinical Study of 1001 Cases and Review of the Literature. Neurosurgery 74: 595-605.
- Ikenaga M, Shikata J, Tanaka C (2005) Radiculopathy of C5 after anterior decompression for cervical myelopathy. J Neurosurg Spine 3: 210-217.
- Saunders RL (1995) On the pathogenesis of the radiculopathy complicating multilevel corpectomy. Neurosurgery 37: 408-413.
- Yonenobu K, Hosono N, Iwasaki M, Asana M, Ono K (1991) Neurologic complications of surgery for cervical compression myelopathy. Spine 16: 1277-1282.
- Kaneko K, Taguchi T, Morita H, Yonemura H, Fujimoto H, et al. (2001) Mechanism of prolonged central motor conduction time in compressive cervical myelopathy. Clinical Neurophysiology 112: 1035-1040.
- Kaneko K, Kato Y, Kojima T, Imajo Y, Taguchi T (2006) Epidurally recorded spinal cord evoked potentials in patients with cervical myelopathy and normal central motor conduction time measured by transcranial magnetic stimulation. Clinical Neurophysiology 117: 1467-1473.
- Nakanishi K, Tanaka N, Sasaki H, Kamei N, Hamasaki T, et al. (2010) Assessment of central motor conduction time in the diagnosis of compressive thoracic myelopathy. Spine 35: E1593- E1598.
- Imajo Y, Kato Y, Kanchiku T, Suzuki H, Yoshida Y, et al. (2012) Prediction of surgical outcome for proximal-type cervical spondylotic amyotrophy novel mode of assessment using compound action potentials of deltoid and biceps brachii and central motor conduction time. Spine 37: E1444-E1449.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi