Review Article, Arch Med Biotechnol Vol: 1 Issue: 1
The Role of Virus Receptor Recognition in the Determination of Pathogenesis
Aliyu Adamu1,2*, Aminu HA1, Firdausi Aliyu2 and Mienda BS3
1Faculty of Science, Department of Microbiology, Kaduna State University, Tafawa Balewa way, Kaduna PMB 2339, Nigeria
2Faculty of Biosciences and Medical Engineering, Department of Biotechnology and Medical Engineering, Universiti Teknologi Malaysia, Johor Bahru 81310, Johor, Malaysia
3Faculty of Science, Department of Microbiology and Biotechnology, Federal University Dutse, 7156 Ibrahim Aliyu by-pass Dutse, Jigawa State, Nigeria
*Corresponding Author : Aliyu Adamu
Faculty of Science, Department of Microbiology, Kaduna State University, Tafawa Balewa way, Kaduna PMB 2339, Nigeria
E-mail: aliyu.adamu.12@aberdeen.ac.uk
Received: January 13, 2018 Accepted: February 06, 2018 Published: February 13, 2018
Citation: Adamu A, Aminu HA, Aliyu F, Mienda BS (2018) The Role of Virus Receptor Recognition in the Determination of Pathogenesis. Arch Med Biotechnol 1:1.
Abstract
Viral interaction with its cellular receptor on the surface of the host cell initiates a cascade of events that lead to the entry of virus into the cell. The interaction and subsequent attachment of virus with the host cell is specific to the type of cellular receptor expressed on the cell surface. In that way virus discriminately infect certain cell lines or organism species and groups. A cellular receptor can independently mediate viral entry into the host cell, whereas in some cases, other co-receptors or even substances produced as a result of signal transduction are required. Herein, we review the cellular receptors of some medically important viruses and their contributions in the viral infection.
Keywords: Virus cellular receptors; Infection; Pathogenesis
Introduction
All pathogens must adhere to host cells before infection occur; otherwise, they will be washed away by the host clearance mechanism. While adherence to the host cell is usually the first step in bacterial pathogenesis, for viruses and other intracellular pathogens it is prerequisite for invasion [1]. Therefore viral attachment to host cell is one of the major factors that determine viral Pathogenicity. Viruses attach to their host cell in different ways, depending on the structure and compositions of the virus as well as the type of the cell they interact with. Host cell determines, which virus it will interact with via expression of molecules known as virus receptor proteins or glycoproteins on outer membrane surface. For example mucin glycoprotein expressed on surface of the respiratory epithelial cells interacts with as influenza A virus through it terminal sialic acid on the extracellular domain [2]. Virus receptors are specific, in the sense that, they only bind discriminately to only complementary epitopes on the viral surface. Although after the attachment, many processes occur before the virus gain entry into the host cell. A virus may have more than one receptor; similarly, a single receptor may be common for different viruses, for instance coxsackie virus-adenovirus receptor (CAR), which mediates the attachment of both viruses [3].
Although virus-cell interaction via attachment receptor is crucial for the viral invasion, interactions of co-receptors are also required to mediate the viral entry into the host cell [4]. It is impractical to discus the receptors of all viruses with respect to their contribution in pathogenesis, thus, this review focus on few virus groups of utmost medical importance.
Human immunodeficiency virus
Human immunodeficiency virus (HIV) is the causative agent of the popular sexually transmitted pandemic syndrome, acquired immunodeficiency syndrome (AIDS). HIV is a member of retrovirus family with single stranded RNA genome [5]. The principal receptor for HIV is CD4 [6]. However, the interaction of the virus glycoprotein (gp120) with this receptor is not sufficient for the virus to gain entry into the target cell as it only provides attachment. Studies showed several chemokines receptors, termed as co-receptors contribute in fusion, hence, infection of the virus [3]. Although, not all the coreceptors are identified, CXCR4 was shown to mediate the entry of T cell tropic HIV strain. Both CD4 and CXCR4 interact with and effect conformational changes in gp120 of the virus, thus resulting to membrane fusion and subsequent entry into the recognised T cell [7]. It has been shown that transfection of cells with small interfering RNAs that are homologous to a motif in the mRNA that codes for CXCR4 down-regulates the expression of the chemokine receptor on the surface of the cells, and consequently inhibits the fusion of HIV-1 [8]. CCR5 is another chemokine receptor identified to mediate entry of macrophage tropic HIV strain [9]. Binding of CCR5 is reduced by 87% when antibody inhibits the CD4 interaction with the virus gp120; hence CCR5 binding is CD4-dependent [10]. But despite that, CCR5 is required for the viral entry, because mutation by deleting 32bp in gene that encodes CCR5 confers partial resistance to infection with macrophage tropic HIV strain in individuals that carried such mutation [3]. Interactions of gp120 with CD4 and CCR5/CXCR4 induce the metastable envelope subunit, gp41 to undergo the conformational changes required for the viral fusion [11]. The amino acid content and charge of the variable region of gp120 determine the viral tropism for different cell lines [12]. Coreceptor might also determine the tropism of HIV. For example a recent study reported that variations in the amino terminus and the extracellular loop 1 and loop 2 of CCR5 inhibit zoonoses [13]. However, mutations in the viral envelope protein may occur during the progression of the infection, which may change the tropism of the virus, for instance, from macrophage tropic to T cell tropic [3].
Most HIV-1 strains are transmitted horizontally by sexual contact and vertically from mother to infant. This selective transmission may be as a result of selective tropism of CCR5 on dendritic cells and macrophages of mucosal and reproductive tract lining [12]. This idea was supported by the findings of Ostrowski et al., from a crosssectional study carried out to evaluate the expression of the HIV coreceptors, CCR5 and CXCR4 in context of cellular activation marker and stage of the disease by using whole blood of HIV infected and uninfected individuals [14]. Their findings revealed that, in the early stage of the infection, the expression of CCR5 in CD4+ T cells is upregulated and CXCR4 in macrophages is significantly down regulated in HIV infected individual compared to uninfected controls. This may favour the multiplication of macrophage tropic HIV strain.
Apart from attachment mediation, CD4 high affinity for interaction with the virus envelope gp120 triggers the initiation of cyncytium and cyncytium-independent cytopathic effect [15]. Host immune response reacts to that, by producing CD4 specific antibody and soluble CD4 to neutralize the gp120. In an attempt to avoid recognition by this humoral immune strategy, the virus shed its envelope gp120 such that it can binds to the host cells without being neutralised by the immune system. The virus tempers with the functions of the infected cells and renders them susceptible to destruction by cell mediated host immune response such as antibody dependent cellular cytotoxic (ADCC) effectors [15]. Cytopathic effect and the autolysis of infected and non-infected cells, decrease the number of lymphoid cells in infected individuals and hence the immune suppression. Transition from predominance macrophage tropic to predominance T cell tropic virus is important step in progression of the disease [12]. The factors that are responsible for this transition are not clearly understood but may include: the reduction in production of antibodies and killer T cells, escape of T cell tropic virus from neutralizing antibodies and killer T cells, increase in viral replication due to activation of immune system caused by opportunistic pathogens and decrease in synthesis of chemokines, such as SDF-1, that inhibit infection with T cell tropic HIV-1.
Poliovirus
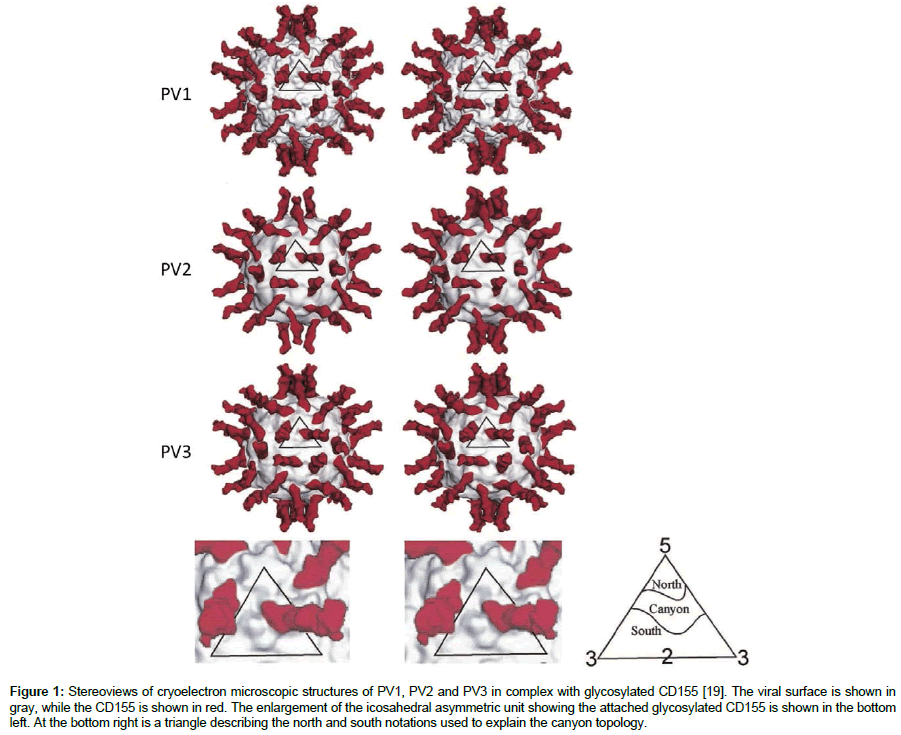
Poliovirus is enteric positive sense single stranded RNA virus with icosahedral symmetry. As a member of enteroviruses, polio virus is transmitted via ingestion through the mouth, then multiply in the lymphoid tissue of alimentary tract from where it spread in the blood to cause viraemia; and lumen of the gut [5]. Poliovirus has three serotypes PV1, PV2 and PV3, and all infect human cells by binding to poliovirus receptor (PVR) also known as CD155, a member of immunoglobulin-like super family [16]. It is composed of polypeptides containing an N-terminal signal sequence, three extracellular immunoglobulin-like domains, a trans membrane and a cytoplasmic tail [17]. The extracellular domain is expressed in four splice variants, designated hPVR-α, β, γ and δ [3]. The capsid of each of the three serotypes consists of sixty copies of each of the three viral surface proteins: VP1, VP2 and VP3 as well as sixty copies of internally located VP4 [18]. The cryoelectron microscopic structures of all three poliovirus serotypes in complex with their cellular receptors, CD155, revealed that the viral receptor binding sites (epitopes) are located in the rim and at the floor of narrow depressions in the capsid known as canyons (Figure 1) [19]. Additionally, Genetic mutation studies identified canyon as the viral binding site for CD155 [20,21]. This formed the basis of the suggestion that, antibody cannot neutralise the virus as the epitopes cannot be recognized in these narrow depressions.
Figure 1: Stereoviews of cryoelectron microscopic structures of PV1, PV2 and PV3 in complex with glycosylated CD155 [19]. The viral surface is shown in gray, while the CD155 is shown in red. The enlargement of the icosahedral asymmetric unit showing the attached glycosylated CD155 is shown in the bottom left. At the bottom right is a triangle describing the north and south notations used to explain the canyon topology.
The infection is initiated by attachment to CD155, during which the β and γ variants of the extracellular domain of the PVR are secreted, whereas α and δ serve as PVR that facilitate the virus entry into the host cell [18]. Poliovirus exclusively infects human and higher primates such as old world monkeys, as PVR is only found in cells of such animals. However, poliovirus was shown to induce neurological defects in PVR-transgenic mice, similar to those observed in man [22]. In man, PVR are expressed in many cells of different tissues, such as liver, spleen, lungs, heart, small intestine, and neurons of the spinal cord. However, the virus is not normally replicating in liver, heart and lungs [3], suggesting cell susceptibility to poliovirus is not only determined by PVR. This assumption might not hold because transmission of the virus to CNS was shown to be via mononuclear cell bearing PVR in the blood [3]. Cells of heart, lungs and other tissues, where the virus is not normally replicating might be infected, but may possess defense mechanism that can alter the activity or even inactivate the virus.
Measles virus
Measles virus (MeV) is a negative-strand RNA virus, which belongs to genus Moribillivirus of Paramyxoviridae family, which causes acute infectious disease characterized by cough, fever, conjunctivitis, generalised maculopapular rash and immunosuppression [23]. The human CD46 is the cellular receptor for MeV. Dörig and colleagues showed that hamster cell lines that expressed CD46 on the surface bind MeV and subsequently get infected, thus producing cyncytia and the viral proteins [24]. CD46 is also known as membrane cofactor protein (MCP) and it is expressed in all human cells except red blood cells [3]. However, CD46 is expressed in the red blood cells of monkey [23]. Apart from CD46, two other receptors were later identified to be associated with the entry of MeV into host cell. CD150 also known as signaling lymphocyte- activation molecule (SLAM), which its expression is restricted to activated lymphocytes; and poliovirus receptor-like protein 4 (PVRL4) otherwise known as nectin-4, also restricted to basolateral surface of epithelial cells [25]. These restrictions of expression of these receptors, is associated with the tropism of the virus in the host. Again, it was reported that C-type dendritic cells (DCs)-specific intercellular adhesion molecule 3-grabbing non-integrin (DC-SIGN) play a role in attachment of DC to the MeV, as neither the attachment nor the infection was observed in the presence of DC-SIGN inhibitor [23]. Thus DC-SIGN acts only as attachment receptor while others mediate the viral entry.
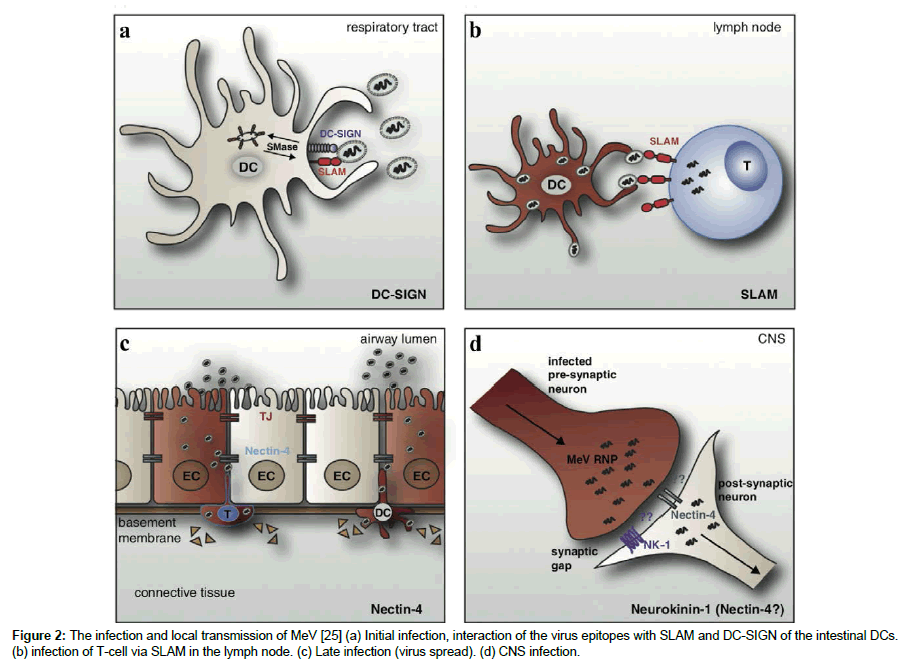
MeV infection and transmission within human host is a multistep process (Figure 2) [25]. Because the main transmission route of the virus is air, therefore the infection is initiated via DCs and the alveolar macrophages. The MeV haemagglutinin (H) attachment protein attaches to DC-SIGN. This result in a signal transduction cascade that leads to the localisation of CD150 to the cell surface through acid sphingomyleinase (SMase), and subsequently the virus gets internalised into the cell. The virus is transferred to local lymph nodes by the infected DCs, where they interact with, activate and infect T cells and B cells via CD150. Infected T cells and B cells disseminate to secondary lymphoid organs from where the virus gain entry into the blood stream to cause viramia and spread into different tissues and organs. In respiratory tract, MeV interacts with the epithelial cells via PVRL4 that they expressed exclusively. The infection can in rare cases reach the nervous system.
Figure 2: The infection and local transmission of MeV [25] (a) Initial infection, interaction of the virus epitopes with SLAM and DC-SIGN of the intestinal DCs. (b) infection of T-cell via SLAM in the lymph node. (c) Late infection (virus spread). (d) CNS infection.
Conclusion
Virus infection into host cell is multifactorial. A receptor can independently mediate entry, whereas in some cases, is a collaborative effort with other co-receptors or even substance produced as a result of signal transduction. Virus infects a cell only when it recognises it receptor on the surface of that cell, failure to do which leads to no interaction and hence no infection.
References
- Boyle EC, Finlay BB (2003) Bacterial pathogenesis: exploiting cellular adherence. Curr Opin Cell Biol 15: 633-639.
- McAuley JL, Corcilius L, Tan HX, Payne RJ, McGuckin MA, et al. (2017) The cell surface mucin MUC1 limits the severity of influenza A virus infection. Mucosal Immunol 10: 1581-93.
- Schneider-Schaulies J (2000) Cellular receptors for viruses: links to tropism and pathogenesis. J Gen Virol 81: 1413-1429.
- Hunter E (1997) Viral Entry and Receptors. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, New York, USA.
- Timbury MC (1995) Notes on Medical Virology (10 edtn) Longman, Singapore.
- Layne SP, Merges MJ, Dembo M, Spouge JL, Nara PL (1990) HIV requires multiple gp120 molecules for CD4-mediated infection. Nature 346: 277-279.
- Scarlatti G, Tresoldi E, Björndal Å, Fredriksson R, Colognesi C, et al. (1997) In vivo evolution of HIV-1 co-receptor usage and sensitivity to chemokine-mediated suppression. Nat Med 3: 1259-1265.
- Zhou N, Fang J, Mukhtar M, Acheampong E, Pomerantz RJ (2004) Inhibition of HIV-1 fusion with small interfering RNAs targeting the chemokine co-receptor CXCR4. Gene Ther 11: 1703-1712.
- Bleul CC, Wu L, Hoxie JA, Springer TA, Mackay CR (1997) The HIV co-receptors CXCR4 and CCR5 are differentially expressed and regulated on human T lymphocytes. Proc Natl Acad Sci U S A 94: 1925-1930.
- Trkola A, Dragic T, Arthos J, Binley JM, Olson WC, et al. (1996) CD4-dependent, antibody-sensitive interactions between HIV-1 and its co-receptor CCR-5. Nature 384: 184-187.
- Alsahafi N, Debbeche O, Sodroski J, Finzi A (2015) Effects of the I559P gp41 change on the conformation and function of the human immunodeficiency virus (HIV-1) membrane envelope glycoprotein trimer. Plos One 10: e0122111.
- Norcross MA (1999) Chemokine receptors and HIV-1 pathogenesis: a viral fatal attraction. HIV and the New Viruses (Second Edition): Elsevier 133-153.
- Platt EJ, Durnin JP, Kabat D (2015) Short Communication: HIV-1 Variants that Use Mouse CCR5 Reveal Critical Interactions of gp120's V3 Crown with CCR5 Extracellular Loop 1. AIDS Res Hum Retroviruses 31: 992-1008.
- Ostrowski MA, Justement SJ, Catanzaro A, Hallahan CA, Ehler LA, et al. (1998) Expression of Chemokine Receptors CXCR4 and CCR5 in HIV-1-Infected and Uninfected Individuals. J Immunol 161: 3195-3201.
- Jakobsdottir GM, Iliopoulou M, Nolan R, Alvarez L, Compton AA, et al. (2017) On the Whereabouts of HIV-1 Cellular Entry and Its Fusion Ports. Trends Mol Med 23: 932-944.
- Rueckert R. (1996) Picornaviridae: the viruses and their replication. Fields virology (3rd edtn) Lippincott-Raven Publishers, Philadelphia, USA.
- Racaniello VR (1996) The poliovirus receptor: a hook, or an unzipper? Structure 4: 769-773.
- He Y, Bowman VD, Mueller S, Bator CM, Bella J, et al. (2000) Interaction of the poliovirus receptor with poliovirus. Proc Natl Acad Sci U S A 97: 79-84.
- He Y, Mueller S, Chipman PR, Bator CM, Peng X, et al. (2003) Complexes of Poliovirus Serotypes with Their Common Cellular Receptor, CD155. J Virol 77: 4827-4835.
- Harber J, Bernhardt G, Lu H-h, Sgro J-Y, Wimmer E (1995) Canyon rim residues, including antigenic determinants, modulate serotype-specific binding of polioviruses to mutants of the poliovirus receptor. Virology 214: 559-570.
- Liao S, Racaniello V (1997) Allele-specific adaptation of poliovirus VP1 BC loop variants to mutant cell receptors. J Virol 71: 9770-9777.
- Deatly AM, Coleman JW, McMullen G, McAuliffe JM, Jayarama V, et al. (1999) Poliomyelitis in intraspinally inoculated poliovirus receptor transgenic mice. Virology 255: 221-227.
- Yanagi Y, Takeda M, Ohno S (2006) Measles virus: cellular receptors, tropism and pathogenesis. J Gen Virol 87: 2767-2779
- Dörig RE, Marcil A, Chopra A, Richardson CD (1993) The human CD46 molecule is a receptor for measles virus (Edmonston strain). Cell 75: 295-305.
- Delpeut S, Noyce RS, Siu RW, Richardson CD (2012) Host factors and measles virus replication. Curr Opin Virol 2: 773-783.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi