Research Article, J Diagn Tech Biomed Anal Vol: 8 Issue: 1
Ultrasound-Guided Axillary Lymph Node Biopsy: Retrospective Analysis
Lucia Veverkova1* and Katherine Vomackova2
1Department of Radiology, University Hospital Olomouc, IP Pavlova 6, Olomouc, 779 00, Czech Republic
2Department of Surgery I, University Hospital Olomouc, Czech Republic, IP Pavlova 6, Olomouc, 779 00, Czech Republic
*Corresponding Author : Lucia Veverkova
Department of Radiology, University Hospital Olomouc, IP Pavlova 6, Olomouc, 779 00, Czech Republic
E-mail: lucia.veverkova@fnol.cz
Received: May 20, 2019 Accepted: June 26, 2019 Published: July 03, 2019
Citation: Veverkova L, Vomackova K (2019) Ultrasound-Guided Axillary Lymph Node Biopsy: Retrospective Analysis. J Diagn Tech Biomed Anal 8:1. doi: 10.4172/2469-5653.1000135
Abstract
Purpose: The aim of this work is a retrospective evaluation of axillary lymph node core biopsy, evaluation of safety of this biopsy method, and its contribution to the diagnosis and staging of breast cancer.
Method: From January 2014 to September 2018, 932 core biopsies were performed and out of this total, 164 biopsies were performed in the axilla. All nodes were evaluated by ultrasound as pathological or suspicious. Data were collected retrospectively. Out of the total number of axillary biopsies, 148 biopsies were performed in women with newly diagnosed breast cancer or with a history of breast cancer. Other biopsies were indicated based on an accidentally suspicious or pathological finding on screening mammography or at the request of the attending physician after another examination (e.g., PET/CT for another indication with a suspicious node in the axilla). Axillary core biopsy was performed. Data were evaluated using pivot tables in MS Excel.
Results: Out of the total number of axillary node biopsies, 148 biopsies were performed in patients associated with breast cancer. In these patients, metastasis of breast cancer was found in 124 cases, with the remainder having benign changes in the lymph nodes. The group included 16 patients with no history of breast cancer. No post-biopsy complications were reported in any of the cases.
Conclusion: Core biopsy of axillary lymph nodes is safe procedure and should be used routinely to diagnose metastases. The introduction of the core biopsy to the routine practice can contribute to the implementation of targeted axillary dissection.
Keywords: Axilla; Lymph node; Core biopsy; Breast; Cancer; Staging
Introduction
Breast cancer most commonly metastasizes into ipsilateral axillary lymph nodes. Axillary lymph node metastases are associated with tumor size at diagnosis, tumor grade, lymph vascular invasion, and multifocality or multicentricity of the tumor. Axillary nodal status is an important prognostic factor.
Breast cancer staging involves sonographic evaluation of the axillary nodes [1-9]. According to the findings, the ultrasound examination is evaluated as normal, suspicious, or clearly pathological. All clearly pathological nodes are not necessarily verified by biopsy [5].
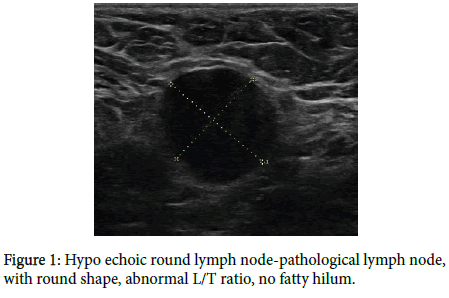
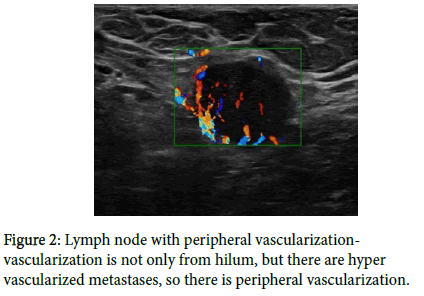
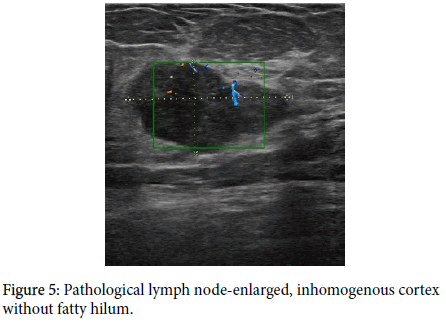
An ultrasonographic image of a pathological lymph node is based on the development of metastases that are deposited sub cortically [3] and then hypo echoic cortex becomes expanded on ultrasonographic features. The fatty hilum of the lymph node disappears (Figures 1 and 2). Metastases in the cortex are hyper vascularized; therefore, there is a peripheral type of vascularization in the lymph node on Doppler imaging.
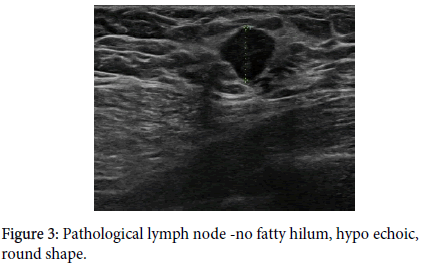
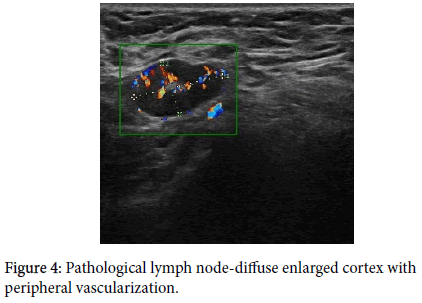
A clearly pathological lymph node is enlarged, hypo echoic, with no fatty hilum, and with peripheral vascularization (Figures 1-4) on an ultrasound image [1-4,8-11].
However, these clearly pathological lymph nodes must be differentiated from those that are not yet so morphologically altered, thus presenting a greater diagnostic challenge. The ultrasound image could be considered as suspicious and must always be followed by histological verification of these nodes. Minimal morphological changes may not correlate with metastasis [4,12].
There are two ways to verify histological changes in the nodes. Fine needle aspiration is reported in the literature [6,12,13], but due to its lower sensitivity, we use core biopsy only. At our Department, we use fine needle aspiration to diagnose metastases in axillary lymph nodes only in at-risk cases. This risk of per procedural bleeding can usually be eliminated by using Doppler imaging of the vascular structures [5], the patient’s position, and the choice of an appropriate access pathway [2,3,12].
Method
At our Department, a total of 932 core biopsies were performed from January 2014 to September 2018. Out of this total, 164 biopsies were performed in the axilla. Node morphology was assessed by ultrasound imaging and the nodes were subsequently labeled as clearly pathological or suspicious. Data were collected retrospectively. 148 biopsies were performed in women with a newly diagnosed or a history of breast cancer. Other axillary lymph node biopsies were indicated based on accidentally suspicious or pathological features on screening mammography or at the request of the attending physician after another examination (e.g., PET/CT in another indication with a suspicious node in the axilla).
In our study, axillary node biopsy was always guided by ultrasound navigation using the so-called “free-hand” technique. Under sterile conditions, after covering with a sterile drape and disinfecting the injection site, about 2 ml of local anesthetic (1% Marcaine) were applied to the skin and to the site of biopsy. A small incision was performed on the skin with a small scalpel. We used a biopsy gun with a needle diameter of 12 or 14 G for core biopsy. The needle was inserted close to the lymph node under sonographic guidance. Next, one to three samples 22 or 15 mm in length were taken. One sample is sufficient if it is representative and the radiologist is sure that it has been taken from a suspicious part of the lymph node. The data were subsequently evaluated using pivot tables in MS Excel.
Axillary node features, metastasis, and other histological findings were evaluated, as was the safety of the core biopsy by counting the number of complications after this procedure.
Results
Out of the total number of axillary node core biopsies, 148 biopsies were performed in breast cancer patients. In these cases, metastasis of breast cancer was found in 124 cases, follicular nodular hyperplasia in 10 cases, histiocytosis in 5 cases, node fibrosis in 1 case, and in 8 cases normal nodal structures with no histological changes were found (Table 1). In two patients, core biopsy ruled out metastases which were then confirmed by surgical sentinel node biopsy (Table 2). This correlates with the low false negativity described in the literature and in our study it was 1.6%. Sensitivity of this biopsy method in our study for metastasis of breast cancer was 98.4%.
| Histopathological features | Women with history of breast cancer | Women without history of breast cancer |
|---|---|---|
| Metastasis of breast cancer | 124 | 0 |
| Metastasis of other tumors | 0 | 2 |
| Lymphoma | 0 | 5 |
| Follicular nodular hyperplasia | 10 | 4 |
| Histiocytosis | 5 | 1 |
| Fibrosis | 1 | 1 |
| Normal lymph node | 8 | 1 |
| Sarcoid granuloma | 0 | 2 |
| Total | 148 | 16 |
Table 1: Two groups, the first includes women with newly diagnosed breast cancer or with breast cancer history, the second includes women without breast cancer history.
| Total amount of biopsies in women with breast cancer history | 148 |
|---|---|
| Women with metastases | 126 |
| Women with metastasis diagnosed by core biopsy | 124 |
| Women with metastasis diagnosed by sentinel node biopsy | 2 |
Table 2: Out of the total number of biopsies in women with breast cancer history metastases were founded in 124 cases by core biopsy, in 2 cases findings from core biopsy were negative and metastases were found by sentinel node biopsy during surgical performance, this points to low false negativity of core biopsy.
We performed 16 biopsies in patients with no history of breast cancer and without newly diagnosed breast cancer. In these cases, indication for core biopsy was based on the presence of a palpable enlargement of an axillary lymph node and its suspicious appearance on ultrasound imaging, or these patients had PET/CT in another indication and there was a suspicious lymph node in the axilla; or it was a patient with lymph node syndrome and biopsy was performed to exclude hemato-oncological malignancy and there were some reasons not to perform whole node extirpation. Among these patients, lymphoma was confirmed in 5 cases. These were patients who needed confirmation or exclusion of diagnosis of a hemato-oncological malignancy and they were not able to undergo surgical extirpation of the whole node. In 4 patients, follicular hyperplasia was found; in 2 cases metastasis of a primary tumor other than breast cancer was found; in 2 cases there were changes in the type of sarcoid granuloma; in one case fibrosis was present; in 1 there was histiocytosis; and in one case a normal node with no other histological features was present (Table 1).
Patients with breast cancer and with metastasis in an axillary lymph node underwent neoadjuvant oncological treatment; lymph node metastasis was subsequently confirmed intraoperative during axillary dissection. Patients in whom metastasis was not confirmed intraoperative were considered to have a complete pathological response to neoadjuvant oncological therapy. There were 24 breast cancer patients with benign features in biopsied lymph nodes; they underwent surgical treatment of breast cancer and sentinel node biopsy. In two of these patients, metastasis in an axillary lymph node during sentinel node biopsy was discovered intraoperative.
This suggests a low false negative rate of axillary lymph node core biopsy. Patients without breast cancer history with benign axillary node biopsy were followed up; in one of them, lymphoma was confirmed by surgical extirpation of the whole lymph node – this could point to the false negative rate of axillary lymph node core biopsy in this indication to exclude hemato-oncological malignancy (but in this study, there was a very small sample of population in this indication). Sarcoidosis was confirmed in one patient with sarcoid granuloma in histological feature. A second patient with this histological finding of sarcoid granuloma was referred to her general practitioner for further examination, but we have no further information about her. In the other patients with benign axillary lymph node biopsy after 6 and 12 months, follow-up ultrasound was performed and there was a stationary image or regression of morphological changes in the axillary lymph nodes.
No complications were seen in any of the cases, and manual compression for 5 minutes was performed in all patients as a prevention of hemorrhage. If minor bleeding occurred, we used manual compression for 5 to 10 minutes with respect to the severity of the bleeding. The patients were under supervision for another 15 to 30 minutes after the procedure, with none reporting any complications during that time. Only one patient had a small hematoma – its size was about 15 mm. No post biopsy complications were recorded long time after the procedure and no patients reported local pain after biopsy.
Discussion
Axillary node staging is an important prognostic factor in the diagnosis of breast cancer as well as one of the crucial factors in determining the type of primary therapy [2,4,8,9,12]. Axillary ultrasonography plays an important role in this staging.
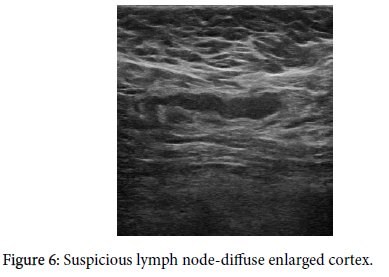
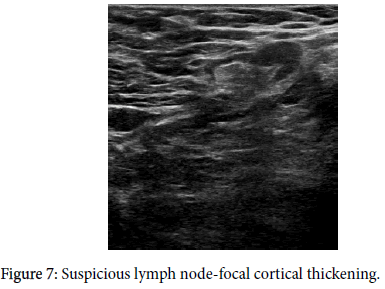
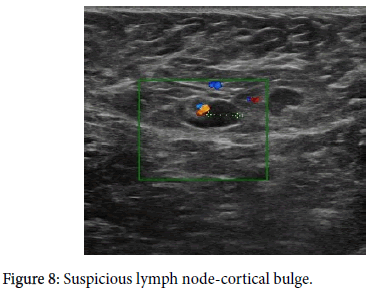
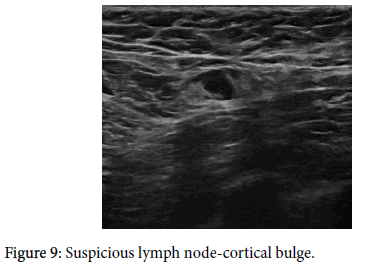
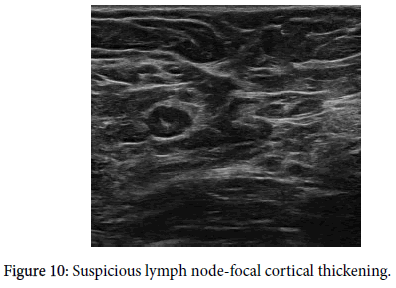
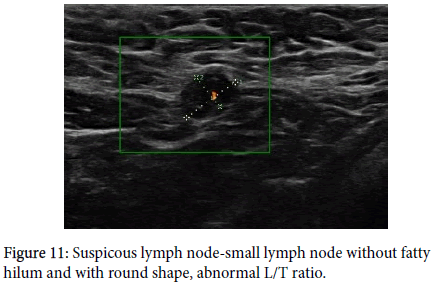
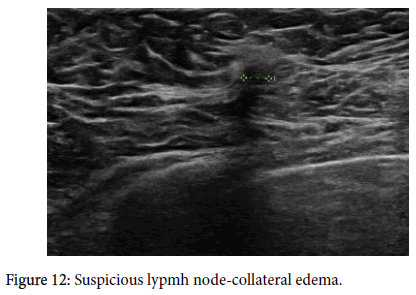
Axillary lymph nodes have a precisely described image on ultrasound. A benign node has an oval shape, an L/T ratio greater than 2, it has a preserved hyper echoic fatty hilum [1-3,8,9,10,12], its cortex is thin and regular, and there is polar type of vascularization (from the hilum). A clearly pathological lymph node is enlarged, hypo echoic, with no fatty hilum, and with peripheral vascularization (Figures 1-5) on an ultrasound image [1,3,4,8-11]. In the case of so-called “reactive nodes” of benign origin, the sonographic appearance may be different. These nodes may have an irregularly distributed cortex (Figure 6), sometimes with focal thickening or with cortical bulge (Figures 7-10), or inhomogeneity in an otherwise hypo echoic cortex [1,2,9,12-14], but usually the fatty hilum and the polar type of vascularization remain. The node may be small but round and hypo echoic, and the L/T ratio is abnormal (Figure 11). If found in patients without breast cancer and if side-symmetrical, these nodes are usually not considered suspicious. Another situation occurs when the finding of a node with an irregularly distributed cortex or with a cortical bulge is one-sided and there is an already known cancer diagnosis in the ipsilateral breast. These signs should be viewed in correlation with the contralateral axilla, including aggressiveness and size of the breast tumor. In this case, it also depends on the experience of the investigating radiologist whether or not he or she will evaluate the node as suspicious and order biopsy. Sometimes, a hyper echoic rim around the node (Figure 12) that corresponds to edema [9,12] may be a clue to performing biopsy. Biopsy should be indicated if the nodes are pathological, have a cortex wider than 4 mm, the hilum is diminished or the nodes are round and their L/T ratio is less than 2 (Figure 11). In some cases of breast cancer and clearly pathological nodes in the ipsilateral axilla, we leave the decision whether or not to perform node biopsy up to the oncologist. For instance, in cases where distant metastases of breast cancer have been confirmed and primary breast cancer has been confirmed locally, it is not necessary to verify pathological nodes in the ipsilateral axilla.
Ultrasound-guided core biopsy is the gold standard in breast cancer diagnosis as well as in the diagnosis of axillary nodal metastases. It is performed under local anesthesia with 16 to 12G needles. In order to confirm or exclude metastasis of breast cancer and in detecting breast cancer metastases, core biopsy is very accurate, with sensitivity between 88% and 100% [1,6,12,15].
In our study, ipsilateral lymph node biopsy was indicated in 148 breast cancer patients. Out of these 148 biopsies, metastases of breast cancer were confirmed in 124 cases. In two patients, core biopsy ruled out metastases which were then confirmed by surgical sentinel node biopsy. This correlates with the low false negativity described in the literature and in our study it was 1, 6%. Sensitivity of this biopsy method in our study for metastasis of breast cancer was 98, 4%.
Moreover, ultrasound-guided axillary lymph node core biopsy is a very safe method [2,3,5,6,8,12]. If we choose an appropriate access pathway, which may be influenced by the patient’s position and use Doppler imaging to differentiate vessels, we can perform this biopsy without any bleeding complications. Indeed, we did not notice any serious bleeding or infectious complications in our study.
Other indications for this biopsy method which exhibits high sensitivity and safety are to exclude the presence of metastases of another primary tumor or to confirm changes in a node of another histological origin. In the case of confirmation or exclusion of a hemato-oncological malignancy, extirpation of the whole node is preferred for diagnosis at our Department. Core biopsy can be used in this indication especially in diagnosis of lymphoma [16-18]. We use it due to practice at our Department when there are some reasons not to perform surgical extirpation of the whole node. For instance, if a node is placed deep and the patient is not able to undergo general anesthesia possibly due to internal causes [16,17]. In our study, five patients were shown to have lymphoma. In one patient, despite a negative core biopsy, suspicion of hemato-oncological malignancy persisted and it was subsequently confirmed by surgical extirpation the whole node.
When core lymph node biopsy and fine needle aspiration are compared, it should be noted that the literature reports a higher sensitivity of core biopsy; hence it should be preferred to verify the origin of morphological changes in axillary nodes [8,15]. But other studies describe that differences between sensitivity of these two methods in detection of metastases of breast cancer are not statistically significant [8]. According to habits at our Department we use fine needle aspiration when core biopsy is very risky in terms of bleeding complications.
Conclusion
Core biopsy is the gold standard in diagnosing of breast cancer. If there are doubts about morphological changes in the ipsilateral axillary lymph nodes in breast cancer patients and if these cannot be considered clearly benign, core biopsy is the method of choice for staging and treatment determination. Core biopsy is preferred over fine needle aspiration.
In other indications, axillary node core biopsy is also preferred and is very accurate in diagnosing metastasis of another primary tumor or in determining the histological nature of morphological changes in a benign node. It is also possible to indicate core biopsy to exclude or confirm a hemato-oncological malignancy if surgical extirpation of the whole node cannot be performed or if it is risky.
In addition, this type of biopsy is very safe with minimal risk of per procedural complications. The introduction of the core biopsy to the routine practice can contribute to the implementation of targeted axillary dissection.
Conflict of Interest
None declared. This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
References
- Dialani V, James DF, Slanetz PJ (2015) A practical approach to imaging the axilla. Insights Imaging 6: 217-229.
- Kim KH, Son EJ, Kim EK, Ko KH, Kang H, et al. (2008) The safety and efficiency of the ultrasound-guided large needle core biopsy of axilla lymph nodes. Yonsei Med J 49: 249-254.
- Abe H, Schmidt RA, Sennett CA, Shimauchi A, Newstead GM (2007) US-guided core needle biopsy of axillary lymph nodes in patients with breast cancer: Why and how to do it. Radiographics 27: 91-99.
- Pinheiro DJP, Elias S, Nazário ACP (2014) Axillary lymph nodes in breast cancer patients: sonographic evaluation. Radiol Bras 47: 240-244.
- Nori J, Bazzocchi M, Boeri C, Vanzi E, Nori Bufalini F, et al. (2005) Role of axillary lymph node ultrasound and large core biopsy in the preoperative assessment of patients selected for sentinel node biopsy. Radiol Med 109: 330-344.
- Marie AG, Margarita LZ, Gordon SA, Amy HL, Amy EK, et al. (2014) Ultrasound guided core biopsy versus fine needle aspiration for evaluation of axillary lymphadenopathy in patients with breast cancer. ISRN Oncology Article ID 703160: 9 pages.
- Jatoi I, Hilsenbeck SG, Clark GM, Osborne CK (1999) Significance of axillary lymph node metastasis in primary breast cancer. J Clin Oncol 17: 2334-2340.
- Houssami N, Turner RM (2014) Staging the axilla in women with breast cancer: the utility of preoperative ultrasound-guided needle biopsy. Cancer Bio Med 11: 69–77.
- Dudea SM, Lenghel M, Botar-Jid C, Vasilescu D, Duma M (2012) Ultrasonography of superficial lymph nodes: benign vs. malignant. Med Ultrason 14: 294–306.
- Rahbar H, Partridge SC, Javid SH, Lehman CD (2012) Imaging axillary lymph nodes in patients with newly diagnosed breast cancer. Curr Probl Diagn Radiol 41: 149-158.
- Houssami N, Ciatto S, Turner RM, Cody HS 3rd, Macaskill P. (2011) Preoperative ultrasound-guided needle biopsy of axillary nodes in invasive breast cancer: meta-analysis of its accuracy and utility in staging the axilla. Ann Surg 254: 243–251.
- L.Veverková, R.Dusíková (2016) The status of core-needle biopsy of axillary lymph nodes in the breast cancer diagnosis. Ces Radiol 70: 100-104.
- Suvi R, Amro M, Mazen S, Anna S, Outi P, et al. (2013) Axillary lymph node biopsy in newly diagnosed invasive breast cancer: comparative accuracy of fine-needle aspiration biopsy versus core-needle biopsy. Radiol 269: 54–60.
- Sahoo S, Sanders MA, Roland L, Pile N, Chagpar AB (2007) A strategic approach to the evaluation of axillary lymph nodes in breast cancer patients: analysis of 168 patients at a single institution. Am J Surg 194: 524–526.
- Vidya R, Iqbal FM, Bickley B (2017) Pre-operative axillary staging (2011) should core biopsy be preffered to fine needle aspiration cytology. ecancer 11: 724.
- Chun-Nan C, Tsung-Lin Y (2014) Application of ultrasound-guided core biopsy in head and neck. J Med Ultrasound 22: 133-139.
- Vandervelde C, Kamani T, Varghese A, Ramesar K, Grace R, et al. (2008) A study to evaluate the efficacy of image-guided core biopsy in the diagnosis and management of lymphoma: Results in 103 biopsies. European J Radiol 66: 107-111.
- Allin D, David S, Jacob A, Mir N, Giles A, et al. (2017) Use of core biopsy in diagnosing cervical lymphadenopathy: A viable alternative to surgical excisional biopsy of lymph nodes? Ann R Coll Surg Engl 99: 242–244.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi