Research Article, Int J Glob Health Vol: 2 Issue: 1
Assessment of Carbon Monoxide Exposure among Women Cooking With Biomass Fuels and Their Children Living in Sudan
Suliman AK1,5, Saleh MM2, Malek AA2, Karrar AFI3, Kitch D1, Sznajder KK4*, King TS4, Tsay AJ5 and Warren WS1,5
1Department of Paediatrics, Penn State Hershey Children’s Hospital, Hershey, PA, USA
2Department of Paediatrics, Redsea University Faculty of Medicine, Port Sudan, Redsea State, Sudan
3Department of Anatomy, Redsea University Faculty of Medicine, Port Sudan, Redsea State, Sudan
4Department of Public Health Sciences, Penn State College of Medicine, Hershey, PA, USA
5Penn State College of Medicine, Hershey, PA, USA
*Corresponding Author : Kristin Sznajder
PhD, Department of Public Health Sciences, Penn State College of Medicine, Hershey, PA, 17033, USA
Tel: 717- 531-0003-285639
E-mail: ksznajde@phs.psu.edu
Received: September 09, 2018 Accepted: September 22, 2018 Published: January 24, 2019
Citation: Suliman AK, Saleh MM, Malek AA, Karrar AFI, Kitch D, et al. (2019) Assessment of Carbon Monoxide Exposure among Women Cooking With Biomass Fuels and Their Children Living in Sudan. Int J Glob Health 2:1.
Abstract
Objective: Questionnaires were used to assess levels of household air pollution [HAP], defined as inhalation of smoke from biomass fuels. Women and their children in developing countries are at higher risk of HAP exposure due to the disproportionate time spent indoors exposed to smoke from biomass fuels used for cooking. HAP contains multiple substances including [CO], which attaches to red blood cells causing hypoxia. CO combined with other inhaled substances such as particulate matter 2.5 (PM2.5), can lead to poor respiratory health among the affected households. Monitoring of HAP has traditionally measured the [CO] exposure within the living environment; however exposure in dwellings may not accurately measure an individual’s exposure. This study demonstrates that CO, a major component of HAP, can be measured using a COoximeter and that subjects’ CO levels represent a biomarker and a surrogate for overall HAP exposure.
Methods: Women of low socioeconomic status and of reproductive age and their children were invited to six women’s centers in Port Sudan, Sudan. At a single visit, data on dwelling size, cooking fuels and health information were collected and CO levels of mothers and children were measured using the Masimo Rad-57 Rainbow Pulse CO-oximeter. Anthropomorphic measurements of children were made using standard equipment.
Results: Data were collected on 373 women, 100 were excluded due to inaccurate CO-oximetery readings. Headache, cough, weakness, dyspnea, fatigue, as well as lung infections and infants perceived to be small at birth were reported more often among women with higher CO levels. Four hundred thirty one children were enrolled and 87 excluded because the children or their mothers had invalid CO-oximetry or missing data. Mean CO levels were higher in all age groups among children exposed to biomass cooking fuels compared with gas. These levels were consistent with the maternal levels. Children with moderate [10-15%] and high CO levels [16-25%] had more symptoms associated with CO poisoning, more chest infections and were smaller at birth and smaller when compared to WHO standards.
Conclusion: CO-oximetry is a valuable tool to assess individuals exposed to high levels of HAP and the resulting CO levels are an important biomarker for these exposed individuals.
Keywords: Household air pollution; Carbon-monoxide; Sudan; Respiratory health
Introduction
Household air pollution [HAP], is a global issue and is at its highest levels among households that use biomass fuels for cooking. The major source of HAP in developing countries is the use of biomass fuels for cooking and heating in poorly ventilated dwellings. In March 2014 report the World Health Organization [WHO] estimated that HAP causes 4.3 million deaths annually worldwide. Low and middle income countries had the highest pollution burden in 2012 with an estimated 3.3 million deaths linked to indoor air pollution [1]. The numbers have not improved. In a 2016 report the WHO estimates that there will be 6.5 million deaths annually attributable to exposure to air pollution [2].
Infant mortality has been linked to HAP [3] and is reported to be 47.6/1000 live births in Sudan [4]. Monitoring of indoor air pollution has been carried out primarily by measuring indirect exposure in dwellings and on the clothes of the residents [5]. These techniques measure the primary products of biomass fuel combustion; carbon monoxide, sulfur dioxide, nitrogen dioxide and particulate matter [6] and are associated with variations of exposure within households and subgroups [7]. Nevertheless, the WHO’s capability to characterize the extent of HAP and its health related effects continues to rely on these estimates of exposure [5,8]. Carbon Monoxide [CO], a primary product of partial combustion of biomass fuels is absorbed into the body after inhalation. Blood levels can be measured. CO has previously been used as a tracer for HAP exposure [9]. Thus, CO levels could be considered to be a surrogate marker for total HAP exposure. Health related effects similar to those reported with HAP exposure have been reported with CO poisoning involving: the heart [10], central nervous system, [11,12] placental dysfunction [13].
In Sub Saharan Africa and Sudan 95% of the population use biomass fuels for cooking which results in high levels of HAP [14]. Port Sudan, Sudan was chosen for this study for several reasons: an interest in the proposed research by the Redsea University Faculty of Medicine, and members of the faculty interested in participating in the research that had access to a population of lower socioeconomic women and children who cook with biomass fuels. This population of lower socioeconomic women and their children was chosen because it was postulated that they would live in small poorly ventilated dwellings; cook in open fires with biomass fuels such as wood, charcoal, dung, and crop residues; and have high levels of HAP exposure which would result in elevated CO levels. Some cultural issues which were not recognized prior to starting the study affected the data: the use of henna dye on the hands and feet of married women in Sudan affected the CO-oximetry results, and the fact that prepartum and postpartum women did not cook for several months before and after delivery. This may have resulted in an underestimation of exposure and lower CO levels in some subjects.
The goal of this pilot study was to demonstrate that the CO levels as measured by the CO-oximeter are a biomarker and a surrogate for overall HAP exposure.
Methods
The study area chosen was in the periphery of Port Sudan, a large city located in eastern Sudan where there is a population of low income women and children up to five years of age and cooking in one to three room dwellings, the study population. These women use biomass fuels as their main energy source for cooking in open fires although some slightly more affluent women cook with gas. Many of these women attend women’s centers where they learn to read and write. This cross-sectional study targeted the women who attended the women’s centers and their young children.
Orientation materials were developed to explain the purpose of the study, why a consent form was necessary and why children under five years of age were to be included. Each of these issues was reviewed with prospective subjects by Arabic speaking research assistants. Because the potential subjects were illiterate or semiliterate, a verbal consent form was written using the Penn State Milton S. Hershey Medical Center Institutional Review Board guidelines. Permission to include children was part of this verbal consent form. Questionnaires for women and their children were developed and aimed at obtaining a history of HAP exposure due to a variety of factors such as cooking fuels [wood, charcoal, gas, dung, crop residues], tobacco use, incense use, and dwelling size. A second questionnaire aimed at obtaining a history of potential health effects of HAP exposure was also developed. All documents were translated into Arabic and the translations were verified by third parties not involved in the study. CO-oximetry was performed on mothers and children. Anthropomorphic measurements on enrolled children were carried out as follows: head circumference for children up to two years of age using standard disposable paper measuring tapes; length for children up to two years using a standard length board; height for children over two years using tape measures attached to a wall and a level; weights for children up to two years in the mothers’ arms using a Homedic dual display digital bathroom scale; and for children over two years using a Detecto 1130 analog bathroom scale. Weights recorded in pounds were converted to kilograms. A Sudanese study team which included a field director, six female physicians in training and six counselors at women’s centers were recruited, as research assistants. The six female physicians were divided into three teams and supervised by the PI and field director.
Ethical approval
The study was approved by the Penn State Milton S. Hershey Medical Center Institutional Review Board, the Ethics Committee of the Redsea University Faculty of Medicine as well as the Minister of Health Redsea State.
Participant recruitment and enrollment
Prior to the study, the Sudanese members of the field research team were oriented to the symptoms of CO poisoning, the health related effects of HAP exposure, the consent process, the questionnaires and how to use a CO-oximeter. Counselors at six women’s centers in Port Sudan were trained by the research team to recruit potential subjects. Each women’s center represented a target community. At the women’s centers all potential subjects recruited by the counselors were offered an opportunity to participate in the research. The orientation materials were reviewed including an explanation for enrolling children up to five years of age. The consent form was explained and the subject asked to check or initial the form if she agreed to participate. All subjects recruited by the counselors who consented were included in the study with the exception of several women who appeared to be severely ill who were excluded and referred for hospital care. Following the consent process, demographic and subjective data on dwelling size, cooking fuels and health information were collected. CO levels were measured in duplicate using the Masimo Rad-57 pulse CO-oximeter.
The initial goal of the study was to demonstrate that the Masimo CO-oximeter was an effective tool to determine the CO levels of women exposed to high levels of HAP. On the advice of our statistician, it was thought that approximately one hundred women who reported high levels of exposure [cooking inside with biomass fuels] would be sufficient to demonstrate significantly elevated CO levels.
Equipment
Three Masimo Rad-57 pulse CO-oximeters together with adult and pediatric digital sensors were provided by the Masimo Corporation, Irvine, California. The Masimo CO-oximeter is a noninvasive monitoring platform which uses eight wavelengths of light to measure oxygen and CO saturation. The Masimo corporation reports that the standard deviation for accuracy of the CO-oximeter is +/- 3% [Operator’s manual, Masimo] while Zaouter and Zavorsky [15] report readings of +4% to -6% when the true COHb% is in the 10-14% range. The CO-oximeters and sensors were tested prior to starting and daily prior to resuming the study. The CO determinations for the three instruments were consistent with values ranging from 2-4% on the investigators. Normal CO values as measured by the CO-oximeter for individuals with no risk factors such as tobacco use are up to 5%. Levels of 10% or more are considered to be distinctly abnormal and may be associated with symptoms.
Statistical analysis
Analyses comparing the frequency of symptoms or conditions between two groups were performed using the chi-square test, and results were reported using frequencies, percentages and chi-square p-values. Analyses comparing the average CO% between two groups were performed using two group t-tests and the results were reported in terms of means, standard deviations, and t-test p-values. When comparisons of frequencies involved more than two groups, the chisquare test was also used and comparisons of means among more than two groups were performed using analysis of variance. Statistical significance was determined as p<0.05.
Results
Adult data
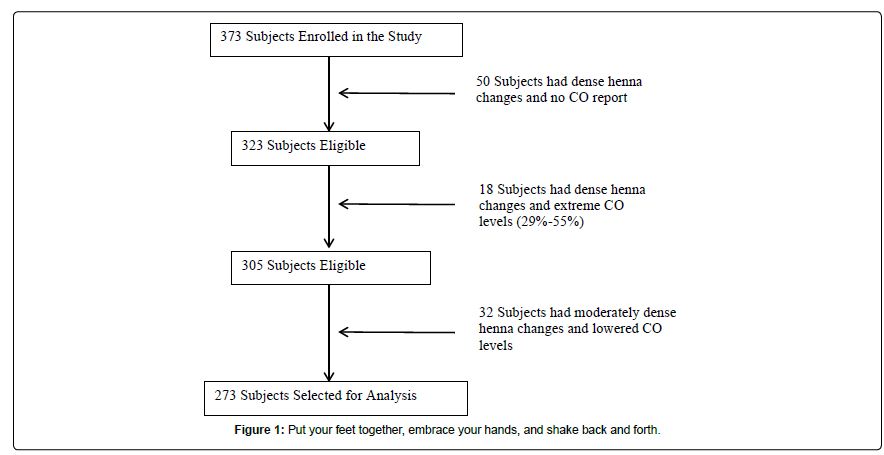
Three hundred seventy three women were enrolled. Henna dye applied to the hands and feet of married women was found to affect CO-oximetry results. Those subjects with dense henna changes were stratified into three groups (Figure 1). Even moderately dense henna changes had a significant effect. The CO level for subjects with moderately dense henna changes was 12% as compared to those with low henna of 16% [p<0.0001]. A total of 100 adult subjects were excluded from the analysis due to moderate or high henna changes. Therefore, 273 subjects with low or minimal henna changes were available for the analyses. The 188 women cooking with biomass fuels had a mean CO level of 18% compared with the 85 women using gas whose mean CO level was 12% [p<0.0001]. The mean CO levels did not change when the women who cooked outside were eliminated. The mean CO level for the 9% of women who reported cooking with biomass fuels outside was 18% [p=0.23].
Dwelling size had a significant effect on CO levels. The mean CO levels for women cooking with gas or biomass fuels living in one room dwellings was 17% compared with 14% for those living in two or more rooms [p<0.0001] Similarly, the mean CO level for children living in one room as compared to three rooms was 16% compared to 11% [p<0.0001].
Headache, dyspnea, weakness, fatigue and cough are symptoms commonly reported by individuals with high HAP exposure and by people with elevated CO levels. In this study all of these symptoms were more often reported by women who cooked with biomass fuels as compared to those who cooked with gas [data not shown, p-values ranged from <0.0001 to 0.028]. When one looks at the CO levels and symptoms of all 273 subjects, for each symptom there was a significant difference in the CO levels between those women reporting a symptom and those who did not report a symptom regardless of cooking fuel [data not shown, p-values ranged from <0.0001 to 0.007], and the number of women that reported symptoms rose as the CO levels increased [p-values ranged from <0.001 to 0.012] (Table 1).
| Symptoms | Number of Subjects n (%) |
Low CO 0-10% |
Moderate CO 10-15% | High Co>15% | p-Value |
|---|---|---|---|---|---|
| Headache | 156 (57%) | 12 (8%) | 28 (17%) | 116 (75%) | <0.001 |
| Dyspnea | 61 (22%) | 4 (7%) | 9 (15%) | 48 (79%) | 0.005 |
| Fatigue | 57 (21%) | 2 (4%) | 12 (21%) | 43 (75%) | 0.008 |
| Weakness | 54 (20% | 3 (6%) | 9 (17%) | 42 (78%) | 0.012 |
| Cough | 151 (55%) | 8 (5%) | 31 (21%) | 112 (74%) | <0.001 |
Table 1: Increasing frequency of symptoms with reported rising of CO levels (total n=273).
Women who cooked with biomass fuels reported some health related effects consistent with high levels of HAP exposure and CO poisoning. For example, the mean CO levels were significantly higher for women reporting red eyes, infants perceived to be small at birth, infant mortality, and with three or more lung infections [data not shown, p-values ranged from <0.001 to 0.029] while the difference for spontaneous abortion was not statistically significant [data not shown, p=0.43]. Among 273 subjects, reporting health related effects typical of HAP exposure and CO poisoning, the number of reports were higher as the CO levels increased (Table 2).
| Health Effects | Number of Subjects n (%) |
Low CO 0-10% |
Moderate CO 10-15% | High Co>15% | p-Value |
|---|---|---|---|---|---|
| Red eyes | 64 (23%) | 2 (3%) | 19 (30%) | 43 (67%) | 0.004 |
| Small newborns | 208 (76%) | 39 (19%) | 51 (25%) | 118 (57%) | 0.011 |
| Infant mortality | 21 (8%) | 2 (10%) | 5 (24%) | 14 (67%) | 0.43 |
| Lung infection | 186 (68%) | 29 (16%) | 44 (24%) | 113 (61%) | 0.117 |
| 3 or more lung infections | 42 (15%) | 1 (2%) | 13 (13%) | 28 (67%) | 0.023 |
Table 2: Increasing frequency of health effects with reported rising of CO levels (total n=273).
Pediatric data
Four hundred thirty-nine children, [226 boys] 0-5 years of age, accompanied their mothers to the women’s centers and were enrolled. Three hundred forty-four mother child pairs had complete data sets. There was no difference in the average CO levels of boys versus girls [15% vs. 15%, p=0.8]. Table 2 depicts the mean CO levels associated with biomass or gas cooking fuels among mothers and children in several age groups. The CO levels of children in the 0-6 month group and their mothers were low regardless of cooking fuel because mothers did not cook in the immediate postpartum period. As the mothers began to cook again, babies in the 7-12 month age group were exposed and mothers re-exposed to smoky air and their mean CO levels began to increase. Children in the 13-24 month age group continued to have rising mean CO levels while the mothers’ mean levels stabilized. As the children became more ambulatory, their mean CO levels stabilized. Mean CO levels were consistently higher in all age groups for children and their mothers who cooked with biomass fuels [p<0.001] (Table 3). Increasing amounts of HAP exposure reflected by increasing mean CO levels among the pediatric population were associated with increasing reports of clinical symptoms cough, dyspnea and fatigue [data not shown, p-values ranged from <0.001 to 0.024] and health related effects: chest infection and small at birth with p-values of <0.001 and 0.02 respectively. Anthropomorphic values stratified by mean CO levels were not significantly different (Table 4) although the head circumference values almost meet the 0.05 alpha level of significance. However, when the study children were compared to The WHO international child growth standards, there were significant differences in each measurement (Table 5).
| Age | Gas n=49 n (%) |
Biomass n=292 n (%) |
Mother’s gas mean CO level (CI) |
Mother’s biomass mean CO level (CI) |
Child’s gas mean CO level (CI) | Child’s biomass mean CO level (CI) | p-Value mother’s Gas vs. Biomass CO |
p-Value child’s Gas vs. Biomass CO | p-Value gas CO Mother vs. Child | p-Value biomass CO Mother vs. Child |
|---|---|---|---|---|---|---|---|---|---|---|
| 0-6* | 10 (20%) | 27 (9%) | 8.9 (6.3-11.5) | 15.0 (13.4-16.1) | 7.1 (4.2, 10.0) | 14.7 (11.5-18.0) | <0.001 | <0.01 | 0.31 | 0.88 |
| 7-12* | 10 (20%) | 30 (10%) | 11.1 (8.2-14.1) | 18.3 (16.5-20.1) | 8.5 (6.1, 10.9) | 16.9 (14.5-19.3) | <0.001 | <0.001 | 0.14 | 0.34 |
| 13-24 | 14(29%) | 83 (28%) | 12.3 (9.9, 14.7) | 16.9 (15.9, 17.9) | 11.1 (8.5, 13.7) | 16.3 (15.3-17.3) | <0.001 | <0.001 | 0.47 | 0.39 |
| 25-36* | 7 (14%) | 54 (18%) | 9.4 (6.7, 12.2) | 16.9 (15.6-18.3) | 7.6 (4.7, 10.4) | 15.3 (14.4-16.3) | <0.001 | <0.001 | 0.28 | 0.05 |
| 37-60 | 8 (16%) | 98 (34%) | 10.5 (4.5, 16.6) | 17.2 (16.3-18.2) | 9.9 (6.0, 13.7) | 15.6 (14.7-16.5) | <0.001 | <0.001 | 0.84 | 0.01 |
Table 3: Mother and child exposure to biomass fuels and associated CO level.
| Anthropomorphic values | Low<10% mean (SD) |
Moderate CO 10-15% mean (SD) | High Co>15% mean (SD) |
p-Value |
|---|---|---|---|---|
| Height | 92.5 ( ± 8.2) | 89.6 ( ± 7.7) | 91.1 ( ± 15.0) | 0.76 |
| Length | 69.1 ( ± 9.5) | 68.2 ( ± 6.1) | 69.8 ( ± 7.8) | 0.72 |
| Weight | 10.0 ( ± 3.6) | 10.2 ( ± 3.1) | 11.1 ( ± 3.6) | 0.35 |
| Head Circumference | 46.0 ( ± 3.7) | 46.8 ( ± 3.4) | 47.2 ( ± 3.2) | 0.09 |
Table 4: Anthropomorphic values stratified by CO levels among children less than 2 years of age.
| M | F | |||||
|---|---|---|---|---|---|---|
| No. Responses | Mean ± SD | p-Value | No. Responses | Mean ± SD | p-Value | |
| Weight for Age (kg) | 164 | <0.001 | 182 | 0.01 | ||
| Study | 10.9 ± 3.8 | 11.0 ± 3.3 | ||||
| WHO | 12.9 ± 3.7 | 12.1 ± 3.7 | ||||
| Length/Height for Age (cm) | 163 | <0.001 | 183 | 0.01 | ||
| Study | 83.4 ± 17.0 | 83.0 ± 13.2 | ||||
| WHO | 90.0 ± 14.9 | 86.7 ± 15.0 | ||||
| Head Circumference (cm) | 158 | 0.01 | 182 | 0.05 | ||
| Study | 46.6 ± 3.6 | 47.3 ± 3.2 | ||||
| WHO | 47.8 ± 3.2 | 46.6 ± 3.1 | ||||
Table 5: Mean anthropomorphic measurements of children 0-5 years of age in the study compared to WHO standards.
Discussion
The goal of this study was to assess the utility of the Masimo CO-oximeter to evaluate effects of HAP exposure on Sudanese women and their children. The data presented confirms that CO-oximetry can provide important biomarker data in women and children exposed to high levels of HAP and suggests that the CO levels reported by the CO-oximeter may be a useful surrogate for levels of exposure. Prior studies have relied on exposure data and related this information to various disease states. However, exposure to carbon monoxide has not proved to be a consistently valid surrogate measure of exposure to other HAP components such as PM2.5. Exposure studies combined with CO levels have not been done [5].
The study has several limitations. CO measurements were made at a single visit. Measurement on several visits might have permitted us to track day to day, seasonal or individual variations as reported by Bruce and colleagues in 1998 [6]. A second limitation involved the use of questionnaires to determine the degree of HAP exposure and to learn about the symptoms and health related effects of the exposure. However, the CO levels of those cooking with biomass fuels or gas were similar to those reported by others [16]. Further, the mothers’ reports of health related effects and symptoms of CO poisoning increase as their CO levels increase and are in agreement with those which they reported in their children.
The presence of henna dye on the fingers of married Sudanese women resulting in the exclusion of 100 subjects was also a limitation. The traditional dye is made from the finely ground powder of the henna plant [Lawsonia inermis]. Black henna may contain not only the natural dye but other dyes, metal salts, waxes, oils, and chemicals such as phenylenediamine [17]. We conjecture that the subjects with low henna used either the natural dye or another dye which was wearing off. The moderate henna subjects with low CO levels may have had fresh application of the natural henna or some mixture of black henna which was fading permitting a compromised COoximetry reading. The subjects with very dense henna changes and extremely high CO levels may have had one of the many black henna mixtures prepared by the local artists which altered the ability of the light waves to pass through the arterioles. The dye was so intense on the fingers of 50 subjects that the CO-oximeter did not register a result. In another 18 subjects with very dark henna changes the subjects had extremely high CO levels, [29%-55%] but did not report more symptoms of CO poisoning than subjects in the minimal henna group [p=0.9]. When this finding was reviewed with the director of research at Masimo and his team, they were unable to explain this result. Thirty-two more subjects had sufficient henna changes to alter in part the ability of the light waves to pass through the digital arterioles resulting in an artificially low CO level. It may be that even minimal henna changes artificially lowered the CO levels so that all of the levels reported in this study are underestimates. The henna effect observed is consistent with data from Yamamoto that heavy layers of nail polish may interfere with oximetry which utilizes two light waves versus eight light waves with CO-oxmetry. In the case of the Sudanese subjects, not only the nails were deeply pigmented but the palmer surface of the fingers was deeply pigmented as well. Subjects designated to have low henna changes continued to have visible henna changes in the nails and on the palmer surface which may have had more effect on the CO-oximetry results than is reported with usual amounts of nail polish [18].
The issue of abortion was another limitation. Abortion is illegal in Sudan. Therefore, subjects may not have been willing to candidly discuss the issue and to distinguish between spontaneous and induced abortion. In this study the CO levels of 18% could have played a significant role for the subjects who reported one or more abortions. Most authorities agree that high levels of CO are able to cause fetal death [19]. However, two 2015 papers from Sudan examined the causes of recurrent spontaneous abortion in Sudan [20,21]. Thrombophilia, heavy metals, and tobacco use were noted to be associated. HAP exposure associated with cooking fuels was not mentioned, but tobacco smoke may add to cumulative HAP exposure [22]. Other cultural issues may have impacted the CO levels. Almost all of the subjects enrolled used incense regularly in their dwellings. Many Sudanese women perform a sauna ritual [dukhan] with scented smoke. Precise data on these practices was not collected.
Multiple studies of HAP have measured exposure in dwellings over a period of several days to determine levels of exposure but have provided no concomitant biomarker information [22-24]. In a recent report which looked at CO exposure as a surrogate measure of exposure to particulate matter the authors concluded: 1. Accurate exposure assessment is imperative for evaluating exposure and 2. Exposure to CO is not a consistently valid surrogate for exposure to PM2.5 [5]. The CO-oximetry test was chosen for this study because of its efficiency, the fact that it had not been previously used for an epidemiologic study and provided biomarker data. Completion of the questionnaires and CO-oximetry tests in this study required less than one hour per subject, resulted in a substantial number of enrolled subjects, suggested levels of individual exposure, and provided important biomarker data. Lam and colleagues [25] reported an acute exposure study in which they compared the CO-oximeter and an exhaled breath monitor. This study does provide biomarker data, CO levels. But, the exhaled breath test is effort dependent and requires a 20 second breath hold to perform an accurate test. In spite of careful training by the Arabic speaking physicians, obtaining consistent reliable results in the mothers and children in our study would be unlikely. Additionally, the results of an exhaled breath test may not be accurate in subjects at risk for an element of chronic lung disease [26].
The symptoms and health related effects associated with HAP exposure and CO poisoning reported in this study are consistent with those reported by other workers in the field [11,23]. The CO levels reported in this study are similar to those reported by Behera et al. who used double wavelength spectrophotometry to estimate the CO levels [16]. The consistency continued when analyzing the symptoms and health related effects which were reported by more women cooking with biomass fuels. Women reporting a health related effect had higher CO levels than those who did not report the effect. Similarly, higher CO levels among the children were associated with more symptoms and health related effects consistent with the report of Smith and colleagues [3].
Elevated CO levels in women who reported cooking with gas may be explained by several factors in addition to dwelling size: ninety percent of women use incense regularly in their houses; many Sudanese women perform a sauna ritual [dukhan] with scented smoke periodically; exposure to biomass fuel combustion when gas is unavailable; failure to clean the holes in the burners of the gas stove which results in partial combustion and emission of CO [16].
The prevalence of low birth weight infants in Sudan is reported to be 13/2% [27] in contrast to 17% reported by study mothers with a mean CO level of 18%. The higher the children’s CO level the more frequently their mothers reported them to be small at birth. This finding is consistent with the findings reported by Smith and colleagues [3]. Two studies which reported risk factors for low birth weight in Sudan included maternal anemia, hypertension, renal disease and educational level but did not consider HAP exposure [27,28]. Clearly many factors come together to affect the outcome of a pregnancy and CO levels may be one of many [3].
In our study we were unable to distinguish between small for date and preterm infants due to the fact that our subjects were unable to provide actual birth weight data. But the results are consistent with the literature; high CO levels are associated with more reports of small newborns [4,29]. An association between ambient air pollutant levels, pre term birth and low birth weight has been reported [30,31]. However, ambient air pollution in the neighborhoods studied was primarily due to the use of biomass fuels for cooking. The usual causes of ambient air pollution such as automotive and industrial exhausts did not exist in this area.
The association between CO exposure among smokers and fetal growth was studied by Cornelius and Day [6]. They found that the average birth weight of newborns born to smoking mothers was lower than the birthweight of newborns of nonsmoking mothers. Although the mean carboxyhemoglobin level [COHb%] for smoking mothers was only 1.92%, the correlation coefficient between birth weight and COHb% was significant. Studies of pregnant animals exposed to high concentrations of CO with COHb% comparable to the mean CO levels of our subjects reported low birth weights: Astrup reported that the birth weights of newborn rabbits whose mothers were exposed to increasing levels of CO were significantly lower, and there was an inverse relationship between levels of exposure and birth weights [31]. Wylie and colleagues [32] studied prenatal exposure to particulate matter and CO and reported thromboses of the placenta which occurred in a dose dependent manner. Clemente and associates suggest that hypoxia due to the greater affinity of CO compared with oxygen for hemoglobin binding sites results in decreased placental mitochondrial DNA and may explain the relationship between prenatal air pollution exposure and low birth weight [33]. These reports of progressively lower birth weights as the CO exposure rises in laboratory animals correlate with the significantly higher CO levels in mothers who reported small infants at birth in this study as well as the increased reports of small size at birth as the CO levels increased in study children. Finally, the data from these studies support the concept that CO crosses from the blood to the fetus and in part explains the elevated CO levels in the 0-6 month old study children whose mothers did not cook during their postpartum convalescence.
Twenty-one women 5.6% reported children dying in the first year of life. The reported mortality rate among children in the first year of life living with women who cooked with biomass fuels was 9% vs. 4% for women cooking with gas. The CO levels were similarly elevated in both groups [17% vs. 16%] suggesting a possible role for CO in the death of these infants. The smoke from partial combustion of biomass fuels may have other components such as PM2.5 which may contribute to the mortality. Children of mothers who cooked with gas may have been in a slightly higher socioeconomic group, had better nutrition, lived in larger dwellings, and may not have been exposed to other components of HAP which might have put them at lower risk for more severe lower respiratory tract infections and death in the first year of life. In addition we do not know how long the subjects had cooked with gas and over what period the deaths had occurred. The number of subjects in each cooking group was small, which makes it difficult to make meaningful comparisons; however, the average mortality rate for Sudan is 43.7 per 1000 live births similar to the rate of women who cooked with gas. On the other hand the 9% mortality for infants in the first year of life whose mothers cooked with biomass fuels is consistent with other neighboring countries where biomass fuels are used extensively for cooking, Chad 73.4 per 1,000 live births and the Central African Republic 87.6 per 1,000 live births.
More women cooking with biomass fuels reported treatment for three or more lung infections and their CO levels were significantly higher. However there is little data to suggest that the CO alone causes respiratory tract toxicity [34]. Thus the CO levels may represent a biomarker and surrogate for levels of HAP exposure experienced by the subjects and it’s the other products of partial biomass combustion such as PM2.5 which affected the lungs resulting in frequent infections and ultimately chronic obstructive pulmonary disease.
The adult and pediatric data were separately analyzed by different statisticians. Yet the reported data points [symptoms, health related effects and CO levels] between mothers and children throughout are consistent. Although the children’s anthropomorphic values did not appear to be affected by increasing CO levels, when compared to WHO standards there were significant differences for both males and females. These differences may reflect not only lifetime HAP exposure but fetal exposures, nutritional deficiencies and various chronic infections such as intestinal parasitic infection [35,36].
Conclusion
The hypothesis that levels of HAP exposure by history can be confirmed by CO-oximetry is supported by this study data. The CO levels obtained are an important biomarker for the symptoms and health related effects associated with HAP and a surrogate for levels of HAP exposure. Using CO levels to track various disease states could expand our understanding of the health related effects of HAP beyond death. Clark et al. and Rylance et al. in 2013 papers [34,36] emphasize the need for improved exposure assessment and studies into biomarkers of exposure to HAP. Our data suggest that future well designed studies of populations at risk for HAP could be carried out using questionnaires, exposure measurements and CO-oximetry.
References
- WHO (2012) Burden of disease from household air pollution.
- Smith KR, Samet JM, Romieu I, Bruce N (2000) Indoor air pollution in developing countries and acute lower respiratory infections in children. Thorax 55: 518-532.
- World Bank (2015) World Development Indicators 2015 report.
- Carter E, Norris C, Dionisio KL, Balakrishnan K, Checkley W, et al. (2017) Assessing exposure to household air pollution: A systematic review and pooled analysis of carbon monoxide as a surrogate measure of particulate matter. Environmental Health Perspectives 125: 076002.
- Cornelius MD, Day NL (2000) The effects of tobacco use during and after pregnancy on exposed children. Alco Res Health. 24: 242-249.
- Bruce N, Neufeld L, Boy E, West C (1998) Indoor biofuel air pollution and respiratory health: the role of confounding factors among women in highland Guatemala. Int J Epidemiol 27: 454-458.
- WHO (2014) Burden of disease from household air pollution for 2012.
- Naeher LP, Smith KR, Leaderer BP, Neufeld L, Mage DT (2001) Carbon monoxide as a tracer for assessing exposures to particulate matter in wood and gas cook stove households of highland Guatemala. Environ Sci Technol 35: 575-581.
- WHO (1999) Environmental Health Criteria 213, Carbon Monoxide second edition W.H.O.I.P.o.C. Safety.
- Townsend C, Maynard R (2002) Effects on health of prolonged exposure to low concentrations of carbon monoxide. Occup Environ Med 59: 708-711.
- Prockop LD, Chichkova RI (2007) Carbon monoxide intoxication: An updated review. Journal of the Neurological Sciences 262: 122-130.
- Farrow JR, Davis GJ, Roy TM, McCloud LC, Nichols GR (1990) Fetal death due to nonlethal maternal carbon monoxide poisoning. J Forensic Sci 35: 1448-1452.
- UNICEF (2016) Clear the air for children. U.N.I.C.s.R. Fund.
- Zaouter C, Zavorsky GS (2012) The measurement of carboxyhemoglobin and methemoglobin using a non-invasive pulse CO-oximeter. Respir Physiol Neurobiol 182: 88-92.
- Behera D, Dash S, Yadav SP (1991) Carboxyhaemoglobin in women exposed to different cooking fuels. Thorax 46: 344-346.
- Kang IJ, Lee MH (2006) Quantification of para-phenylenediamine and heavy metals in henna dye. Contact Dermatitis 55: 26-29.
- Yamamoto LG1, Yamamoto JA, Yamamoto JB, Yamamoto BE, Yamamoto PP (2008) Nail polish does not significantly affect pulse oximetry measurements in mildly hypoxic subjects. Respir Care 53: 1470-1474.
- Norman CA, Halton DM (1990) Is carbon monoxide a workplace teratogen? A review and evaluation of the literature. Ann Occup Hyg 34: 335-347.
- Adam KM, Abdaltam SA, Noreldeen AM, Alseed WA (2015) Relationship between maternal blood lead, cadmium, and zinc levels and spontaneous abortion in Sudanese women. Public Health Res 5: 171-176.
- Babker AMAAA, Elzaki SG, Dafallah SE (2013) An observational study of causes of recurrent spontaneous abortion among Sudanese women. Int J Sci Res 4: 1435-1438.
- BMJ Editorial (1973) Smoking Hazard to the Fetus. BMJ 5850: 369.
- Ezzati M, Kammen DM (2002) The health impacts of exposure to indoor air pollution from solid fuels in developing countries: Knowledge, gaps, and data needs. Env Health Persp 110: 1057.
- Northcross A, Chowdhury Z, McCracken J, Canuz E, Smith KR (2010) Estimating personal PM2.5 exposures using CO measurements in Guatemalan households cooking with wood fuel. J Environ Monit 12: 873-878.
- Lam N, Nicas M, Ruiz-Mercado I, Thompson LM, Romero C, et al. (2011) Non-invasive measurement of carbon monoxide burden in Guatemalan children and adults following wood-fired temazcal (sauna-bath) use. J Environ Monit 13: 2172-2181.
- Guyatt AR, Kirkham AJ, Mariner DC, Cumming G (1988) Is alveolar carbon monoxide an unreliable index of carboxyhaemoglobin changes during smoking in man? Clinical Sci 74: 29-36.
- Ahmed HA, Ibrahim AMF, Mahmood EAA, Abdu-Allah TOA (2014) Risk factors of low birth weight at three hospitals in Khartoum State, Sudan. Sudan J Paediatr 14: 22.
- Elhassan EM, Abbaker AO, Haggaz AD, Abubaker MS, Adam I (2010) Anaemia and low birth weight in Medani, Hospital Sudan. BMC Research Notes 3: 181.
- Hao H, Chang HH, Holmes HA, Mulholland JA, Klein M, et al. (2016) Air pollution and preterm birth in the U.S. State of Georgia (2002-2006): Associations with concentrations of 11 ambient air pollutants estimated by combining Community Multiscale Air Quality Model (CMAQ) simulations with stationary monitor measurements. Environ Health Perspect 124: 875-880.
- Salam MT, Millstein J, Li YF, Lurmann FW, Margolis HG, et al. (2005) Birth outcomes and prenatal exposure to ozone, carbon monoxide, and particulate matter: Results from the Children’s Health Study. Environ Health Perspect 113: 1638.
- Astrup P (1972) Some physiological and pathological effects of moderate carbon monoxide exposure. Br Med J 4: 447-452.
- Wylie BJ, Matechi E, Kishashu Y, Fawzi W, Premji Z, et al. (2017) Placental pathology associated with household air pollution in a cohort of pregnant women from Dar es Salaam, Tanzania. Environ Health Perspect 125: 134-140.
- Clemente DB, Casas M, Vilahur N, Begiristain H, Bustamante M, et al. (2016) Prenatal ambient air pollution, placental mitochondrial DNA content, and birth weight in the INMA (Spain) and ENVIRONAGE (Belgium) birth cohorts. Environ Health Perspect 124: 659-665.
- Rylance J, Gordon SB, Naeher LP, Patel A, Balmes JR, et al. (2013) Household air pollution: A call for studies into biomarkers of exposure and predictors of respiratory disease. Am J Physiol Lung Cell Mol Physiol 304: 571-578.
- Opara KO, Udoidung NI, Opara DC, Okon OE, Edosomwan EU, et al. (2012) The impact of intestinal parasitic infections on the nutritional status of rural and urban school-aged children in Nigeria. Int J MCH AIDS 1: 73-82.
- The World Bank (2015) "Mortality rate, infant (per 1,000 live births).”
- Clark ML1, Peel JL, Balakrishnan K, Breysse PN, Chillrud SN, et al. (2013) Health and household air pollution from solid fuel use: the need for improved exposure assessment. Environ Health Perspect 121: 1120-1128
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi