Research Article, J Virol Antivir Res Vol: 10 Issue: 5
The Role of Emerging Tools Used in COVID-19 in India
Rinka Juneja, Anil Ahuja and Vishal M. Balaramnavar*
School of Pharmacy & Research Center, Sanskriti University, Chhata Mathura, India
*Corresponding Author: Vishal M. Balaramnavar
School of Pharmacy & Research Center, Sanskriti University, Chhata Mathura, India
E-mail: v.balaramnavar@gmail.com
Received: August 19, 2021 Accepted: September 17, 2021 Published: September 24, 2021
Citation: Juneja R, Ahuja A, Balaramnavar VM (2021) The Role of Emerging Tools Used in COVID-19 in India. J Virol Antivir Res 10:5.
Abstract
The discovery of SARS-CoV-2 and related disease Covid-19 showed a negative impact on health and lifestyle. Covid 19 is a disease that causes destruction on India which will have the largest population in world Currently Covid-19 is caused by the Severe Acute Respiratory Syndrome (SARS-CoV-2). In India social distancing and lockdown rules were imposed but they show a harmful effect on economy, health life. To identify the importance of biological and immunological process that underlines the clinical manifestations of Covid-19. SARS-CoV-2 is transmitted through droplets into the air also with a faecal contamination pathway but it still unproven. The onset of symptoms appears within 4-5 days after the infection. Recent findings stated the role of emerging tools used in Covid-19 in India. To understand the pharmacology of SARS-Cov-2 it is important to diagnose the target proteins and therapeutics that used in Covid-19.
Keywords: Covid-19; SARS-Cov-2; Fecal contamination; Target proteins
Introduction
The current pneumonia outbreak which come into force on 1 December, 2019 nearby Wuhan city, Hubei Province, China that triggered by the novel corona virus (ncoV) termed as ‘2019-nCoV or 2019 Novel Corona Virus or Covid-19’by World Health Organization (WHO) [1]. Covid-19 is a virus that can cause disease. Bats are seems to be the Covid-19 carrier, but the intervening host has yet to be identified. Corona Virus divided into four types: Gamma corona Virus, Delta corona Virus, Beta corona Virus and Alpha corona Virus [2]. The first two type’s species mostly infect the birds whereas third and fourth types mostly infect mammals. Severe Acute Respiratory Syndrome (SARS) is a form of acute respiratory syndrome. Covid-19 disease is caused by SARS-CoV-2). SARS-CoV-2 is 79.6% identical to SARS-CoV by sequence and 96% similar to bat derived SARS-like corona virus [3]. SARS-CoV and MERS-CoV is highly pathogenic virus that transmits from bats and now to humans. Coronaviruses are enveloped with RNA Viruses with a crown shaped structure which can ranges in size from 60 nm to 140 nm in diameter and are found in animals also in humans and birds. It has been shown that Coronaviruses can mutate and recombine that causes illness of respiratory, gastrointestinal, hepatic and neurological systems [4,5].
One million four hundred ninety –eight thousand eight hundred thirty –three cases were confirmed of Covid-19 across the globe with number of deaths 5.8%. The number of deaths was rising continuously which would lead a serious problem for the globe [6]. In order to tackle the spread of corona virus each and every country imposed different methods includes sitting at home, wearing mask, social distancing and washing hands. The government also imposed a lockdown to stave off the citizens to move freely. Covid -19 is not an only a factor to slaughtered the individuals but peoples may affect with mental disintegration that results to unemployment and famine [7].
It has been shown that Coronaviruses can mutate and recombine that causes illness of respiratory, gastrointestinal, hepatic and neurological systems [8]. Therefore this review focuses on how India is geared to cope up with Covid-19 its harmful consequences on economy, education, mental stress and in human life and as well as different strategies to overcome with pandemic situation in India.
Covid-19 Scenario in India
Initially, Coronaviruses infection in India occurs with the result of international connection instead of transmission within the country. On 30th of January and 3rd of February, the first three cases of infection appeared in Kerala while after they returned from Wuhan, China. Additionally two cases have been reported a couple of months later [9]. On March 3rd one with a travel history from Italy and other from Hyderabad form Dubai. On same day, more cases were reported in Jaipur. To overcome the spread of infection the Ministry of Health and Family Welfare (MoHFW) approved travel warnings similar to the approved in past pandemic such as SARS, Ebola and plague, also imposed the 14 days self-quarantine rules for foreign visitors coming into India [10]. Furthermore travel visa for other countries were strictly restricted till 15th of April. On March 16, 2020 the MoHFW suggested different interventions and tools to control the infection of corona virus such as 1 m social distancing in order to decrease the mortality and morbidity.
On 22nd March, Honorable Prime Minister Narendra Modi advised the citizens of India to maintain the 14 h Janta curfew. In India the first very phase of 21 day lockdown started on 24 March. The government of India declared an extended second phase lockdown till 3rd may and further it extends till 31st May [11]. Government amended Epidemic Disease Act of 1897 that included a quarantine laws in order to make the lockdown and social distancing more efficient. On 30th January, first case confirmed in India [12]. By 15th March no of cases were increased and then percentage of positive cases has already been rising. Covid-19 cases reported in India has increased by ten times in just 15 days between 15 March to 30 March. According to the ICMR, if social distancing and quarantine measures are taken properly, India can reduce the cases by 62%. Covid-19 hotspots were discovered in cities such as Ahmadabad, Benglaru, Bhopal, Chennai, Delhi and Hyderabad, Indore, Jaipur and Kolkata contributing 40% of Covid cases [13].
In India Corona virus has impacted everyone’s daily life. At initial stage the cases that were conformed were brought from abroad. Therefore the government of India has advised to keep a distance in effort to stop transmission. The individuals who is having a lower immunity and older in age were treated with more safety. According to statics the individuals who were infected with Covid-19 is 39 years in age. Individuals somewhat between the age of 21 and 40 were more likely to infect with the Covid infection [14].
Pathophysiology of Covid-19
Respiratory droplets seem to be the most common way to transmit the infection from person to person. The virus has been already identified in sputum, pharyngeal swabs and faces. Vertical transmission of SARS-CoV-2 has been observed with a positive Covid-19 nasopharyngeal swab confirming the disease [15,16]. On average the infection rate of Covid-19 is 5.2 days and most of the patients having symptoms between 11.5 and 15.5 days. Therefore it has been suggested that patient who have already been exposed to infection be quarantined for 14 days. There are three types of corona virus that can multiply and grow in lower respiratory tract and therefore results to pneumonia. With the genetic homology the severe acute respiratory syndrome corona virus-2 resembles with the human corona virus. SARS-Cov-2 is associated with the Beta corona virus [17].
The SARS-CoV -2 infection penetrates to host cells through binding the ACE-2 which is supported by the TMPRSS2 protease for uptake. High infection of virus is connected with the mutation in the receptor site in the S spike protein. The patients who are prone to virus, the virus interacts with ACE2 may decrease the antiinflammatory effect whereas it increases the Angiotensin –II effects. Difficulty in treating with Covid-19 in patients with hypertension, patients have recommended for the use or discontinuation of AT1 receptor blockers and ACE inhibitors [18]. Currently European Society of Cardiology’s Council on Hypertension recommended that patient keep taking antihypertensive drugs as there is no indication that antihypertensive drugs should be stopped and discontinued [19].
The pathogenesis of SARS-CoV-2 is analogues to SARS-CoV. The infection of SARS-CoV-2 in patients is determined by the viral infection and the response of host. The immaculacy of SARS-CoV and MERS-CoV shows a marked pattern of increasing the infection with age [19,20]. In severe Covid-19 cases Acute Respiratory Distress Syndrome is linked with the breathing problems and lower level of oxygen in blood. Consequently a little more than that bacterial and fungal infections may occur in Covid patients. It also lead to respiratory failure which seems to the cause of death [20-22].
Symptoms in Patient with Covid-19
Like other pulmonary diseases, SARS-Cov-2 corona virus may transmit through respiratory droplets. The infection is last up to 4-5 days. In case of infected individuals the symptoms may up to last 11 days or less than 11 days. When a patient is hospitalized the most common symptoms are fever and dry cough although the patient may experience breathing problems, chest pain, joint pain, headache, nausea, vomiting and bleeding in cough [23]. As per as the current studies, fever, fatigue, dry cough, neuropathy and dizziness were the most common symptoms at the beginning of illness whereas headache, abdominal discomfort, diarrhea, nausea and vomiting were least common symptoms [24]. Only a few patients were reported to have only GIT symptoms initially. Within 5-6 days of infection the peak of SARS-CoV-2 takes place and on average of 8-9 days Acute Respiratory Distress Syndrome occurs in severe covid-19 cases [25,26].
As per sources, patients are exhibiting neurological deficits in addition to respiratory symptoms of flu. Furthermore, neurological deficits documented in Covid-19 patients with complicated and uncomplicated infections from hospitals in Wuhan, China, are convincing enough so that neurological deficits might be continuing in the current outbreak without being identified. Covid-19 which is produced by SARS-CoV-2 Virus involves two pathways of entering the brain. The involvement of SARS-Cov-2 involved in brain investigated further in patients of loss of taste, smell and ataxia and convulsions. SARS-CoV-2 was isolated from the brain tissue that showed signs of oedema and neurological degeneration during the examination [27-29].
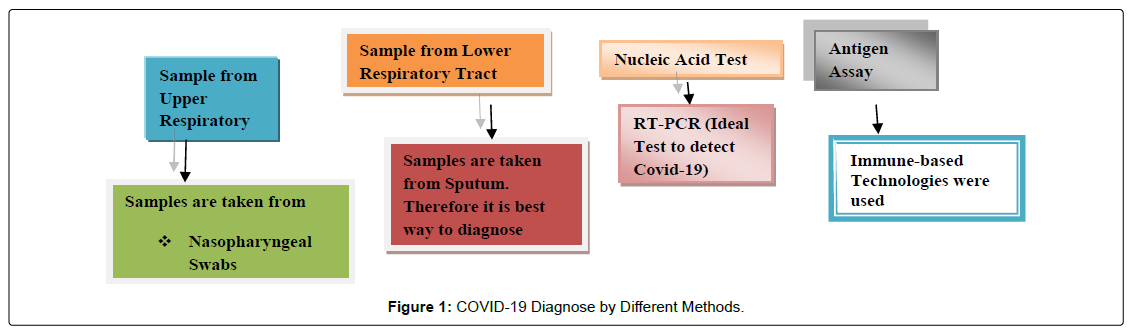
Diagnostic Methods Applied in Covid-19
Covid-19 is a virus that mainly affects the lungs. Virus detection of upper and lower respiratory tract samples has been recommended as primary method for detecting ongoing clinical infection [30]. Currently there are two types of testing methods nucleic acid and antibody testing. Immunological techniques are used to detect the antibody while nucleic acid test explore the RNA virus from either the patient throat or nasal passage. According to the Center of Disease Control and Prevention RT-PCR is highly effective and appreciated [31]. Other types of laboratory testing were suggested such as test based on nucleic acids, antigen and antibody assay. The different methods to diagnose the Covid-19 as shown in Figure 1.
Samples from Upper Respiratory TractClinical sample are collected from the upper respiratory tract oropharyngeal swabs, nasopharyngeal aspirates, and nasopharyngeal swabs. Nasal swabs were collected from the back to the nose [32]. In symptomatic patients or asymptomatic patients having early stage of infections, upper respiratory sample recommended for clinical diagnose of initial disease. Nasopharyngeal swabs (NPS) are a good alternative for nasopharyngeal aspirates (NPA) as it provide a reliable result than oropharyngeal swabs [33,34]. Both nasopharyngeal and oropharyngeal swabs is used to detect the covid-19 to improve the clinical outcomes. Nose swabs, mid-turbinate and saliva specimens are self-collected as they minimize the need of healthcare professionals which is more beneficial when personal protective equipment is in shortage [35-38].
Samples from the Lower Respiratory Tract
Lower respiratory tract samples have greater infection rates and therefore they are more likely to produce positive result than samples of upper respiratory tract. Samples from the lower respiratory tract are considered at the late stage of Covid-19 infection or some with a clinical diagnose of Covid-19 infection [39,40]. To diagnose the lower respiratory tract sputum is the best approach. It has been shown that it improves the efficacy of infection. Specimens of upper respiratory were shown to be less toxic than lower respiratory specimen of Covid-19. Due to the risk of aerosol generated induced sputum is not recommended as it affects the healthcare professionals [41].
Nucleic Acid Test
Nucleic Acid is the laboratory test to detect the current Covid-19 infection. The test based on nucleic acid investigates the RNA virus from the patient’s throat and nostril canal [42]. To diagnoses SARSCov- 2 RNA from the upper respiratory infection real time PCR test (RT-PCR) is preferred [43]. The nucleoplamid, envelop and spike genes as well as open reading frame (orf1a and orf1b) and RNA – polymerase all genes are targeted by NAAT tests. According to centre for disease control and prevention, the common model for RT-PCR assay includes 3 stages collection of sample, Hydrolysis, Purification of RNA and replication [44,45]. In RT-PCR studies, the cycle threshold is the number of cycle needed to multiply RNA to predict the level. Ct value is inversely proportional to the sample of viral RNA. For viral detection, CT-values measurement in RTPCR technique is not standard. When considering CT-values as a reference for clinical treatment physicians should be aware of these limitations. To diagnose the Covid-19 RT-PCR is considered as the ideal test [46]. Studies documented that in most of the patients a positive NAAT confirms the Covid-19. After the onset of symptoms presence of SARS-Cov-2 RNA for weeks confirmed the presence of non-viable virus fragment but does not contagious. A negative NAAT result confines that testing can be done within 24 to 48 hours after the original test and sample of lower respiratory tract can be preserved when there is infection in lower respiratory tract. Negative test result does not indicate the absence of infection but sometimes pre analytical and post analytical procedures showed the negative results [47-50].
Antigen Assays
Antigen detection assay are used to measure SARS-CoV-2 viral protein in respiratory system. Most of the test samples are taken from nasal cavity while saliva has considered as the alternate test for detection of antigen. Antigen detection assay employed immune based technologies for identification such as lateral flow sandwich assay, micro fluidic immunofluorescence technique [51,52]. Generally, to perform the test these lab tests include all the components that are easy to use and utilize for lab testing. As they can produce the outcomes within a limited time period therefore they consider as rapid diagnostic test (RDT). Antigen assay are less accurate than NAATs because it does not target the protein. Generally antigen assay had its own advantages such as it is easy to perform and when NAAT test is in limited source than antigen assay is more effective. When NAAT is not available, SARS-Cov-2 antigen assay performed in such situation [53,54].
Antibody Detection
Immunity is described as the defense mechanism to protect the body against sickness. Innate immunity and Adaptive immunity are the two main forms of immunity. Innate Immunity is a type of immunity that provides a swift response within minutes. Acquired immunity works on the effector cell and memory cell [55-57]. The role of antibody is to identify the small components of antigens [58]. There are five forms of immunoglobin: IgG, IgM, IgA, IgD and IgE and their role were depicted in Table 1.
| S.No | Immunoglobin | Immunoglobin functions | References |
|---|---|---|---|
| 1. | IgG | IgG divided into: Ig1, Ig2, Ig3 and Ig4. Generally Ig1, Ig3 and Ig4 traverse the placenta more efficiently and also plays a pivotal role in fetus development. | [59] |
| 2. | IgE | This type of antibodies produce anaphylactic shock engaged in hypersentivity reactions. It plays an important role in allergic reactions to produce histamine. | [59,60] |
| 3. | IgD | It does not attack antigens directly but it can target the antigen. It triggers the both mast cell and basophile to produce antimicrobial species. | [61,62] |
| 4 | IgM | IgM is the initial therapy to fight against the infection or virus. When there is large production of IgG it can eradicate the foreign particle at the initial stage of B-cell humoral immunity. | [63] |
| 5 | IgA | It prevents the growth of foreign particle in respiratory tract, stomach lining and in genital area. It is also available in oral saliva, breast milk. Also IgA plays an activating role in mucous cell membrane that seems to be the primary penetration site. | [64] |
Table 1: Role of Immunoglobulin’s.
To detect the antibody test of SARS-Cov-2 it mainly targets on two antigens: the nucleoplasmid (N) and spike protein(S) [59-61]. ELISA (Enzyme Linked Immunosorbant Assay) and chemiluminescence immunoassay (CLIA) is often used as a laboratory based test to detect the Covid-19 in patients. These tests reveal the antibodies such as IgG, IgM and IgA. Generally, immunoglobin testing IgG is more comprehensive than IgM and IgA [62-65]. When the infection risk is considered to be low false negative test is reported. To enhance the laboratory test, Center for Disease Control and Prevention US guidelines recommended two steps. Firstly it involves two separate antibodies in which original test is confirmed by second antibody [65- 70].
Medications Available in COVID-19
Currently, supplies of vaccines are available in India. Earlier anti HIV, anti-Hepatitis C, anti-malarial drugs are given to the patients to treat the Covid-19. The drugs that used to treat Covid-19 were depicted in Table 2.
| S.No | Drugs used to treat Covid-19 | Indications | References |
|---|---|---|---|
| 1 | Hydroxychloroquine (HCQ) and Chloroquine (CQ) |
The aminoquinolines Chloroquine and Hydroxychloroquine were used to treat Malaria and autoimmune disorder over the past than 50 years. In vitro assay reveal that CQ and HCQ both are beneficial against SARS-CoV-2. Studies reported that Chloroquine outperformed on the basis of clinical recovery and lengthen the recovery of disease and illness. |
[66-70] |
| 2 | Lopinavir (LPV ) and Ritonavir (RTV) |
The antiviral protease agents such as Lopinavir and Ritonavir were used to treat HIV infection. In elderly patients who are admitted in hospital with severe case of covid-19, studies found that there was no satisfaction treatment with LPV/RTV. |
[71] |
| 3 | Remdesivir (RDV) | Remdesivir is used as a novel antiviral medication for the treatment of SARS-CoV-2. In-Vitro study stated that Remdesivir suppressed SARS -CoV-2 viruses. Earlier several studies are ongoing to improve the toxicology of Remdesivir in Covid-19 patients. |
[72,73] |
| 4 | Favipiravir (FPV) | From 2014, FPV is used as guanine analogue that inhibits the RNA –virus. It is also used to treat the influenza virus As FPV hinder SARS-CoV-2 virus therefore it is used as a treatment in patients with the severe cases Covid-19. | [74] |
| 5 | Tocilizumab | As stated by the recent clinical investigation patients in ICU has increased the blood levels. Therefore Tocilizumab is a monoclonal antibody and also behaves as the IL-6 antagonist. According to US Food and Drug Administration Tocilizumab is approved for the patients of covid-19 in severe cases. |
[75] |
Table 2: Current Medicines used in Covid-19.
Vaccines
Currently many of the vaccine candidates showed a successful result and also showed that vaccine bears a good fruit to the candidates. Generally Vaccine of Covid-19 has developed in India. Currently, Vaccines available in India [70-76].
Covisheild by Serum Institute of India
It is also known as Astra ZencaAdeno virus that based on AZD1222. Several studies reported that in month of January company will manufacture 100 million doses at each month [77-79].
Covaxin by Bharat Biotech
Covaxin was designed and sold by Bharat Biotech International Limited in collaboration with Indian Council of Medical Research and National Institute of Virology. In vitro Covaxin was generated as an inactive virus that combines with aluminum hydroxide gel. At the month of August 2021 the company will deliver approx. 150 million doses and 700 by the close of this year [80,81].
Conclusion
Covid-19 is the most public health issue across the globe. China, Europe and United States showed a number of incidents and death rates. SARS-CoV-2 is related with betacoronavirus that correlate with the bat corona virus. The virus can enter into the cell through Angiotensin converting enzyme -2 receptor causes alterations in respiratory systems and neurological systems. Fever and dry cough are the most common symptoms. Other symptoms such as neurological, gastrointestinal and cardiac were also reported. Patients who are having the old age are more prone to infection. Currently most of the drugs are available in India to treat the Covid-19. Now – a –days several clinical trial of vaccines are ongoing.
Conflict of Interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
References
- Azer SA (2020) COVID-19: pathophysiology, diagnosis, complications and investigational therapeutics. Elsevier 37: 100738.
- Sohrabi C, Alsafi Z, Neil NO, Khan M, Kerwan A, et al. (2020) World Health Organization declares global emergency: A review of the 2019 novel coronavirus (COVID-19). Int J Surg 76: 71-76.
- Aritra G, Srijita N, Tapas K, Mallick (2020) How India is dealing with COVID-19pandemic. Sensors Inter 1: 100021.
- Singhal T (2020) A review of corona virus disease-2019 (COVID-19). Ind J Pediatr 87(4): 281-286.
- Zhou T, LouYang X , Wang XG, Hu B, Zhang L, et al. (2020) A pneumonia outbreak associated with a new corona virus of probable bat origin. Nature 579: 270-273.
- Sujath R, Jyotir Moy C, Aboul Ella H (2020) A machine learning forecasting model for COVID-19 pandemic in India. Stochastic Environ Res and Risk Assess 34: 959–972.
- Pulla P (2020) Covid-19: India imposes lockdown for 21 days and cases rise. BMJ 368.
- Rahmandad H, Lim TY, Sterman J (2020) Estimating the global spread of COVID-19. Med Rxi 1.
- Patrikar S, Poojary D, Basannar DR, Kunte R (2020) Projections for novel coronavirus (COVID-19) and evaluation of epidemic response strategies for India. Med J Armed Forces Ind 76(3): 268-275.
- Kumar A (2020) A perspective on India's fight against COVID – 19. Epidemiol Int 5.
- (2020) What Is Janata Curfew: Self Isolation by the People, for the People to Prevent Coronavirus, The Times of India.
- The Lancet (2020) India under COVID-19 lockdown. Lancet 395(10233).
- Nomani MZM, Rauf M, Ahmed Z, Faiyaz T, Khan SA, et al. (2020) Quarantine law enforcement & corona virus (COVID-19) pandemic in India. J Xidian Univ 14.
- Elmousalami HH, Hassanien AE (2020) Day level forecasting for Coronavirus Disease (COVID-19) spread: analysis, modeling and recommendations. ARXiv preprint.
- Amico FD, Baumgart DC, Danese S, Peyrin-Biroulet L (2020) Diarrhea during COVID-19 infection: pathogenesis, epidemiology, prevention and management. Clin Gastroenterol Hepatol 18(8): 1663-1672.
- Li M, Chen L, Zhang J, Xiong C, Li X, et al. (2020) The SARS-CoV-2 receptor ACE2 expression of maternal–fetal interface and fetal organs by single-cell transcriptome study. PLoS One 15. 15(4): 0230295.
- Cheng Lai C, Tzu-Ping S (2020) Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) and coronavirus disease-2019 (COVID-19): The epidemic and the challenges. Int J Antimicrob Agents 55(3): 105924.
- Wenhui Li, Michael JM, Natalya V, Jianhua S, Swee K, et al. (2003) Angiotensin converting enzyme 2 is a functional receptor for the SARS coronavirus. Nature 426(6965): 450-454.
- European Society of Cardiology (2020) Position statement of the ESC Council on hypertension on ACE-inhibitors and angiotensin receptor blockers.
- Chiappelli F, Khakshooy A, Greenberg G (2020) Covid-19 immunopathology and immunotherapy. Bioinformation 16(3): 219-222.
- Anant P (2021) COVID-19: Current understanding of its Pathophysiology, Clinical presentation and Treatment. BMJ Journals 97(1147): 312-320.
- Zhang B (2020). Clinical characteristics of 82 death cases with covid-19. Preprint at medRxiv. 15(7): 0235458
- Chu KH (2005) Acute renal impairment in corona virus-associated severe acute respiratory syndrome 67(2): 698-705.
- Chen N, Zhou M, Dong X, Qu J, Gong F, et al. (2020) Epidemi-ological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study. Lancet 395.
- Xu XW, Wu XX, Jiang XG, Xu KJ, Ying LJ, et al. (2020) Clinical findings in a group of patients infected with the 2019 novel corona virus (SARS-Cov-2) outside of Wuhan, China: retrospective case series. BMJ 368.
- Wang D, Hu B, HuC (2020) Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus–infected pneumonia in Wuhan, China. JAMA. Epub ahead of print.
- Hui DSC, Zumla A (2019) Severe acute respiratory syndrome: historical, epidemiologic, and clinical features 33(4): 869-889.
- Yeh EA, Collins A, Cohen ME (2004) Detection of corona virus in the central nervous system of a child with acute disseminated encephalomyelitis. Pediatrics.
- Hui DSC, Zumla A (2019) Severe acute respiratory syndrome: historical, epidemiologic, and clinical features. Infect Dis Clin North Am 33: 869–889.
- Mao L, Wang M, Chen S (2020) Neurological manifestations of hospitalized patients with COVID-19 in Wuhan, China: a retrospective case series study. medRxiv. Epub ahead of print 25 February.
- World Health Organization (2020) Diagnostic testing for SARS-CoV-2. Interim guidance.
- Meagan Esbin N, Oscar Whitney N, shasha C, Anna M, Xavier D, et al. (2020) Overcoming the bottleneck to widespread testing: a rapid review of nucleic acid testing approaches for COVID-19 detection 26(7): 771-783.
- Spencer S, Thompson MG, Flannery B (2019) Comparison of respiratory specimen collection methods for detection of Influenza virus infection by reverse transcription-PCR: a literature review. J Clin Microbiol 57(9).
- Chan KH, Peiris JSM, Lim W (2008) Comparison of nasopharyngeal flocked swabs and aspirates for rapid diagnosis of respiratory viruses in children. J Clin Virol: Off Publ Pan Am Soc Clin Virol 42(1): 65-69.
- Zou L, Ruan F, Huan M (2020) SARS-CoV-2 viral load in upper respiratory specimens of infected patients. N Engl J Med 382(12): 1177-1179.
- Wong SCY, Tse H, Siu HK (2020) Posterior oropharyngeal saliva for the detection of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), Clin Infect Dis.
- Sutjipto S, LeeP H, Tay JY (2020) The effect of sample site, illness duration, and the presence of pneumonia on the detection of SARS-CoV-2 by real-time reverse transcription PCR. Open forum Infect Dis 7(9).
- Ek P, Bottiger B, Dahlman D (2019) A combination of naso- and oropharyngeal swabs improves the diagnostic yield of respiratory viruses in adult emergency department patients. Inf Disp 51(4): 241-248.
- Lai CKC, Chen Z, Lui G (2020) Prospective study comparing deep-throat saliva with other respiratory tract specimens in the diagnosis of novel coronavirus disease (COVID-19) J Infect Dis 222(10): 1612-1619.
- World Health Organization (2020) Clinical management of COVID-19 (Interim Guidance) World Health Organization.
- Wong MC, Huang J, Lai C (2020) Detection of SARS-CoV-2 RNA in faecal specimens of patients with confirmed COVID-19: a meta-analysis. J Infect 81(2): 31-38.
- Yu F, Yan L, Wang N (2020) Quantitative detection and viral load analysis of SARS-CoV-2 in infected patients. Clin Infect Dis. Off Publ Infect. Dis Soc Am 71(15): 793-798.
- Li SY, Cheng QX, Li XY, Zhang ZL, Gao S, et al. (2018) CRISPR-Cas12a-assisted nucleic acid detection. Cell Discov 4: 1-4.
- Lu R, Wu X, Wan Z, Li Y, Zuo L, et al. (2020) Development of a novel reverse transcription loop-mediated isothermal amplification method for rapid detection of SARS-CoV- 2. Virol Sin 35(3): 344-347.
- Chu DKW, Pan Y, Cheng SMS (2020) Molecular diagnosis of a novel coronavirus (2019-nCoV) causing an outbreak of pneumonia. Clin Chem 66(4): 549-555.
- Hong KH, Lee SW, Kim TS (2020) Guidelines for laboratory diagnosis of coronavirus disease 2019 (COVID-19) in korea. Ann Lab Med 40(5): 351-360.
- Pasteur I (2020) Protocol: Real-time RT-PCR assays for the detection of SARS-CoV-2. 1-3.
- Centers for Disease Control and Prevention (2020). CDC 2019-novel coronavirus (2019-nCoV) real-time RT-PCR diagnostic panel for emergency use only instructions for use. CDC, Atlanta.
- Carter LJ, Garner LV, Smoot JW (2020) Assay techniques and test development for COVID-19 diagnosis. ACS Cent Sci 6(5): 591-605.
- Feng W, Newbigging AM, Le C (2020) Molecular diagnosis of COVID-19: challenges and research needs. Anal Chem 92(15): 10196-10209.
- Lambert-Niclot S, Cuffel A, LePape S, Alexander JM (2020) Evaluation of a rapid diagnostic assay for detection of SARS-CoV-2 antigen in nasopharyngeal swabs. J Clin Microbiol 58(8): 00977.
- Us Food and Drug Administration. Emergency Use Authorizations.
- World Health Organization (2020) Antigen-detection in the diagnosis of SARS-CoV-2 infection using rapid immunoassays. Interim Guideline.
- Weiss A, Jellingsø M, Sommer MOA (2020) Spatial and temporal dynamics of SARS-CoV-2 in COVID-19 patients: a systematic review and meta-analysis. E Bio Med 58: 102916.
- Dinnes J, Deeks JJ, Adriano A (2020) Rapid, point-of-care antigen and molecular-based tests for diagnosis of SARS-CoV-2 infectio. Cochrane Database Syst. Rev 3(3): CD013705.
- Immunity (2011) In Pathology Illustrated (Seventh Edition).
- McComb S, Thiriot A, Krishnan L, Stark F (2013) Introduction to the immune system. Methods Mol Biol 1061: 1–20.
- Owen JA, Punt J, Stranford SA (2013) Kuby Immunology, WH Freeman & Company, New York, USA.
- Novaretti MCZ, Dinardo CL (2011) Clinical applications of immunoglobulin: update. Rev Bras Hematol. Hemoter 33(3): 221–230.
- Pier GB, Lyczak JB, Wetzler LM (2004) Immunology, Infection, and Immunity, ASM Press, ISBN 978-1-55581-246-1.
- Lee ML, Gale RP, Yap PL (1997) Use of intravenous immunoglobulin to prevent or treat infections in persons with immune deficiency. Annu Rev Med 48: 93–102.
- Schroeder HW, Cavacini L (2010) Structure and function of immunoglobulins. J Allergy Clin Immunol 125: 41–52.
- Chen K, Xu W, Wilson M, He B, Miller NW, et al. (2009) Immunoglobulin D enhances immune surveillance by activating antimicrobial, proinflammatory and B cell stimulating programs in basophil. Nat Immunol 10(8): 889–898.
- Geisberger R, Lamers M, Achatz G (2006) The riddle of the dual expression of IgM and IgD Immunology 118(4): 429–437.
- Underdown BJ, Schiff JM (1986) Immunoglobulin A: strategic defense initiative at the mucosal surface. Annu Rev Immunol 4(1): 389–417.
- Deeks JJ, Dinnes J, Takwoingi Y (2020) Antibody tests for identification of current and past infection with SARS-CoV-2. Cochrane Database Syst Rev (6): CD013652.
- Liu J, Cao R, Xu M (2020) Hydroxychloroquine, a less toxic derivative of chloroquine, is effective in inhibiting SARS-CoV-2 infection in vitro. Cell Discov 6: 16.
- Yao X, Ye F, Zhang M (2020) In vitro antiviral activity and projection of optimized dosing design of hydroxychloroquine for the treatment of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). Clin Infect Dis.
- Gautret P, Lagier JC, Parola P Hydroxychloroquine and azithromycin as a treatment of COVID-19: results of an open-label nonrandomized clinical trial. Int J Antimicrob Agent 56(1): 105949.
- Yazdany J, Kim AHJ (2020) Use of hydroxychloroquine and chloroquine during the COVID-19 pandemic: what every clinician should know. Ann Intern Med 172(11): 754-755.
- Cortegiani A, Ingoglia G, Ippolito M, Giarratano A, Einav S, et al. (2020) A systematic review on the efficacy and safety of chloroquine for the treatment of COVID-19. J Crit Care 57: 279–283.
- Cao B, Wang Y, Wen D, Liu W, Wang J, et al. (2020) A trial of lopinavir-ritonavir in adults hospitalized with severe covid-19. N Engl J Med 382(19).
- Tp S, Ac S, Sr L (2020) Comparative therapeutic efficacy of remdesivir and combination lopinavir, ritonavir, and interferon beta against MERS-CoV. Nat Commun 11(1): 222.
- De Wit E, Feldmann F, Cronin J (2020) Prophylactic and therapeutic remdesivir (GS-5734) treatment in the rhesus macaque model of MERS-CoV infection. Proc Natl Acad Sci USA 117(12): 6771–6776.
- Furuta Y, Komeno T, Nakamura T (2017) Favipiravir (T-705), a broad spectrum inhibitor of viral RNA polymerase Proc Jpn Acad Ser B Phys Biol Sci 93(7): 449–463.
- Luo P, Liu Y, Qiu L, Liu X, Liu D, et al. (2020) Tocilizumab treatment in COVID-19: a single center experience. J Med Virol 92(7): 814-818.
- Velayudhan M (2021) Strategy for COVID-19 vaccination in India: the country with the second highest population and number of cases. Vaccines.
- Liu Y, Wang K, Massoud TF, Paulmurugan R (2 020) SARS-CoV-2 vaccine development: an overview and perspectives. ACS Pharm. Transl Sci 3(5): 844–858.
- Voysey M (2021) Safety and efficacy of the ChAdOx1 nCoV-19 vaccine (AZD1222) against SARS-CoV-2: an interim analysis of four randomised controlled trials in Brazil, South Africa and the UK. Lancet 397 (10269): 99–111.
- Vaccine information, ICMR New Delhi—COVID-19 vaccine.
- Zhang Y. Safety tolerability, and immunogenicity of an inactivated SARS-CoV-2 vaccine in healthy adults aged 18–59 years: a randomised, double-blind, placebo-controlled, phase 1/2 clinical trial. Lancet 21(2): 181-192.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi