Research Article, J Spine Neurosurg Vol: 6 Issue: 3
Transforaminal Endoscopic Decompression for Painful Degenerative Conditions of The Lumbar Spine: A review of One Surgeon’s Experience with Over 10,000 Cases Since 1991
Yeung AT*
University of New Mexico School of Medicine, Albuquerque, New Mexico, Desert Institute for Spine Care Phoenix, Arizona, USA
*Corresponding Author : Dr. Anthony T. Yeung
Voluntary professor, University of New Mexico School of Medicine, Albuquerque, New Mexico, Desert Institute for Spine Care, Phoenix, Arizona, USA
Tel: +1 602-944-2900
E-mail: ayeung@ sciatica.com
Received: December 09, 2016 Accepted: February 15, 2017 Published: February 21, 2017
Citation: Yeung AT (2017) Transforaminal Endoscopic Decompression for Painful Degenerative Conditions of The Lumbar Spine: A review of One Surgeon’s Experience with Over 10,000 Cases Since 1991. J Spine Neurosurg 6:3. doi: 10.4172/2325-9701.1000266
Abstract
Background: Endoscopic Spine Surgery has evolved rapidly in the past 5 years, and is getting less pushback by traditional spine surgeons who have no exposure or training in endoscopic spine surgery in their training. The author has dedicated his last 25 years to transforaminal endoscopic surgery. He has previously reported on his 12 years’ experience in 2007 in IJAS Surgery, but 13 years later, continued evolution of the transforaminal approach aided by new endoscopes and endoscopic instrumentation has allowed the author and experienced endoscopic surgeons adopting this approach, with the ability to treat 90 percent of painful degenerative conditions of the lumbar spine after the learning curve is conquered.
Methods: Three different methods have evolved. Yeung’s “insideout” philosophy and technique, Hoogland’s “outside in” philosophy technique, and a hybrid targeted technique combining inside out and outside in, that depends on targeting the patho-anatomy. This paper reviews Yeung’s philosophy and technique backed by over
10,000 procedures in the past 25 years
Results: After a learning curve for each pathology targeted, an overall 90% good to excellent success rate measured by VAS, Oswestry, patient satisfaction, and minimal minor complications of less than 3 percent can be achieved. A detailed review of Yeung’s experience supplements his 12 year report in the International Journal of Spine Surgery in 2007.
Conclusion: The transforaminal endoscopic technique has evolved to be the least invasive, most effective method to surgically address the pain generators in the lumbar spine. The technique allows for earlier treatment of painful conditions that fail nonsurgical treatment. This monograph can be used as a guide for new surgeons who want to treat patients with “surgical pain management” that incorporates pain management with surgical treatment that will help decrease the escalating cost of spine care all over the world.
Keywords: spine
Introduction to Transforaminal Endoscopy: Philosophy and the YESS Technique
I was a trained as a general orthopedic surgeon in 1971 when orthopedic surgery included arthrocopic surgery of painful joints, joint replacement, and lumbar translaminar decompression. Open translaminar decompression was a standard procedure for herniated discs, and stenosis. Fusion was reserved for instability after decompression. In 1991, I took Parviz Kambin’s course in arthroscopic spine surgery that soon sparked an interest that has not waned. It seemed natural to me to utilize the endoscope to examine patho-anatomy in the spine. I quickly decided to not just visualize the nucleus being removed as emphasized initially by Kambin, but also visualize the epidural space through the foramen after decompression of the disc with the “inside out” philosophy and technique to obtain confirmation that the spinal nerves were decompressed. In 1991, the laser was also introduced for the lumbar spine, and I became a fan of laser as a surgical tool for decompression under endoscopic visualization because it had the tissue effect of decreasing bleeding and ablating soft tissue and bone. This provided relief not just for sciatica, but back pain. My experience caused me to develop my own endoscopic system in conjunction with Richard Wolf Surgical Instrument system, and marketed the FDA approved system in 1997, called the Yeung Endoscopic Spine System (YESS) (Figure 1). I emphasize that the surgery I describe is a surgical procedure that is best performed by surgeons with extensive open surgery experience since surgical complications as well as potential complications are best recognized by trained spine surgeons with experience in handling complications and not appropriate for specialties without training in surgical complications and techniques. It should not be confused by pain management philosophy just because needle procedures are an integral part of operating on the patho-anatomy.
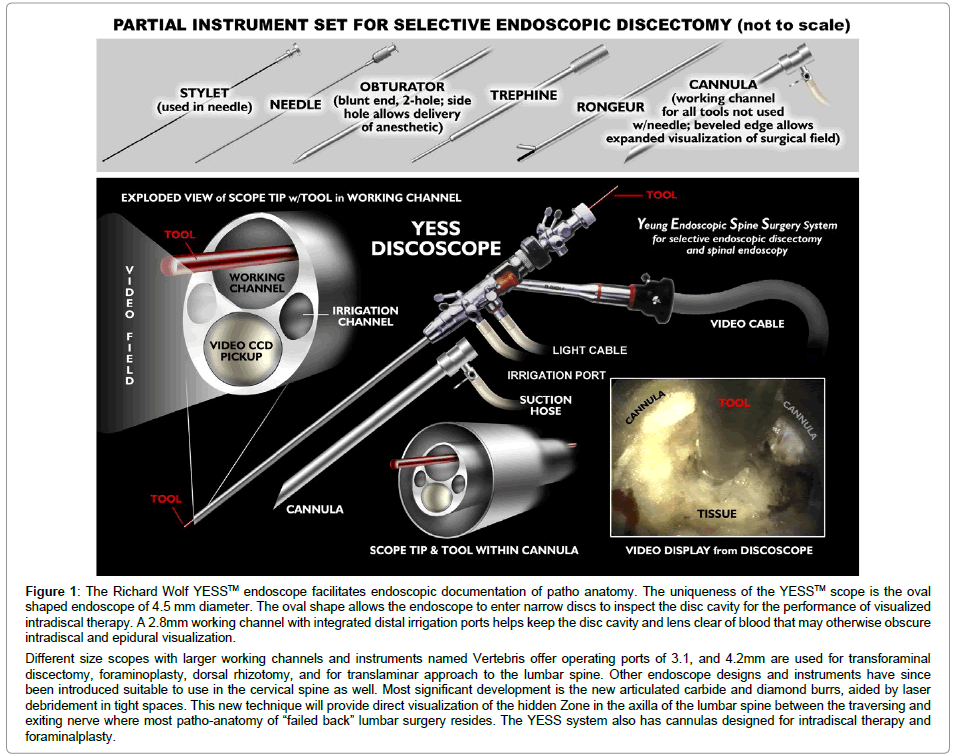
Figure 1: The Richard Wolf YESSTM endoscope facilitates endoscopic documentation of patho anatomy. The uniqueness of the YESSTM scope is the oval shaped endoscope of 4.5 mm diameter. The oval shape allows the endoscope to enter narrow discs to inspect the disc cavity for the performance of visualized intradiscal therapy. A 2.8mm working channel with integrated distal irrigation ports helps keep the disc cavity and lens clear of blood that may otherwise obscure intradiscal and epidural visualization.
Different size scopes with larger working channels and instruments named Vertebris offer operating ports of 3.1, and 4.2mm are used for transforaminal discectomy, foraminoplasty, dorsal rhizotomy, and for translaminar approach to the lumbar spine. Other endoscope designs and instruments have since been introduced suitable to use in the cervical spine as well. Most significant development is the new articulated carbide and diamond burrs, aided by laser debridement in tight spaces. This new technique will provide direct visualization of the hidden Zone in the axilla of the lumbar spine between the traversing and exiting nerve where most patho-anatomy of “failed back” lumbar surgery resides. The YESS system also has cannulas designed for intradiscal therapy and foraminalplasty.
My experience in surgical management of back and leg pain for the past 25 years culminated in the need for surgery for painful conditions in my own aging spine. I had a degenerative spinal condition that ultimately resulted in needed surgery for my age related progression to deformity, stenosis, instability, and progressive neurologic deficit from a multi-modal herniated disc. I lived through the symptoms produced by the natural process of my aging spine. I ultimately opted for surgical intervention when I became debilitated from my work as an endoscopic MIS surgeon. In retrospect, with all the available treatment options I provide my patients, I would have been relieved by endoscopic surgical intervention about a year or two earlier. I had prodromal symptoms that I ignored until I had muscle atrophy and a neurologic deficit. I conclude, following my own surgery, that surgical intervention should not be a “last resort”, but placed in the spectrum of care needed and desired by each individual patient for his or her spinal condition as a joint decision by the patient and his surgeon for treatment options. The treatment and procedure(s) available and offered may differ widely, dependent on the state of the art in MIS techniques currently available as well as the technical skills and experience of the surgeon.
My condition, degenerative scoliosis, grade I spondylolisthesis, trimodal HNP, and lumbar spondylosis with facet arthrosis, was acquired due to my spine’s aging process that is traditionally treated by current spine specialists with multi-level decompression, stabilization and fusion as the “evidence based”, and “best practice” intrumented surgical solution backed by EBM literature. This “state of the Art” treatment is now promoted and defended by traditional spine surgeons as the proper algorithm for surgical intervention with emphasis on correcting femoral lumbar and saggital alignment.
Framed by this backdrop of my spinal condition, I remained active and productive, practicing endoscopic surgery until the day before my first surgery on May 7, 2014 for severe radiculopathy at L4-5. I also had associated three level foraminal spinal stenosis, degenerative scoliosis, and grade I spondylolisthesis that culminated in a staged procedure of Coflex for mild instability and deformity following transforaminal endoscopic decompression. I went back to work 2 days after transforaminal endoscopic decompression of my HNP at L4-5, based on my surgical philosophy of considering the least invasive procedure first. If my condition eventually requires fusion, I can still pursue that option without burning any bridges for surgical options needed for my own individual requirements. I now report, almost three years later, my firsthand experience with this philosophy and surgical techniques designed to address the most symptomatic condition, staged in the backdrop of a progressive and aging degenerative spine. I had the least invasive procedure performed on myself first, under local anesthesia, and without sedation. I trusted my son, whom I trained to utilize the transforaminal endoscopic technique, as the most qualified surgeon available to perform the procedure on me. Still, experience and the development of surgical skills in the context of clinical experience will influence how each MIS spine surgeon will place MIS surgery to the spectrum of treatment offered. This monologue will describe my firsthand experience with the procedure trademarked selective endoscopic discectomy (SED).
The treatment algorhythim in spine is evolving and changing due to a better understanding of the patho-anatomy, well correlated with its pathophysiology. I have published my evolving technique in past publications, first in a series of articles in Surgical Technology International, beginning with the VIII edition, and followed by the XI, XV, and XXI edition. These articles in the literature were followed by three short solicited articles in open access journals [1], “Moving away from fusion” in Spine, [2] “Transforaminal Endoscopic Surgery: Its Role in the Treatment of Painful Conditions of an Aging Spine” [3] and “Intradiscal Therapy and Transforaminal Endoscopic Decompression: Opportunities and Challenges for the future” in Journal of Neurologic Disorders [1-3].
Pain is better understood with in vivo visualization and probing of the pain generators using endoscopic transforaminal access rather than just relying on a symptom diagram and image correlation This culminates in a shared decision making process involving patient and surgeon, focused on a broader spectrum of surgical as well as non-surgical treatments, and not just masking the pain generator. It has moved away from decisions based on symptom diagrams and diagnostic images alone that, while noting the image abnormalities, cannot always explain the pain and disability experienced by each individual patient. Images do not always show variations in nerve supply and patho-anatomy, nor do they quantify the pain experienced by each individual patient. The ability to isolate and visualize “pain” generators in the foramen and treating persistent pain by visualizing inflammation and compression of nerves, serves as the basis for transforaminal endoscopic [TFE] surgery [3,4]. This has also resulted in better pre surgical planning with more specific and defined goals in mind. Diagnostic and therapeutic injections also help predict the result for transforaminal decompression once the anatomic source of pain is determined. There are also various surgical philosophies and techniques proposed by other pioneers in endoscopic surgery for treating these conditions, but I choose to embrace the “inside out” philosophy of TFE surgery as the most safe and precise in trained and good surgical hands. It provides basic access to the disc and foramen that cover a large spectrum of painful pathologies with the least surgical risk.
I begin with a personal account of my own spinal condition that deteriorated rapidly over the past 5 years. It began with an occasional backache that would improve with rest and anti-inflammatory overthe- counter medications. I tolerated the discomfort, as it never debilitated me until the past three months before I opted for MIS surgery when the pain became constant and no longer relieved by the usual rest, activity modification, and anti-inflammatory medications. I never depended on narcotic analgesics. The first X-rays were taken three years previously when I was having a constant low level back ache (VAS 2-3) but required no active treatment. I would experience sclerotomal and dermatome tingling and numbness radiating bilaterally, in no particular dermatome distribution, although predominantly in the right L5 and S-1 distribution. This was relieved by Ibuprofen 600-800 mg. X-rays at that time identified an aging spine with lumbar spondylosis. I continued to self-treat, as I felt that my focus on pain generators as part of my professional work provided me with knowledge of the patho-anatomy and patho-physiology of back pain. A mild degenerative spondylolisthesis then became apparent, not present 5 years previously. I followed my own advice to patients that pain never killed anyone, but when it became bad enough that the activities of everyday living was affected, then it was time to get diagnostic tests to see if surgical intervention was needed. I waited until I was debilitated before getting an MRI.
I was trained to be a general orthopedic surgeon. My mother had unsatisfactory decompressive surgery for spinal stenosis and a central disc herniation when I was an orthopedic resident. She had a congenitally small spinal canal. I recommended surgery with an experienced deformity surgeon when her pain affected her quality of life. The surgery ultimately destabilized her spine, and there was a surgical injury to her traversing nerve. Partly because of my mother’s bad surgical result, I focused on minimally invasive techniques early. I was technically a good surgeon, and I had very good success with decompression of herniated discs and spinal stenosis, even in the face of grade I spondylolisthesis. I was convinced then, that the surgeon factor was the most important factor for the patient to consider when choosing his surgeon. We are not unlike high performance athletes, who have a wide spectrum of surgical abilities.
In spine, I followed the minimally invasive route by first embracing chymopapain and percutaneous procedures such as APLD because the transforaminal access was the least invasive. I reserved fusion for instability and deformity, but performed fusions mostly without hardware, using meticulous techniques for preparing the graft site and an on-lay bone graft with bone harvested from the iliac crest. I recognized then, that a skilled surgeon is the answer to getting the best result possible from surgery for his patient. I did dabble in the use of implants, beginning with Harrington rods and hooks, then Luque instrumentation, followed by Ray cages and BAK cages. When the cage rage era began, followed by pedicle screw instrumented stabilization I was already focusing on MIS spine and alternatives to fusion.
In 1991, I took Parviz Kambin’s course in arthroscopic spine surgery and, with extensive personal experience in joint arthroscopy, began using the endoscope to explore intradiscal anatomy. It seemed natural to me to utilize the endoscope to examine patho-anatomy in the spine. I quickly decided to also visualize the epidural space through the foramen with the “inside out” technique to obtain confirmation that the spinal nerves were decompressed. Also, in 1991, the laser was introduced for the lumbar spine, and I became a fan of laser as a surgical tool under endoscopic visualization because it had the tissue effect of decreasing bleeding and ablating soft tissue as well as bone through a 2mm working cannula or working channel scope. I became an instructor for Laserscope, a company promoting the KTP laser that complemented Kambin’s percutaneous Arthroscopic discectomy (AMD). Then I adopted the use of a chromophore that not only stained the degenerative nucleus for easier visually guided “selective” discectomy, it enhanced the KTP laser’s ability to thermally modulate the disc. I initially used the chromophore indigo carmine to enhance the KTP laser, but soon learned that the laser with the chromophore mostly stained the loose degenerative, fragmented portions of the nucleus, while sparing the more normal nucleus 10. It also provided a bloodless surgical field that enabled me to see not just inside the disc but allowed for visualization of the epidural space, an area that Kambin discouraged entering because of the fear of epidural scarring. I noted also that thermal modulation of the disc annulus also seemed to help back pain. My experience caused me to develop my own designed endoscopic system in conjunction with Richard Wolf Surgical Instrument system, and marketed the FDA approved system in 1997. I called it the Yeung Endoscopic Spine System (YESS) (Figure 1). I also switched to the Holmium: YAG laser by Trimedyne since it provided better visualization and ablation.
I have not deviated from this surgical philosophy since then, except to constantly evolve the technique, updating my experience in the literature, adding new scopes and instrumentation, aided by my recognition of spinal patho-anatomy [3-11]. I had customized surgical instrumentation that I developed to facilitate decompression, ablation, and irrigation of the pain generator [6,7]. I documented patho-anatomy identified by the endoscope and monitored the effect from probing, decompression, ablation or transection. The original endoscope design is still the state of the art configuration for intradiscal therapy, where the degenerative process begins. It has been copied to some extent in the endoscope designs of competing endoscope companies. As the procedure evolved, different scope sizes, designs and working channels were augmented by better endoscopic instrumentation and modified approach techniques.
Methods
In 1997, after freezing the YESS scope design for commercial production, it became FDA 510 K approved through the Richard Wolf Instrument Company in the US. In 2001 Yeung and Satish Gore, my first spine fellow, published the then evolving YESS technique and methodology with emphasis on skin markings, trajectory to the foramen, and visualization of the basic pathology of annular tears, which caused the symptoms of discogenic pain. In 2002, Yeung and Tsou, another Yeung spine fellow, described the YESS transforaminal (TFE) technique with specialized instruments able to treat all forms of disc herniation by the transforaminal approach. We reported on 309 consecutive patients using the YESS philosophy and technique. The technique was always performed under local anesthesia and was constantly evolving. It did not selectively stratify patients, which will provide patient selection criteria dependent on the skill and experience of each individual surgeon. I also initiated a retrospective study comparing 100 consecutive SED patients using local anesthesia compared with 100 patients operated on with neuromonitoring, and identified no advantage or need for neuromonitoring when local anesthesia was used.
The use of 1% lidocaine permits generous use for pain control but still allows the patient to feel pain when the inflamed nerve root is manipulated or stimulated with RF. The patient, under a local anesthetic, usually remains comfortable during the entire procedure, with the exception of periods such as during Evocative Chromo-Discography, annular fenestration, or when ablation tools or instruments are manipulated next to the foraminal nerves.
Evocative Chromo-discography is performed in every surgical case to get anatomic feedback from the discogram pattern fluoroscopically and by “evoking” the patient’s response of pain as positive, negative, concordant, similar, or discordant. This facilitates interpretation of the disc degenerative pattern and labels the degenerative disc for guided extraction or no extraction endoscopically. Degenerative disc is stained by a 10% mix of indigocarmine dye with a non-ionic radiographic agent like ISO-VUE 300. As the degenerative acidic blue stained nucleus is removed, [13] the inner annulus is visualized for loose degenerative intradiscal disc material, including disc embedded in the torn annulus. The annulus is inspected for tears and modulated as needed. Intradiscal thermal modulation then treats the tear intradiscally from the ventral annulus. The endoscope is then withdrawn to inspect the foramen, the exiting nerve, the traversing nerve and the epidural space as needed. Extruded disc fragments in the annular layers dorsally are accessed by foraminoplasty of the ventral facet from the foramen with a foraminoplasty scope and its accompanying instruments, extending the decompression to the exiting nerve and the axilla. Probing the epidural space to the traversing nerve in the axilla of “hidden zone” confirms adequate removal of all mechanical lesions. Tissue, such as disc fragments, synovial cysts, or osteophytes in the axilla between the exiting and traversing nerve is removed. Frequently, nerves not observed or missed by traditional surgeons are found in the foramen and axilla that are painful when probed. These finding and its contribution to the pain complex are still not clear. When possible, I just decompress and not ablate or resect these nerves unless they are already partially transected by foraminal access. Blunt technique to gain access to the foramen will mitigate inadvertent transection of these nerves, which I label “furcal” nerves. 80 mg of Depomedrol and 2cc .5% Marcaine is deposited in the foramen at the end of the procedure. In cases where we suspect inadequate decompression of the lateral canal or hidden zone of Macnab, we go in the subarticular zone and decompress the axilla, and the roof of the upper foramen.
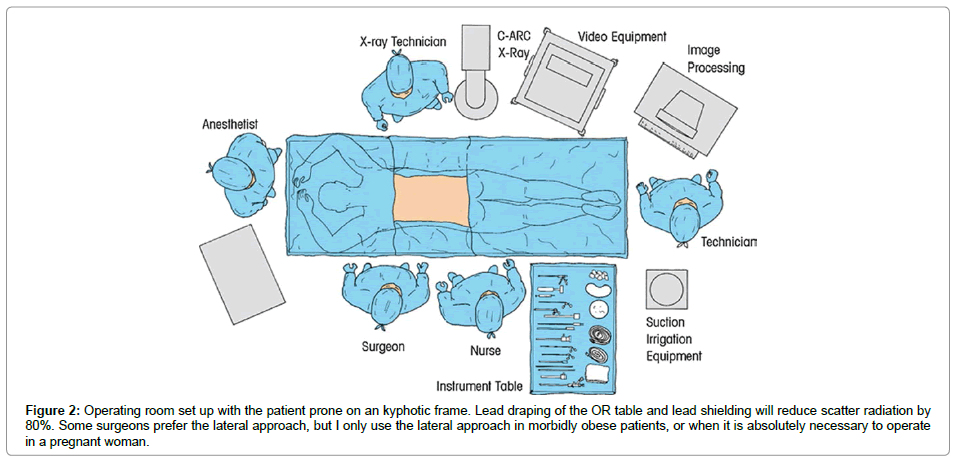
A standardized technique with a crystallized learning objective of precise needle placement at pain the generator of TFE, is described as the YESS technique. A Yeung describes the original YESS system in detail [6-8]. The procedure is carried out in an operating room, using local anesthesia and conscious sedation attended by an anesthesiologist. The patient is prone, positioned on a radiolucent frame, with the position of the imaging equipment, instrument table, and operating room personnel in relation to the surgeon (Figure 2). The original technique has also evolved to address the variations in anatomy and severity of the conditions treated surgically.
Figure 2: Operating room set up with the patient prone on an kyphotic frame. Lead draping of the OR table and lead shielding will reduce scatter radiation by 80%. Some surgeons prefer the lateral approach, but I only use the lateral approach in morbidly obese patients, or when it is absolutely necessary to operate in a pregnant woman.
The conscious patient is instructed to report any unusual painful sensations to the operating surgeon while the procedure is in progress. Although some surgeons utilize the lateral position, the prone position is preferred, as the prone position allows a biportal (bilateral) approach if necessary. This helps with real time visualization of large and flexible working instruments in the disc while the operating endoscope in the other port [foramen] simultaneously directs smaller instruments through the operating channel during discectomy. It is more common to use a uniportal approach as it is sufficient to address most issues with the new instrument and techniques available.
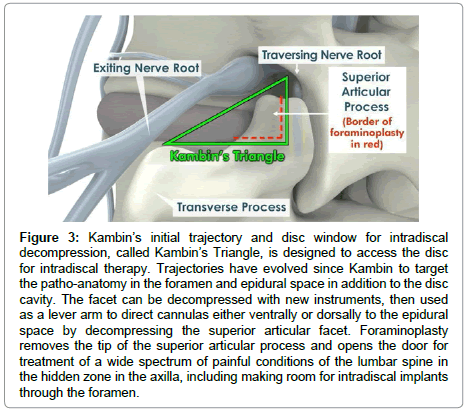
Biplane intraoperative fluoroscopic images are employed for percutaneous guidance. The approach trajectory starts from an optimally located skin window entering the disc through the foraminal annular window. This starting point is calculated by drawing the trajectory on the skin by a protocol described as part of the YESS technique for needle placement. We are between longissimus and psoas (Figure 3).
Figure 3: Kambin’s initial trajectory and disc window for intradiscal decompression, called Kambin’s Triangle, is designed to access the disc for intradiscal therapy. Trajectories have evolved since Kambin to target the patho-anatomy in the foramen and epidural space in addition to the disc cavity. The facet can be decompressed with new instruments, then used as a lever arm to direct cannulas either ventrally or dorsally to the epidural space by decompressing the superior articular facet. Foraminoplasty removes the tip of the superior articular process and opens the door for treatment of a wide spectrum of painful conditions of the lumbar spine in the hidden zone in the axilla, including making room for intradiscal implants through the foramen.
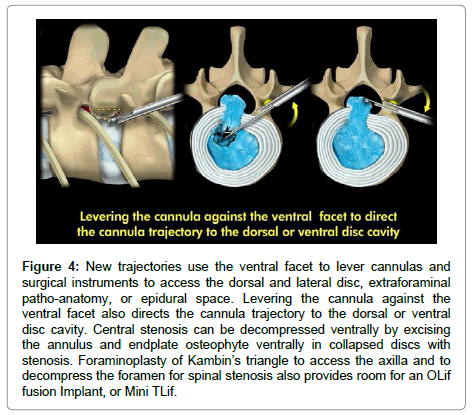
First a needle is inserted into the disc without causing leg pain, then the exiting nerve is retracted from harm’s way by blunt manual manipulation or tapping the obturator past the nerve root with a dilator. Then, by using a beveled or other configured cannula as a tubular retractor and a surgical access tunnel, the disc is entered. If an “inside-out” technique is used, a cavity in the disc must be created for viewing and manipulating the endoscopic tools intradiscally. Some endoscopic surgeons target the extruded herniated fragment directly by placing a guide wire to the disc fragment, then dilating cannulas and cutting instruments into the epidural space to address the herniation Cutting the facet first 15 also known as the “outside in” technique In comparing the transforaminal approach to the traditional translaminar approach, the surgeon only has to learn foraminal anatomy and get used to viewing the patho-anatomy from the foramen. The “inside out” technique emphasizes safety by accessing the annulus of the foramen, taking the time to view the nerves and vasculature on the foramen before cutting, while the “outside in” or targeted technique is a simplified concept of targeting and serial dilating to reach the patho-anatomy. This technique may be over simplified, as it ignores the pitfalls of anomalous nerves in the foramen and all the variables in the foramen anatomy that may be responsible for axial and discogenic back pain. Yeung’s “inside- in technique“ enters Kambin’s Triangle and the disc just ventral to the facet, but decompresses the foramen under direct endoscopic vision with cutting instruments inside the specially designed “medium or long tang cannula anchored against the ventral facet, exposing it for bone removal with trephines, kerrisons, and burrs (Figure 4) [13-19].
Figure 4: New trajectories use the ventral facet to lever cannulas and surgical instruments to access the dorsal and lateral disc, extraforaminal patho-anatomy, or epidural space. Levering the cannula against the ventral facet also directs the cannula trajectory to the dorsal or ventral disc cavity. Central stenosis can be decompressed ventrally by excising the annulus and endplate osteophyte ventrally in collapsed discs with stenosis. Foraminoplasty of Kambin’s triangle to access the axilla and to decompress the foramen for spinal stenosis also provides room for an OLif fusion Implant, or Mini TLif.
Patho-physiologically, the initial change in most cases of disc degeneration is loss of tissue turgor resulting in an annular tear. Whether degeneration begins from outside the annulus or inside the disc is still not clear. The tear in the annulus may leak the cytokines from a degenerating nucleus and cause inflammation around the annular tear, adjacent nerves, spinal nerve root, or DRG. Nuclear fragmentation may give rise to herniation through this tear. Thus, we target the fragment and the annular tear, the granulation tissue surrounding the leak from the tear, and physiologically, the inflammation giving rise to mechanosensitization of the root and DRG, which in addition may have compression on spinal nerves needing decompression. Access to these targets is initially by use of a needle, which may suffice if only an anti-inflammatory drug or steroid is injected at the site. If surgery is contemplated, this access pathway is then DILATED to a larger diameter by use of various sized access cannulas, the current standard being a 6mm obturator and cannula, which makes way for a working sheath that accommodates an endoscope. As a result introduction of an endoscope without any tissue cutting is possible. The landing of the instruments is in the foramen below the facet in the safe triangle as described by Kambin. The needle and instrument trajectories will be adjusted for targeting central, paracentral, or foraminal herniations. The landing point is adjusted in the subarticular zone between the medial and lateral pedicular border. Foraminoplasty, as opposed to simple foraminotomy, is performed to provide the required trajectory angle to access the patho-anatomic target site (Figure 4).
The surgery does not need general anesthesia, blood transfusion, or skin sutures, and thus is greatly simplified, especially when performed under local anesthesia with conscious sedation. Only small adhesive skin strips are needed to close the skin. A single suture may be used to close the .8 cm incision, but because of its size, sutures are not necessary. Any bleeding from the incision site will stop by the time the patient becomes supine and then brought to the recovery room. Suture less surgery is preferred because bleeding will eventually stop from tissue tamponade, and any bleeding through the skin incision mitigates hematoma formation in the foramen and epidural space. As I have personally experienced, sedation is also not absolutely needed in very experienced surgical hands [20-22].
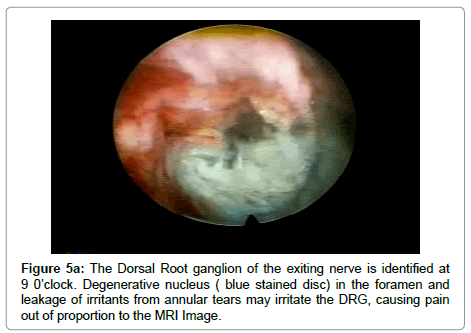
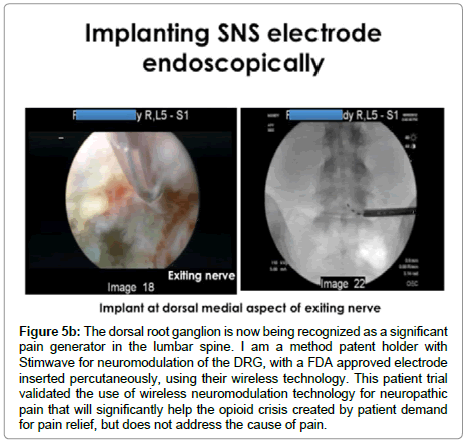
This modified access technique is to target superior facet and walk the needle down the ventral bony facet, hugging the facet to avoid the exiting nerve and using the facet as a fulcrum to change trajectories of endoscopic cannulas and instruments for removal of large fragments (Figure 4b). A special long or medium tang cannula can also be used to isolate the ventral facet for foraminoplasty, and a long awl, made custom by Maxmore, named after Tom Hoogland’s terminology as the “Yeung-Shidi” needle can start the foraminoplasty, then aided by the articulating carbide or diamond burr. Both techniques provide access to the DRG, the major pain generator responsible for severe pain that may be out of proportion to the imaging study, but is the target of decompression in the foramen. (Figure 5).
Figure 5b: The dorsal root ganglion is now being recognized as a significant pain generator in the lumbar spine. I am a method patent holder with Stimwave for neuromodulation of the DRG, with a FDA approved electrode inserted percutaneously, using their wireless technology. This patient trial validated the use of wireless neuromodulation technology for neuropathic pain that will significantly help the opioid crisis created by patient demand for pain relief, but does not address the cause of pain.
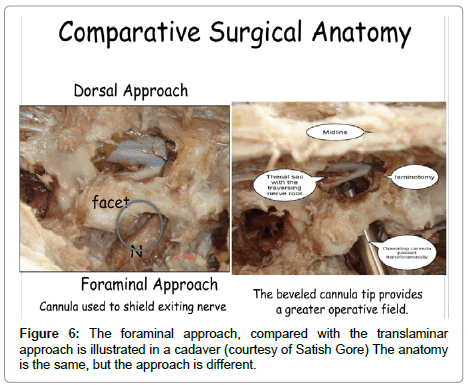
The anatomy of the spinal segment is the same whether the trans laminar or transforaminal surgical approach is utilized. The transforaminal endoscopic approach views that anatomy through a posterolateral access cannula and views the anatomy from a different angle (Figure 6).
Currently available equipment include HD video, high resolution rod lens operating endoscopes, beveled cannulas, trephines, endoscopic kerrisons, articulated rongeurs, flexible shavers, bipolar RF electrodes, straight and side firing Holmium-YAG laser, and a high speed diamond and articulated carbide burr. Straight and flexible shavers aid rapid decompression of loose fragments of degenerative nucleus. Each tool has a unique role in performing special surgical tasks with various sized access cannulas and scopes. The recent addition of a hook and flexible curette designed by Gore, articulated pituitaries that access the foramen through a working channel endoscope, along with multiple sized cutting and grasping rongeurs, can extend the reach to the epidural area and migrated fragments (Figure 6). These instruments help expose the hidden zone and decompress the DRG and axilla spinal segment Figure 7a,b). Having these instruments, and the addition of new surgical instruments such as powered rasps and affordable laser tips, will facilitate the development of transforaminal MIS surgery. All endoscopic companies entering this developing field will contribute to the advancement of endoscopic transforaminal surgery.
Results
Yeung and Tsou, in 2002, reported TFE on 307 consecutive patients with disc herniations that were candidates for transcanal microdiscectomy [11]. The study included intracanal and extracanal herniations. Recurrent herniations and patients with previous surgery at the same level were not excluded. Results were reported after a 1-year follow up. Ninety-one percent of the patients were satisfied with their results and would opt to undergo the procedure again if they had the same diagnosis and symptoms. The overall complication rate was reported to be 3.5%. Since 2002, the YESS technique has evolved and improved further with the experience gained by the senior author from this initial study.
Tsou and Yeung, 2002 separated out a subgroup of 219 patients with non-contained herniations and reported the results at 1 year [12]. Patient satisfaction was 91%. TFE could provide equivalent results to reported results of open microdiscectomy, even with noncontained, extruded herniations.
YEUNG reported in 2013 50 Cases of Micro Lumbar Discectomy in his group practice compared with SED performed by his group at DISC that included his son, Chris Yeung and associate Justin Field Both surgeons were trained by A Yeung, but all MLD cases were performed by C. Yeung and Field. The levels were L4-5=15,L5-S1=35. The average VAS decreased from 6.5 to 1.7. Average ODI decreased from 44% to 30%. Complications included 1 seroma and 1 durotomy. Patients receiving MLD were usually for extruded, migrated, or sequestered HNP believed better suited for MLD, even in the experienced hands of transforaminal surgeons in the same spine group. These cases were deemed to be more difficult for SED because the success for complete disc excision was less due to anatomic factors limiting access or sequestration and migration of a disc fragment.
SED™ included extruded, migrated, sequestered HNP at all levels felt possible by all three surgeons. Most were by ATY, who had the most experience with SED. The SED group numbered 250 of the 300 consecutive levels operated. The average VAS was decreased from 6.6 to 2.5 and ODI decreased from 46% to 32%. 37 patients in the SED group (15%) developed temporary dysesthesia in the 2-week postoperative period. Even with dysesthesia, however, improvement in VAS and ODI and patient satisfaction was comparable to MLD.
I have continued to report on the evolution of my work at numerous national and international peer reviewed meetings, and have written invited book chapters to update my work.
I now have experienced, first hand, my own spinal condition of a tri-modal HNP at L4-5, with three level stenosis in the face or degenerative scoliosis and grade one spondylolisthesis. I had my endoscopic decompression as a staged procedure, performed only under local anesthetic and no sedation before undergoing the three level decompression and coflex implant. Each surgical procedure provided the relief anticipated, and in stages required at the time.
Yeung’s firsthand experience with transforaminal endoscopic (SED) and Coflex surgery.
My Personal Experience undergoing 2 Selective Endoscopic Decompressive Procedures followed by an open three level decompression and Coflex interlaminar stabilization as a staged procedure only served to provide me more experience and strengthen my philosophy of giving the patient an option of electing the least invasive procedure first, to address the patho-anatomy of the pain generator.
My son, Chris Yeung, performed my surgery, a fellowship trained Spine surgeon who had 10 years’ experience with foraminal endoscopic disc surgery, although less extensive experience. He had great technical skills. I had no sedation as I wanted to feel what my patients would feel during surgery. My recovery was remarkable. I was able to return to work in 2 days after recovering over the weekend with very little muscle soreness.
I was able to tell that the most sensitive area of the spinal segment was in the axilla between the traversing and exiting nerve, called the “hidden zone of MacNab”. This solidified my experience focusing on decompressing this axillary and foraminal zone for Failed Back Surgery syndrome.
After successful endoscopic surgery for sciatica and radiculopathy, I still had to address my bilateral foraminal stenosis at L4-5, and left foraminal stenosis at L5-S1 along with a small cephalad extrusion at L3-4 R. Pain from bone impingement is different than disc impingement, it can be painless unless the instability causes bony impingement of the exiting nerve in the sub articular zone.
I elected to have a three level decompression with dynamic stabilization with a Coflex implant. The recovery of back pain was three to four times longer with the open three level procedure, but each procedure provided the anticipated surgical relief. I still have to deal with the contralateral stenosis at L5-S1, which was partially relieved, however painful with twisting movements. I anticipate that with time, my spine will stabilize on its own. It has improved yearly. If not, one last MIS stabilization procedure may be needed.
This personal surgical experience serves to solidify my bias toward transforaminal decompression as an option for first line surgical decompression for discogenic pain in the experienced hands of a trained MIS surgeon, even though it was not the ultimate definitive procedure. Because SED has the ability to address the source of pain with the least surgical morbidity and fastest recovery, this may have some influence in the algorhythim of surgical MIS intervention in the future of spine care. The surgical options will be a joint decision by the patient and surgeon. I emphasize appropriate training, as there are no academic centers in the United States that I am aware of who train endoscopic spine surgeons. Most centers practicing endoscopic surgery are relatively recent, and adopted by surgeons who may have taken a few courses from the early MIS surgeons around the world. The establishment of an endoscopic spine center at the University of New Mexico is the first such effort.
Discussion
My personal experience undergoing the YESS procedure is a valuable first-hand experience most spine surgeons will not have the opportunity to experience. It now serves to provide me the opportunity and motivation to report this experience, and to augment my other articles in Surgical Technology International, Inc.
The technique and equipment has undergone a slow, progressive evolution over the past thirty years with integration of added instrumentation and decompressive techniques over the past 15 years. The major change has been new working channel scopes and new instruments to increase the reach and effectiveness at the targeted patho-anatomy through the endoscope. Techniques by various pioneer spine surgeons may differ, and the students of the original pioneers will likely help evolve endoscopic surgery further, but the results should be the same when the surgery is successful. Techniques and its indications have also continued to improve and evolve, expanding indications to include a wider spectrum of painful degenerative painful conditions of the lumbar spine. As a result of combining adjunctive minimally invasive technologies such as nucleus augmentation and biologics, progress will fill the gap bridging clinical efficacies of conventional trans canal open decompression and pain management.
While arthroscopic lumbar discectomy is a term first proposed by Parviz Kambin, through a working “triangular zone”, the evolution and expansion of Kambin’s technique since 1991 has made the term selective endoscopic discectomy™, annuloplasty, and foraminoplasty more appropriate in describing Yeung’s transforaminal “inside out” approach to the disc and foramen.
There have been many variations in the techniques of transforaminal spine endoscopy, which have been based on instrument designs and treatment philosophy. Due to a relative absence of access related complications and alteration of normal anatomy, all surgeons adopting TFE access should recognize it as the best surgical option, limited only by individual surgical skills and experience. TFE surgery is greatly dependent on the “surgeon factor”. All surgeons are trained to perform laminectomy/ discectomy, but few have the training and experience in endoscopic treatment of the degenerating functional spinal unit reaching all of its pathological variations. Each operating surgeon then places his own self-directed method and an independent emphasis on his version of TFE surgery as it evolves.
Indications
Current Indications for TFE are:
1. Annular tears with discogenic lumbar pain as determined by evocative discography, both positive and false negative
2. All disc herniations and protrusions accessible through the foramen whether contained, extruded, or sequestered
3. Foraminal and extraforaminal herniations
4. Central disc herniations (contained or uncontained) with stenosis
5. Foraminal and extraforaminal stenosis
6. Foraminal osteophytosis
7. FBSS from foraminal fibrosis, recurrent HNP, and subarticular lateral recess stenosis
8. Mild and soft tissue central spinal stenosis
9. Discitis
10. Juxtafacet and pedunculated cysts
These indications are dependent on surgeon experience, each patient’s individual anatomy, and the patho-anatomy being addressed surgically. Therefore, the reader is cautioned to recommend to their patient the transforaminal endoscopic approach only if the surgeon has training and experience. By being informed, patients will need to know that transforaminal endoscopic surgery is amenable for certain conditions that are usually more difficult for traditional approaches or not severe enough to warrant a more invasive procedure. Examples of these conditions will be illustrated in this article as a surgical procedure by trained endoscopic surgeons. Disc herniations at levels from T10 to S1 meet the inclusion criteria.
The ideal indications are foraminal and extraforaminal disc herniations and discitis. These types of herniations and conditions have greater approach related surgical morbidity with the traditional translaminar open median and even paramedian posterior approach. With central disc herniations, results from open discectomy are mixed, because decompressing the disc material centrally will weaken the herniation site excessively, and recurrence, residual back pain from removing and destabiling of the disc annulus make outcomes less predictable. This makes many spine surgeons hesitant to operate on central disc herniations. Transforaminal discectomy does not produce such paradoxical effects as severely, and the results are more uniformly good. Therefore, while not making central HNP an absolute indication, it is preferable to open discectomy.
My recent experience identifying the most common causes of failed back surgery suggests that FBSS is from both a technical failure and an unrecognized patho- anatomic anomalous condition. The technical failures will decrease with surgeon skill, access to surgical instruments and endoscopes, and experience.
Transforaminal visualization of the hidden zone between the traversing and exiting nerve has identified this anatomic zone as the area to inspect to determine adequate decompression in both endoscopic and open procedures. The endoscopic approach has also given an edge over traditional approach to failed back surgery, as now we are able to visualize the hidden zone in the axilla in the subarticular area, not just the subarticular area.
Anomalous anatomy
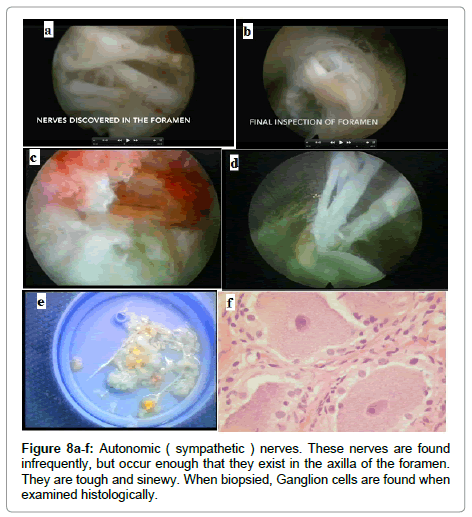
Anomalous nerves in the foramen certainly contribute to the pain complex that cannot be explained by current imaging studies, yet exist when identified with transforaminal visualization of pathoanatomy (Figure 8a-f). They certainly contribute to the pain complex in enough patients that make taking care of patient with axial and neuropathic pain challenging. These nerves produce pain when probed, and these nerves may contribute to post op dysesthesias experienced by patients. Fortunately, most are transient, but residual pain and weakness may also result.
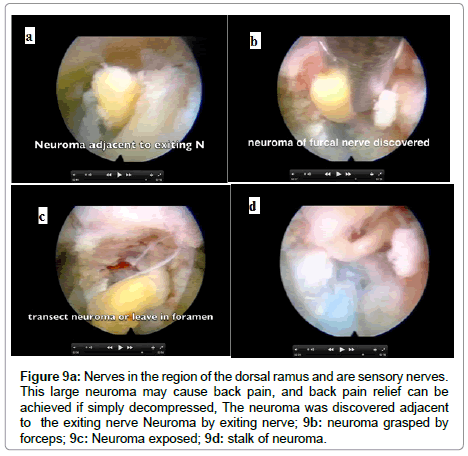
These anatomical anomalies or variations visualized in the foramen while not diagnosable by traditional imaging techniques, contribute to the pain complex. Examples include furcal nerves, foraminal nerves, autonomic nerves, and the dorsal ramus and its branches. It is difficult to sort out except by excision biopsy and study of histologic slides. These nerves, including neuromas, can be found in the tissue extracted during transforaminal discectomy. When encountered, I try to avoid injuring the nerves, but often it is not possible to avoid when it is in the surgical pathway. Transecting these nerves do not always cause symptoms, especially if they happen to be a pure sensory nerve, such as the branches of the dorsal ramus known to innervate facet joints, but can be visualized on the foramen before the nerves exit to the innervate facet joint (Figure 9a-d).
Figure 9a: Nerves in the region of the dorsal ramus and are sensory nerves. This large neuroma may cause back pain, and back pain relief can be achieved if simply decompressed, The neuroma was discovered adjacent to the exiting nerve Neuroma by exiting nerve; 9b: neuroma grasped by forceps; 9c: Neuroma exposed; 9d: stalk of neuroma.
Contraindications
Contraindications are relative, and depend on the individual patient’s individual anatomy and the variable factors involved for foraminal access. Endoscopic removal of disc herniation is only limited by the accessibility of endoscopic cannulas and instruments to reach the patho-anatomy site. The location of the disc herniation, the extent of the extrusion and sequestration, the concomitant foraminal stenosis, and the experience level of the surgeon are vital considerations for patient selection. Some patients with high iliac crests, horizontal L5-S1 disc spaces, or degenerative scoliosis make surgical access through the foramen more difficult. Special techniques may require a trans-iliac approach, as described by Osmon, but if this is an issue, simply opt for the translaminar approach. There is no real advantage to forcing a transforaminal approach for sake of the approach. Most spine surgeons unfamiliar with TFE may not even attempt to remove extruded, sequestered disc herniations because they are much more familiar and adept with the traditional transcanal approach. They reserve the approach for contained and foraminal or extraforaminal disc herniations because in their hands, traditional techniques provide more predictable surgical results. This is not a barrier to the experienced endoscopic surgeon, who individualizes each indication by selecting the best approach for the patient. The “outside in” access does target the intra-canal fragment directly after cutting the facet blindly and is practiced by experienced surgeons. The far lateral access also targets foramen the extruded fragment in epidural space. All these variations in approach have evolved from basic technique.
Complications and avoiding pitfalls
The risks of serious complications or injury are low, as with most visualized endoscopic approaches—approximately 1% or less in the authors’ experience. As with any surgery, surgical competence is important. There are the usual risks of infection, nerve injury, dural tears, bleeding, and scar-tissue formation. Transient dysesthesia, the most common postoperative complaint, occurs in approximately 5%-15% of cases, and is almost always transient. Its cause remains incompletely understood and may be related to nerve recovery, operating adjacent to the dorsal root ganglion of the exiting nerve, furcal nerves or a small hematoma adjacent to the ganglion of the exiting nerve, as it can occur days or even weeks after surgery.
There are also anomalous nerve fibers in the annular tissue, which may be furcal nerves, or nerves growing into an inflammatory membrane in the area of the foramen that is not the traversing or exiting nerve. It could show up in the surgical specimen without permanent effect on the patient, but may cause temporary dysesthesia. At this time, when anonymous nerves are encountered, it is not known whether they play a role in the pain complex. I usually try to avoid injuring these nerves, especially when they are more than 1-2 mm in diameter, and try to work around these nerves while decompressing the disc, especially when the patient reports pain when the nerve is probed or stimulated. If these nerves are branches of the pure sensory dorsal ramus, it is usually located on the ventral lateral aspect of the superior articular process in the foramen before it exits through the intertransverse ligament to innervate the facet. Probing or ablation of these nerves only denervates the facet and reduces axial back pain, but there can be motor fibers from the ventral ramus connected by the communicating branch.
Using blunt techniques to dilate the annular fibers has limited surgical morbidity but dysesthesia cannot be avoided completely. It can occur even when there were no adverse intraoperative events and in cases where the continuous electromyography (EMG) and somatosensory evoked potentials (SEP) did not show any nerve irritation. The symptoms are sometimes so minimal that most endoscopic surgeons do not report it as a “complication.”
The more severe dysesthetic symptoms are similar to a variant of complex regional pain syndrome, but usually less severe, and without the skin changes. Post-operative dysesthesia is treated with transforaminal epidurals, sympathetic blocks, and the off-label use of Pregabalin or Gabapentin (Pfizer, Inc., New York, New York, USA) titrated to as much as 1800-3200 mg/day. Gabapentin is FDAapproved for post-herpetic neuralgia, but effective in the treatment of neuropathic pain. High doses of vitamin B-12 may help.
Politically, it is not credible for surgeons or expert witnesses who do not personally perform transforaminal decompressive surgery to criticize or to fight it in turf wars or in medical/legal or quasi judicial situations as not appropriate because they simply cannot do it, choose not to do it and/or personally won’t accept it. In the past, medical/ legal claims experts with no experience and knowledge have offered personal opinions inappropriately as expert testimony. Experts in malpractice claims hide behind the second amendment simply by giving “personal opinion”.
The conditions ideally treated with transforaminal decompression
The conditions ideally treated with transforaminal decompression are:
1. Painful annular tears: The Ability to visualize inside the disc cavity. Pathologic processes of aging and degeneration start intradiscally. Intradiscal therapy will add effective means to treat disc herniations earlier in the disease process. Discogenic pain from annular tears may progress to herniation.
Granulation tissue in annular defects (tears) can be visualized and ablated and closed using a RF electrode. It is best identified by discography, not always on MRI. Endoscopy can successfully identify and treat painful annular tears visually and is much more effective than the fluoroscopically image guided techniques of IDET and variations of surgical disc decompression.
2. Central disc herniation: it is less invasive to target and remove a central herniated disc fragment by transforaminal access staying inside the disc. Even large contained disc herniations are more effectively and treated less invasively when the weakened annulus is not fenestrated at its weakest point [23].
3. Foraminal and extraforaminal HNP: Direct access to this type of herniation produces much less tissue trauma and does not require laminectomy and facectectomy
4. Lateral recess stenosis: The lateral recess, similar to access to foraminal disc herniations is more effectively decompressed with all the new instrumentation available commercially. Previously many of the surgical tools were custom made for a few high volume pioneer endoscopic surgeons. In addition to the early trephines and rasps, aided by laser, different endoscopic companies have developed special endoscopes, kerrisons, straight and articulated burrs and rasps for rapid foraminal boney decompression.
5. Spondylo-Discitis: The disc space is thoroughly debrided, infected tissue is excised, and antibiotics can be injected directly into the disc cavity in high concentration. Auto fusion results rapidly, and the infection is resolved much more rapidly than with antibiotics alone. There is little chance of spreading the infection when the tissues of the spinal canal do not have to be cut.
6. Upper lumbar and low thoracic HNP: The foraminal approach is more versatile than the posterior approach the more cephalad the herniation level because less or no bony decompression of the facets are required. From T-10 to L-4, the foraminal posterolateral approach offers the greatest and most flexible access to the lumbar upper discs without the need for laminectomy.
Endoscopic ablation of nerves causing axial back pain
While endoscopic transforaminal decompression and ablation is very effective for painful degenerative conditions in the lumbar spine, it is much simpler and safer to target the nerves innervating the facet with visualized dorsal endoscopic rhizotomy. The procedure was developed in 2006 and reported in 2007 at the ISMISS international spine meeting in Zurich. A rhizotomy set was developed with Richard Wolf GmBH for commercial sale. It is very effective, but has not caught on yet.
Results in the Literature
Overall, the endoscopic techniques have shorter operating times, less blood loss, less operative site pain, and faster postoperative rehabilitation/shorter hospital stay/faster return to work than the microsurgical techniques. There were no significant differences in the main clinical outcome criteria between the endoscopic and the microsurgical techniques in any of the trials. All 5 studies, however, demonstrate fewer complications with the endoscopic technique, and this was statistically significant in 2 of the studies. One study shows a lower rate of revision surgeries requiring arthrodesis with the endoscopic technique.
All 5 studies originate from experienced investigators and all 4 RCTs came from one group [22,23]. This limits the transferability of their results to surgeons less experienced in endoscopic disc surgery.
In a review of 2014 literature 14 TFE Vs. MLD, Patients in the PELD group had less blood loss (p<0.01), shorter hospitalization hours (p<0.01) and smaller surgical wounds (p<0.01) than the patients that underwent traditional OD surgery. MacNab criteria based evaluation showed levels of satisfaction were above 90% in both groups post-operative six months. There was no significant difference in pain index between the two groups (p>0.05). Furthermore, the levels of CRP, CPK and IL-6 in the PELD group were all lower than those in the OD group with a significant difference (p<0.01). The PELD had less damage to tissues than the traditional OD.
Three RCTs Hermantin, Krappel, Mayer and three retrospective studies Kim, Lee and Lee studies give TFE and MD comparison [14,17-21].
There is Level 4 and 5 evidence that transforaminal endoscopy {TFE} is not superior to micro discectomy [MLD] for back pain.
There is Level 4-5 evidence from two RCTs (n=80) that there is no difference in the proportion of people who return to work 18,19 Unpublished data, presented at international spine meetings from the author (A. Yeung) demonstrated faster return to work in self employed patients who were not awarded a set time frame for return work for employed patients.
There is Level 5 evidence from two RCTs and three nonrandomized studies (n=1,109) that operative time 18,20,21,22 are not different; mean operative time was 55.2 min for TFE and 60.3 min for Microdiscectory.
There is Level 5 evidence from three RCTs 15,18,19 (total n=160) of no difference in reoperation rate, but low quality evidence that TFE results in more reoperations when two non-randomized, retrospective studies 21,22 (total n=1,129) are included.
One RCT 19 (n=40) reported that TFE may be more expensive than MD.
Mixed RCTs
Two RCTs Ruetten (n=200 and n=100) allocated patients either to IL E /TFE surgery (interlaminar or transforaminal approach) or to MD [23,24]. FE interlaminar, IL approach was generally used for herniations inside the spinal canal and TF for intra- and extra-foraminal herniations, although the final decision was at the discretion of the surgeon.
One study included patients with first time disc herniations, the other only patients with recurrent disc herniations who previously had discectomy surgery at the same level [23,24]. These studies were both judged to have a high risk of bias. As such, all pooled analyses provide low quality evidence. There is Level 4 evidence that the effects of minimally invasive surgery (IL or TF) for patients with first time and recurrent disc herniations are not different to MD on back pain, leg pain or function at any time point. There is low quality evidence that more patients are satisfied with MIS and low quality evidence that Oswestry score is lower at one year follow up in the MIS group. There is Level 5 evidence that operative time (mean decrease in minutes was reduced compared to MD. Mean MI surgery time was 23 min and MD 50.5 min. There is low quality evidence that complications are reduced compared to MD and low quality evidence that rate of reoperation is not different.
With the results being same or similar, it would be important to know how these results are achieved. It is not possible to ignore the morbidity with open surgery and limitation, which medical comorbidities will have on choice of surgery. In patients with severe medical co morbidities only way we can offer pain relief is surgery under awake and aware status under local anesthesia. My own bias, and when published, will currently be level 5 EBM, but I have had enough experience with transforaminal SED since 1995 that I can usually predict my results if I get good legitimate response to transforaminal diagnostic and therapeutic injections at a 90% level of patient satisfaction for the predicted results of transforaminal decompression. The surgeon factor is important for the conditions I take on [23-24].
Personal Thoughts, Conclusion and Recommendations
The current health care environment is not condusive to the development and acceptance of transforaminal surgery due to the lack of formal academic training programs and lack of adequate reimbursement to reward the surgeon for the extra training it requires specialized surgical skills are required to perform this technically difficult surgical procedure through tubular retractors using an endoscope for direct visualization. This is a surgical procedure and NOT a pain management percutaneous, fluoroscopic guided procedure. Appropriate surgical fees should probably be higher, but at least equivalent, and certainly not less, using existing CPT codes for comparison as payers are trying to reduce re-imbursement. It is hoped that this summary of my expert experience, mature enough that surgeons with proper training can warranty it’s favorable results. Each surgeon can choose the level of expertise he or she acquires. It will serve as a reference for the further development on transforaminal endoscopic surgery and its role in treating painful degenerative conditions of the lumbar spine. If it is not adopted by newly trained specialists to perform transforaminal endoscopic properly, safely, and effectively, our patients will lose out, and our health care system will continue to spiral out of control. Industry will also have to play a role by developing image guidance and robotic enhancement of this highly technical surgical procedure that will serve not only to enhance surgical performance, but reduce radiation exposure. The platform will also enhance the current interest in disc replacement and biologics in a step by step progression, I favor nucleus augmentation as an intradiscal procedure that is less invasive and less costly than disc replacement. This thought is now recently expressed and published in the Journal of Spine and Neurosurgery, the same Journal that has accepted my review article on my 25 years’ experience.
References
- Yeung AT (2015) Moving Away from Fusion by Treating the Pain Generator: The Secrets of an Endoscopic Master. J Spine 4: 6
- Yeung AT (2016) Transforaminal Endoscopic Surgery: Its Role in the Treatment of Painful Conditions of an Aging Spine. Neurol Disorder 4: 6
- Yeung AT, Gore SR (2001) Evolving methodology in treating discogenic back pain by Selective Endoscopic Discectomy (SED) and thermal annuloplasty. Journal of Minimally Invasive Spinal Technique 1: 8–16.
- Gore SR, Yeung AT (2003) Identifying sources of discogenic pain. Journal of Minimally Invasive Spinal Technique 3: 21–24.
- Yeung AT (2002) Tsou Posterolateral endoscopic excision for lumbar disc herniation: surgical technique, outcome, and complications in 307 consecutive cases. Spine (Phila Pa 1976) 27: 722–731
- Yeung AT, Gore SR (2001) In-vivo Endoscopic Visualization of Patho-anatomy in Symptomatic Degenerative Conditions of the Lumbar Spine II: Intradiscal, Foraminal, and Central Canal Decompression. Surg Technol Int XXI: 299-319
- Yeung AT, Yeung CA (2006) In-vivo endoscopic visualization of patho-anatomy in painful degenerative conditions of the lumbar spine. Surg Technol Int 15: 243-256.
- Yeung AT (2007) The Evolution and Advancement of Endoscopic Foraminal Surgery: One Surgeon's Experience Incorporating Adjunctive Techologies. SAS Journal 1: 108-117.
- Yeung AT: Minimally invasive surgery with the Yeung endoscopic spine system (YESS). Surg Tech Int 2000;VIII:267-277
- Yeung AT, Yeung CA (2003) Advances in Endoscopic Disc and Spine Surgery: The Foraminal Approach. Surgical Tech Int 11: 253-261.
- Yeung AT (2003) Percutaneous Endoscopic Discectomy: the Posterolateral Approach, Minimal Access Spine Surgery, 2nd Edition, edited by Drs Regan and Lieberman, Quality Medical Publishing October.
- Kim IS (2005) Indigo Carmine for the Selective Endoscopic Intervertebral Nuclectomy J Korean Med Sci 20: 702-703.
- Yeung AT, Tsou PM (2002) Posterolateral endoscopic excision for lumbar disc herniation: surgical technique, outcome, and complications in 307 consecutive cases. Spine 27: 722–731
- Hermantin FU, Peters T, Quartararo L (1999) A prospective randomized study comparing the results of open discectomy with those of video-assisted arthroscopic microdiscectomy. J Bone Joint Surg 81: 958-965.
- Tsou PM, Yeung AT (2002) Transforaminal endoscopic decompression for radiculopathy secondary to intra-canal non-contained lumbar disc herniations: outcome and technique. Spine Journal 2: 41-48.
- Yeung AT (2013) Presenting author Christopher Yeung, M.D Justin Field, A Comparative Outcome Evaluation of Lumbar Transforaminal Endoscopic Discectomy Versus Micro-Lumbar Discectomy For Lumbar Disc Herniation 2013 poster presented at ISASS Annual Meeting Vancouver.
- Birkenmaier C, Komp M, Leu HF, Egener B, Ruetten S (2013) Review of Controlled Studies Comparing Full Endoscopic Procedures for Disc Herniations to Standard Procedures : Pain Physician 16: 335-344.
- Mayer HM, Brock M (1993) Percutaneous Endoscopic Discectomy: Surgical Technique and Preliminary Results Compared to Microsurgical Discectomy. J Neurosurg 78: 216-225
- Krappel FA, Schmitz R, Bauer E, Harland U (2001) Open or endoscopic nucleotomy? Results of a prospective, controlled clinial trial with independent follow-up, MRI and special reference to cost-effectiveness. Orthopadische Prax 37: 164–16972.
- Lee SH, Chung SE, Ahn Y, Kim TH, Park JY, et al. (2006) Comparative radiologic evaluation of percutaneous endoscopic lumbar discectomy and open microdiscectomy: a matched cohort analysis. Mt Sinai J Med 73: 795-801.
- Lee DY, Shim CS, Ahn Y, Choi YG, Kim HJ, et al. (2009) Comparison of percutaneous endoscopic lumbar discectomy and open lumbar microdiscectomy for recurrent disc herniation. J Korean Neurosurg Soc 46: 515-521.
- Kim MJ, Lee SH, Jung ES, Son BG, Choi ES, et al. (2007) Targeted percutaneous transforaminal endoscopic diskectomy in 295 patients: comparison with results of microscopic diskectomy. Surg Neurol 68: 623–631.
- Ruetten S, Komp M, Merk H, Godolias G (2009) Recurrent lumbar disc herniation after conventional discectomy: a prospective, randomized study comparing full-endoscopic interlaminar and transforaminal versus microsurgical revision. J Spinal Disord Tech 22: 122-129.
- Ruetten S, Komp M, Merk H, Godolias G (2008) Full-endoscopic interlaminar and transforaminal lumbar discectomy versus conventional microsurgical technique: a prospective, randomized, controlled study. Spine (Phila Pa 1976) 33: 931-939.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi