Review Article, Cell Biol Henderson Nv Vol: 7 Issue: 1
The Role of CCL18 protein in Breast Cancer Development and Progression
Sm Faysal Bellah1,2#*, Hameed Akbar2#, S M Saker Billah3 and Divine Mensah Sedzro2
1Department of Pharmacy, Manarat International University, Khagan, Ashulia, Dhaka-1216, Bangladesh
2Laboratory of Cellular Dynamics, School of Life Science, University of Science and Technology of China, Hefei, Anhui-230027, P R China
3Department of Chemistry, Govt. M. M. University College, Jessore-7400, Bangladesh
#These authors contributed equally to this work
*Corresponding Author : Sm Faysal Bellah
Assistant Professor, Department of Pharmacy, School of Engineering, Science and Technology, Manarat International University, Khagan, Ashulia, Dhaka-1216, Bangladesh
Tel: +8801913261838, +8615656529539
E-mail: faysal@mail.ustc.edu.cn, faysal_phku@yahoo.com
Received: July 05, 2018 Accepted: September 07, 2018 Published: September 08, 2018
Citation: Bellah SF, Akbar H, Billah SMS, Sedzro DM (2018) The Role of CCL18 protein in Breast Cancer Development and Progression. Cell Biol (Henderson, NV) 7:1. doi: 10.4172/2324-9293.1000138
Abstract
CCL18 is a CC chemokine ligand 18, which is produced by Tumor-Associated Macrophages (TAMs), that can stimulate the progressiveness, angiogenesis in breast cancer, as well as the underlying pathophysiological mechanisms and it was originally discovered as pulmonary and activation-regulated chemokine (PARC), dendritic cell (DC)-chemokine 1 (DC-CK1), alternative macrophage activation associated CC chemokine-1 (AMAC-1), and macrophage inflammatory protein-4 (MIP-4). The potential functional receptor of CCL18 protein is PITPNM3 that facilitates CCL18 effect and stimulates the intracellular calcium signaling pathway. Worldwide, Breast cancer (BCa) is one of the most common malignancies among the females in both developed and also developing county. This study mainly focused on the role of CCL18 in the production of breast tumor as well as in the invasiveness of breast cancer. The data here suggest that CCL18 protein play an important role in the development of breast tumor and also progression in breast cancer. This knowledge could be helpful for the proposal of new therapeutic approaches particularly in breast cancer
Keywords: Chemokine CCL18; Breast cancer; Breast tumor
Introduction
The CCL18 protein is encoding a polypeptide contain 89 amino acids and a 20 peptide signaling sequence at N-terminal which is cleaved in the endoplasmic reticulum into a 69 peptide which is mature protein [1]. The mature CCL18 protein is almost similar to CCL3 (64% of identical amino acids) & CCL4 (48% of identical amino acids) and located on chromosome 17 [1], together with many other macrophage inflammatory proteins (MIPs). CCL18 protein is CC cytokine which is characterized by two adjacent cysteines, and form two disulfide bonds between Cys10-Cys34 and Cys11-Cys50, which are very much important to show to the function of this protein [2]. This protein also contains a BBXB consensus heparin binding site allowing its binding to glycosaminoglycan [3]. It has 3 exons and 2 introns and it is different from other chemokines, the CCL18 contains 2 pseudo-exons (exons which don’t appear in the final peptide) in first intron [4]. Processed CCL18 isoforms in addition to unglycosylated, intact CCL18 (1-69) was found after isolation of natural human CCL18 from various sources. Certainly, CCL18 (1-68), was copurified with CCL18 (1-69), lacking the COOH terminal Ala from human plasma [5-7]. This protein is produced mostly by antigen-presenting cells of innate immune system which include dendritic cells, monocytes and macrophages [8-10]. Not T-cells, nor B-cells are well known to produce CCL18 [8] and the production is controlled in these cells by cytokines such as IL-4, IL-10 and IL-13 that favor a T-helper 2 type cells response which are normally involved in humoral immunity or immunosuppression. CCL18 is made by fibroblasts exactly by stimulation of collagens that is very much essential in tissue healing and also repairs [10] and highly expressed in lungs, so that CCL18 plays important role in maintaining homeostasis.
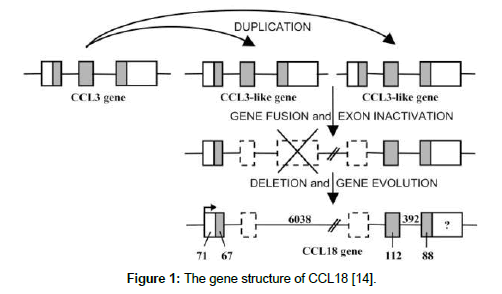
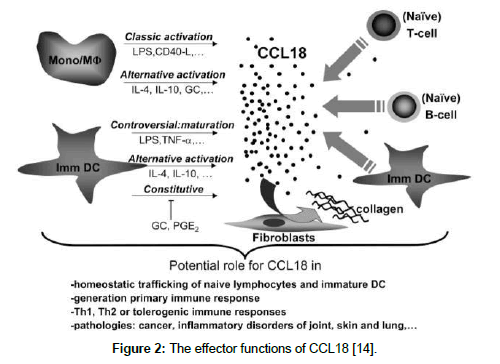
The receptors for CCL18 protein are G-protein coupled receptors (GPCRs). Very recently there are three receptors that have been proposed for CCL18 protein such as PITPNM3, GPR30 (G-protein coupled receptor 30) and CCR8. PITPNM3 is only expressed on breast cancer cells and PITPNM3 –CCL18 binding induces Pyk2 and Src mediated signaling (a cancer related pathway) and succeeding metastasis of breast cancer [11,12]. Excessive production of CCL18 protein in M2 macrophages was correlated with various chronic inflammations and fibroblastic diseases such as Gaucher’s disease and rheumatoid arthritis [13,14]. CCL18 was constitutively expressed in many cancer such as in macrophages infiltrating ovarian cancer, gastric cancer and glioma [15-17]. Though M2 macrophages are most abundant in breast cancer stroma sothat CCL18 was expressed in breast cancer. CCL18 protein abundantly produced from breast TAMs and also it is released by breast TAMs which promotes the invasiveness of breast cancer [18]. As discussed about the gene structure and also the effector functions of CCL18 protein Figure 1 and Figure 2 [14].
Figure 1: The gene structure of CCL18 [14].
Figure 2: The effector functions of CCL18 [14].
CCL18 protein in relation to the risks of Breast tumor angiogenesis and invasiveness
It was confirmed that CCL18 protein is correlated with breast tumor angiogenesis and also expression by immunohistochemical (IHC) staining for CCL18 expression together with anti-CCL18 antibodies and microvascular density (MVD) using anti-CD34/CD31/ von Willebrand Factor (vWF) antibodies in primary invasive ductal carcinoma samples of the breast [19]. CCL18 protein were absent in benign breast tissue but they were infrequently identified in the stroma of ductal carcinomas in situ (DCIS) of the breast, these data confirmed that CCL18 produced by TAMs correlates with the invasiveness without the growth kinetics of breast tumor cells. CCL18 enhances extracellular matrix adherence and migration of breast cancer cells [18]. CCL18 secreted by breast TAMs that reduce the miR98 and miR27b expression [20]. CCL18 was significantly associated with the invasiveness of breast carcinomas and it was high proliferative tumors determined by Ki67, on the basis of estrogen receptor status there was no difference and it was not related to the tumor size, and confirmed the correlation of CCL8 with the invasiveness of the breast cancer but not the growth kinetics of breast tumor cells [21].
Serum level of CCL-18 protein expression and its association with disease prognosis of breast cancer patients
It was investigated that whether CCL-18 protein is a biomarker for poor disease prognosis in the Yanan Shanxi region of China. They established that serum level of CCL-18 was higher in breast cancer patients than that of healthy women and individuals with benign tumors [22]. This result suggested that serum CCL-18 may be a biomarker for breast cancer, and may participate in tumorigenesis. They also found that serum level of CCL-18 is varied among breast cancer patients at different TNM stages; serum CCL- 18 was significantly higher in stage IV patients than stages II and III patients and elevated CCL-18 level can be used as a biomarker for advanced breast cancer [22]. Though, the role of CCL18 protein in cancer progression is controversial. Especially, it was reported that serum CCL18 protein was negatively correlated with clinical cancer stages [23]. Scientists reported that CCL18 protein was participate in immunosuppression of ovarian cancer [24,17], but in gastric cancer CCL18 was associated with prolonged survival in patients [23]. It is possible that the role of CCL18 protein varies due to different immunological mechanisms in different cancers [25]. CCL18 was significantly associated with the invasiveness of breast carcinomas and it was high proliferative tumors determined by Ki67, on the basis of estrogen receptor status there was no difference and it was not related to the tumor size, and confirmed the correlation of CCL8 with the invasiveness of the breast cancer but not the growth kinetics of breast tumor cells.
CCL18 protein promotes the Invasion and Metastasis of Breast Cancers in vivo
For the examination of CCL18 contributed to angiogenesis in breast cancer, it was evaluated the angiogenic effects of CCL18, which does not have a mouse homolog [26], in tumor xenografts derived from MDA-MB-231 (high metastatic potential) or BT-474 (low metastatic potential) breast cancer cells in the mammary fat pads of NOD/SCID mice. Consistent result found with in vitro and significant increase of MVD counts by 7.2-fold (p<0.001) in MDAMB- 231-derived xenografts and by 6.0-fold (p<0.001) in BT-474- derived xenografts, as compared with control mice after intratumoral injection of rCCL18 breast [19].
After intratumor injection of rCCL18 at a dosage of 0.1 mg/kg biweekly for four consecutive weeks at the same dosage, enhanced vascular invasion and penetration of BT-474 cancer cells into adjacent normal tissues. The treatment of BT-474 and MDA-MB-231 cells xenografts with rCCL18 and silencing with PITPNM3 also alleviated the promoting effect of rCCL18 on lung and liver metastases and decreased CCL18-induced reduction in mouse survival and body weight. So, CCL18 promotes invasion and metastasis of breast cancers in vivo in a PITPNM3-dependent manner [25].
Discussion
The CCL18 protein is located on 17q11.2 that encodes a cytokine; it is secreted by various cells such as nucleus cells, macrophages, T cells, B cells, dendritic cells, hemopoietic progenitor cells, fibroblasts, and tumor cell. This protein acts through autocrine and paracrine signaling pathway, and it is responsible for controlling in immunologic and inflammatory processes [27]. A number studies has shown CCL18 has the capability to affect tumorigenesis and also affect the grade and prognosis of ovarian cancer, colon cancer and gastric cancer. It have been established that breast cancer cells co-cultured with carcinoma-associated fibroblast resulted in increased CCL18 secretion, which promoted the invasion and metastasis of breast cancer cells [28]. But Narita et al found that an elevated serum CCL18 levels of breast cancer patients were associated with poor prognosis of disease [29].
TAM (M2 type) and fibroblasts release a high levels of CCL18 and create a CCL18-rich microenvironment for cancer cells and its bind with PITPNM3 [25] and GPR30 receptors [30] expressed on cancer cells. This promotes the cancer cells through Snail1 stabilization, invasion, and migration [25,31]. A potential beneficial effect of CCL18 has been shown in gastric cancer where high levels of CCL18 are associated with prolonged survival of these patients that is not dependent on tumor stage [23]. However, in pancreatic ductal and lung carcinoma, CCL18 positive discrimination of epithelial mesenchymal transition which is correlated with lymph node metastasis, histopathological grade and diminished survival time [31]. CCL18 is an accurate biomarker of bladder cancer and correlates with the severity of cutaneous T-cell lymphoma and with breast cancer metastasis [25].
Conclusion
In conclusion the role of CCL18 is an important TAM-derived cytokine in promoting breast cancer metastasis and also it has shown the tumor angiogenesis in breast cancer. There are many others TAMderived chemokines [32-35] also give expression via another signaling pathway because of complete inhibition of CCL18 did not completely hunk or inhibit breast cancer metastasis by TAMs. So, more studies are necessary to elucidate whether the cytokines released by TAMs can be influence to inhibit the breast tumour progression.
Conflicts of Interest
The authors declare that there is no conflict of interest.
References
- Hieshima K, Imai T, Baba M, Shoudai K, Ishizuka K, et al. (1997) A novel human CC chemokine PARC that is most homologous to macrophage-inflammatory protein-1 alpha/LD78 alpha and chemotactic for T lymphocytes, but not for monocytes. J Immunol 159: 1140-1149.
- Legendre B, Tokarski C, Chang Y, Nadai PD, Rolando C, et al. (2013) The disulfide bond between cysteine 10 and cysteine 34 is required for CCL18 activity. Cytokine 64: 463-470.
- Krohn S, Garin A, Gabay C, Proudfoot AE (2013) The activity of CCL18 is principally mediated through interaction with glycosaminoglycans. Frontiers in Immunology 4: 193.
- Politz O, Kodelja V, Guillot P, Orfanos CE, Goerdt S (2000) Pseudoexons and regulatory elements in the genomic sequence of the beta-chemokine, alternative macrophage activation-associated CC-chemokine (AMAC)-1. Cytokine 12: 120-126.
- Schutyser E, Struyf S, Wuyts A, Put W, Geboes K, et al. (2001) Selective induction of CCL18/PARC by staphylococcal enterotoxins in mononuclear cells and enhanced levels in septic and rheumatoid arthritis. Eur J Immunol 31: 3755-3762.
- Schutyser E, Struyf S, Proost P, Opdenakker G, Laureys G, et al. (2002) Identification of biologically active chemokine isoforms from ascitic fluid and elevated levels of CCL18/pulmonary and activation-regulated chemokine in ovarian carcinoma. J Biol Chem 277: 24584-24593.
- Struyf S, Schutyser E, Gouwy M, Gijsbers K, Proost P, et al. (2003) PARC/CCL18 is a plasma CC chemokine with increased levels in childhood acute lymphoblastic leukemia. Am J Pathol 163: 2065-2075.
- Bellinghausen I, Reuter S, Martin H, Maxeiner J, Luxemburger U, et al. (2012) Enhanced production of CCL18 by tolerogenic dendritic cells is associated with inhibition of allergic airway reactivity. J Allergy Clin Immunol 130: 1384-1393.
- Ferrara G, Bleck B, Richeldi L, Reibman J, Fabbri LM, et al. (2008) Mycobacterium tuberculosis induces CCL18 expression in human macrophages. Scand J Immunol 68: 668-674.
- Schraufstatter IU, Zhao M, Khaldoyanidi SK, Discipio RG (2012) The chemokine CCL18 causes maturation of cultured monocytes to macrophages in the M2 spectrum. Immunology 135: 287-298.
- Li HY, Cui XY, Wu W, Yu FY, Yao HR, et al. (2013) Pyk2 and Src mediate signaling to CCL18-induced breast cancer metastasis. J Cell Biochem 115: 596-603.
- Chen J, Yao Y, Gong C, Yu F, Su S, et al, (2011) CCL18 from tumor-associated macrophages promotes breast cancer metastasis via PITPNM3. Cancer Cell 19: 541-555.
- Martinez FO, Helming L, Gordon S (2009) Alternative activation of macrophages: an immunologic functional perspective. Annu Rev Immunol 27: 451-483.
- Schutyser E, Richmond A, Damme J (2005) Involvement of CC chemokine ligand 18 (CCL18) in normal and pathological processes. J Leukoc Biol 78: 14-26.
- Chang CY, Lee YH, Leu SJ, Wang CY, Wei CP, et al. (2010) CC-chemokine ligand 18/pulmonary activation-regulated chemokine expression in the CNS with special reference to traumatic brain injuries and neoplastic disorders. Neuroscience 165: 1233-1243.
- Leung SY, Yuen ST, Chu KM, Mathy JA, Li R, et al. (2004) Expression profiling identifies chemokine (C-C motif) ligand 18 as an independent prognostic indicator in gastric cancer. Gastroenterology 127: 457-469.
- Zohny SF, Fayed ST (2010) Clinical utility of circulating matrix metalloproteinase-7 (MMP-7), CC chemokine ligand 18 (CCL18) and CC chemokine ligand 11 (CCL11) as markers for diagnosis of epithelial ovarian cancer. Med Oncol 27: 1246-1253.
- Chen J, Yao Y, Gong C, Yu F, Su S, et al. (2011) CCL18 from tumor-associated macrophages promotes breast cancer metastasis via PITPNM3. Cancer Cell 19: 541-555.
- Lin L, Chen YS, Yao YD, Chen JQ, Chen JN, et al. (2015) CCL18 from tumor-associated macrophages promotes angiogenesis in breast cancer. Oncotarget 6: 34758-34773.
- Lin X, Chen L, Yao Y, Zhao R, Cui X, et al. (2015) CCL18-mediated down-regulation of miR98 and miR27b promotes breast cancer metastasis. Oncotarget 6: 20485-20499.
- Narita D, Seclaman E, Ursoniu S, Ilina R, Cireap N (2011) Expression of CCL18 and interleukin-6 in the plasma of breast cancer patients as compared with benign tumor patients and healthy controls. Rom J Morphol Embryol 52: 1261-1267.
- Sun JH, Fan N, Zhang Y (2016) Correlation between serum level of chemokine (C-C motif) ligand 18 and poor prognosis in breast cancer. Genet Mol Res 15: 15038632.
- Leung SY, Yuen ST, Chu KM, Mathy JA, Li R et al. (2004) Expression profiling identifies chemokine (C-C motif) ligand 18 as an independent prognostic indicator in gastric cancer. Gastroenterology 127: 457-469.
- Duluc D, Corvaisier M, Blanchard S, Catala L, Descamps P, et al. (2009) Interferon gamma reverses the immunosuppressive and protumoral properties and prevents the generation of human tumor-associated macrophages. Int J Cancer 125: 367-373.
- Chen J, Yao Y, Gong C, Yu F, Ewen ME et al. (2011) CCL18 from tumor-associated macrophages promotes breast cancer metastasis via PITPNM3. Cancer Cell 19: 541-555.
- Su S, Liu Q, Chen J, Chen F, He C, et al. (2014) A positive feedback loop between mesenchymal-like cancer cells and macrophages is essential to breast cancer metastasis. Cancer Cell 25: 605-620.
- Urquidi V, Kim J, Chang M, Dai Y, Goodison S, et al. (2012) CCL18 in a multiplex urine-based assay for the detection of bladder cancer. PLoS One 7: e37797.
- Peng Q, Zhao L, Hou Y, Sun Y, Liu M, et al. (2013) Biological characteristics and genetic heterogeneity between carcinoma-associated fibroblasts and their paired normal fibroblasts in human breast cancer. PLoS One 8: e60321.
- Nariţa D, Seclaman E, Ursoniu S, Ilina R, Cireap N, et al. (2011) Expression of CCL18 and interleukin-6 in the plasma of breast cancer patients as compared with benign tumor patients and healthy controls. Rom J Morphol Embryol 52: 1261-1267.
- Catusse J, Wollner S, Leick M, Schrottner P, Schraufstatter I, et al. (2010) Attenuation of CXCR4 responses by CCL18 in acute lymphocytic leukemia B cells. J Cell Physiol 225: 792-800.
- Ploenes T, Scholtes B, Krohn A, Burger M, Passlick B, et al. (2013) CC-chemokine ligand 18 induces epithelial to mesenchymal transition in lung cancer A549 cells and elevates the invasive potential. PLoS One 8: e53068.
- Coussens LM, Tinkle CL, Hanahan D, Werb Z (2000) MMP-9 supplied by bone marrow-derived cells contributes to skin carcinogenesis. Cell 103: 481-490.
- Dirkx AE, Egbrink MG, Wagstaff J, Griffioen AW (2006) Monocyte/macrophage infiltration in tumors: modulators of angiogenesis. J Leukoc Biol 80: 1183-1196.
- Goswami S, Sahai E, Wyckoff JB, Cammer M, Cox D, et al. (2005) Macrophages promote the invasion of breast carcinoma cells via a colony-stimulating factor-1/epidermal growth factor paracrine loop. Cancer Res 65: 5278-5283.
- Leek RD, Talks KL, Pezzella F, Turley H, Campo L, et al. (2002) Relation of hypoxia-inducible factor-2 alpha (HIF-2 alpha) expression in tumor-infiltrative macrophages to tumor angiogenesis and the oxidative thymidine phosphorylase pathway in human breast cancer. Cancer Res 62: 1326-1329.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi