Research Article, J Liver Disease Transplant Vol: 12 Issue: 1
The Toxicological Effect of Lead on Liver Histology and Biochemical Parameters: A Systematic Review
Tefera Belsty1, Wubshet Nebiyu1* and Mezgebu Legesse2
1Department of Anatomy, College of Health and Medical science, Haramaya University, Harar, Ethiopia
2Department of Biochemistry, College of Health and Medical Sciences, Haramaya University, Harar, Ethiopia
*Corresponding Author: Nebiyu W
Department of Anatomy, College of Health and Medical science, Haramaya University, Harar, Ethiopia
E-mail: wubenebiyu21@gmail.com
Received date: 13 January, 2023, Manuscript No. JLDT-23-87070;
Editor Assigned date: 16 January, 2023, PreQC No. JLDT-23-87070 (PQ);
Reviewed Date: 30 January, 2023; QC.No. JLDT-23-87070;
Revised Date: 10 February, 2023, Manuscript No. JLDT-23-87070 (R);
Published date: 17 February, 2023, DOI: 10. 4172/2325-9612.1000216.
Citation: Belsty T, Nebiyu W, Legesse M (2023) The Toxicological Effect of Lead on Liver Histology and Biochemical Parameters: A Systematic review. J Liver Diseases 12:1.
Abstract
Introduction: Although lead is the fifth most commonly used metal in the world, it can cause a variety of human health hazards. Lead is an environmentally persistent toxin that can cause neurological, hematological, circulatory, reproductive, liver, and kidney diseases. It has been found to affect virtually every organ of one or more systems in both humans and animals. The aim of this study was to review scientific empirical literature on the toxic effects of lead exposure on liver histology and biochemical parameters. Method: Data were collected from online databases, Google Scholar, PubMed, CINAHAL and Google. Studies were evaluated by focusing on lead dose, exposure duration, and the type of test animals used to measure the effects of lead. The literature reviewed was from published work only. Results: Histological studies showed that lead can induce various changes such as hypertrophy of hepatocytes, portal vein space and central vein dilatation, vacuolation and lymphocytic infiltration. It was also shown that liver enzyme levels were increased in treated rats compared to controls. Conclusion: This study provides a comprehensive rationale for recent updates describing the health effects of lead exposure, relevant biomarkers, and the mechanisms involved in lead toxicity
Keywords: Histological alterations; Lead acetate; hepatocyte; Toxicity; Hematology
Abbreviations
ALT: Alanine Transaminase; AST: Aspartate Transaminase; ALP: Alkaline Phosphatase
Introduction
Lead is commonly found as lead sulphide from subsurface earth and is widely diffused from the atmosphere due to various human activities. Lead is the fifth most used metal worldwide. Because of its chemical properties, lead is used in various industries such as the battery industry, mining and smelting activities that can emit lead into the atmosphere. Once distributed in the atmosphere, lead has a persistence capacity in the global atmosphere [1]. World Health Organization report Lead as one of ten toxic substances in human health and US Agency on Toxic Substances and Disease Registries also report that lead is the second most dangerous toxic substance in the world [2]. Lead can also be found in air, dust, soil and water in varying degrees. Any route of the human route can induce exposure by ingestion or inhalation and dermal absorption. Human exposure can be assessed directly by measuring body exposure or indirectly by measuring environmental lead levels [3].
Lead is an environmentally persistent toxin that causes neurological, hematological, gastrointestinal, reproductive, circulatory, and immunological pathologies. Liver and kidney damage has been reported in some cases [4,5]. Lead absorbed from the environment binds to erythrocytes and can reach soft tissues such as the kidneys, liver, brain, muscles, heart and spleen via the blood. Eventually, most of it gets deposited in the bones and teeth [6]. The liver is histologically divided into microscopic masses of cells functionally arranged around the terminal veins in an acinar unit. Blood flows from the terminal portal veins into the sinusoids and comes in contact with hepatocytes of liver cells that are 13- 30 in diameter and have eight or more surfaces. The hepatocyte of the microvilli faces the perisinusoidal space and extends into the pericellular space [7].
The half-life of lead in the blood of adult humans has been estimated to be 28 to 36 days [8,9]. Almost 99% of lead in red blood cells; the remaining 1% is in the blood plasma. However, it is blood plasma that transfers lead between the blood compartment and the soft and mineralizing tissues and is therefore potentially more biologically significant [10,11]. Many lead-based products still pose a health risk. One of the many life-sustaining functions of the liver is to filter all circulating toxins from the bloodstream. The liver is one of the first major organs to be exposed to ingested chemicals due to its portal blood circulation. The liver does not filter lead toxins from the body when lead toxins are overexposed. Therefore, this study article provides comprehensive information on lead toxicity to the liver that could generate widespread public awareness.
Materials and Methods
Databases such as Google Scholar and PubMed were searched to repossess the papers for this study. Selected works date between 2009 and 2022. The search methodology can be verified as follows: after selecting a set of search terms and generating reliable combinations, we used electronic research databases to search for related articles. The search strategy was used to find all relevant studies in the search database by using major Boolean operators with initial keywords (effect OR toxicity AND lead AND liver). Posts that did not meet any of the search criteria were excluded. The reference lists and bibliographies of all articles were then checked for additional published and unpublished studies. Then we selected a maximum of 16 papers that met the selection criteria.
Inclusion criteria in this review
Systemic review was performed on experimental studies that were standardized, valid, and reliable histological techniques to identify alternation of liver histology and biochemical parameters. At least one functional alteration of the liver due to lead toxicity. Papers not related to the chosen topic, without a brief notice and not in English were excluded. Full-text articles in English published between 1976 and 2022 were included.
Results
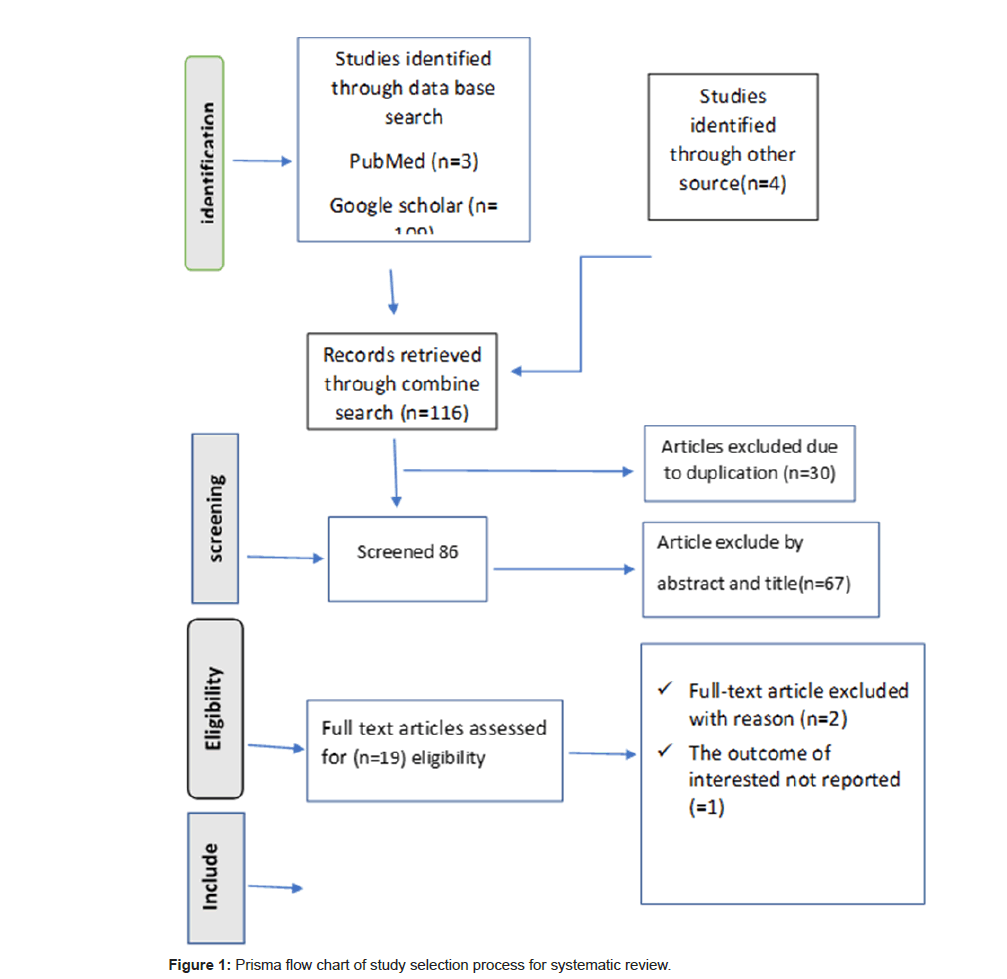
Search A total of 116 articles from multiple sources were identified in our literature using a lead liver toxicity search strategy on PubMed, Google Scholar, CINHAL and other non-publications on Google. Of these 30 duplicates, the article was removed with ENDNOTE and manual tracking. After removing duplicates, 86 studies remained. 86 records were searched by their abstract and title and 68 record articles were excluded. A full-text review was performed for 19 articles and 3 articles were excluded. The remaining 16 articles met the eligibility criteria and were included in the studies of this literature review (Figure 1).
Effect of lead exposure on liver and biochemical parameters
Regarding effect of lead exposure on liver several researchers using different animal models, and using various methodologies have been conducted on the effect of lead exposure on histology and function of the liver. Some of these studies have been reviewed and analyzed in this study paper (Table 1).
| SI.No | Animal Model | Route of exposure | Dose | Duration | Observation | Reference |
|---|---|---|---|---|---|---|
| 1 | albino mice | Orally | Variable | 28 days | The finding indicated that more fatty degeneration, a large area of the hepatic lobule, vacuolar degeneration and hemorrhage between hepatic cords, Tissues were very scanty | [12] |
| 2 | Rats | Orally | Variable | 28 days | Significant decrease in body weight Gross liver was enlarged, pale, swelling of hepatocytes, granular degeneration Increased eosinophilic granularity of hepatocytes cytoplasm Diffuse infiltration of mononuclear cells around the portal triad | [13] |
| 3 | Rats | Administered in drinking water | Variable | >10 month | Chronic portal triad and the periportal zones inflammation appeared, Kupffer cells hyperplasia, Binucleation | [14] |
| 4 | Rats | Orally | 8.5 mg/l | 20 days | vacuolation and fatty degeneration in the hepatocytes, congestion within central veins and sinusoids, the sinusoidal Kupffer cells showed a prominent appearance | [15] |
| 5 | Rats | Orally | Variable | 8 weeks | More loss of hepatic architecture, hepatic cells necrosis, Moderate increase in the amount of the periportal fibrous tissue and also radiating in between the cord of hepatocyte | [7] |
| 6 | Rats | Orally | 2 g/l | 35 days | liver weight increases defused disorganization of hepatic cords, vascular congestion, mild lymphoid and mononuclear infiltration, and a significant increase in plasma ALT and AST concentration | [16] |
| 7 | Rats | Orally | Variable | 4 weeks | Diffuse affection of hepatic lobule, highly vacuolation of cytoplasm nuclei. | [17] |
| 8 | Chicken | feeding | 50,100,200 ppm | 3 weeks | Live hemorrhage was observed. AST and ALP were increased | [18] |
| 9 | Rats | orally | 150 mg Pb/kg | ALT, AST, and ALP increase a significant effect at the higher dose | [2] | |
| 10 | Men | Work Exposed to lead | - | 6 month | ALP, ALT, and AST were significantly elevated | [19] |
| 11 | Men | Work expose to lead | - | - | ALP and ALT levels significant elevation but AST | [20] |
| 12 | Japanese quails(birds) | Orally | 0.4 mg Pb/kg | 21days | AST and ALT significantly increased | [21] |
| 13 | Rat | Orally | 60 mg pb/kg | 45 days | Histopathology of liver changes like micro vesicular fatty changes, focal loss of hepatocytes and bile ductular hyperplasia and in kidney desquamated renal tubular epithelium, disruption of renal tubules and inter tubular hemorrhages | [22] |
| 14 | Human | Work exposure | - | More than six hours per day for 2-20 years | Blood lead and urine lead level significantly increased. AST, ALT, ALP, and GGT levels were significantly increased. Serum total protein, serum albumin, globulin and A/G ratio were decreased whereas, serum creatinine and serum uric acid level were declined | [23] |
| 15 | Wistar albino rats | Orally | Variable | 7 day | Alteration in hepatocytes mainly anisokaryosis, neacuar vasculation, bineaculation, cytoplasmic inclusion, cytoplasmic swelling, necrosis and redaction in glycogen level was observed. Additionally, kupffer cell hyperplasia and no cirrhosis was detected. | [24] |
| 16 | albino rats | Orally | 0.5 g/100ml | Two months | The amount of serum Aspartate Aminotransferase (AST), Alanine Aminotransferase (ALT), Alkaline Phosphate (ALP), Gamma Glutamyltransfrase (GGT), Lactate Dehydrogenases (LDH) and bilirubin and cholesterol level were increased as compare to control group but total protein and erythrocyte account was decreased compare to control group | [25] |
Table 1: Lead and its toxicity in different experimental studies.
Discussion
Possible mechanism of lead toxicity to liver
Below The scientific literature reviewed in this article showed that exposure to lead produces the histopathological effect, hepatotoxicity, biochemical change and impairment of enzyme activities and impairment of liver habituation [12-15].
Lead and serum enzyme
The histopathological effect of lead on the liver showed that serum Alanine Aminotransferase (ALT), Aspartate Transaminase (AST) and Alkaline Phosphatase (ALP) increased compared to the control group. ALT, AST, and ALP elevations in animals appeared to be directly proportional to lead strength [2,16,17].
Lead and body weight
The evaluation of the body weight of lead-poisoned rats was significantly lower than that of the healthy group (control group). Body weight gain was reduced with increasing lead exposure [13,16]. The reduced body weight can be linked to several factors; one of the factors can be the imbalance of metabolic enzymes.
Lead and liver weight
The increase in organ weight was thought to be due to reticular cell hyperplasia as a defense mechanism, necrosis and apoptosis, which could be attributed to the accumulation of lipids in the organ [13,17].
Lead and histopathologic changes
The most common histopathologic findings in the liver were vacuolation, fatty degeneration, congestion in the central veins, and loss of normal structure in the hepatocytes [12,16,18-20]. According to these researchers, the changes may be due to lead acetate decreasing the enzyme. The review conducted by Negm showed different degrees of histological changes in liver tissue in experimental groups compared to the control group; some of the hepatocytes appeared with acidophilic, slightly vacuolated granular cytoplasm, others showed markedly vacuolated cytoplasm, and others with hypereosinophilic cytoplasm [17]. Mononuclear cell infiltration has been observed in the portal vein [7,13,21,22]. According to researchers, these changes were duration-dependent; the longer the duration, the greater the damaging effects. Histological analysis of the liver revealed several changes, such as B. Hepatocyte hypertrophy and disorganization of the liver cords. The hypertrophy was due to swelling of intracellular organs, particularly the mitochondria and endoplasmic reticulum. In addition, liver toxicity manifests itself in the form of cellular vacuolation, which is a cellular defense mechanism against harmful substances [13,16]. After treatment with lead, lymphocytic infiltration and sinusoidal congestion are signs of liver damage. The lymphocytic infiltrates observed in this review after lead treatment showed evidence of cell irritability, inflammation and hypersensitivity to the toxin used [23,24].
Hematological effect of lead exposure
An experimental review conducted in Egypt indicates that lead exposure can lead to a decrease in hemoglobin levels and the total amount of erythrocytes. As a result, lead-induced anemia can occur due to the reduced lifespan of erythrocytes [25]. The decrease in hemoglobin and erythrocyte count is due to the toxic effect of lead and is related to the increase in plasma bilirubin as a result of induction of heme oxygenase [26].
Effect of lead exposure on cholesterol levels
Various researchers report that lead exposure to the liver increases the body’s total cholesterol levels [24,25]. The increase in cholesterol can be either an increase in the synthesis or a decrease in the removal of lipoproteins. The decrease in lipoprotein removal is due to the change in the surface cell receptor for lipoprotein [25]. Other researchers also suggest that lead had the ability to quench cytochrome p-450, allowing it to limit bile acid biosynthesis, which is an important pathway for the removal of cholesterol from the body, as suggested by Alvares et al. [27].
Effect of lead exposure on kupffer cells and glycogen amount
Studies show that lead had the ability to activate the phagocytic activity of sinusoid cells by increasing Kupffer cells. This could be due to autophagy of liver tissue to remove excess lead from the body as suggested by Neyrinck AJ et al. [28]. Various studies have confirmed that lead exposure in the liver can reduce the amount of glycogen, which may be due to lead’s effect on glucose absorption and enzymes involved in glycogenesis [24].
Conclusion
In summary, lead has no recognized biological function in the body. The results of various studies considered in the present publication report that lead causes considerable damage to liver by accelerating induced hepatic weight, vacuolation, fatty degeneration, occlusion within a central vein and hypertrophy of hepatocytes and disorganizing hepatic marrow. All studies examined showed similar toxic effects of lead in the histology of the liver, regardless of the dose used and the duration of the different experiments.
Acknowledgements
The authors acknowledge the support of Haramaya University for helping with the overall outcome of the manuscript.
Author’s Contributions
Tefera Belsty continued the literature review and authored the paper in addition to that Wubshet Nebiyu and Mezgebu Legesse provides guidance, literature organization, critical assessment and peer review of the writing. All authors have given their final approval for the publication of this version. All authors read and approved the final manuscript.
References
- Nakhaee S, Amirabadizadeh A, Brent J, Mehrpour O (2019) Impact of chronic lead exposure on liver and kidney function and haematologic parameters. Basic Clin Pharmacol Toxicol 124(5):621-628.
[Crossref] [Google Scholar] [Pub Med]
- Andjelkovic M, Buha Djordjevic A, Antonijevic E, Antonijevic B, Stanic M, et al., (2019) Toxic effect of acute cadmium and lead exposure in rat blood, liver, and kidney. Int J Environ Res Public Health 16(2):274.
[Crossref] [Google Scholar] [Pub Med]
- Karrari P, Mehrpour O, Abdollahi M (2012) A systematic review on status of lead pollution and toxicity in Iran; Guidance for preventive measures. DARU J Pharm Sci 20(1):1-7.
[Crossref] [Google Scholar] [Pub Med]
- Sharma V, Sharma S, Pracheta SS, Sharma SH (2011) Lead induced hepatotoxicity in male swiss albino mice: The protective potential of the hydromethanolic extract of Withania somnifera. Int J Pharmaceu Sci Rev Res 7(2):116-121.
- Shalan MG, Mostafa MS, Hassouna MM, El-Nabi SH, El-Refaie A (2005) Amelioration of lead toxicity on rat liver with vitamin C and silymarin supplements. Toxicology 206(1):1-5.
[Crossref] [Google Scholar] [Pub Med]
- Meyer PA, Brown MJ, Falk H (2008) Global approach to reducing lead exposure and poisoning. Mutat Res 659(1-2):166-175.
[Crossref] [Google Scholar] [Pub Med]
- Hegazy AM, Fouad UA (2014) Evaluation of lead hepatotoxicity; histological, histochemical and ultrastructural study. Frn Med Antmy Res 2(03):70.
- Griffin TB, Coulston F, Wills H (1975) Biological and clinical effects of continuous exposure to airborne particulate lead. Arh Hig Rada Toksikol 26(Supplement):191-208.
- Rabinowitz MB, Wetherill GW, Kopple JD (1976) Kinetic analysis of lead metabolism in healthy humans. J Clin Invest 58(2):260-270.
[Crossref] [Google Scholar] [Pub Med]
- deSilva PE (1981) Determination of lead in plasma and studies on its relationship to lead in erythrocytes. Occup Environ Med 38(3):209-217.
[Crossref] [Google Scholar] [Pub Med]
- Everson J, Patterson CC (1980) "ULtra-clean" isotope diultion/mass spectrometic analyses for lead in human blood plasma indicated that most reported values are artificially high. Clin Chem 26(11):1603-1607.
[Crossref] [Google Scholar] [Pub Med]
- A El-Nouri A, H Jankeer M (2009) Histological study of the liver and kidney of albino mice Mus musculus exposed to lead. JSN 20(3):42-51.
- Suradkar SG, Vihol PD, Patel JH, Ghodasara DJ, Joshi BP et al., (2010) Patho-morphological changes in tissues of Wistar rats by exposure of Lead acetate. Vet World 3(2):82-84.
- Bharali MR (2013) Effect of acute lead acetate exposure on liver of mice. J Glob Biosci 2(5):121-125.
- Dehkordi KK, Dehkordi SK, Dehkordi RA (2015) Histopathological study of the rat liver exposed with lead acetate as a microscopic survey. Animal Vet Sci 3(5):141-143.
- Haouas Z, Sallem A, Zidi I, Hichri H, Mzali I, et al., (2014) Hepatotoxic effects of lead acetate in rats: Histopathological and cytotoxic studies. J cytol Histol 5(5):1-6.
- Negm FA, El-din RAS, Nabil EM (2015) Anatomical and histological study of the effect of lead on hepatocytes of albino rats. Int j biomed mater res 3(4): 34-45.
- Ashrafizadeh M, Rafiei H, Ahmadi Z (2018) Histological changes in the liver and biochemical parameters of chickens treated with lead acetate II. Iran J Toxicol 12(6):1-5.
- Onyeneke EC, Omokaro EU (2016) Effect of occupational exposure to lead on liver function parameters. Int J Pharm Med Sci 6:15-9.
- Alsaeed AH, Shaik AP, Faiyaz-ul-Haque M, Tabassum S, Alsaeed AM et al., (2017) Evaluation of blood lead levels and their association with haematological and liver function test parameters in Saudi workers from Riyadh region, KSA. Biomed Res 28(4):1-4.
- Hamidipour F, Pourkhabbaz H, Banaee M, Javanmardi S (2016) Bioaccumulation of lead in the tissues of Japanese quails and its effects on blood biochemical factors. Iran J Toxicol 10(2):13-21.
- Paul A, Sujatha K, Srilatha CH, Kumar NV (2022) Effect of Lead toxicity on liver and kidney in Wistar rats and its amelioration with Linum usitatissimum (flaxseed) and Emblica officinalis (amla). 46(1):49-58.
- Dongre Nilima N, Rathi SA (2010) Occupational Lead Exposure In Automobile Workers In North Karnataka (India): Effect On Liver And Kidney Functions.
- Jarrar BM, Taib NT (2012) Histological and histochemical alterations in the liver induced by lead chronic toxicity. Saudi J Biol Sci 19(2):203-210.
[Crossref] [Google Scholar] [Pub Med]
- Hanan AA, Riham MR (2012) Effect of lead toxicity on cytogenisity, biochemical constituents and tissue residue with protective role of activated charcoal and casein in male rats. Aust J Basic Appl Sci 7(6):497-509.
- Ibrahim NM, Eweis EA, El-Beltagi HS, Abdel-Mobdy YE (2012) Effect of lead acetate toxicity on experimental male albino rat. Asian Pac J Trop Biomed 2(1):41-46.
[Crossref] [Google Scholar] [Pub Med]
- Alvares AP, Fischbein A, Sassa S, Anderson KE, Kappas A (1976) Lead intoxication: Effects on cytochrome P-450-mediated hepatic oxidations. Clin Pharmacol Ther 19(2):183-190.
[Crossref] [Google Scholar] [Pub Med]
- Neyrinck AJ (2004) Modulation of Kupffer cell activity: Physio-pathological consequences on hepatic metabolism. Bull Mem Acad R Med Belg 159(5-6):358-366.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi