Research Article, Endocrinol Diabetes Res Vol: 5 Issue: 1
Efficacy of Metformin in Vitamin B12 Deficient Liver Cells
May Oo Khin1*, Antonysunil Adaikalakoteswari2,3, Philip Voyias2, Ponnusamy Saravanan2,4
1National Guideline Alliance, Royal College of Obstetricians and Gynaecologists, 27, Sussex Pl, Regent’s Park, London, United Kingdom
2Population Evidence and Technologies, Warwick Medical School, University of Warwick, Coventry, United Kingdom
3School of Science and Technology, Nottingham Trent University, Nottingham, NG11 8NS, United Kingdom
4Department of Diabetes and Endocrinology, George Eliot Hospital, Nuneaton, United Kingdom
*Corresponding Author : May Oo Khin
National Guideline Alliance, Royal College of Obstetricians and Gynaecologists, 27, Sussex Pl, Regent’s Park, London, United Kingdom
Tel: +44 20 7772 6484
E-mail: mkhin@rcog.org.uk
Received: March 13, 2018 Accepted: December 20, 2018 Published: January 24, 2019
Citation: Khin MO, Adaikalakoteswari A, Voyias P, Saravanan P (2019) Efficacy of Metformin in Vitamin B12 Deficient Liver Cells. Endocrinol Diabetes Res 5:1. doi: 10.4172/2470-7570.1000140
Abstract
Objective: The anti-diabetic drug, metformin, is associated with progressive decrease in serum vitamin B12 levels whereas vitamin B12 deficiency is related to increased insulin resistance and dyslipidaemia by altered methylation. As the risk of vitamin B12 deficiency is increased among metformin users, it is of utmost importance to examine how metformin response in vitamin B12 deficient population.
Research design and methods: We investigated the cellular mechanism of metformin in vitamin B12 insufficient human hepatocellular cell line (HepG2). HepG2 was cultured in different vitamin B12 conditions (0, 10, 100, 1000 nM) for four passages over 24 days. Then, they were treated with metformin 2 mM for 24 h. Protein and RNA extracts were quantified for AMP-activated protein kinase (AMPK) and its downstream signals.
Results: In HepG2 culture, there was a decreased gene expression level of Fatty Acid Synthase (FAS) and 3-Hydroxy 3-Methylglutaryl CoA Reductase (HMGCR) enzymes in 1000 nM B12 compared to 0 nM B12 condition. In metformin-treated hepG2 cells, phosphorylations of AMPK and acetyl-CoA carboxylase (ACC) were increased in 1000 nM B12 compared to 0 nM B12 condition. Similarly, there was a decreased gene expression level of FAS and HMGCR with metformin activation and the effects was more pronounced in B12 supplemented cultures than 0 nM B12 condition.
Conclusion: Our preliminary results indicate that metformin phosphorylation of AMPK together with its downstream signals were reduced in low B12 differentiated hepatic cells. This noble finding highlights the importance of vitamin B12 sufficiency for full potency of metformin.
Keywords: Metformin; Vitamin B12; Liver cells; HepG2 culture
Introduction
Metformin is a well-established anti-diabetic drug, used in diabetes and because of its role in lowering blood lipid levels; it is increasingly used for obese patients. One of its side-effects includes a decrease in serum vitamin B12 levels. It has been reported that long-term metformin use in type 2 diabetes is associated with progressive decrease in blood vitamin B12 levels [1]. In fact, vitamin B12 deficiency among metformin users is two times higher than non-metformin users [2]. Thus, it is not uncommon to find vitamin B12 deficiency among metformin users. Metformin primarily works by activating AMPK (adenosine monophosphate-activated protein kinase, a master energy regulator of the cell. Zang et al. [3] also reported that the lipid lowering effects of metformin occurs through AMPK activation [3]. Activated AMPK in liver then subsequently phosphorylates downstream enzymes important in lipid metabolism. Phosphorylation at serine 79 of ACC (Acetyl-CoA Carboxylase) by AMPK could inhibit its action on lipogenesis while favouring fatty acid oxidation [3]. In addition, AMPK also inhibits SREBP (sterol regulatory element binding protein) which is nuclear transcription factor for lipid synthesis enzymes and thereby, decreases expression of 3-hydroxy-3-methyl glutaryl coenzyme A reductase (HMGCR) and fatty acid synthase (FAS), which are rate-limiting enzymes in cholesterol and triglyceride synthesis [4]. It is thus clear that AMPK phosphorylation is essential for the metabolic benefits of metformin. At the same time, it has been reported that vitamin B12 deficiency is associated with impaired lipid metabolism [5,6]. So far, it is suggested to occur through altered methylation by the fact that adipocyte cultured in vitamin B12 deficient culture revealed increased intracellular cholesterol levels [7]. It is well-established that vitamin B12 plays an indispensable cofactor in energy metabolism. However, the importance of vitamin B12 deficiency in energy regulation is not very clear yet. In fact, it is a clinically interesting question to answer how metformin acts in vitamin B12 deficient and sufficient cells. In order to examine this further, we have chosen human hepatoma cell (HepG2) line as a model to conduct our experiment. Liver is the storage organ of vitamin B12 in the body and it also plays a major role in the regulation of blood lipid levels. Moreover, the recommended culture media of HepG2 (MEM, Invitrogen) does not contain vitamin B12. The only source of vitamin B12 in HepG2 culture media is from the supplemented fetal bovine serum which was stated to have 120 pM levels of vitamin B12 [8]. Considering the fact that these cells are replicated successively in culture media without vitamin B12 over time, it can be assumed that vitamin B12 in these cells are lower than the required amount for optimal cellular function. It could therefore be postulated that longterm in-vitro culture of HepG2 can induce vitamin B12 deficient hepatocyte model. The passage number in this experiment started from passage 4 HepG2 as to ensure the replication of experimental hepatocytes in media lack of vitamin B12. We chose the vitamin B12 supplementation model rather than subtraction study as it resembles much more closely to the physiological condition.
The aims of this study are to examine the efficacy of metformin in vitamin
To examine how low vitamin B12 affects the lipid metabolism in liver cell line
To examine whether vitamin B12 supplementation augments metformin action in liver cell line
Materials and Methods
Materials
Metformin (1,1-dimethylbiguanide), cyanocobalamin, Antirabbit antibodies conjugated to horseradish peroxidase and dimethyl sulphoxide (DMSO) were purchased from Sigma. Phospho-AMPKα (Thr-172), total AMPK against AMPKα1 and AMPKα2, phospho- Akt (Ser-473), Akt, phospho-GSK3α/β, GSK3β, phospho-ACC (Ser-79), total-ACC, β-actin antibodies were purchased from cell signalling. Eagle’s minimum essential medium (MEM) and fetal bovine serum were from Invitrogen. The custom taqman gene expression assays and housekeeping gene 18s rRNA from Applied Biosystems. The gene assays with their catalogue numbers included are: SREBF1 (Hs01088691_m1), SREBF2 (Hs01081784_m1), FAS (Hs00236330_m1) and HMGCR (Hs00168352_m1). The gene primers and polymerase chain reaction (PCR) test kits were from life technologies. Modified Eagle’s Medium (MEM), fetal bovine serum, sodium pyruvate and antibiotics (penicillin and streptomycin) were from Invitrogen.
Culture of HepG2 cells:
HepG2 cells were kindly contributed by Dr Ming Zhan Xue, who originally purchased them from American Type Culture Collection (ATCC). The cells were grown in MEM supplemented with 10% foetal bovine serum (FBS), L-Glutamine (2 mM), sodium pyruvate (100 mM) and penicillin-streptomycin (100 units/mL) in T-75 or T-175 flasks (corning cell culture flask surface area 75 cm2 or 175 cm2 respectively). The cells were maintained as sub-confluent monolayer at 37°C and 5% CO2. The media was changed the next morning and then it was changed every alternate day.
Experimental treatment of HepG2 cells with different concentration of vitamin B12:
HepG2 Cells were seeded at a density of 6000 cells/cm2 and treated with different concentrations of vitamin B12 (≥ 98% cyanocobalamin, Sigma-Aldrich) (10 nM, 100 nM and 1000 nM) for 24 days (passaged on every 6th day for 4 passages) and incubated at 37°C in 5% CO2. The dose concentrations are approximated from the previous studies [8]. The culture plates were kept in serum free medium overnight before extraction or before any treatment. A control culture plate with respective vitamin B12 concentration was kept alongside together with any treatment. Three independent experiments were conducted for each treatment.
Western blot analysis
Cells cultured in 6 well plates were rinsed three times with icecold phosphate-buffered saline (PBS), then were scraped into 0.5 ml ice-cold RIPA buffer (20 mM Tris-HCl (pH 7.5), 150 mM NaCl, 1 mM EDTA, 1% Nonidet P40, 0.5% deoxycholate, 0.1% SDS, 5 mM NaF, and 0.1 mg/ml PMSF). Extracts were quantitated by the Bio-Rad Protein Assay (Bio-Rad Laboratories, Hercules, and CA) and were stored at -80oC. Equal amounts of protein (5 ug) were resolved by 7.5% sodium dodecyl sulfate–polyacrylamide gels for electrophoresis (SDS-PAGE), were transferred to nitrocellulose membranes and were blocked one hour in PBS containing 0.05% Tween 20 and 0.2% i-block. Membranes were incubated with primary antibodies for 1 h at RT, followed by three 5 min washes in PBS/Tween 20. Blots were then incubated for 1 h with secondary antibody/PBS/Tween 20. Following three 5 min washes in PBS/Tween 20, immune complexes were visualized by chemiluminescence. Autoradiographs were semi-quantified using 2D densitometry software (ImageQuant, GE healthcare, UK). The bands were first normalised as a function of the loading control (protein of interest/β-actin) or total expression of the proteins (for phosphoproteins), then be converted to fold change compared with controls.
RNA isolation and quantitative real-time PCR (qRT-PCR) analysis
RNA isolation was performed using test kits (Qiagen). Quantitative real-time PCR analysis was used to validate the mRNA expression levels of selected genes using an ABI 7500 real-time PCR Sequence Detection system. Pre-optimized quantitative primer and probe sequences for genes were utilized (Applera, Cheshire, UK). All reactions were multiplexed with the housekeeping gene 18S, provided as a pre-optimized control probe (Applera), enabling data to be expressed in relation to an internal reference to allow for differences in relative threshold efficiency. Data obtained as cycle threshold (Ct) values according to the manufacturer’s guidelines (the cycle number at which logarithmic PCR plots cross a calculated threshold line) were used to determine ΔCt values (ΔCt=Ct of 18 S housekeeping gene subtracted from Ct of gene of interest). Measurements were carried out as triplicates for each sample. To exclude potential bias due to averaging data which has been transformed through the equation 2-ΔΔCt, all statistics were performed at the ΔCt stage.
Statistics
Data were expressed as mean ± standard error of the mean (SEM). A two-tailed unpaired Student’s t test was used to analyse the data. P<0.05 were considered to be statistically significant.
Results
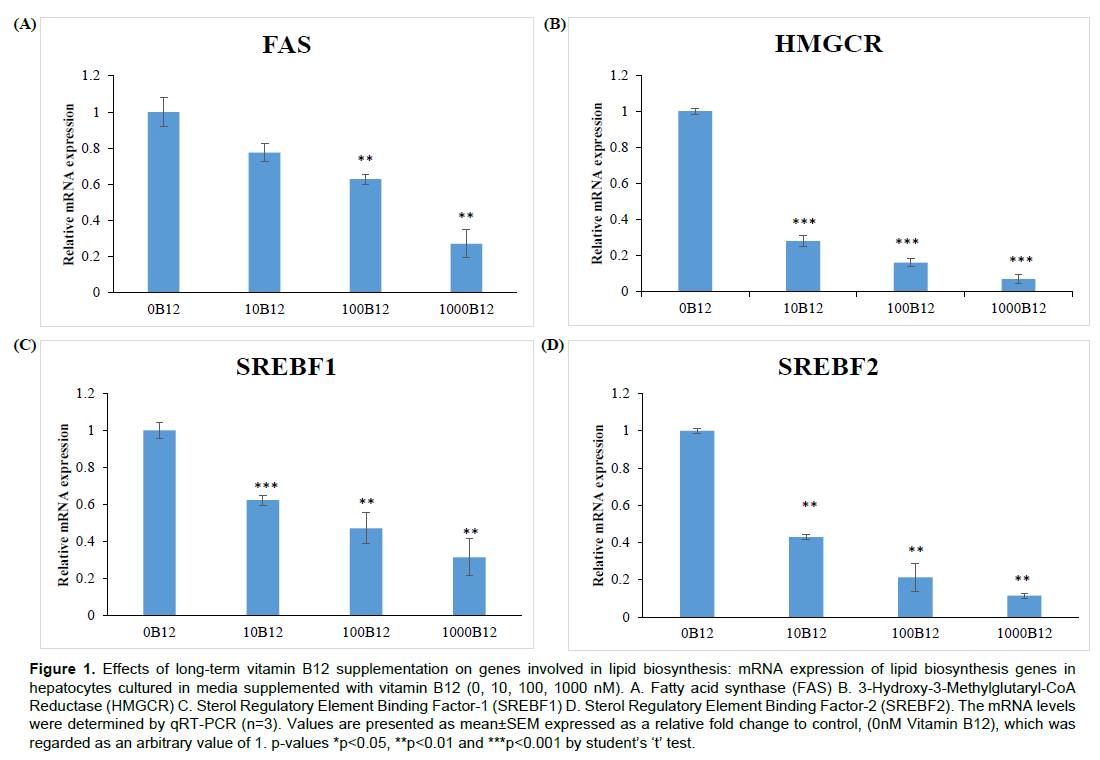
Lipogenic genes were down-regulated by vitamin B12 supplementation in hepatocyte cell line cultured in long-term low vitamin B12 (0 nM) media. The mRNA expression levels of key enzymes of triglyceride and cholesterol biosynthesis (FAS and HMGCR, respectively) and their regulatory genes (Sterol regulatory element binding factors (SREBFs) were measured by qRT-PCR. SREBFs are conditional transcriptional factors resided in endoplasmic reticulum and can be stimulated when intracellular lipid levels are low (SREBF1 mainly for triglyceride and SREBF2 mainly for cholesterol). When activated and released by protease, nuclear SREBFs bind to the promoter region of more than 30 target enzymes dedicated to increased synthesis and uptake of lipids including FAS and HMGCR. We have found that vitamin B12 supplementation significantly down-regulated the mRNA expressions of FAS and HMGCR enzymes in 100 and 1000 nM vitamin B12 conditions compared to 0 nM vitamin B12 condition (Figure 1 A and B). Furthermore, their transcriptional regulators such as SREBF1 and SREBF2 also showed decreased expression with increasing vitamin B12 concentrations with the lowest in 1000 nM B12 condition (Figure 1 C and D). Phosphorylation of AMPK and ACC enzymes induced by metformin was increased in vitamin B12 supplemented culture compared to low B12 culture (0 nM)
Figure 1. Effects of long-term vitamin B12 supplementation on genes involved in lipid biosynthesis: mRNA expression of lipid biosynthesis genes in hepatocytes cultured in media supplemented with vitamin B12 (0, 0, 100, 1000 nM). A. Fatty acid synthase (FAS) B. 3-Hydroxy-3-Methylglutaryl-CoA Reductase (HMGCR) C. Sterol Regulatory Element Binding Factor-1 (SREBF1) D. Sterol Regulatory Element Binding Factor-2 (SREBF2). The mRNA levels were determined by qRT-PCR (n=3). Values are presented as mean±SEM expressed as a relative fold change to control, (0nM Vitamin B12), which was regarded as an arbitrary value of 1. p-values *p<0.05, **p<0.01 and ***p<0.001 by student’s ‘t’ test.
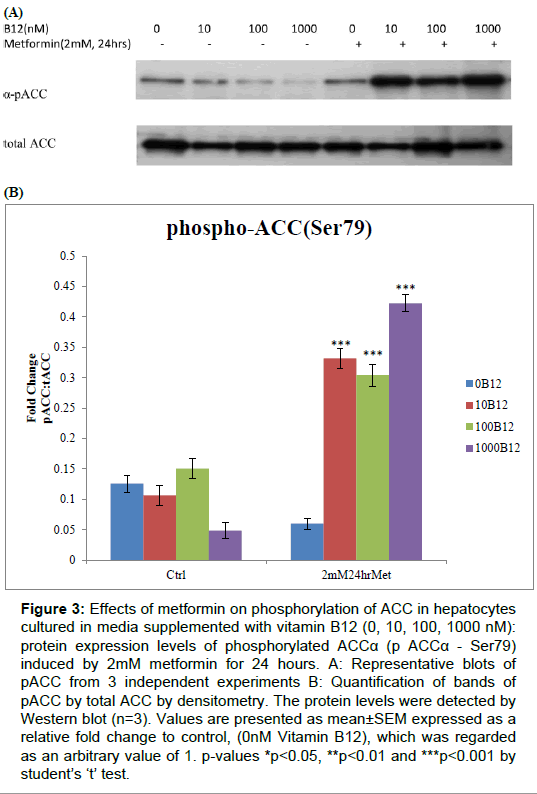
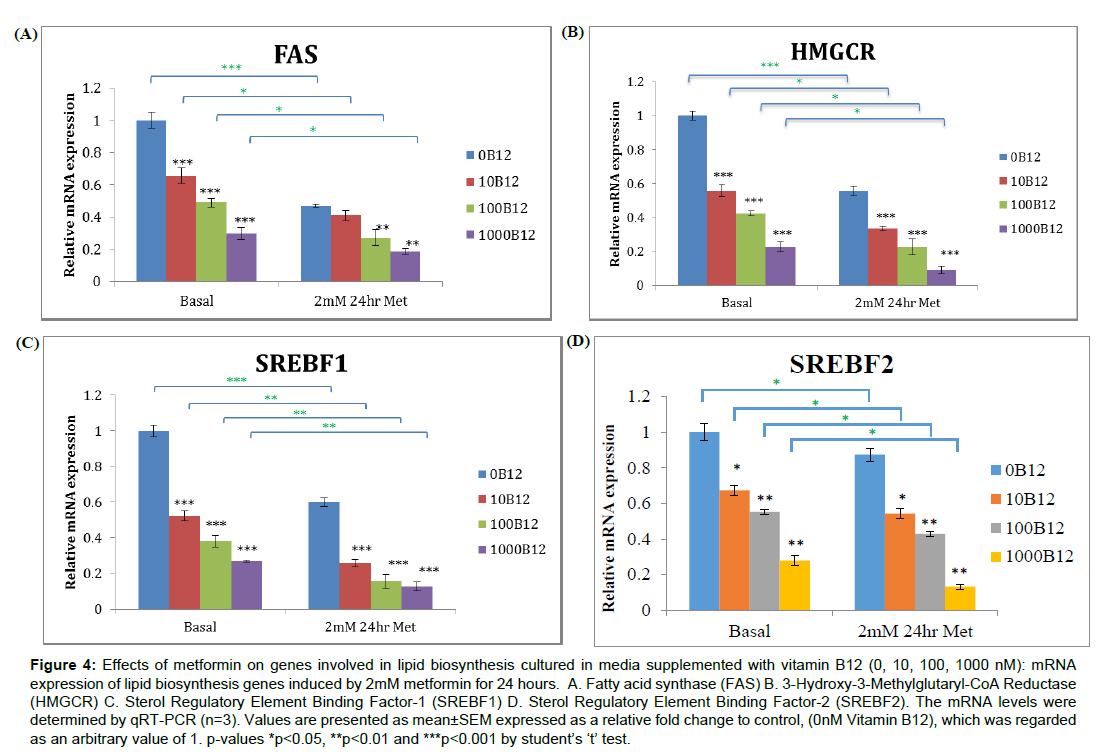
Zhou et al. [9] have reported that metformin works by activating AMPK in liver. Metformin partially blocks the complex I of mitochondrial oxidative respiration thereby, altering AMP: ATP ratio which phosphorylates AMPK at threonine 172 [9]. Subsequently, the target enzymes of AMPK, such as ACC, FAS and HMGCR, are suppressed and intracellular lipid levels are decreased. Metformin-induced phosphorylation of AMPK is reported to be the greatest with 2 mM metformin for 24 h treatment. We found that the protein expression levels of metformin-stimulated phosphorylated AMPK and ACC ratios were lowest in 0 nM vitamin B12 condition compared to other vitamin B12 concentrations (10, 100, 1000 nM) (Figure 2 and 3). Moreover, the down-regulated mRNA levels of FAS and HMGCR together with their transcriptional factors, SREBF1 and SREBF2 respectively, induced by vitamin B12 supplementation were significantly further decreased with metformin stimulation significantly compared to their vitamin B12 counterparts (Figure 4).
Figure 2: Effects of metformin on phosphorylation of AMPK in hepatocytes cultured in media supplemented with vitamin B12 (0, 10, 100, 1000 nM): protein expression levels of phosphorylated AMPK (pAMPK - Thr172) induced by 2mM metformin for 24 hours A: Representative blots of pAMPK from 3 independent experiments B: Quantification of bands of pAMPK by total AMPK by densitometry. The protein levels were detected by Western blot (n=3). Values are presented as mean±SEM expressed as a relative fold change to control, (0nM Vitamin B12), which was regarded as an arbitrary value of 1. p-values *p<0.05, **p<0.01 and ***p<0.001 by student’s ‘t’ test.
Figure 3: Effects of metformin on phosphorylation of ACC in hepatocytes cultured in media supplemented with vitamin B12 (0, 10, 100, 1000 nM): protein expression levels of phosphorylated ACCα (p ACCα - Ser79) induced by 2mM metformin for 24 hours. A: Representative blots of pACC from 3 independent experiments B: Quantification of bands of pACC by total ACC by densitometry. The protein levels were detected by Western blot (n=3). Values are presented as mean±SEM expressed as a relative fold change to control, (0nM Vitamin B12), which was regarded as an arbitrary value of 1. p-values *p<0.05, **p<0.01 and ***p<0.001 by student’s ‘t’ test.
Figure 4: Effects of metformin on genes involved in lipid biosynthesis cultured in media supplemented with vitamin B12 (0, 10, 100, 1000 nM): mRNA expression of lipid biosynthesis genes induced by 2mM metformin for 24 hours. A. Fatty acid synthase (FAS) B. 3-Hydroxy-3-Methylglutaryl-CoA Reductase (HMGCR) C. Sterol Regulatory Element Binding Factor-1 (SREBF1) D. Sterol Regulatory Element Binding Factor-2 (SREBF2). The mRNA levels were determined by qRT-PCR (n=3). Values are presented as mean±SEM expressed as a relative fold change to control, (0nM Vitamin B12), which was regarded as an arbitrary value of 1. p-values *p<0.05, **p<0.01 and ***p<0.001 by student’s ‘t’ test.
Discussion
The association of vitamin B12 deficiency with blood dyslipidaemia has been reported by several clinical studies [5-7,10]. The proposed mechanistic model is that it occurs through reduced methylation potential and hypo-methylation of lipogenic enzymes in vitamin B12 deficient condition and which has partly elucidated by a study done on adipocyte culture [7]. As liver plays a central role in both uptake and transport of lipids in the circulation, examining the changes of lipid metabolism in vitamin B12 deficient will enable to understand how serum vitamin B12 deficiency can lead to abnormal lipid profiles and, more importantly, whether vitamin B12 supplementation can reverse this. Similar to the previous adipocyte study [7] and in vivo studies [11], we have found that there was an increased expression of hepatic lipogenic enzymes in low vitamin B12 condition. The study also suggested that vitamin B12 deficiency could impair the efficacy of metformin whereas vitamin B12 supplementation can augment it. Thus, the novel finding in this study is that vitamin B12 supplementation could improve the efficacy of metformin.
Effects of low vitamin B12 levels on lipid synthesis
Firstly, we decided to look at the expression of enzymes important for cholesterol and triglyceride synthesis in liver cells, which have been passaged in different vitamin B12 conditions successively for several times. We included enzymes for triglyceride in this study because liver is central regulator of blood triglyceride levels. Moreover, in Wister rat study done by Kumar and Frenkel groups [11], it was independently observed that vitamin B12 restriction in mothers increased lipid accumulation in viscera and altered adipose tissue composition in the offspring by increasing the activities of hepatic FAS and ACC [11]. We chose FAS rather than ACC because FAS is highly correlated with ACC and the rate of fatty acid synthesis [11]. In this study, HMGCR and FAS mRNA levels were downregulated in the liver supplemented with vitamin B12. The decreased lipogenic enzyme levels are in coordination with their transcription factors (SREBF1 and SREBF2). The effects of maternal B12 deficiency on lipid synthesis of progeny’s liver have been studied in animals over time and they have consistently reported that maternal vitamin B12 deficiency increased liver lipid content, triglyceride, total cholesterol, cholesterol ester and free cholesterol, in their offspring [12-14]. Furthermore, an adipocyte cell culture model indicated increased cholesterol synthesis in no or low vitamin B12 conditions [7]. Accordingly, the possible findings of intracellular lipids in our study could be decreased with vitamin B12 supplementation.
Lipid-lowering effects of metformin in vitamin B12 deficient hepatocytes
At the basal condition without metformin treatment, there was no change in phosphorylated AMPK (Thr172) levels among HepG2 culture with vitamin B12 addition. When metformin was given, the increased phosphorylation of AMPK was observed in all HepG2 cells. The effect was more pronounced in vitamin B12 supplemented cells compared with 0B12 condition. This might indicate that metformin-induced AMPK phosphorylation was impeded in low B12 (0B12) condition and cells with vitamin B12 supplementation could improve the situation. We have also looked at metformin effect on the phosphorylation of ACC at Ser79, the downstream signal of AMPK. Similarly, once AMPK activated by metformin, the ACC phosphorylation was increased with vitamin B12 concentrations. We also determined the ratio of ACC phosphorylation levels by dividing with total ACC protein levels and there was increased phospho: total-AMPK or phospho: total-ACC with increasing B12 conditions. This might suggest that vitamin B12 supplementation could help augment the effects of metformin. Theoretically, in vitamin B12 deficiency, MMA is accumulated and impedes Coenzyme Q biosynthesis. Thus, it is possible that vitamin B12 sufficient culture helps relieve these MMA and thereby enhancing mitochondrial respiration, which is the primary site of metformin action. This work should be further confirmed by future study with vitamin B12 on mitochondrial respiration. It should also be noted that once ACC is phosphorylated, triglyceride synthesis was inhibited. Moreover, vitamin B12 supplementation can help fatty acyl transfer into mitochondria by acting at carnitine parmitoyltransferase-1 (CPT-1) Thus; we concluded that vitamin B12 supplementation helps improve lipid lowering effects of metformin, possibly by regulating energy metabolism within the cells. We also examined other important downstream enzyme expression of AMPK, important for lipogenesis, FAS and HMGCR together with their transcriptional factors SREBF 1 and 2. AMPK activation by metformin or other drugs/metabolites are reported to reduce the expression of these enzymes with their transcriptional factors in liver [15]. Likewise, when metformin was given, we have observed that HMGCR and FAS expression levels were further reduced by metformin treatment and this was accompanied by reduction of their transcriptional factors. The levels are in accordance with increasing concentrations of vitamin B12. Thus, it is likely that in vitamin B12 deficient cells, metformin-induced lipid lowering effects of metformin can be enhanced by addition of vitamin B12.
Strengths and Limitation
The strength of the study is that this is the first pioneer study to support that cells with supplemented vitamin B12 can improve the efficacy of metformin on AMPK activation and improve lipid profiles. Moreover, the use of liver cell culture could help extrapolation of the mechanism to clinical studies. In addition, the inclusion of 4 different vitamin B12 concentrations will help for future successive studies. However, these data are supported by long-term vitamin B12 supplementation and thus, the metformin effect on liver cell with shorter duration of treatment would be of advantage before implementing the supplementation of vitamin B12 to vitamin B12 deficient patients at metformin initiation. Although we have reported the AMPK signalling pathway of metformin affected by vitamin B12 concentrations, the findings should be confirmed by inhibitor studies using blockers or transfection of AMPK. It is also worthwhile to confirm the findings in animal model of vitamin B12 supplementation with metformin treatment before future implication.
Suggestions for future study
The future study should look at other key enzymes in lipid metabolism such as ACC, HMGCS and LDLR together with intracellular lipid content. Then, we need to explore the other factors contributing to elevated VLDL outputs such as increased fatty acid influx, mainly from lipolysis of adipocytes. Amstead and his group have predicted that the enzyme for conversion of free to esterified cholesterol might be impaired in vitamin B12 deficiency [13] possibly suggesting involvement of lipid esterification enzyme, whereas Khaire et al. [16] has reported that the lipid fractions in the liver are affected by vitamin B12. Thus, we suggest that the individual fatty acid fractions of increased lipid content (such as total cholesterol and triglyceride levels) due to vitamin B12 addition should be included. In addition, a recent study of vitamin B12 supplementation to wistar rats during pregnancy has suggested that there is increased plasma triglyceride levels in offspring of vitamin B12 supplemented rats with decreased hepatic eicosapentaenoic acid (EPA) accumulation which regulates triglyceride metabolism [16]. Thus, the relationship between omega3 fatty acid with vitamin B12 on lipid metabolism should be further explored. Furthermore, the role of vitamin B12 in mitochondrial dysfunction and metabolic dysregulation need to be fully established. In addition, as the lipid composition of the tissues and the circulation can be differed by gender [14], it is of importance to report findings from the clinical studies looking at the lipid profile in B12 deficiency by gender.
Clinical implications
Vitamin B12 is vital for normal lipid homeostasis. However, because of high storage in liver and low prevalence of vegetarian population, its significance is usually ignored. On the other hand, having aware of importance of folic acid in neuronal development, folic acid fortification is everywhere from elderly diet to antenatal care. It should be noted that B12 deficiency is quite common in pregnancy due to high demand. In fact, one prospective study has reported that the intakes of low B12 high folate diet in pregnant mothers have high risk of small-for-gestational-age [17]. The possible mechanism could be that the associated dyslipidaemia and high homocysteine levels in vitamin B12 deficient mothers disturb with placenta blood flow, thereby, altering the size of intrauterine baby [10,18]. Although adequate vitamin B12 levels can help improve these outcomes by providing enough methyl groups for normal fetal programming, there is limited data to support that vitamin B12 supplementation are beneficial. Thus, considering the importance of vitamin B12 in pregnancy, epidemiological and mechanistic studies looking at vitamin B12 deficiency and supplementation are highly demanded before implementation of vitamin B12 at population levels.
In conclusion, vitamin B12 deficiency can impede methylation of lipid synthesis enzymes as well as the efficacy of metformin, particularly its lipid lowering effects; can be impaired in vitamin B12 deficiency. Thus, before metformin is given, it should be considered to check vitamin B12 levels.
Guarantor’s Statement
Dr. May Oo Khin is the guarantor of this work and, as such, had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Author Contributions
S.P conceived the research question. M.O.K conducted all the experiments and drafted the manuscript. All the authors contributed to data analysis and writing discussion and editing the manuscript.
Funding
This work was supported by research student fund of Warwick Medical School.
References
- de Jager JA, Kooy P, Lehert MG, Wulffele J, van der KD, et al. (2010) Long term treatment with metformin in patients with type 2 diabetes and risk of vitamin B-12 deficiency: Randomised placebo controlled trial. BMJ 340: 2181.
- Reinstatler LP, Qi RS, Williamson JV, Garn JP, Jr Oakley, et al. (2012) Association of biochemical B(1)(2) deficiency with metformin therapy and vitamin B (1) (2) supplements: The National Health and Nutrition Examination Survey 1999-2006. Diabetes Care 35: 327-333.
- Zang MA, Zuccollo X, Hou D, Nagata K, Walsh H, et al. (2004) AMP-activated protein kinase is required for the lipid-lowering effect of metformin in insulin-resistant human HepG2 cells. J Biol Chem 279: 47898-47905.
- Li YS, Xu MM, Mihaylova B, Zheng X, Hou B, et al. (2011) AMPK phosphorylates and inhibits SREBP activity to attenuate hepatic steatosis and atherosclerosis in diet-induced insulin-resistant mice. Cell Metab 13: 376-388.
- Adaikalakoteswari AR, Jayashri N, Sukumar H, Venkataraman R, Pradeepa K, et al. (2014)Vitamin B12 deficiency is associated with adverse lipid profile in Europeans and Indians with type 2 diabetes. Cardiovasc Diabetol 13: 129.
- Mahalle NV, Kulkarni MK, Garg SS, Naik (2013) Vitamin B12 deficiency and hyperhomocysteinemia as correlates of cardiovascular risk factors in Indian subjects with coronary artery disease. J Cardiol 61: 289-294.
- Adaikalakoteswari AS, Finer PD, Voyias CM, McCarthy M, Vatish J, et al. (2015) Vitamin B12 insufficiency induces cholesterol biosynthesis by limiting s-adenosylmethionine and modulating the methylation of SREBF1 and LDLR genes. Clin Epigenetics 7: 14.
- Oltean S, Banerjee (2003) Nutritional modulation of gene expression and homocysteine utilization by vitamin B12. J Biol Chem 278: 20778-20784.
- Zhou GR, Myers Y, Li Y, Chen X, Chen J, (2001) Role of AMP-activated protein kinase in mechanism of metformin action. J Clin Invest 108: 1167-1174.
- Adaikalakoteswari AM, Vatish A, Lawson C, Wood K, Sivakumar PG, et al. (2015) Low maternal vitamin B12 status is associated with lower cord blood HDL cholesterol in white caucasians living in the UK. Nutrients 7: 2401-2414.
- Kumar KA, Lalitha D, Pavithra IJ, Padmavathi M, Ganeshan KR, et al. (2013) Maternal dietary folate and/or vitamin B12 restrictions alter body composition (adiposity) and lipid metabolism in Wistar rat offspring. J Nutr Biochem 24: 25-31.
- Moore JH, Doran (1962) Lipid metabolism in the normal and vitamin B12-deficient chick embryo. Biochem J 84: 506-513.
- Armstead EE (1971) The incorporation of acetate-2-14C and mevalonate-2-14C into cholesterol during vitamin B12 deficiency. Proc Soc Exp Biol Med 136: 911-915.
- Icayan EE, Chow BF (1962) Effect of vitamin B12 deficiency on cholesterol metabolism. J Nutr 78: 109-114.
- Lee YM, Uhm ES, Lee J, Kwon SH, Park, et al. (2008) AM251 suppresses the viability of HepG2 cells through the AMPK (AMP-activated protein kinase)-JNK (c-Jun N-terminal kinase)-ATF3 (activating transcription factor 3) pathway. Biochem Biophys Res Commun 370: 641-645.
- Khaire AR, Rathod A, Joshi (2015) Vitamin B12 and omega-3 fatty acids together regulate lipid metabolism in Wistar rats. Prostaglandins Leukot Essent Fatty Acids 99: 7-17.
- Dwarkanath PP, Barzilay T, Thomas A, Thomas S, Bhat AV, et al. (2013) High folate and low vitamin B-12 intakes during pregnancy are associated with small-for-gestational age infants in South Indian women: a prospective observational cohort study. Am J Clin Nutr 98: 1450-1458.
- Hogeveen M, Blom, den Heijer (2012) Maternal homocysteine and small-for-gestational-age offspring: Systematic review and meta-analysis. Am J Clin Nutr 95: 130-136.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi