Research Article, Cell Biol Henderson Nv Vol: 6 Issue: 2
Treatment Options for the Patients with Diabetic Retinopathy
Nataliia Sych1*, Mariya Klunnyk1, Iryna Matiyashchuk1, Mariya Demchuk1, Olena Ivankova1, Andriy Sinelnyk1, Khrystyna Sorochynska2 and Marina Skalozub3
1Clinical Department, Cell Therapy Center EmCell, Kyiv, Ukraine
2Stem Cells Bank, Cell Therapy Center EmCell, Kyiv City, Ukraine
3Laboratory and Biotechnology Department, Cell Therapy Center EmCell, Kyiv City, Ukraine
*Corresponding Author : Nataliia Sych
Clinical Department, Cell Therapy Center EmCell Kyiv City, Ukraine
Tel: +380688898989
E-mail: nataliia_sych@ukr.net
Received: September 09, 2017 Accepted: September 18, 2017 Published: September 18, 2017
Citation: Sych N, Klunnyk M, Matiyashchuk I, Demchuk M, Ivankova O, et al. (2017) Treatment Options for the Patients with Diabetic Retinopathy. Cell Biol (Henderson, NV) 6:2. doi: 10.4172/2324-9293.1000134
Abstract
Objective: to elaborate the methods of treatment for the patients with diabetic retinopathy (DR) by means of both conventional treatment (medical or surgical) and preparations with extracted fetal stem cells (FSCs) in suspensions containing stem cells of fetal liver, brain and fetal eye.
Materials and methods: A comparative study was conducted on 15 patients with diabetic retinopathy including 7 women and 8 men aged from 27 to 61 years. All patients were administered conventional treatment, laser coagulation and, only over 6 months following the latter, FSCs suspensions containing cells separated from human embryonic tissues of fetal liver, brain and fetal eye were used for treatment of the patients under study.
Results: Authors proved effectiveness and safety of treatment using FSCs in patients suffering from DR. Based on the study we recorded improvements of glycemic and lipids profile as well as overall ophthalmology status among the patients.
Conclusion: FSCs use in complex treatment of patients with DR stabilizes disease compensation, can improve laboratory values and ophthalmology status.
Keywords: Diabetes mellitus; Diabetic retinopathy; Fetal stem cells
Introduction
Diabetes mellitus (DM) is considered to be a significant medical and social problem. A number of patients suffering from DM tend to annually increase and in accordance with data of the WHO over 1 million of patients (which constitute around 2% of population) have been affected in Ukraine [1]. Nowadays, diabetic retinopathy (DR) – a damage of the vessels of eye globe retina remains one of the most frequent and prognostically unfavorable signs in DM patients which often can result in severe loss of vision, blindness and invalidity among the patients. Vision complications are observed in 85% of patients suffering from type 1 DM (T1DM) during 20 years and over. Immediately after type 2 DM (T2DM) diagnosis in people of middle and elderly age in over 50% of likely cases the patients revealed damage of vessels responsible for eyes blood supplying [2].
The main causes of DR occurrence are immunology, metabolic, hormonal, blood rheological, hypoxic, genetic, and the other factors which cause lesions of walls in capillaries of eye retina and disturbances of vessels permeability. Swelling of macular zone is the principal cause of eyesight impairment in cases of DR disease. Microangiopathy along with occlusion of capillaries consists in DR pathogenesis [3-6]. Altogether, such disturbances lead to leakage in micro vessels and cause disruption of blood-retina barrier resulting in retinal hemorrhage, exudates and edema, as well as occurrence of edema in macular area. Subsequently, microvascular occlusion and ischemia contribute to “cotton wool” maculae, changes in capillaries, in particular; resulting in development of arteriovenous shunts and neovascularization. Elevation of vascular endothelial growth factor (VEGF) can be regarded as one of the principal angiogenic causes which are implicated within pathogenesis of DR [7,8].
Among the factors of risk for DR disease development one can emphasize the following: persistent control of blood pressure ranges (<130/80 mmHg), constant glycaemia control (HbA1C <7%), undeviating assessment of lipids level (total cholesterol <4.0 mmol/L, low-density lipoproteins (LDL cholesterol) <2.0 mmol/L, highdensity lipoproteins (HDL cholesterol) >1.0 mmol/L) and levels of triglycerides <1.7 mmol/L.
Persistent hyperglycemia can result in loss of endothelial cells, dysfunction in vascular smooth muscle cells as well as pericytial regions leading to hypoxia. Such changes are remarkable for both T2DM (insulin non-dependent) and T1DM (insulin-dependent). In particular, endothelial cells and pericytial capacity to self-renewal can be affected in case of DM; their turnover potential is subsequently disrupted [3] after which acellular capillaries become non-perfused, and adjacent retina appears to be hypoxic. Such hypoxia environment upregulates vascular endothelial growth factor (VEGF), which contributes to higher vascular permeability resulting in diabetic macular edema [4,5] and, eventually, visual function loss occurs [6] as well as neurodegeneration, including neuronal apoptosis and reactivity of glial cells [9].
At present one can emphasize two main directions (medicinal and operative) among the existing methods of treatment for DR. Despite of enormous possibilities of therapy for retina by means of laser coagulation in patients suffering from DR, there are still some limitations of this therapy mode as it can lead to a different extent of complications. To absolute contraindications for laser coagulation in patients with DR we can refer large areas of occlusion of capillaries; in particular – within a central zone of ocular fundus, as well as intensive neovascularization, marked glial cells proliferation, vitreomacular traction, preretinal and vitreal hemorrhages. Among complications of laser coagulation in DR, intense swelling of eye macule; hemorrhages to the retina and vitreous body as well as exudative retinal detachments most frequently occur. Though laser coagulation remains the main method of DR treatment for today it does not affect the pathogenetic mechanisms of this disease, but it is rather used only on a local level with the aim of stabilization of pathology process and inhibiting further progressive loss of vision acuity [10].
Collaborators proved effectiveness and safety of treatment for DM by use of FSCs [11]. So far, no therapy has been established yet that could support regeneration of the damaged vessels of retina resulting from a long-term hyperglycemia. Treatment using stem cells might become a feasible option for a purpose of both neurovascular damage prevention as well as promoting retinal damage regeneration, which is supported by the evidence from the recent studies by use of several types of stem cells.
Contemporary approach has been recently developed for treatment of early and moderate stage of DR [12-15]; that is specifically based on capacity of mesenchymal stem cells (MSCs) to production of neurotrophic and neuro-protective factors including a potential of endothelial progenitor cells (EPCs) for repairing the vessels, or likely ability of adipose stromal cells (ASCs) to assure both of the above mentioned functions.
Material and Methods
Study was performed for 15 patients suffering from DR including 7 women and 8 men with their mean age between 27 to 61 years. All patients were given conventional treatment, laser coagulation and, over 6 months following the latter, FSCs preparations with inclusion of separated stem cells were administered for the patients in suspensions containing stem cells, specifically cells of fetal liver, brain and fetal eye.
DR diagnosis was established by means of imaging analysis and with consideration of past history of DM. All patients were tested by way of the following blood laboratory parameters as glycosylated hemoglobin (HbA1c), C-peptide, insulin and fasting blood glucose ranges, including lipidogram profile. The patients have been suffering from T2DM during a period of 3-15 years; average disease duration constituted around 7 ± 3.12 years. From among 15 patients a diagnosis of non-proliferative DR was confirmed in 8 individuals (16 eyes), whereas in 4 patients (8 eyes) – we recorded pre-proliferative stages of DR and 3 of the patients (6 eyes) under study reported proliferative stages of DR disease.
Ocular organs examination in all patients was performed following a generally accepted method. In this respect a complex of measures for diagnosis consisted in: optical coherence tomography (OCT) study, automated perimetry for macula threshold, as well as measurement of macular zone thickness was performed. The patients were examined at baseline, over 3-4 months and following 6-8 months after beginning of FSCs therapy.
Stem cells procedure
All preparations are produced as cryopreserved suspensions of separated stem cells: one of suspensions containing stem cells of fetal liver; the second one – stem cells of fetal brain and the third suspension with inclusion of cells of fetal eye. For this purpose, EmCell collaborators received FSCs material, specifically separated from 8 weeks gestation human fetal liver and fetal brain, following strict aseptic and antiseptic rules in surgery premises. The fetuses were sacrificed as a result of medical abortion in cases when pregnancy in women was terminated owing to social indications; on provision of absence of any developmental pathology or infection in the embryo as well as signed written informed consent by the woman-donor. Extracted fetal tissues are then placed into sterile transport medium with Hanks solution. While in sterile environment fetal tissues are separated and homogenized inside of Hanks solution. All obtained suspensions are exposed to filtration and cryopreservation. Dimethyl sulfoxide is used as cryoprotectant. Later readily made suspensions are immersed to the test-tubes in volume of 0.1-1.0 mL. Cryopreservation of cell suspensions is performed inside the chamber of a controlledrate freezer in accordance with the specially adjusted program. Suspensions containing stem cells of fetal liver, fetal brain as well as stem cells separated from fetal eye are preserved in the cryobank with liquid nitrogen at minimally low temperature of (-196°Ð¡).
For a purpose of safety, both women-donors as well as readilymade stem cell suspensions are tested for viral, mycotic, bacterial infections and parasites such as hepatitis B virus, hepatitis C virus, human immunodeficiency virus 1 and 2, cytomegalovirus, herpes simplex virus 1 and 2, Epstein–Barr virus, rubella, syphilis (Treponema pallidum), toxoplasmosis (Toxoplasma gondii), Mycoplasma genitalium, Ureaplasma urealyticum, Ureaplasma parvum, and Chlamydia trachomatis.
Stem cell procedure was undertaken by transplantation of cryopreserved FSCs in the form of suspension following conventional conventional premedication with infusion of diphenylhydramine 30 mg and prednisolone 15 mg on treatment day 1. Subsequently, all patients were administered a specially prepared solution during the treatment day 2. Patients suffering from DR were performed dripfeed intravenous infusion of fetal liver cells in a volume of 3.2 ± 0.52 mL containing nucleated stem cells ranging from 1.0 to 54 × 106 / mL as well as the levels of progenitor cells CD34+ were 11 ± 2.6 × 105 /mL and amount of CFU constituting from 0.72 ± 0.34 × 106 /mL. During the second day the patients received treatment using suspensions with cells extracted from fetal brain and administered subcutaneously in a volume from 3.2 ± 1.3 mL and suspensions containing fetal eye stem cells which were injected into retrobulbar tissue in amount of 0.5 ± 0.2 mL.
To control overall health, prior to fetal stem cell treatment (FSCT), over 3 and 9 months after therapy general instrumental investigations as well as laboratory tests were performed for the patients. All patients signed their informed consent agreement before they could enter our procedure with FSCs administration.
Our study was conducted according to the ethical guidelines and regulations on good clinical practice, in conformity with Helsinki declaration as well as the ethical standards of practice by use of human fetal tissues tissues, approved by the Ministry of Health of Ukraine.
The principal aim of the study was improvement of methods of treatment for the patients with DR by means of a combination with classical therapy using medicines or surgery approach along with transplantation of FSCs in suspensions with separated cells of fetal liver, fetal brain and stem cells of fetal eye.
Statistical analysis
In a process of this investigation doctors applied a complex of statistic programs Statistica 6.0 (StatSoft, Tulsa, Oklahoma, USA). Samplings for all the results obtained were checked-up for a normal distribution by means of Kolmogorov-Smirnov test. With account for the data under study having different laws of distribution, mean difference and sample standard deviation were applied for a purpose of presenting specific characteristics. For calculations we used the methods for parametric (Student’s t-criterion with normal distribution) and non-parametric assessments: U - Mann-Whitney rank test (for independent sample), W - Wilcoxon signed rank (for dependent sample) following a distribution different from normal. Fischer’s exact test we applied to control a significant difference by all qualitative characteristic features. In all clinical cases such such a difference was regarded as significant when it reached the level of confidence <0.05. Correlations were calculated with the help of a test for non-parametric Spearman coefficient – as all studied parameters were relevant to the law of distribution, different from the normal ranges. Acquired correlation was considered to be statistically significant if Ñ€ <0.05.
Results
All individuals revealed a syndrome of early post-infusion improvements which were presented as better physical and emotional state in the patients. After administration of the suspensions containing cryopreserved stem cells of fetal liver we did not record any single evidence of complication or adverse reaction after therapy; no single case of graft-versus-host disease (GVHD) was recorded among the patients who received transplanted FSCs.
Both immediate and remote results of our study demonstrated efficacy of the above suggested method of FSCs therapy.
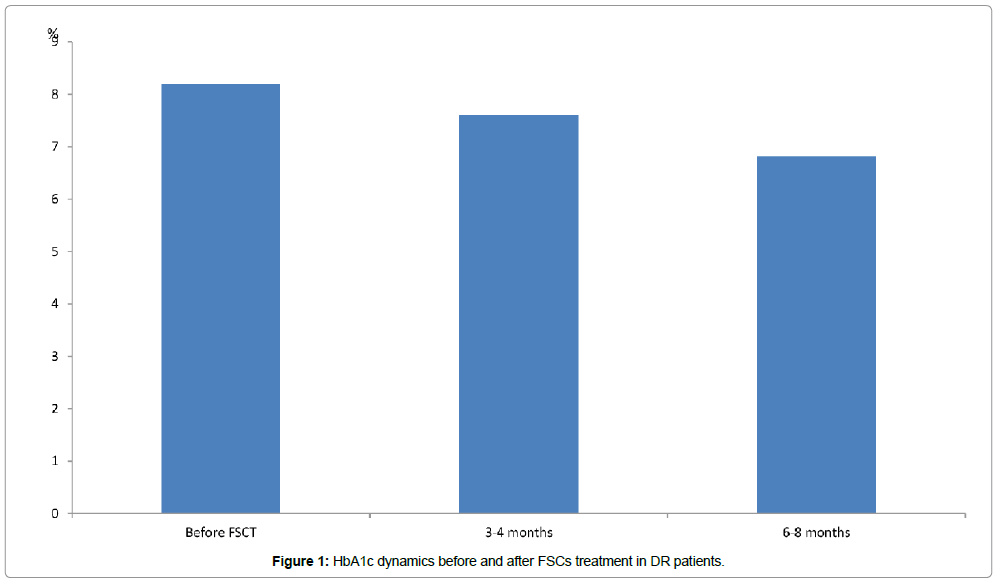
Thus, a tendency to decreasing in ranges of glycosylated hemoglobin (HbA1c) over 3-4 months after treatment was established as well as significant reduction in the levels of HbA1c within 6-8 months following FSCs administration (6.81 ± 0.21 % versus (8.2 ± 0.25 %), Ñ€<0.05 (Figure 1).
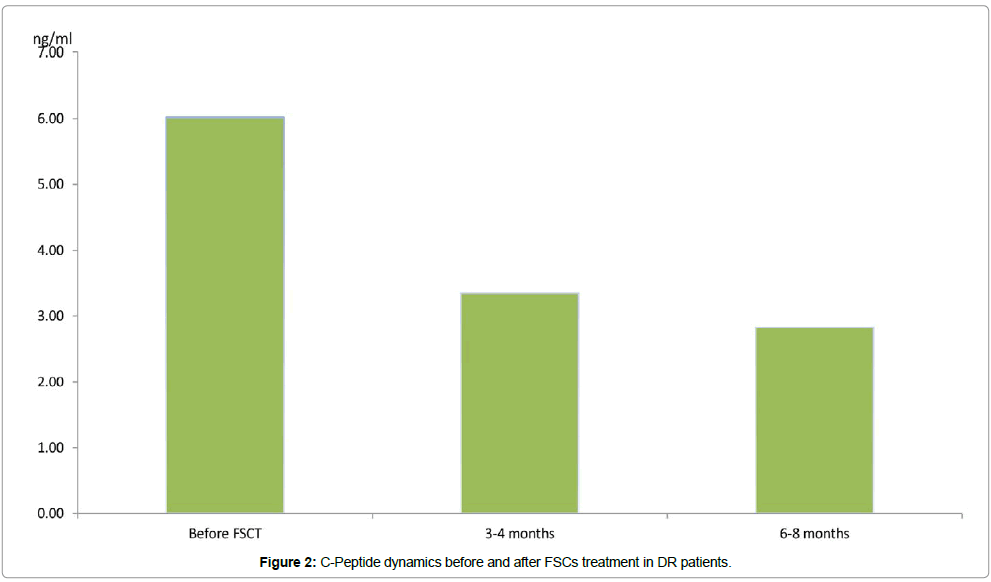
Over 3-4 months following FSCT a decreased value of blood serum C-peptide was remarkable in the patients under observation (on average by 6.02 ± 0.43, Ñ€<0.05). The same score remained stable in 9 patients (60.0%) over 6-7 months after FSCs transplantation (3.34 ± 0.31 on average, Ñ€<0.05); whereas the same range over 6-8 months constituted (2.82 ± 0.25 on average, Ñ€<0.05) (Figure 2).
Assessment of lipids profile was made and the results of such observation are described in the table presented below (Table 1).
| Values | Before treatment | Over 3-4 months | Over 6-8 months |
|---|---|---|---|
| Cholesterol, mmol/L | 6.4 ± 0.02 | 5.9 ± 0.04* | 5.2 ± 0.9** |
| Triglycerides, mmol/L | 0.35 ± 0.05 | 0.42 ± 0.05 | 0.51 ± 0.05 |
| HDL Cholesterol, mmol/L | 1.18 ± 0.02 | 1.21 ± 0.04 | 1.42 ± 0.05** |
| LDL Cholesterol, mmol/L | 5.2 ± 0.12 | 4.7 ± 0.13 | 3.5 ± 0.06** |
| Atherogenic index (AI) | 4.4 ± 0.11 | 3.8 ± 0.11 | 2.5 ± 0.07** |
* р<0.05 before treatment and following 3 months after FSCs therapy
**Ñ€<0.05 before treatment and following 6 months after FSCs therapy
Table 1: Dynamics of blood lipid levels among the DR patients before and following FSCs therapy.
In summary, based on the results presented in Table 1 we can observe significant improvement of lipids profile by 6 months following FSCs therapy. Significant lowering of total cholesterol was recorded over 3 months after FSCT (5.9 mmol/L versus 6.4 mmol/L), р<0.05. By 6 months following FSCs therapy significant increase in HDL levels was demonstrable (1.42 mmol/L) compared to the levels baseline (1.18 mmol/L), р<0.05 as well as significant decrease of the LDL ranges over 6 months after treatment if compared with the values baseline (3.5 mmol/L and 5.2 mmol/L respectively, р<0.05). The same changes were characteristic for the Atherogenic index (AI) (2.5 and 4.4, respectively, р<0.05).
In accordance with data of OCT, 53.33% of patients revealed higher thickness in the layer of retinal neural fibers throughout a peripapillary zone of both the right and left eye. Thickness of the right eye became higher by 5-7% on average, whereas the left eye thickness increased by 4-6%.
We observed insignificant increase in the mean thickness of macular retinal zone in both eyes (by 8-10 μm) in 60% of patients.
According to the results of automated perimetry for Macula Threshold in 73.33% of patients we recorded increased light perception by macular cells both in the right eye and in the left, whereas a reduction of relative scotoma (almost disappearance) was noteworthy in both eyes.
Nowadays, scientists proved few effective methods of therapy for DR focused on prevention, which include tight glycemic and cholesterol control promoting a sustained protection from disease ongoing progression [16-19]. If likely disease progresses to sightthreatening macular edema or proliferative diabetic retinopathy (PDR), laser photocoagulation helps reaching disease regression by means of peripheral retina destruction and thus, reduced demand in oxygen [20-23].
Retinal vessels degeneration is considered to be a criterion for earlier DR stage, which corresponds to non-proliferative diabetic retinopathy (NPDR). In the diabetic environment characterized by elevated levels of reactive oxygen species (ROS), vascular progenitors switch to producing pathologic cytokines such as tumor necrosis factor (TNF)-α, IL-8, resulting in higher expression of pathologic inducible nitric oxide synthase (iNOS) instead of endothelial nitric oxide synthase (eNOS) [24]. As a result, diabetic EPCs have reduced bioavailable nitric oxide (NO) due to either decreased activity of eNOS or increased generation of ROS via upregulated NADPH oxidase [25]. NO-mediated signaling events are significant for mobilization of EPCs from the bone marrow and homing in the ischemia zones [26,27]. As shown by the Grant’s group, the function of diabetic EPCs can be partially restored by increased expression of eNOS, either by using NO donors, or by reducing NADPH oxidase-dependent ROS production [28,29]. In addition, increased ROS and reduced bioavailable NO, as well as several other molecular alterations have been detected in dysfunctional diabetic EPCs, including decreased cathepsin L activity [30] and elevated expression of thrombospondin-1 [31].
In regard to proliferative diabetic retinopathy disease at advanced stages, or PDR, contemporary studies prove that high amounts of bone marrow-derived EPCs constitute a major factor in the development of such serious complications as pathologic neovascularization of ischemic tissues. Although increased numbers of circulating CD34+CD45−endothelial colony-forming cells (ECFCs) were found in patients with PDR compared to the control group [32], the same cells could be defective in capacity to migrate towards SDF-1, incorporate into and form vascular tubes. Such results suggest that even though the ECFCs from PDR patients are mobilized into the circulation, they are unable to properly migrate and repair damaged vascular endothelium [32].
Despite such interpretations, cell-based treatment could be presented as effective alternative strategy for the new approaches to treatment of end-stage DR similar to the other ischemic retinopathies. Stem cell therapy is targeted at early and intermediate stages of vascular degeneration by promoting vessels repair, contributing to reversed ischemia, reducing hypoxic or inflammatory signaling, as well as preventing progression to advanced and eyesight-threatening stages of DR [33]. Likely approach is proven to be successful if autologous progenitors are modified to function properly. In recent years, a number of methods have been developed to reverse defects of EPCs in diabetic patients, including improved mobilization for the EPCs and cells homing with granulocyte colony-stimulating factor (G-CSF) [34] and stromal derived factor (SDF-1) [35], or use of an NO donor to alleviate defects of SDF-1-mediated migration [36]. Subsequently, some data confirm that diabetic EPCs dysfunction could be improved or corrected by treatment using peroxisome proliferator-activated receptor (PPAR)-δ and -γ agonists GW501516 [37], rosiglitazone [38], or atorvastatin. Additionally, it was found that the levels of transforming growth factor (TGF)-β1 were significantly higher both in the EPCs and in the serum of type 2 diabetic patients. TGF-β1 inhibition in CD34+ cells enhanced overall cells survival, NO release, and in vivo vascular reparative ability, suggesting that this approach can be used for improved potential of dysfunctional diabetic CD34+ cells to regenerate cells by means of autologous therapy [36].
If one considers diabetic eye disease at large, stem cells might contribute to future treatment not only for retinal vasculature, but also for diabetic corneal disruptions known as diabetic keratopathy. Severity of likely changes, e.g. of neuropathy, can be correlated with corresponding severity of retinopathy [37]. A significant decrease in the expression of a number of putative stem cell markers in the compartment of corneolimbal epithelial stem cells has been recently established [38]. Since corneal epithelium could be renewed using limbal stem cells, this explains clinically a delay which could be observed at healing for diabetic wounds, for example, after epithelial debridement for vitrectomy [39] or following refractive surgery [40]. Adenoviral gene therapy with overexpression of c-met protooncogene and/or silencing of matrix metalloproteinase-10 and cathepsin F promotes stabilized epithelial wound healing due to stem cell marker expression in human organ-cultured diabetic corneas [38- 41]. It is essential that gene therapy by means of limbal stem cell niche could only produce the same normalization of expression for stem cell marker and healing of lesions [42]. In the future, gene therapy or replacement of ailing stem cells by the cultured normal cells similar to those made from iPSC [43] could become an alternative therapy option for alleviating diabetic corneal disease.
To summary everything above, fetal stem cells can facilitate new trends for retarding DR progression and, simultaneously, alleviating the symptoms of DR disease. Nowadays, their use is supported by an evidence of stem cells effect at early stages of this disease; long before the onset of PDR. Some of these cells by way of secreting special growth factors can serve for neuroprotection inside of diabetic retina. New strategies to normalize functions of diabetic progenitors offer ways to use them for autologous therapy. Endothelial progenitor cells are used for prevention and/or repair for capillary closure and will facilitate reduction in pericyte dropout. Established technologies by administration of ESCs and iPSC can also help generating bankable and renewable sources of stem cells being capable for differentiation in order to raise regeneration of retinal cells in a diabetic patient.
Conclusion
Use of fetal stem cells in combined treatment of the patients with DR can be characterized as safe and effective method contributing to compensation of carbohydrates and lipids metabolism among the patients.
The following significant clinical effects of FSCs have been observed
1. Syndrome of early post-infusion effects.
2. Advantages for overall physical and emotional states of the patients were recorded in 86.7% of cases.
3. Advantages in regard to blood glucose ranges.
Gradual reduction in fasting blood glucose levels, postprandial glycaemia and HbA1c have been initially demonstrable among the patients as early as by 3-4 months following FSCs administration; whereas the same values shown a significant lowering only by 6-8 months after FSCs administration.
4. Hypolipidemia therapeutic effects. We have recorded a significant improvement in parameters of lipids profile over 6-8 months after treatment administered.
5. Ophthalmology status among the patients. In conformity with results of OCT enhanced thickness of retinal neural fibers in peripapillary zone of the eye was characteristic.
We also observed insignificant increasing in moderate thickness of macular region of eyes retina bilaterally (by 8-10 μm) in among 60% of patients. Due to the results of automated perimetry for Macula Threshold, increased light perception by macular cells in both eyes was observed in 73.33% of individuals.
References
- Tao Z, Shi A, Zhao J (2015) Epidemiological Perspectives of Diabetes. Cell Biochem Biophys 73: 181-185
- Kajiwara A, Miyagawaa H, Saruwatari J, Kita A, Sakata M, et al. (2014) Gender differences in the incidence and progression of diabetic retinopathy among Japanese patients with type 2 diabetes mellitus: a clinic-based retrospective longitudinal study. Diabetes Res Clin Pract 103: 7–10.
- Wong TY, Klein R, Islam FM, Cotch MF, Folsom AR, et al. (2006) Diabetic retinopathy in a multiethnic cohort in the United States. Am J Ophthalmol 141: 446-455.
- Knudsen LL, Lervang HH, Lundbye-Christensen S, Gorst-Rasmussen A (2006) The North Jutland County Diabetic Retinopathy Study: population characteristics. Br J Ophthalmol 90:1404-1409.
- Caroline Day (2001) The rising tide of type 2 diabetes. Br J Diabetes Vasc Dis 1: 37-43.
- Nguyen TT, Wong TY (2006) Retinal vascular manifestations of metabolic disorders. Trends Endocrinol Metab 17: 262-268.
- Caldwell RB, Bartoli M, Behzadian MA, El-Remessy AE, Al-Shabrawey M, et al. (2005) Vascular endothelial growth factor and diabetic retinopathy: role of oxidative stress. Curr Drug Targets 6: 511-524.
- Nguyen QD, Tatlipinar S, Shah SM, Haller JA, Quinlan E, et al. (2006) Vascular endothelial growth factor is a critical stimulus for diabetic macular edema. Am J Ophthalmol 142: 961-969.
- Yau JW, Rogers SL, Kawasaki R, Lamoureux EL, Kowalski JW, et al. (2012) Global prevalence and major risk factors of diabetic retinopathy. Diabetes Care 35: 556-564.
- El-Asrar AM (2012) Role of inflammation in the pathogenesis of diabetic retinopathy. Middle East Afr J Ophthalmol 19: 70-74.
- Demchuk M, Ivankova O, Klunnyk M, Matiyashchuk I, Sych N, at al. (2016) Efficacy of Fetal Stem Cells Use in Complex Treatment of Patients With Insulin-resistant Type 2 Diabetes Mellitus. J Stem Cell Res Ther 6: 2-7.
- Ljubimov AV, Shaw LC, Li Calzi S, Kielczewski JV, Caballero S, Boulton ME, Grant MB. The vascular stem cell. In: Dartt DA, Besharse JC, Dana R, et al. (2010) Encyclopedia of the eye, San Diego, CA: Elsevier 289–99.
- Shaw LC, Neu MB, Grant MB (2011) Cell-based therapies for diabetic retinopathy. Curr Diab Rep 11: 265-274
- Kojima H, Kim J, Chan L (2014) Emerging roles of hematopoietic cells in the pathobiology of diabetic complications. Trends Endocrinol Metab 25: 178–187.
- Megaw R, Dhillon B (2014) Stem cell therapies in the management of diabetic retinopathy. Curr Diab Rep 14: 498.
- Nathan DM, Genuth S, Lachin J, Cleary P, The DCCT Research Group (1993) The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. N Engl J Med 329: 977-986.
- Robert Turner (1998) Effect of intensive blood-glucose control with metformin on complications in overweight patients with type 2 diabetes (UKPDS 34). UK Prospective Diabetes Study (UKPDS) Group. Lancet 352: 854-865.
- Barr CC (2001) Retinopathy and nephropathy in patients with type 1 diabetes four years after a trial of intensive insulin therapy. N. Engl. J. Med 342: 381-389.
- UK Prospective Diabetes Study (UKPDS) Group (1998) Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). Lancet 352: 837-853.
- Photocoagulation for diabetic macular edema (1985) Early Treatment Diabetic Retinopathy Study report number 1. Early Treatment Diabetic Retinopathy Study research group. Arch Ophthalmol 103: 1796-1806.
- Early Treatment Diabetic Retinopathy Study Research Group (1991) Early photocoagulation for diabetic retinopathy. ETDRS report number 9. Early Treatment Diabetic Retinopathy Study Research Group. Ophthalmology 98: 766-785.
- Aiello L (2003) Perspectives on diabetic retinopathy. Am J Ophthalmol 136: 122–135.
- Tan K, Lessieur E, Cutler A, Nerone P, Vasanji A, et al. (2010) Impaired function of circulating CD34(+) CD45(-) cells in patients with proliferative diabetic retinopathy. Exp Eye Res 91: 229–37.
- Shaw LC, Neu MB, Grant MB (2011) Cell-based therapies for diabetic retinopathy. Curr Diab Rep 11: 265-274.
- Cho HJ, Kim HS, Lee MM, Kim DH, Yang HJ, et al. (2003) Mobilized endothelial progenitor cells by granulocyte-macrophage colony-stimulating factor accelerate reendothelialization and reduce vascular inflammation after intravascular radiation. Circulation 108: 2918-2925.
- Butler JM, Guthrie SM, Koc M, Afzal A, Caballero S, et al. (2005) SDF-1 is both necessary and sufficient to promote proliferative retinopathy. J Clin Invest 115: 86-93.
- Hwang JH, Kim SW, Park SE, Yun HJ, Lee Y, et al. (2006) Overexpression of stromal cell-derived factor-1 enhances endothelium-supported transmigration, maintenance, and proliferation of hematopoietic progenitor cells. Stem Cells Dev 15: 260-268
- Segal MS, Shah R, Afzal A, Perrault CM, Chang K, et al. (2006) Nitric oxide cytoskeletal-induced alterations reverse the endothelial progenitor cell migratory defect associated with diabetes. Diabetes 55: 102-109.
- Jarajapu YP, Grant MB (2010) The promise of cell-based therapies for diabetic complications: challenges and solutions. Circ Res 106: 854-869
- Urbich C, Dernbach E, Rossig L, Zeiher AM, Dimmeler S (2008) High glucose reduces cathepsin L activity and impairs invasion of circulating progenitor cells. J Mol Cell Cardiol 45: 429–36
- Ii M, Takenaka H, Asai J, Ibusuki K, Mizukami Y, et al. (2006) Endothelial progenitor thrombospondin-1 mediates diabetesinduced delay in reendothelialization following arterial injury. Circ Res 98: 697–704
- Tan K, Lessieur E, Cutler A, Nerone P, Vasanji A, et al. (2010) Impaired function of circulating CD34(+) CD45(-) cells in patients with proliferative diabetic retinopathy. Exp Eye Res 91: 229-237
- Cheang WS, Fang X, Tian XY (2013) Pleiotropic effects of peroxisome proliferator-activated receptor ? and d in vascular diseases. Circ J 77: 2664–2671.
- Sorrentino SA, Bahlmann FH, Besler C, Müller M, Schulz S, et al. (2007) Oxidant stress impairs in vivo reendothelialization capacity of endothelial progenitor cells from patients with type 2 diabetes mellitus: restoration by the peroxisome proliferator-activated receptor-gamma agonist rosiglitazone. Circulation 116: 163-173
- Mohler ER, Shi Y, Moore J, Bantly A, Hamamdzic D, at al. (2009) Diabetes reduces bone marrow and circulating porcine endothelial progenitor cells, an effect ameliorated by atorvastatin and independent of cholesterol. Cytometry A 75: 75–82
- Bhatwadekar AD, Guerin EP, Jarajapu YP, Caballero S, Sheridan C, et al. (2010) Transient inhibition of transforming growth factor-ß1 in human diabetic CD34+ cells enhances vascular reparative functions. Diabetes 59: 2010–2019.
- Rosenberg ME, Tervo TM, Immonen IJ, Muller LJ, Gronhagen-Riska C, at al. (2000) Corneal structure and sensitivity in type 1 diabetes mellitus. Invest Ophthalmol Vis Sci 41: 2915–2921.
- Saghizadeh M, Soleymani S, Harounian A, Bhakta B, Troyanovsky SM, et al. (2011) Alterations of epithelial stem cell marker patterns in human diabetic corneas and effects of c-met gene therapy. Mol Vis 17: 2177-2190.
- Chen WL, Lin CT, Ko PS, Yeh PT, Kuan YH, et al. (2009) In vivo confocal microscopic findings of corneal wound healing after corneal epithelial debridement in diabetic vitrectomy. Ophthalmology 116: 1038-1047
- Simpson RG, Moshirfar M, Edmonds JN, Christiansen SM (2012) Laser in-situ keratomileusis in patients with diabetes mellitus: A review of the literature. Clin Ophthalmol 6: 1665–1674.
- Saghizadeh M, Epifantseva I, Hemmati DM, Ghiam CA, Brunken WJ, et al. (2013) Enhanced wound healing, kinase and stem cell marker expression in diabetic organ-cultured human corneas upon MMP-10 and cathepsin F gene silencing. Invest Ophthalmol Vis Sci 54: 8172–8180.
- Saghizadeh M, Dib CM, Brunken WJ, Ljubimov AV (2014) Normalization of wound healing and stem cell marker patterns in organ-cultured human diabetic corneas by gene therapy of limbal cells. Exp Eye Res 129: 66–73.
- Sareen D, Saghizadeh M, Ornelas L, Winkler MA, Narwani K, et al. (2014) Differentiation of human limbal-derived induced pluripotent stem cells into limbal-like epithelium. Stem Cells Transl Med 3: 1002-1012
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi