Case Report, J Otol Rhinol Vol: 8 Issue: 1
A Case of Pembrolizumab Induced Autoimmune Sensorineural Hearing Loss
Kealan Hobelmann* and Dennis Fitzgerald
Deptartment of Otolaryngology, Thomas Jefferson University, PA 19107, USA
*Corresponding Author : Kealan Hobelmann
Department of Otolaryngology-Head and Neck Surgery, Thomas Jefferson University, 925 Chestnut Street, 6th Floor, Philadelphia, PA 19107, USA
E-mail: kealan.hobelmann@jefferson.edu
Received: February 05, 2019 Accepted: March 18, 2019 Published: March 27, 2019
Citation: Hobelmann K, Fitzgerald D (2019) A Case of Pembrolizumab Induced Autoimmune Sensorineural Hearing Loss. J Otol Rhinol 8:1. doi: 10.4172/2324-8785.1000365
Abstract
increasingly used to treat a variety of cancers by enhancing the immune response against cancer cells. Pembrolizumab is a humanized antibody which prevents tumor cells from deactivating host T-cells. Like most forms of immunotherapy, many of the adverse effects are autoimmune in origin due to the heightened immune response. Case: We present a rare case of Pembrolizumab induced autoimmune hearing loss in a patient with invasive melanoma which was responsive to steroids. A 67-year-old male presented with metastatic melanoma of the toe. He underwent amputation followed by treatment with IV pembrolizumab 2 mg/kg every 3 weeks. After a single dose, he experienced severe bilateral sensorineural hearing loss. His treatments were stopped and he was started on oral steroids. He later also underwent intratympanic steroid injection. He ultimately achieved improvement to normal-to-mild hearing loss levels in conversational frequencies with subjective return to baseline hearing. According to the FDA, this is one of 14 reported cases of autoimmune hearing loss from Pembrolizumab. Conclusion: Autoimmune hearing loss is a rare, but significant adverse effect of Pembrolizumab treatment for advanced melanoma which is responsive to steroids started up to 4 weeks after onset. While heightened awareness of this response is necessary, we do not recommend routine audiograms in patients undergoing treatment.
Keywords: Pembrolizumab; Hearing loss; Antibody; Metastatic melanoma
Introduction
In the last decade, immunotherapy has emerged as an important therapy option for a variety of malignancies. Pembrolizumab is a humanized antibody against the programmed cell death 1 (PD-1) receptor, part of a larger class of immune checkpoint inhibitors (ICIs). In 2014, Pembrolizumab was initially approved by the FDA for treatment of advanced metastatic melanoma, but has since expanded to other cancers including non-small cell lung cancer and squamous cell carcinoma of the head and neck among others. The adverse events related to ICIs differ somewhat by drug and by cancer type, although they are generally related to a heightened inflammatory response among normal cell types. The most common adverse events for Pembrolizumab in treatment of advanced melanoma include dermatitis, diarrhea, thyroid dysfunction, and arthralgias, though others have been reported as well [1,2].
In this report, we describe a patient treated with Pembrolizumab for metastatic melanoma who subsequently developed rapid onset of autoimmune sensorineural hearing loss which responded to systemic steroids and intratympanic steroid injections.
Case Report
A 67 year old man initially presented with a blister on his toe in December 2017. Biopsy revealed invasive melanoma. He underwent amputation and lymph node dissection with 12/20 positive nodes. Afterwards, he was scheduled to undergo treatment with IV pembrolizumab 2 mg/kg every 3 weeks. Approximately 1 week after his first infusion, the patient began to notice bilateral hearing loss. His symptoms were thought to be related to congestion and a referral was made to a local otolaryngologist for further evaluation. The decision was made to proceed with the second infusion as planned.
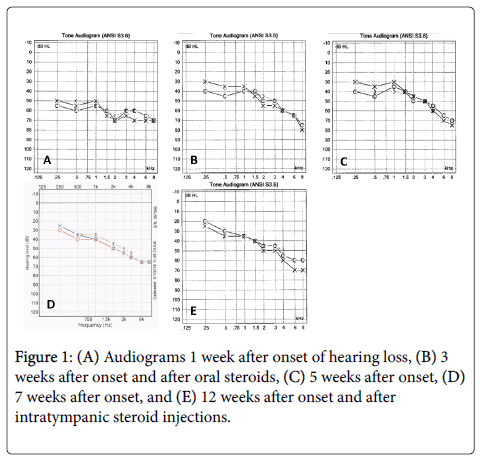
Soon after receiving his second infusion, the patient presented to an otolaryngologist with a primary complaint of progressive bilateral hearing loss. He described a dull quality to his hearing with poor word comprehension in conversation over the prior two weeks. Otoscopic exam revealed normal appearance of the external and middle ear bilaterally with Weber test midline and Rinne test with air conduction greater than bone conduction bilaterally. Audiologic testing at the time revealed symmetric moderate to severe sensorineural hearing loss (SNHL), type A (normal) tympanograms bilaterally, and absent otoacoustic emissions (OAEs) at all frequencies. Word recognition (WRT) was 72% in the left ear and 68% in the right ear.
Based on the findings of SNHL, the patient’s symptoms were attributed to the recent infusion of pembrolizumab and the decision was made to stop further treatment with the agent. He received 2 infusions total. The patient was then started on PO prednisone 60 mg daily for 5 days followed by a slow taper over 10 days. In total, treatment was started 4 weeks after the onset of SNHL.
The patient was seen 2 weeks later and reported significantly improved subjective hearing. Otoscopic exam was again normal. Audiogram at that time showed significant symmetric improvement in the lower frequencies to mild hearing loss levels with continued severe SNHL in the higher frequencies and type A tympanograms. WRT was 68% in the left ear and 88% in the right ear. The patient was noted to have an adequate response to medication and was not prescribed any additional steroids.
The patient was seen another 2 weeks later and reported no change in subjective hearing. Audiometry was largely unchanged since the prior visit with continued mild to severe SNHL, mostly in the high frequencies. WRT was 80% in the left ear and 100% in the right ear. The patient was given the option of hearing aids and a referral was made to an otologist due to concern for future recurrent hearing loss.
About 2 weeks later, the patient was evaluated in an otology clinic. He reported stable hearing loss since his prior visit, bilateral tinnitus and aural fullness. Audiologic testing showed slight improvement across all frequencies bilaterally, a type A’s right tympanogram and type A left tympanogram, and absent OAEs at all frequencies bilaterally. WRT was 92% on the left and 96% on the right. The patient underwent intratympanic injection of 0.4 cc of dexamethasone 10 mg/ml on the right side. A week later, he returned for injection of the left ear as well.
Finally, 5 weeks later (roughly 12 weeks after treatment), the patient returned to his primary otolaryngologist with subjective resolution of his hearing loss. Audiometry revealed stable symmetric mild SNHL in the low frequencies and moderate SNHL in the high frequencies. WRT was 84% on the left and 95% on the right. The patient was reassured and no further intervention was made.
Discussion
It has been shown that some tumors block the natural immune response by the expression of PD-L1 and PD-L2 which bind the PD-1 receptor on T-cells, causing deactivation [3]. Pembrolizumab is a humanized antibody which binds the PD-1 receptor on T-cells to inhibit binding to tumor PD-L1 and PD-L2 ligands and prevent T-cell deactivation. By preserving T-cell function, Pembrolizumab allows for enhanced natural host immune response against tumor cells [3].
Melanocytes are expressed most abundantly in the skin, but are also present in other tissues including the uvea of the eye, the stria vascularis of the inner ear, and the meninges [4]. It is postulated that enhanced immune response to melanocytic cells is responsible for both ocular and audiovestibular symptoms in patients treated for melanoma [5,6]. In fact, melanoma alone has been shown to be associated with retinopathy due to the induced host response to tumor cells [7].
A corollary to the autoimmune hearing loss associated with Pembrolizumab is Vogt-Koyanagi-Harada disease, an autoimmune granulomatous disease characterized by panuveitis, hearing loss, and vitiligo. Melanocytes in the skin, eye, and inner ear are targeted by T-cells, typically after a viral infection. Hearing loss is reported in 70% of cases while tinnitus is present in 42%. Vestibular involvement is rare. Treatment generally consists of oral steroids, often up to 6 months [8].
This case demonstrates the acute onset of autoimmune sensorineural hearing loss following a single dose of IV pembrolizumab 2 mg/kg, successfully treated with oral prednisone 60 mg daily for 5 days followed by a slow taper over 10 days. Despite a delay of 4 weeks until treatment by an otolaryngologist, the patient achieved subjective return to baseline hearing. Of particular interest, the patient demonstrated neither ocular impairment nor vertiginous symptoms during his course of treatment.
There is only one other case report of autoimmune hearing loss related to pembrolizumab. Zibelman et al. describe a patient with metastatic melanoma involving the posterior nasopharynx with skull base erosion and orbital extension treated first with ipilimumab and later switched to pembrolizumab after progression of disease. The patient developed autoimmune SNHL after 2 doses of pembrolizumab. He was treated with twice weekly intratympanic steroid injections for a total of 6 injections on the right and 4 injections on the left with subjective resolution of his hearing loss [6]. As pointed out by Nader et al., the anterior skull base involvement of that case makes leptomeningeal metastasis another important etiology to consider, though the response to steroids makes drug reaction more likely [9].
Our case is the first report of steroid-responsive SNHL in a patient treated with pembrolizumab alone. Audiologic testing demonstrated return of hearing to mild to moderate hearing loss levels at conversational frequencies after the steroid course. Over the next 6 weeks, hearing continued to improve to normal to mild hearing loss levels at conversational frequencies without any additional intervention. These results suggest that in addition to being steroid-responsive, autoimmune hearing loss caused by pembrolizumab involves some degree of spontaneous resolution (Figure 1).
While there is limited data on sudden sensorineural hearing loss (SSNHL) related to immunotherapy, it is well established that recovery from idiopathic SSNHL is correlated with time elapsed from presentation to treatment with oral or intratympanic steroids. The optimum window is generally accepted to be 10-14 days from onset [10,11]. However, additional studies have shown response to steroids started up to 2 months from onset [12].
Conclusion
SNHL is a rare, but notable adverse effect related to the use of pembrolizumab in the treatment of melanoma, most likely due to the autoimmune involvement of the stria vascularis in the cochlea. Patients may present without vertiginous symptoms. Recovery is possible with corticosteroids started as late as 4 weeks after symptom onset, though we recommend treatment within 10 days for optimum response. We also recommend cessation of pembrolizumab when possible to allow for maximal recovery, though this decision must be made in light of treatment goals and alternative therapies. With cessation of the inciting agent, there is a degree of spontaneous recovery. While heightened awareness of this adverse effect is necessary, we do not recommend routine audiograms in patients undergoing treatment. These findings also highlight the importance of melanocytic cells in the function of the cochlea and inner ear.
References
- FDA Adverse Event Reporting System (FAERS) Public Dashboard.
- Spain L, Diem S, Larkin J (2016) Management of toxicities of immune checkpoint inhibitors. Cancer Treat Rev 44: 51-60.
- Patnaik A, Kang SP, Rasco D, Papadopoulos KP, Elassaiss-Schaap J (2015) Phase I study of pembrolizumab (MK-3475; Anti-PD-1 Monoclonal Antibody) in patients with advanced solid tumors. Clin Cancer Res 21: 4286-4293.
- Steel KP, Barkway C (1989) Another role for melanocytes: Their importance for normal stria vascularis development in the mammalian inner ear. Development 107: 453-463.
- Seaman BJ, Guardiani EA, Brewer CC, Zalewski CK, King KA, et al. (2012) Audiovestibular dysfunction associated with adoptive cell immunotherapy for melanoma. Otolaryngol Head Neck Surg 147: 744-749.
- Zibelman M, Pollak N, Olszanski AJ (2016) Autoimmune inner ear disease in a melanoma patient treated with pembrolizumab. J Immunother Cancer 4: 8.
- Barozzi S, Ginocchio D, Socci M, Alpini D, Cesarani A (2015) Audiovestibular disorders as autoimmune reaction in patients with melanoma. Med hypotheses 85: 336-338.
- Lavezzo MM, Sakata VM, Morita C, Rodriguez EE, Abdallah SF, et al. (2016) Vogt-Koyanagi-Harada disease: Review of a rare autoimmune disease targeting antigens of melanocytes. Orphanet J Rare Dis 11: 29.
- Nader ME, Myers JN, Gidley PW (2017) Sudden hearing loss in a melanoma patient on pembrolizumab: An etiology not to be omitted in the differential diagnosis. J Immunother Cancer 5: 24.
- Edizer DT, Celebi O, Hamit B, Baki A, Yigit O (2015) Recovery of idiopathic sudden sensorineural hearing loss. J Int Adv Otol 11: 122-126.
- Stachler RJ, Chandrasekhar SS, Archer SM, Rosenfeld RM, Schwartz SR, et al. (2012) Clinical practice guideline: Sudden hearing loss. Otolaryngol Head Neck Surg 146: 1-35.
- Anyah A, Mistry D, Kevern E, Markiewicz K (2017) Idiopathic sudden sensorineural hearing loss: Average time elapsed before presentation to the otolaryngologist and effectiveness of oral and/or intratympanic steroids in late presentations. Cureus 9: e1945.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi