Editorial, Expert Opin Environ Biol Vol: 1 Issue: 1
Closing the Circle: A Plea for State of the Art Microscopy Supporting Environmental Biodiversity Data from PCR Based Approaches
| Martin Pfannkuchen* | |
| Ruder Boskovic Institute, Croatia | |
| Corresponding author : Martin Pfannkuchen, MD Ruder Boskovic Institute, Croatia Tel: 099 8121 728 E-mail: Martin.Andreas.Pfannkuchen@irb.hr |
|
| Received: June 16, 2012 Accepted: June 18, 2012 Published: June 22, 2012 | |
| Citation: Pfannkuchen M (2012) Closing the Circle: A Plea for State of the Art Microscopy Supporting Environmental Biodiversity Data from PCR Based Approaches. Expert Opin Environ Biol 1:1. doi:10.4172/2325-9655.1000e101 |
Abstract
Closing the Circle: A Plea for State of the Art Microscopy Supporting Environmental Biodiversity Data from PCR Based Approaches
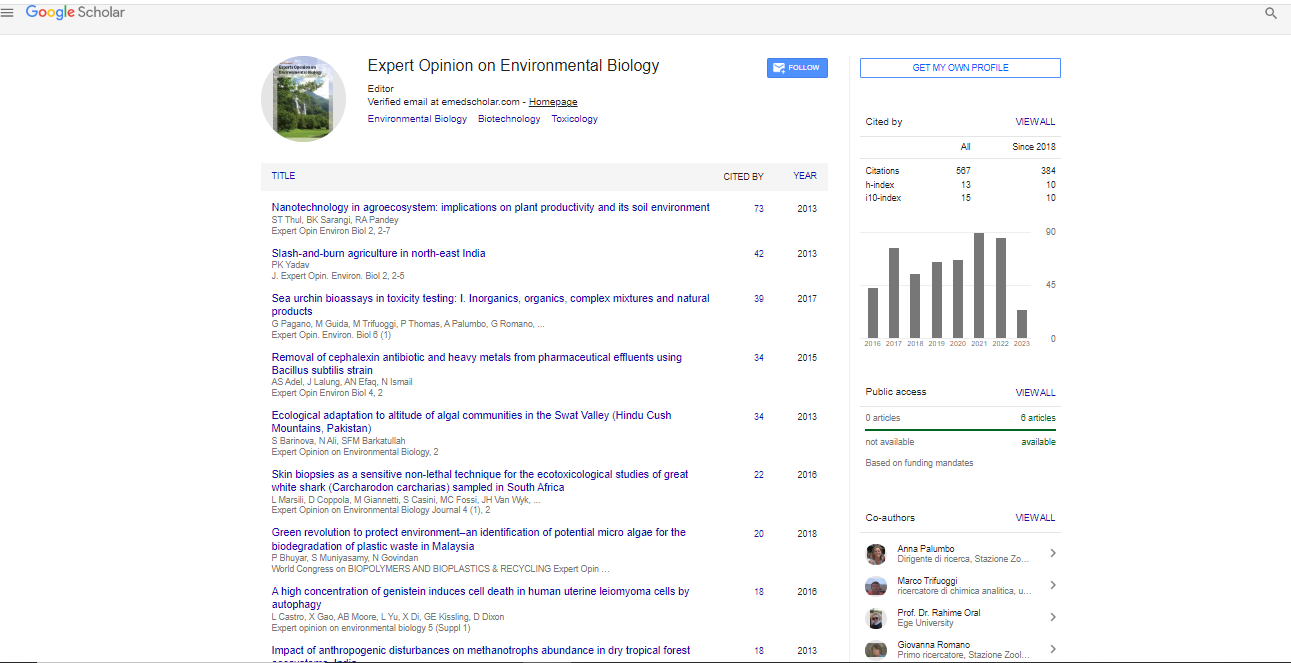
Advanced microscopy is a reemerging tool in many fields of natural sciences from materials research to cell biology. Especially after the discovery of fluorescent molecules and their heterologous expression, fluorescence microscopy has become a key tool in the research of cellular regulation and transport mechanisms.
| Advanced microscopy is a reemerging tool in many fields of natural sciences from materials research to cell biology. Especially after the discovery of fluorescent molecules and their heterologous expression, fluorescence microscopy has become a key tool in the research of cellular regulation and transport mechanisms. The application of fluorescently labeled antibodies, oligonucleotide probes or fluorescent proteins allows the localization and quantification of target molecules in whole organisms or in cells with sub micrometer resolution. In environmental biology however the biodiversity of unicellular organisms and viruses is with increasing intensity assessed by molecular methods. PCR based approaches in combination with modern and affordable sequencing technologies are employed to determine or even monitor the biodiversity of microorganisms and viruses [1-8]. The challenges of the aforementioned approaches in terms of data storage and analysis are widely discussed. However the pitfalls and limitations are less prominently discussed. An increasing number of research papers reports sequences retrieved from environmental samples. They most often aim at getting an overview of the species diversity in the sample by amplifying and sequencing taxonomically informative marker genes. A second aim is to retrieve information about metabolic pathways active or possible in the sample by amplifying and sequencing key genes for the activity of interest. In the majority of reports however, the interpretation of the results is not a mere listing of amplifiable sequences from environmental sequences, but a conclusion suggesting information about the entire diversity of targeted sequences in the sample, in many cases the results are even interpreted quantitatively. | |
| At this point it is important to remind ourselves, that PCR is a process that is introducing mistakes in the resulting sequences. Now if a clean and uniform template sequence is used, there do exist methods to identify and eliminate those mistakes. For environmental samples with an unknown diversity of templates however this is not the case. PCR introduces sequences alterations are virtually unidentifiable for environmental samples. While the aforementioned problem might be counteracted by thresholds in diversity analyses, PCR based methods face a more serious problem in environmental samples. PCR is a highly competitive process. In a mixture of different templates, given a set of PCR conditions, easier amplifiable or more abundant templates outcompete more difficultly amplifiable templates. We should also keep in mind, that the aforementioned particularities are superimposed on more upstream challenges. The extractability of DNA or RNA differs from species to species, which effects the yields in a species specific manner. And finally each environment or environmental condition provides its own set of chemicals which are known to change PCR results in even sequence specific ways. Not all of those chemicals are readily eliminated during DNA extraction and purification. Given the above considerations, a PCR based analysis of environmental samples does results in a set of extractable, amplifiable and sequencable sequences. And in all likelihood the resulting sequence (a) diversity can be found in the environmental sample. But this approach has to be considered as a minimum approach towards the true sequence diversity within the examined environmental sample. This kind of approach can readily be used to screen for new sequences of taxonomical or biotechnological interest. However, given the above considerations on the limitation of PCR based approaches, ecological interpretations are highly speculative. As both, the chemical composition as well as the taxonomic composition of the environmental sample does influence the quantitative and qualitative outcome of PCR based approaches in an unpredictable way, neither qualitative nor quantitative aspects of the results can be compared between samples. They cannot even be considered to be complete within one single sample. | |
| As a prerequisite for environmental interpretation (e.g. qualitative or quantitative) of PCR based results in the broader sense of metagenomic approaches, further controls of the results are required to estimate the quantitative and qualitative information within the resulting datasets. Already in 1999 Manz et al. [9] suggested a procedure to overcome the limitations of PCR based methods: The full circle approach. As briefly mentioned in the introductory part, modern fluorescence microscopy in combination with in situ hybridization techniques allows to localize and quantify target molecules (proteins and DNA/RNA) with high specificity and high spatial (sub cellular) resolution. While it is admittedly laborious, it is possible to localize and quantify each sequence resulting from PCR based analysis of environmental samples in environmental samples. And this is possible with the accuracy up to a single base mismatch in target sequences [9]. | |
| The full circle approach consists of 5 principle steps: | |
| 1. DNA Isolation from environmental samples | |
| 2. Recovery of sequence information from environmental DNA or RNA | |
| 3. Sequence analysis and design of specific hybridization probes against the retrieved sequences | |
| 4. In situ hybridization with fluorescently labeled specific probes | |
| 5. Localization of the sequences of interest in environmental samples via fluorescence microscopy (Figure 1) | |
| Figure 1: during infection, but how the host responds, one might determine which of these responses are important for viral survival, and exploit them for antiviral purposes. | |
| The second half of the full circle approach (steps 3-5) allows to localize the retrieved sequences in environmental samples. In that way the experimental setup controls, whether the retrieved sequences are actually present in the environmental sample. Steps 4 and 5 also give additional valuable information about the relative and absolute abundances of the respective sequences in the environmental sample and depending on the sample preparation even on their spatial organization [10,11]. Only closing the full circle approach by following all 5 steps represents a fully controlled experimental setup. | |
| Environmental interpretations of PCR based sequence datasets from environmental samples do almost always require a quantitative aspect of data analysis. Microscopic examinations of environmental samples did already radically correct the ecological conclusions drawn from PCR based results [12]. To avoid erroneous conclusions from environmental sequences data sets, we have to make sure, that the sequences of interest are actually of quantitative relevance in environmental samples. Complementing PCR based sequence retrievals from environmental samples with well documented microscopic proof will ensure the accuracy of future interpretations of such datasets. This is a demanding challenge for experimental setups as well as for reviewers in the future. It will require a combined and interdisciplinary effort to guarantee the necessary accuracy in the performance and documentation of the methodological spectrum required for the full circle approach. However what is the good practice standard in molecular ecology since at least 1999 has to finally become the standard in present day publications as well. | |
References |
|
|
|
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi