Research Article, J Appl Bioinforma Comput Biol Vol: 7 Issue: 2
Ab Initio Modeling and Garcinia Biflavonoids-Binding Study of Tyrosinase: The Signature Enzyme of Melanogenesis
1Centre for Bio-computing and Drug Development, Adekunle Ajasin University, Akungba-Akoko, Ondo State, Nigeria
2Department of Biochemistry, Adekunle Ajasin University, Akungba-Akoko, Ondo State, Nigeria
*Corresponding Author : Ogunwa Tomisin Happy
Centre for Bio-computing and Drug Development, Department of Biochemistry, Adekunle Ajasin University, Akungba-Akoko, Ondo State, Nigeria
E-mail: tomisin.ogunwa@aaua.edu.ng, ogunwatomisin@gmail.com
Received: February 26, 2018 Accepted: March 28, 2018 Published: April 04, 2018
Citation: Ogunwa TH (2018) Ab Initio Modeling and Garcinia Biflavonoids-Binding Study of Tyrosinase: The Signature Enzyme of Melanogenesis. J Appl Bioinforma Comput Biol 7:2. doi: 10.4172/2329-9533.1000149
Abstract
Hyperpigmentation in humans can result from sun damage, inflammation, or other skin injuries. Tyrosinase, a crucial enzyme which catalyzes the main and rate limiting steps in melanogenesis, is a validated target in the treatment of skin hyperpigmentary disorders. Previously, employing in vitro analysis, inhibitory activities of Garcinia biflavonoid complex (kolaviron) and morelloflavone (a Garcina dulcis biflavonoid) on tyrosinase were observed. In the current study, ab initio modeling was carried out for tyrosinase and the resultant structure was employed in molecular docking to investigate the precise interaction pattern of these biflavonoids on the protein’s putative binding site. The results obtained revealed that the biflavonoids occupied the binding pocket (tropolone binding site) with one of its monoflavonoid subunits and completely barricaded the entrance of the active site with the other subunit. In this way, the biflavonoids could successfully prevent L-tyrosine and DOPA (substrates) access into the active site. Also, the biflavonoids subunit interferes with one of the two copper ions found within the binding site, an indication of their moderate inhibitory activity. Among the ligands studied, morelloflavone possessed the highest affinity (-8.5 kcal/mol) while GB1 displayed the lowest affinity (-7.6 kcal/mol) to the enzyme. Kolaflavanone and GB2 also showed modest affinity and good interaction pattern with the enzyme. These binding energy values further validate the moderate inhibitory potential of the ligands against the protein. Interestingly, these data are consistent with previous in vitro reports and thus, verify that the Garcinia biflavonoids are unique naturally-occurring competitive inhibitors of tyrosinase. Hence, their chemical scaffolds might contribute to the design and development of new tyrosinase inhibitors for combating hyperpigmentation.
Keywords: Hyperpigmentation; Tyrosinase; Garcinia bioflavonoids; Molecular docking; Melanogenesis
Introduction
Melanin, a major skin pigment molecule, plays an essential role in the protection of human skin from harmful damages caused by UV radiation from the sun. However, when an abnormal amount of melanin accumulates in different specific parts of human skin, it can result in a number of known pigmented patches which are often viewed as an esthetic problem [1]. Although still controversial, excess melanin in the body is said to be toxic through its binding to numerous drugs which can lead to ocular toxicity such as retinopathy. Hence, the pharmaceuticals may elicit toxic effects through accumulation in tissues. Also, forming complexes with melanin can easily prolonged their exposure, apart from the possible interference with melanin protective role. Furthermore, excessive production of melanin can yield highly reactive compounds like quinones as intermediate metabolites and they can initiate cell damage or death [2,3].
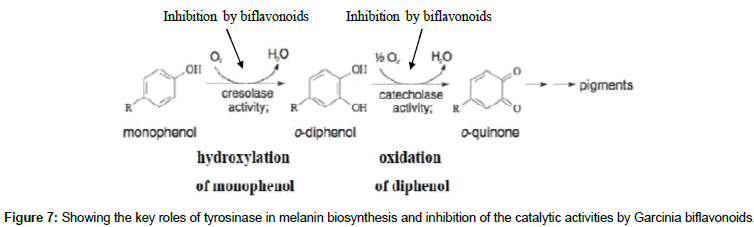
Apart from the photoprotective role of melanin in the skin, the pigment also contributes to the determination of a person’s phenotypic appearance [1,4]. Melanin is a large biological pigment and its formation involves a combination of chemical and enzymatically catalyzed reactions. It is produced through a biosynthetic pathway utilizing L-tyrosine as a precursor. Tyrosinase (E.C.1.14.18.1) is the enzyme that catalyzes the conversion of L-tyrosine to 3,4-dihydroxyphenylalanine (L-DOPA) which is the committed step in the pathway, and also oxidizes L-DOPA to form dopaquinone [5]. Hence, the enzyme controls the rate-limiting step in melanin biosynthesis. In succinct details, tyrosinase can be described as a copper-containing metalloenzyme which is multifunctional and glycosylated and, exhibits both monophenolase activity and diphenolase activity during mammalian melanogenesis [6]. The tyrosinase protein is specifically implicated in two particular reactions in melanin biosynthesis pathway, the hydroxylation of a monophenol and the conversion of an O-diphenol to the corresponding O-quinone. The spontaneous polymerization of this O-quinone which is a highly reactive compound yields melanin [4-6].
Over the years, there have been unrelenting efforts by researchers in the search for tyrosinase inhibitors from both natural and synthetic origin simply because of undesirable hyperpigmentary disorders like melasma, lentigo, freckles, age spot, senile lentigines and ephelide in human skin and, their application in the enzymatic browning associated with beverages, fruits and vegetables [7-10]. The compounds sought are expected to be potent and safe while used medicinally in skin whitening treatment, agriculturally as antibrowning in farm products and for cosmetic purposes. The current tyrosinase inhibitors such as hydroquinone, mercury and steroids have been associated with undesirable cutaneous and systemic side effects including ochronosis, irritant contact dermatitis and renal complications due to long-term use [11-13]. Therefore, discovery of new chemical scaffolds as potent and less toxic inhibitors of tyrosinase is still encouraged.
For the last few decades, chemical investigations of various Garcinia species gained the attention of researchers resulting in an extensive evaluation being carried out on their phytochemical and biological activities [14]. Garcinia is a very large genus of Clusiaceae family which is well-known for interesting bioactivities and comprises of around 450 species of herbs, shrubs and small trees distributed in Tropical Africa, America, Australia, Madagascar and Asia. Members of Garcinia L. are used in traditional system of medicine to treat dysentery, menstrual problems, obesity, diabetes, fever, cancer, HIV and inflammation [14,15]. Garcinia kola is one of the well-documented species among the Garcinia genus. Just like the other valuable species, G. kola is a rich source of secondary metabolites such as phenolic compounds, lactones, flavonoids, xanthones and benzophenones [16]. In addition, some biflavonoids have been isolated from Garcina species. These compounds are dimers of monoflavonoids, flavone-flavone, flavanone-flavonol, flavanone-flavone and flavanone-flavanone subunits, linked together with a C-C or C-O-C bond [17]. The natural biflavonoids have been associated with numerous bioactivities such as antioxidative, anticancer, antiviral, antimicrobial and anti-inflammatory effects [17-21]. A biflavonoid complex obtained from the extract of Garcinia kola seeds is called kolaviron. The identified constituents of kolaviron include the Garcinia GB1, GB2 and kolaflavanone [19]. As a complex, various chemical and biological investigations have been carried out on kolaviron for the last few years [22-24]. Morelloflavone is another biflavonoid comprising of luteolin and apigenin as its subunit monoflavonoids. It has been reported in many Garcinia species such as G. edulis, G. cambogia, G. multiflora, G. morella and, possesses potent pharmacological potentials such as antifungal, anti-hepatotoxic, anti- HIV, antimalarial and anti-inflammatory effects [25,26].
Evidence abound in the literature to show the various pharmacological properties of kolaviron including antidiabetes, antioxidant, antiviral, antihypercholesterolemia, antimicrobial, antiprotozoal, antiobesity, anti-inflammatory and neuroprotective effect [21-24,27-29]. Not only that, kolaviron has been reported to inhibit the catalytic activity of some enzymes such as alpha amylase, aldose reductase, alpha glucosidase, tyrosinase and acetylcholinesterase in vitro [21,30,31]. On the other hand, morelloflavone possesses in vitro inhibitory potential against HMG-COA reductase, papain and cruzain (cysteine proteases), trypsin (serine peptidase), HIV-1 reverse transcriptase and phospholipase A2 obtained from snake venom [26,32-34]. Despite these reports, there exists a scarcity of evidence to show the precise pattern of molecular interaction between these proteins and the Garcinia biflavonoids. Considering the importance of tyrosinase as a target for the pharmaceuticals in the treatment of hyperpigmentation and in browning process of agricultural producce, it would be interesting to provide information on the interaction mechanism of the Garcinia biflavonoids with the enzyme. Therefore, the current in silico study was carried out to analyze the precise interaction pattern and inhibitory potential of morelloflavone and kolaviron constituents (Garcinia biflavonoids) with tyrosinase in order to study their potency as tyrosinase inhibitors and the molecular mechanism of inhibition.
Materials and Method
Ligand preparation and optimization
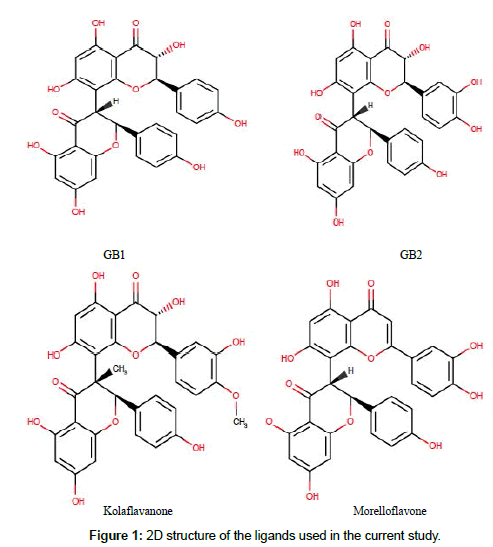
A total of four (4) ligands used in this docking study were selected from the literature [30,34]. The compounds were biflavonoids isolated from Garcinia species. GB1, GB2 and kolaflavanone are isolated from kolaviron, a biflavonoid complex obtained from Garcinia kola seeds while morelloflavone is isolated from Garcinia dulcis. The chemical structure of these compounds was prepared using Marvinsketch. 2D-coordinate of the ligands was sketched using ChemAxon software (https://www.chemaxon.com), and, with the conformers suit of Marvin-Sketch, the 2D structures were converted to 3D geometry using the Merck molecular force field (MMFF94). The 3D format (sdf) of the compounds was docked into the tyrosinase model using the AutoDock 4.2. The best results in terms of conformation and binding energy values were chosen for further analysis.
Preparation of target protein
The starting coordinate of tyrosinase used in this study was retrieved from the Brookhaven protein data bank (http://www.rcsb.org/pdb). Crystal structure of tyrosinase with PDB ID: 2Y9X was downloaded and visualized using Pymol. The protein was bound to tropolone as deposited by Ismaya et al. [35]. The “FASTA” file (Accession: 2Y9X_A GI: 339717372) for tyrosinase was then retrieved from Genbank and employed in ab-initio modeling as done on the iTASSER Server (https://zhanglab.ccmb.med.umich.edu/ITASSER/). The active site of the protein was identified with reference to the co-crystallized ligand. Except the copper ions, all other cocrystallized ligands and water molecules were then deleted from protein crystal before molecular docking procedures.
Structural evaluation and validation of the protein model
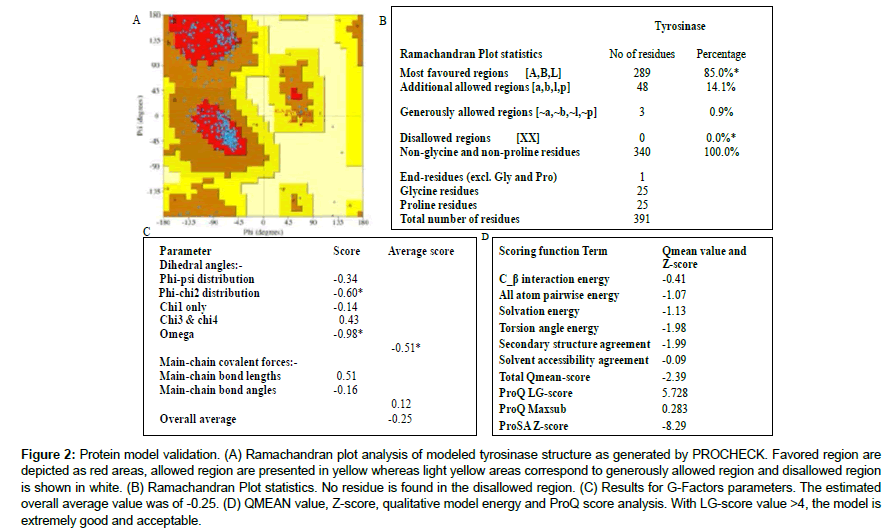
The quality of modeled tyrosinase was evaluated using PROCHECK validation. Ramachandran plot was obtained using Pdbsum database of the European Bioinformatic institute (EMBLEBI) (http://www.ebi.ac.uk/) while the Ramachandran statistic plot values, LG-score, Maxsub, Qmean score and Z score were also estimated with the aid of relevant online servers.
Molecular docking and scoring
For ligand docking, scoring and visualization, Autodock Vina suite on PYMOL was used [36,37]. First, based on the already present co-crystallized ligand in the pdb file, the inhibitor binding site was defined with grid parameters set at x=120, y=120 and z=120 while the coordinate of origin (x, y and z) was set at -7.41, -23.63 and -32.64 to include all the amino acid residues at the active site. This gives enough space to enhance adequate ligand rotation and translation. The spacing between grid points was maintained at 0.375 angstroms. All optimized ligands were docked to the active site of the protein. While the rotatable bonds of the ligands were set to be free, the protein molecule was treated as a rigid structure. Throughout this in silico investigation, ten (10) docking runs were performed for each ligand with the number of modes set to 10 so as to achieve more accurate and reliable results.
Data analysis
The protein-ligand complexes as well as the molecular interaction were all visualized using PYMOL and snapshots were taken.
Results and Discussion
Herein, in silico tools were employed to prepare a model of tyrosinase protein on which studies were carried out to evaluate the binding signature and molecular interaction of GB1, GB2, kolaflavanone and morelloflavone (Garcinia biflavoinoids) as inhibitors of the protein. The choice of this study was based on the fact that hyper-pigmentation disorders still remain very common among Africans coupled with an extensive use of medicinal plant products among the rural people of the continent [38,39]. It is envisaged that the results of this work will contribute to the design and development of new pharmaceuticals for the management of hyperpigmentation challenges. In addition, the so-far observed increasing demand for novel products for depigmentation, cosmeceutical, and skin lightening purposes which is a result of the recent expansion of global market makes this study very relevant. Furthermore, African farmers might prefer cheap natural agents to prevent enzymatic browning in raw vegetables and fruits. The chemical structure of the ligands used in this study is given in Figure 1 and their calculated physicochemical properties are presented in Table 1 which suggest them as biologically active compounds.
| Ligand | Chemical formular | Molecular weight | No of HB donor | No of HB acceptor | Plant sources |
|---|---|---|---|---|---|
| GB1 | C30H22O11 | 558.495 | 7 | 11 | Garcinia kola |
| GB2 | C30H22O12 | 574.494 | 8 | 12 | Garcinia kola |
| Kolaflavanone | C31H24O12 | 588.521 | 7 | 12 | Garcinia kola |
| Morelloflavone | C30H20O11 | 556.101 | 7 | 11 | Garcinia dulcis, G. cambogia, G. morella, G. multiflora |
Table 1: Ligands physicochemical properties and plant sources.
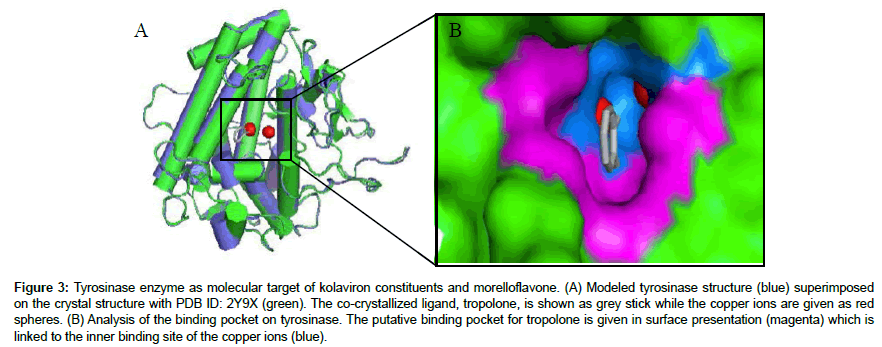
The tyrosinase protein used in the current in silico experiment is a 128 kDa tetrameric protein [40] which has been proved as a major molecular target for anti-hyperpigmentation drugs [41]. Albeit crystallographic structure for human tyrosinase has not been deposited yet in the protein data bank, several authors have used the available structure of mushroom tyrosinase to investigate the plausible interaction of drugs with the enzyme [1,10,12,42-44]. Thus, this protein crystal was retrieved from the data bank (PDB ID: 2Y9X) and visualized in PYMOL. However, it is not unknown that protein crystal structures found online at the databanks may harbor hidden errors such as missing residues and broken chains/linkages in available X-ray or NMR crystal structures which require correction. Therefore, the most appropriate and acceptable methodology is to build a starting protein model for an in silico experiment using appropriate coordinates. The two famous methods of protein modeling are the homology modeling and the ab initio modeling. In the present study, the later was adopted to build the protein structure to be targeted by Garcinia biflavonoids towards unveiling the binding poses and molecular interaction. The resultant protein structure was validated by analyzing dihedral angles by Ramachandran plot analysis (Figure 2a). All the amino acid residues of the protein model were found within the allowed region with none of the residues at the disallowed region. Moreover, LG score (5.728), maxsub score (0.28), Z-score (-0.829) and G. factor (0.25) value obtained for the model prove the good quality and hence, acceptability of the protein structure for further docking studies (Figure 2b-2d). It is expected that models built this way would be more suitable for examining ligand interaction and binding affinity [45]. Furthermore, superimposition of the model with the crystal structure revealed the binding sites as visualized on PYMOL. The surface representation of the enzyme structure is given in Figure 3, revealing the “tropolone binding site and the copper ions binding pocket”. Many reported inhibitors of tyrosinase are known to bind to this active site [41-43]. While some may penetrate deeper into the binuclear cupper-binding groove, other inhibitors stayed within the entrance sharing similar mechanism of inhibition to tropolone. To date, research on inhibition of tyrosinase has attained significant level because they have potential applications not only in medicine but also in cosmetics (as whitening agents) and in agriculture (as bioinsecticides). Hence, it is an all-time requirement to design new and potent inhibitors of this enzyme.
Figure 2: Protein model validation. (A) Ramachandran plot analysis of modeled tyrosinase structure as generated by PROCHECK. Favored region are depicted as red areas, allowed region are presented in yellow whereas light yellow areas correspond to generously allowed region and disallowed region is shown in white. (B) Ramachandran Plot statistics. No residue is found in the disallowed region. (C) Results for G-Factors parameters. The estimated overall average value was of -0.25. (D) QMEAN value, Z-score, qualitative model energy and ProQ score analysis. With LG-score value >4, the model is extremely good and acceptable.
Figure 3: Tyrosinase enzyme as molecular target of kolaviron constituents and morelloflavone. (A) Modeled tyrosinase structure (blue) superimposed on the crystal structure with PDB ID: 2Y9X (green). The co-crystallized ligand, tropolone, is shown as grey stick while the copper ions are given as red spheres. (B) Analysis of the binding pocket on tyrosinase. The putative binding pocket for tropolone is given in surface presentation (magenta) which is linked to the inner binding site of the copper ions (blue).
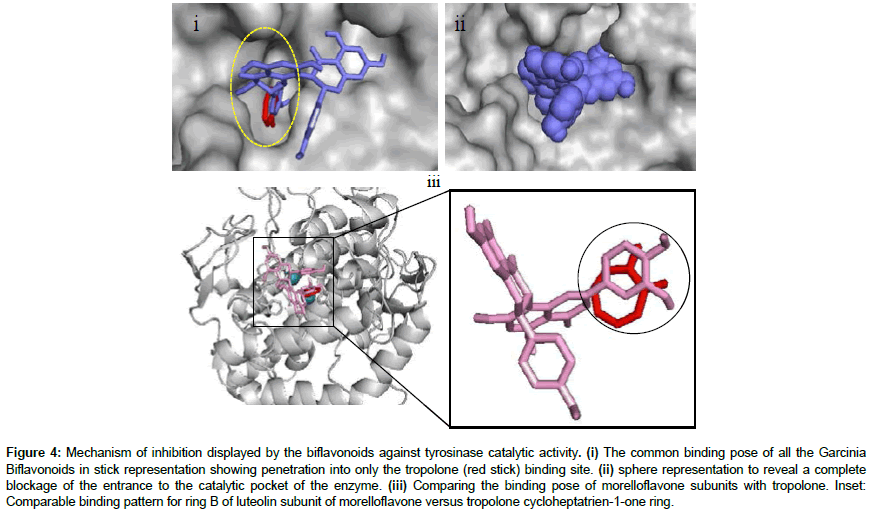
According to this study, the Garcinia biflavonoids have the required potential to bind with varied affinity to the active site of tyrosinase in a manner similar to that of tropolone (Figure 4). Tropolone, a chemical compound with unusual electronic structure, is one of the most effective inhibitors of mushroom tyrosinase with IC50 4μM [46] which compete against the dihydroxyphenols for binding to the copper ions at the active site of the protein [35]. The compound can chelate the metal ions and made them unavailable for the enzyme use. Tropolone also deeply penetrated the binding pocket where it enjoyed hydrophobic interaction with residues Phe264, Phe292, Gly281, His259, Val283, Ser282, His61, His263, Ala286 and Met280, cation-π interaction with residue His263 and hydrogen bonding with copper B (Cu-B) [45,47]. As revealed in Figure 4, after molecular docking experiment, all the biflavoinoids shared common binding pose at the binding site and were found inserting one of their monoflavonoid subunits into the “tropolone binding pocket” while the other monoflavonoid appeared “hooked” to the solvent-exposed region around the active site (Figure 4i-4iii). This result forms part of the evidence for the existence of two types of substrate binding sites on the enzyme, one type for the phenolic substrate and another type for the dioxygen binding [35]. Further detailed observation of this interaction showed that the biflavonoids can completely block the entrance to the active site after binding (Figure 4ii) and prevent substrate assess to the site as a competitive inhibitor. Hence, the size of the ligands might be responsible for the interaction pattern observed with tyrosinase in the current study. Biflavonoids are relatively large phytochemicals and their large size always plays a significant role in their binding pattern to any protein target. In addition, the type of linkage joining the monoflavonoid subunits of a biflavonoid is also an important factor in their binding orientation to any protein [18,26]. Morelloflavone and the other ligands used in this study are dimeric-flavonoids joined at 2’,8” linkage position. Moreover, it is a common occurrence that biflavonoids utilize their monoflavonoid components to interact with protein for inhibitory activities [26]. The positions of the numerous hydroxyl and methoxyl moieties found on the biflavonoids also tend to determine their binding configuration and fitness within the binding pocket which eventually dictate their inhibitory potential against the enzyme [18]. The docking grid and parameters adopted for the current study was intentionally made large due to the size of the biflavonoids and to allow for adequate sampling of all possible binding orientation within the binding pockets available on the protein structure (Tables 2 and 3).
Figure 4: Mechanism of inhibition displayed by the biflavonoids against tyrosinase catalytic activity. (i) The common binding pose of all the Garcinia Biflavonoids in stick representation showing penetration into only the tropolone (red stick) binding site. (ii) sphere representation to reveal a complete blockage of the entrance to the catalytic pocket of the enzyme. (iii) Comparing the binding pose of morelloflavone subunits with tropolone. Inset: Comparable binding pattern for ring B of luteolin subunit of morelloflavone versus tropolone cycloheptatrien-1-one ring.
| Receptor (PDB ID) | Centre X (Å) | Centre Y (Å) | Centre Z (Å) |
|---|---|---|---|
| 2Y9X | -7.41 | -23.63 | -32.64 |
Table 2: Grid center coordinates (X, Y and Z) used for the docking runs.
| Receptor (PDB ID) | Spacing (Å) | X point (Å) | Y point (Å) | Z point (Å) |
|---|---|---|---|---|
| 2Y9X | 0.375 | 120 | 120 | 120 |
Table 3: Grid box spacing and parameters (X, Y and Z points) used in this study for the docking.
The lowest binding affinity among the Garcinia biflavonoids was serendipitously observed for GB1 in this study with binding energy value -7.6 kcal/mol. Interestingly, this compound has previously been reported to possess low inhibitory potential on tyrosinase [30]. Morelloflavone, however, displayed the highest binding affinity to tyrosinase having energy value of -8.5 kcal/mol. The lesser ΔG value indicates a relatively higher inhibitory potential of the compound against the enzyme. This result is in agreement with the report of Mulholland et al., [48]. The GB2 and kolaflavanone also have remarkably good affinity to the modeled tyrosinase with energy score of -7.8 kcal/mol and -8.1 kcal/mol respectively (Table 4). Analysis of the molecular interaction of the biflavonoids with tyrosinase was done using PYMOL. The results revealed the significance of hydrogen bond and hydrophobic interaction between the tyrosinasebiflavonoid complexes (Figure 5). Kolaflavanone established two hydrogen bonds with residue His263 and Met280 with distance of 2.4 Å and 2.7 Å respectively and, hydrophobic interactions with residues Val283, Val248, Asn81 and Ser282 (Table 4). Morelloflavone also formed two hydrogen bonds with amino acid residues Met280 and His263 but with a relatively longer bond length (2.5 Å and 2.7 Å respectively). Hydrophobic interaction with residues Phe264, Ala286, Asn260, Val248, Val283 and Glu322 were also observed for this biflavonoid. The higher affinity predicted for morelloflavone compared to kolaflavanone on the active site may not be unconnected with an extra hydrophobic interaction coupled with the extended length of the hydrogen bond. This results indicate the crucial role of hydrophobic interaction in tyrosinase inhibition. The amino acid residues that are involved in these molecular bonds are therefore essential for inhibition of tyrosinase catalytic functions [45]. Hence, they should be targeted in the design and development of novel inhibitors of the protein. These observations are in agreement with previous reports that morelloflavone is an effective melanin production inhibitor which suppressed melanogenesis and decreased the melanin content of MeWo cells [47]. The earlier report also showed a derivative of morelloflavone (morelloflavone - 7″-sulphate) as the more potent inhibitor of tyrosine. This is possibly due to the extra – SO3H moiety present on the compound which may participate in additional hydrogen bond [48,49].
Figure 5: Binding configuration and molecular interaction analysis of the ligands. Binding poses and interaction pattern of (A) GB1, (B) GB2, (C) kolaflavanone and (D) morelloflavone were visualized with Pymol. Residues involved in hydrogen bond formation are presented as green stick while the bonds are shown as red dashed lines. The copper ions at the active site are given as spheres (cyan). Only important residues are shown in stick representation (grey). Other amino acid residues are displayed in line representation (grey). The biflavonoids interacted with only one out of the two copper ions in the active site via hydrogen bond.
| Ligand | Energy value (kcal/mol) | No of H-Bond | Amino acid residues involved in Hydrogen bond | Distance (Å) | Bond length to Cu2+ (Å) | Amino acid residues involved in hydrophobic interaction |
|---|---|---|---|---|---|---|
| GB1 | -7.6 | 1 | Glu322 | 2.5 | 2.7 | Val283, Val248 |
| GB2 | -7.8 | 1 | His85 | 2.3 | 3.0 | Val283, Val248, Asn81, Asn260 |
| Kolaflavanone | -8.1 | 2 | His263, Met280 | 2.4 2.7 |
3.2 | Val283, Val248, Asn81, Ser282 |
| Morelloflavone | -8.5 | 2 | Met280, His263 | 2.5 2.7 |
3.2 | Phe264, Ala286, Asn260, Val248, Val283, Glu322 |
Table 4: Molecular interaction analysis and binding energy value of kolaviron constituents and morelloflavone on tyrosinase putative binding site.
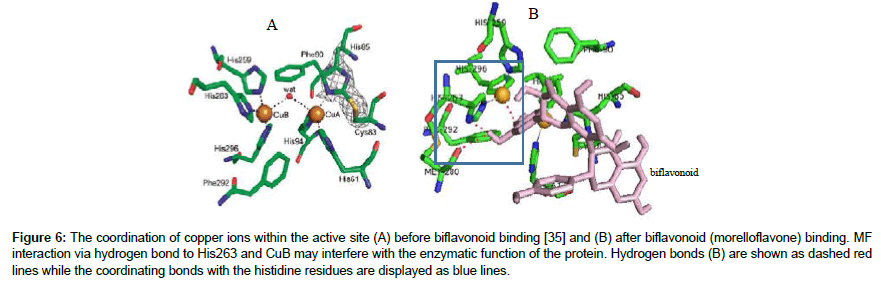
The Garcinia GB1 and GB2 are constituents of kolaviron which have been reported to inhibit tyrosinase activity in vitro [30]. In the current study, a binding energy value of -7.8 kcal/mol for GB2 compared to -7.6 kcal/mol for GB1 (Table 4) suggests a higher inhibitory potential of GB2 against the protein. These results are consistent with the reports of previous study carried out by Okunji et al. claiming an IC50 value of 582 μM for GB2 but very weak inhibition for GB1 against tyrosinase [30]. The data of current study therefore corroborates the report and reveal the direct interaction of these biflavonoids with the protein. Both compounds interacted with residues at the active site via a single hydrogen bond. However, while GB2 engaged in hydrogen bond with amino acid residue His85 (Figure 5), GB1 formed hydrogen bond with Glu322, an amino acid a bit far from the active site. As depicted in Figure 6, the biflavonoids interfered with the coordination of the second copper ion (Cu-B) by the His263, His259 and His296 residues. The minor difference in the binding energy values of GB2 versus. GB1 may be attributable to comparable binding pattern. However, the increased potency of GB2 may not be unconnected with the hydrophilic interaction with His85 for which there are evidence of inhibitor interaction in the literature. Also, this residue His85 appears to participate in the coordination of copper A ion. Hydrogen bonding with His85 by GB2 may interfere with the Cu-A availability for enzymatic functions of the protein. Furthermore, kolaflavanone and morelloflavone interacted directly with the imidazole ring of His263 via polar bonds. In the absence of the biflavonoids, each copper ion is coordinated by three histidine residues. The first copper ion (Cu-A) is bonded to atoms of His61, His85 and His94 whereas Cu-B is connected to atoms of His259, His263 and His296 [35]. The formation of hydrogen bond with Cu-B and imidazole ring of His263 may alter enzyme affinity for the metal. Such interference might distort the biological function of the protein. However, the biflavonoids interacted with only one of the cupper ions.
Figure 6: The coordination of copper ions within the active site (A) before biflavonoid binding [35] and (B) after biflavonoid (morelloflavone) binding. MF interaction via hydrogen bond to His263 and CuB may interfere with the enzymatic function of the protein. Hydrogen bonds (B) are shown as dashed red lines while the coordinating bonds with the histidine residues are displayed as blue lines.
Among the biflavonoids that are constituents of kolaviron, kolaflavanone possessed the best affinity and conveniently bind with lesser ΔG value. The results of the current study indicate that these biflavonoids possess at least moderate inhibition against the enzyme and can block melanin production as shown in Figure 7, which is compatible with earlier reports obtained in in vitro experiments [30]. The reduced potency of the biflavonoids against tyrosinase might be due to their inability to interact with both copper ions found at the binding site. Usually, chemicals that form complexes with copper atoms at the active site of tyrosinase are potent inhibitors of tyrosinase activity [35,46]. As earlier hinted, the great size of the compounds contributes immensely to this property. Since tyrosinase inhibitors are majorly sought and studied for their benefits in skin medications and cosmetics [50], this work hence provides information which validates the application of medicinal plant extracts containing these biflavonoids for cosmetic purposes. It is important to emphasize that this study represents the first in silico report on tyrosinase-Garcinia biflavonoids interaction.
Conclusion
The current study has been carried out to unravel the molecular interaction and inhibitory potential of four Garcinia biflavonoids obtained from G. kola seeds and G. dulcis leaves against tyrosinase enzyme, the signature protein in melanogenesis. Based on the molecular docking results, the mechanism of interaction of the ligands was suggested to involve complete obstruction of entrance to the enzyme active site with interference with only one of the binuclear copper ions within the binding site. This observation formed the basis of their moderate inhibitory effect on the enzyme. Study of protein– ligand interactions revealed information about the hydrogen bonding interactions between residues Glu322, His85 and His263 with the hydroxyl moieties of the monoflavonoid subunit of the biflavonoids as well as hydrophobic interaction with residues forming the binding pocket play key role in the biflavonoids-tyrosinase complex formation and stability. This work corroborates earlier in vitro reports and contributes to information on the molecular mechanisms of the bioactivity of the plant species, kolaviron and morelloflavone. Therefore, Garcinia biflavonoids might be considered promising candidates for use in the development of phytopharmaceutical, nutraceutical formulations and therapeutic agents for dermatological disorders associated with melanin hyperpigmentation.
Conflict of Interests
The author declares that no conflict of interest exists.
Acknowledgement
The author is grateful to all researchers at the Centre for Bio-computing and Drug Development (CBDD), Adekunle Ajasin University, Akungba-Akoko, Ondo state, Nigeria for the assistance and technical support received from them.
References
- Bae SJ, Ha YM, Kim J, Park JY, Ha TK, et al. (2013) A novel synthesized tyrosinase inhibitor: (E)-2-((2,4-Dihydroxyphenyl)diazenyl)phenyl 4-methylbenzenesulfonate as an azo-resveratrol analog. Biosci Biotechnol Biochem 77: 65-72.
- Rees JL (1996) The melanoma epidemic: Reality and artefact.BMJ 312: 137-138.
- ArianayagamS,Ryan TJ (2014) Human pigmentation: A side effect adapted from a primitive organism's survival. Part 2: The melanocyte as mentor of the keratinocyte. Indian Dermatol Online J 5: 328-333
- Kim DH, Kim SJ, Ullah S, Yun HY, Chun P, et al. (2017) Design, synthesis, and antimelanogenic effects of (2-substituted phenyl-1,3-dithiolan-4-yl)methanol derivatives. Drug Des Devel Ther 11: 827-836
- Olivares C, Solano F (2009) New insights into the active site structure and catalytic mechanism of tyrosinase and its related proteins. Pigment Cell Melanoma Res 22: 750-760.
- Ashraf Z, Rafiq M, Seo S, Kwon KS, Babar MM, et al. (2015) Kinetic and in silico studies of novel hydroxy-based thymol analogues as inhibitors of mushroom tyrosinase. Eur J Med Chem 98: 203-211
- Kubo I,Yokokawa Y,Kinst-Hori I (1995) Tyrosinase inhibitors from Bolivian medicinal plants. J Nat Prod 58: 739-743.
- Quispe YN,Hwang SH,Wang Z,Lim SS (2017) Screening of peruvian medicinal plants for tyrosinase inhibitory properties: identification of tyrosinase inhibitors in Hypericum laricifolium Juss. Molecules 22: 402
- Di Petrillo M, González-Paramás AM, Era B, Medda R, Pintus F, et al. (2016) Tyrosinase inhibition and antioxidant properties of Asphodelus microcarpus extracts. BMC Comp Alter Med 16: 453.
- Qin H, Shang Z, Jantan I, Tan OU, Hussain MA, et al. (2015) molecular docking studies and biological evaluation of chalcone based pyrazolines as tyrosinase inhibitors and potential anticancer agents. RSC Adv 5: 46330-46338.
- Dlova NC, Hendricks NE, Martincgh BS (2012) Skin-lightening creams used in Durban, South Africa. Int J Dermatol 5: 51-53.
- Padariya M, Kalathiya U, Baginski M (2014) Docking simulations, Molecular properties and ADMET studies of novel Chromane6,7diol analogues as potential inhibitors of Mushroom tyrosinase. Gene Ther Mol Biol 16: 201-218.
- Franco DCZ, Carvalho GSG, Rocha PR, Teixeira RS, daSilva AD, et al. (2012) Inhibitory effects of resveratrol analogs on mushroom tyrosinase activity. Molecules 17: 11816-11825.
- Hemshekhar M, Sunitha K, Santhosh MS, Devaraja S, Kemparaju K, et al. (2011) An overview on genus Garcinia: Phytochemical and therapeutical aspects. Phytochem Rev 10: 325-351.
- Ekene EN, Erhirhie EO (2014) Garcinia kola: A review of its ethnomedicinal, chemical and pharmacological properties. Int J Cur Res Rev 6: 1-7.
- Kumar S,Sharma S,Chattopadhyay SK (2013) The potential health benefit of polyisoprenylated benzophenones from Garcinia and related genera: ethnobotanical and therapeutic importance. Fitoterapia 89: 86-125.
- Chen J, Chang HW, Kim HP, Park H (2006) Synthesis of phospholipase A2 inhibitory biflavonoids. Bioorgoranic Med Chem Lett 16: 2373-2375
- Lee J, Jung K, Woo E, Kim Y (2008) Docking study of biflavonoids, allosteric inhibitors of protein tyrosine phosphatase 1b. Bull Korean Chem Soc 29: 1479-1484.
- Lin YM, Flavin MT, Schure R, Chen FC, Sidwell R, et al. (1999) Antiviral activities of biflavonoids. Planta Med 65:120-125.
- Bagla VP, McGaw LJ, Elgorashi EE, Eloff JN (2014) Antimicrobial activity, toxicity and selectivity index of two biflavonoids and a flavone isolated from Podocarpus henkelii (Podocarpaceae) leaves. BMC Comp Alter Med 14: 383
- Iwu MM, Igboko OA, Okunji CO, Tempesta MS (1990) Antidiabetic and aldose reductase activities of biflavanones of Garcinia kola. J Pharm Pharmacol 42: 240-292.
- Abarikwu SO, Farombi EO (2012) Pant AB: Kolaviron biflavanoids of Garcinia Kola seeds protect atrazine-induced cytotoxicity in primary cultures of rat leydig cells. Int J Toxicol 31: 407-415.
- Adedara I, Vaithinathan S, Jubendradass R, Mathur P, Farombi EO (2013) Kolaviron prevents carbendazim-induced steroidogenic dysfunction and apoptosis in testes of rats. Environ Toxicol Pharmacol 35: 444-453.
- Farombi EO, Adedara IA, Ajayi BO, Ayepola OR, Egbeme EE (2013) Kolaviron, a natural antioxidant and anti-inflammatory phytochemical prevents dextran sulphate sodium-induced colitis in rats. Basic Clin Pharmacol Toxicol 113: 49-55.
- Pinkaew D, Cho SG, Hui DY, Wiktorowicz JE, Hutadilok-Towatana N, et al. (2009) Morelloflavone blocks injury-induced neointimal formation by inhibiting vascular smooth muscle cell migration. Biochim Biophys Acta 1790: 31-39.
- Ogunwa TH, Ayenitaju FC (2017) Molecular binding signatures of morelloflavone and its naturally occurring derivatives on HMG-COA reductase. Int J Biol Sci Appl 4: 74-81.
- Farombi EO, Tahnteng JG, Agboola AO, Nwankwo JO, Emerole GO (2000) Chemoprevention of 2-acetylaminofluorene-induced hepatotoxicity and lipid peroxidation in rats by kolaviron-a Garcinia kola seed extract. Food Chem Toxicol 38: 535-541.
- Onasanwo SA, Singh N, Olaleye SB, Palit G (2011) Anti-ulcerogenic and proton pump (H+, K+ ATPase) inhibitory activity of Kolaviron from Garcinia kola Heckel in rodents. Indian J Exp Biol 49: 461-468
- Olaleye SB, Farombi EO, Adewoye EO, Onasanwo SA, Owoyele BV, et al. (2000) Analgesic and anti-inflammatory effects of kolaviron (a Garcinia kola seed extract). Afr J Biomed Res 3: 163.
- Okunji C, Komarnytsky S, Fear G, Poulev A, Ribnicky DM, et al. (2007) Preparative isolation and identification of tyrosinase inhibitors from the seeds of Garcinia kola by high-speed counter-current chromatography. J Chromatography A 1151: 45-50.
- Ijomone OM, Obi AU. Kolaviron (2013) isolated from Garcinia kola, inhibits acetylcholinesterase activities in the hippocampus and striatum of wistar rats. Ann Neurosci 20: 42-46.
- Gil B, Sanz MJ, Terencio MC, Gunasegaran R, Paya M, et al. (1997) Morelloflavone, a novel biflavonoid inhibitor of human secretory phospholipase A2 with anti-inflammatory activity. Biochem Pharmacol 53: 733-740.
- Pereañez JA, Patiño AC, Núñez V, Osorio E (2014) The biflavonoid morelloflavone inhibits the enzymatic and biological activities of a snake venom phospholipase A2. Chem Biol Interact 220: 94-101.
- Vanessa SG, Jaqueline PJ, Wagner AJ, Alyne AA, Ingridy RC, et al. (2015) Morelloflavone and its semisynthetic derivatives as potential novel inhibitors of cysteine and serine proteases. J Med Plant Res 9: 426-434.
- Ismaya WT,Rozeboom HJ,Weijn A,Mes JJ,Fusetti F,et al. (2011) Crystal structure of Agaricus Bisporus mushroom tyrosinase: identity of the tetramer subunits and interaction with tropolone. Biochemistry 50(24): 5477-5486.
- Seelinger D, de Groot BL (2010) Ligand docking and binding site analysis with PYMOL and Autodock/Vina. J Comput Aided Mol Des 24: 417-422.
- Trott O, Olson AJ (2010) AutoDock Vina: Improving the speed and accuracy of docking with a new scoring function, efficient optimization and multithreading. J Comput Chem 31: 455-461.
- Cayce KA, McMichael AJ, Feldman SR (2004) Hyperpigmentation: An overview of the common afflictions. Dermatol Nursing2004: 16
- Summers B (2006) A lightening tour of skin-brightening options. South Afri Pharma Cosmetic Rev 33: 29-30
- Haghbeen K, Jazii FR, Karkhane AA, Borojerdi SS (2004) Purification of tyrosinase from edible mushroom. Iranian J Biotechnol 2: 189-194
- Yin S, Si Y, Wang Z, Wang S, Oh S, et al. (2011) The effect of thiobarbituric acid on tyrosinase: inhibition kinetics and computational simulation. J Biomol StructDyn 29: 463-470
- da Silva AP, Silva NDF, Andrade EHA, Gratieri T, Setzer WN, et al. (2017) Tyrosinase inhibitory activity, molecular docking studies and antioxidant potential of chemotypes of Lippia origanoides (Verbenaceae) essential oils. PLoS ONE 12: e0175598.
- Asadzadeh A, Fassihi A, Yaghmaei P, Pourfarzam M (2015) Docking studies of some novel kojic acid derivatives as possible tyrosinase inhibitors. Biomed Pharmacol J 8: 535-545
- Senol FS, Khan MTH, Orhan G, Gurkas E, Orhan IE, et al. (2014) In silico approach to inhibition of tyrosinase by ascorbic acid using molecular docking simulations. Curr Topics Med Chem 14: 1469-1472
- Omotuyi OI, Ueda H (2013) A novel unified ab initio and template-based approach to GPCR modeling; A case of EDG-LPA receptor. Curr Bioinformatics 8: 1-12.
- Kahn V, Andrawis A (1985) Inhibition of mushroom tyrosinase by tropolone. Phytochem 24: 905-908.
- Asadzadeh A, Sirous H, Pourfarzam M, Yaghmaei P, Fassihi A (2016) In vitro and in silico studies of the inhibitory effects of some novel kojic acid derivatives on tyrosinase enzyme. Iran J Basic Med Sci 19: 132-144
- Mulholland DA, Mwangi EM, Dlova NC, Plant N, Crouch NR, et al. (2013) Non-toxic melanin production inhibitors from Garcinia livingstonei (Clusiaceae). J Ethnopharmacol 149: 570-575.
- Dlova N, Mashamba-Thompson T (2014) Mechanism of melanin inhibition of Morelloflavone- 7″- sulphate and Sargaol extracts from Garcinia livingstonei (Clusiaceae): Homology modelling; molecular docking and molecular dynamics Simulations. Int J Bioeng Life Sci 8.
- Parvez S, Kang M, Chung H, Bae H (2017) Naturally occurring tyrosinase inhibitors: mechanism and applications in skin health, cosmetics and agriculture industries. Phytother Res 21: 805-816.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi