Research Article, Endocrinol Diabetes Res Vol: 3 Issue: 2
Embryo Implantation and Inhibin Dimers (Activin) as Diagnosis of Ectopic Pregnancy
Adil Omar Bahathiq*
Department of Physiology, Faculty of Medicine, Um-Alqura University, Saudi Arabia
*Corresponding Author : Adil Omar Bahathiq
Department of Physiology, Faculty of Medicine, Um-Alqura University, Saudi Arabia
Tel: +966 12 550 1000
E-mail: drbahathiq@hotmail.com
Received: March 29, 2017 Accepted: May 22, 2017 Published: May 29, 2017
Citation: Bahathiq AO (2017) Embryo Implantation and Inhibin Dimers (Activin) as Diagnosis of Ectopic Pregnancy . Endocrinol Diabetes Res 3:2. doi: 10.4172/2470-7570.1000120
Abstract
Embryo implantation is necessary for the successful formation of pregnancy. Ectopic implantation external the uterine cavity and the growth of ectopic pregnancy (EP) is a major cause of maternal morbidity and occasionally mortality during the first trimester. EP may be encouraged by failure of tubal transport and/or increased tubal receptivity. Activin A and related proteins (inhibins, follistatin [FS], follistatin-related gene [FLRG], endometrial bleeding associated factors [ebaf]) are involved in the complex mechanisms letting the formation and the upkeep of pregnancy. Pathological expression of activins and their binding protein, follistatin, was observed in tissue and serum samples collected from EP. Numerous studies with different designs studied the diagnostic value of a single measurement of serum activin-A in the differentiation between normal intrauterine and failing early pregnancy and the results are controversial. Nevertheless, the diagnostic value of activins in EP, including the other activin isoforms (activin-B and -AB) and follistatin, merits further research. The local derangement of activin A pathway in some pregnancy disorders (incomplete and complete miscarriages, recurrent abortion, and ectopic pregnancy [EP]) further sustains the hypothesis that activin A and its related proteins play a relevant role in the formation of pregnancy. This review appraises the data to date researching the role of activins or inhibin dimers in the formation of normal pregnancy and, pathogenesis and diagnosis of tubal EP.
Keywords: Activin; Inhibin dimers; Uterine tube; Endometrium; Implantation; Ectopic pregnancy; Early diagnosis
Introduction
The Fallopian tube plays a vital role in successful human reproduction. It transports the embryo to the uterus for Implantation and helps pre-implantation improvement of the embryo with the aid of supplying nourishment and mediating maternal-embryo signals Tubal function is controlled by local interactions among exclusive cellular types, mediated by using paracrine mechanisms. While present in the Fallopian tube. Human endometrium is a tremendously specialized tissue, offering a finest environment for implantation of the semiallogeneic embryo. Following the estrogenbrough on proliferation, progesterone involves the cyclic adenosine monophosphate (cAMP) signaling pathway and induces differentiate activities within all compartments of the endometrium, producing a receptive atmosphere for blastocyst attachment and invasion. The complex process of implantation and trophoblast invasion is crucial for the successful formation of pregnancy [1-3]. The embryo generally implants in the endometrium during the implantation window and uterine receptivity for implantation is maximal during this period [3]. Endometrial receptivity includes the expression of molecular factors that govern the feto-maternal dialogue and initiate decasualization of the endometrial stroma [1,2]. Candidate molecules, which include cytokines and growth factors, are secreted via endometrial and tubal epithelial cells and had been described as most important regulators of blastocyst implantation [4]. Ectopic Pregnancy (EP) is a top notch reason of maternal bleakness and is in charge of pregnancy associated passing inside the principal trimester 1. Diagnosing an EP is a mission to the clinician in light of the fact that there is no complete nonsurgical analytic test while the determination is dubious through routine blood checks and ultrasound, and finding frequently requires following up patients over various visits [5]. Ectopic pregnancy (EP) is a shape of strange pregnancy in which the prepared ovum embeds outside the intra-uterine hole and the ampullary area of the fallopian tube is the most well-known site of implantation [6]. EP speaks to 1-2% of all pregnancies in created nations and discharge from an EP due to tubal break remains the most widely recognized reason for maternal mortality in the main trimester of pregnancy in those nations [7-9]. Preparatory information is thought about the reasons why fetuses in a few pregnancies embed inside the fallopian tube while most embed eutopically inside the endometrium of the uterus [4,10]. Ectopic implantation has for quite some time been credited to disappointment of the tubal transport component. Either/or the musical smooth muscle constriction and ciliary beat action are accepted to flop in this way prompting an EP. An option clarification for EP would be an expansion in tubal epithelial receptivity because of postponement in the tubal transport and debilitated endometrial receptivity with ectopic implantation happening following disappointment of the ordinary organic associations between endometrium, fallopian tube and developing life [4,10]. Presently, the main biomarker utilized routinely in clinical practice is human chorionic gonadotropin (hCG), however it isn’t symptomatic and can just help conclusion in blend with ultrasound use with hCG observing being utilized to take after patients until finish determination of the EP [7,8,11,12]. EP can be dealt with surgically by laparotomy or laparoscopy, restoratively by methotrexate (MTX) infusion and incidentally by perception alone [8]. MTX is the most financially savvy and has comparable impact as laparoscopic administration on planned fruitfulness and repeat of EP. Be that as it may, it requires early recognition of EP as it is just reasonable in haemodynamically steady patients, with insignificant or no manifestations, starting serum hCG ≤ 3000 IU/L, EP estimate <4 cm and nonattendance of fetal heart movement [13-15]. It is trusted that just about half of EP cases are at first misdiagnosed in spite of the utilization of high-determination transvaginal sonography and touchy measures for β-hCG [7,8,16,17]. Clinicians should likewise catch up patients more than a few days to seven days, which builds the danger of tubal break what’s more, perilous discharge. The postponement in the conclusion lead the need to grow new markers and calculations that give a more delicate and particular device for the determination of EP [11,18,19]. Activins furthermore, their coupling protein, follistatin, are copiously communicated in the female reproductive tract and they have recently been proposed as potential sensitive and specific markers for the diagnosis of ectopic pregnancy by several research groups [20-22]. The current review summarizes the physiological actions of activins and follistatin in the endometrium, fallopian tube, embryo implantation and their clinical value in the diagnosis of normal and abnormal early pregnancies.
Structure and Function
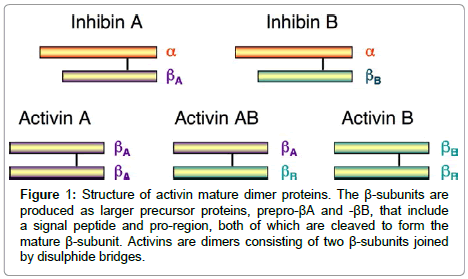
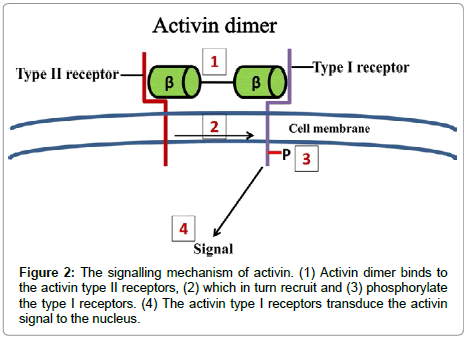
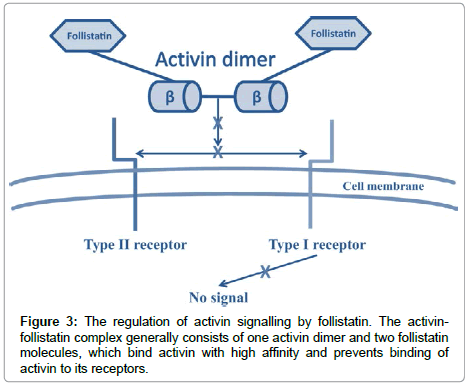
Inhibin and Activin-Are dimeric glycoproteins that are characterized by their activities on pituitary gonadotrophic cells as either restraining (inhibins) or invigorating (activins) the generation of FSH. They act through serine/threonine kinase trans membrane receptors, coming about in the phosphorylation of intracellular middle people, S-MAD (proteins identified with the Drosophila quality moms against deca-penta-plegic) (1). The neighbourhood bioactivity of activin is firmly controlled by the arrange blend of its coupling protein, follistatin, which ties and kills activin with high partiality, by averting communication with the sort II receptors (2). Follistatin (2) is perceived to be a vital controller of cell separation and emission through it is powerful capacity to tie activins. The creation of follistatin is, thus, managed by activin in the pituitary, gonads, and somewhere else. Activins were later observed to be emitted by all organs of the female regenerative framework where they work as paracrine and autocrine elements to control a assortment of regenerative capacities [21,23,24]. Activins are homodimers or, on the other hand heterodimers of two β-subunits (βA and βB), and the unique dimerization of subunits by a disulphide bond offers ascend to three develop proteins named activin-A (βA-βA), activin-B (βB- βB) and activin-AB (βA-βB) (Figure 1) [23]. The human activin βA-subunit what’s more, βB-subunit qualities are situated on chromosome7 locus7p14-p15 what’s more, chromosome 2qcen-q13, separately [25]. The mRNA of both subunits encodes a pre-proportion, their develop districts show around 70% grouping homology and them two need perceived glycosylation destinations [26]. Activins start their organic reactions by restricting particular receptors on the surface of target cells. The Activins dimer helpfully ties two sort I and II serine/threonine kinase receptors, coming about in a ternary receptor complex [23]. Sharing the normal b-subunit, the inhibins can vie for the Activins receptors, and go about as extracellular controllers of Activins signalling. Extra extracellular control is given by the group of follistatin and follistatin like proteins [27]. Activins signalling begins with the official and initiation of a serine/threonine kinase sort II receptor, ActRIIA or, on the other hand ActRIIB and can tie their individual receptor sort II when communicated alone, yet neglect to tie to sort I receptor in the nonattendance of sort II receptor [27]. Be that as it may, both sort I and II receptors are important to produce a high-proclivity complex with the ligand, as well concerning signalling (Figure 2) [28]. The planned union of follistatin with activin is the fundamental controller of the neighbourhood bioactivity of activin since official of activin to follistatin is practically irreversible [23]. The Activins-follistatin complex for the most part comprises of one activin dimer and two follistatin particles [29]. When all is said in done, activin-A,-Abdominal muscle, and - B tie to follistatin with comparative partiality (Figure 3) [30]. Both activin subunits, activin sort II receptors and follistatin were beforehand restricted inside the human fallopian tube [23,31], endometrium [32-34] and placental [35] tissues proposing a part for these proteins in the direction the physiological capacities of these tissues. Along these lines these proteins have been proposed as potential delicate and particular markers to screen the advance what’s more, result of pregnancy [36,37].
Figure 1: Structure of activin mature dimer proteins. The β-subunits are produced as larger precursor proteins, prepro-βA and -βB, that include a signal peptide and pro-region, both of which are cleaved to form the mature β-subunit. Activins are dimers consisting of two β-subunits joined by disulphide bridges.
Roles of activins in normal early pregnancy events
Activins are potential factors for maternal-embryo interactions, dueto their roles in regulating cell proliferation, differentiation and apoptosis,and their abundant expression and actions in remodeling tissues,embryogenesis and organogenesis in a variety of species [34,38].
Activins in tubal physiology
The uterine tube is the site of fertilization and early embryogenesis,what’s more, it appears to be likely that the internal epithelial covering of the uterine tube (the endosalpinx) is associated with the science of gametes and early developing life, giving emissions into the tubal lumen that frame the microenvironment of the gametes at the season of treatment [23,38]. The prepared ovum goes through the fallopian tube under the control of tubal ciliary beat recurrence and tubal smooth muscle withdrawal to reach to the intrauterine depression for implantation inside 48-72 h after preparation [39]. Impeded tubal transportation and secretory capacities can happen after outside or inward irritation, prompting tubal harm, ectopic pregnancy and barrenness [40]. Early reports on the declaration of activin subunits in the fallopian tube were produced from various creature species. Initially, just βA-subunit articulation was recognized at the protein and quality levels in rodent [41] and cow-like [42] tubal tissues. Follistatin articulation was excluded in either think about [41,42]. Reliably, the outflow of activins and follistatin by the human endosalpinx in pre-and postmenopausal ladies has been shown [23,31]. In all tissues considered, the βA-and βB-subunits were expressed with their sort II receptors and restricting protein, recommending that the activin dimer has a neighbourhood paracrine or autocrine part inside the tube [23,31]. Another investigation has later announced that the declaration of both activin subunits mRNA and protein differ in a cycle-subordinate way in the oviduct of nonpregnant cycling mice [34]. The overwhelming wellspring of activin β-subunits amid the oestrous cycle and pre-implantation stage were the epithelial cells covering the oviduct and endometrium [34]. A comparable example of articulation by the tubal epithelium amid the menstrual cycle has likewise been accounted for in human fallopian tube and the outflow of activin βA-and βB-subunits, activin sort II receptors, and follistatin was most astounding in the luteal stage at the quality and protein levels. These come about recommend that activins and their related particles have a part in tubal physiology and early embryonic advancement [38]. The mouse what’s more, human oocyte is equipped for accepting an activin signal created by encompassing cumulus cells, yet not of transmitting one [43]. This is additionally upheld by the discoveries that early mouse and human developing lives, from the four-cell stage to the morula organize, can’t incorporate activins as no mRNA for these proteins has been distinguished at these phases of improvement [41,44]. In any case, the statement of these particles expanded detectably at the blastocyst organize, which may proposed that the statement of the βA-subunit, activin sort I receptor and follistatin is reliant on embryonic formative stage and initiation of the embryonic genome in mouse and human [44]. Subsequently it has been hypothesized that activins, which tie to the activin sort I and sort II receptors on the oocyte and preimplantation developing life, might be gotten from the encompassing tissue, for example, the tubal epithelium [34,44]. Right now, there is no proof about a part for activins in the direction of tubal ciliary beat recurrence and smooth muscle withdrawal [6,38]. Be that as it may, a few examinations have demonstrated that Activin-A actuates the generation of nitric oxide, a powerful inhibitor of tubal peristalsis, in a focus subordinate way in an assortment of tissues and cells [45-47]. Moreover, it has appeared that activin-A diminished the outflow of oxytocin and HoxA-10 mRNA by human myometrial cells in vitro and it diminished oxytocin also, thromboxaneA2 instigated amassing of intracellular Ca+2 [48]. In this way, activin-A could relaxingly affect human fallopian tube smooth muscle by fortifying the generation of nitric oxide as well as by decreasing accumulation of intracellular Ca+2 [6].
Activins in endometrial physiology
The endometrium performs morphological and secretory changes amid the menstrual cycle to shape a responsive soil for the arriving blastocyst in the late luteal stage [1,2,4]. These changes incorporate the presence of pinopodes in the epithelial layer, decidualization of the endometrial stroma and vascular multiplication, which are total prerequisites for effective implantation and development of interchanges with the fetus [1,2,4]. These alterations happen under the direction of ovarian hormones to give a microenvironment rich in cell bond particles, cytokines, chemokine’s and development factors [1,4,49,50]. Activins have been depicted as essential controllers of decidualisation what’s more, endometrial receptivity following the recognizable proof of their articulation in the endometrium of a few animal groups at the quality and protein levels. Activin subunits are communicated in the endometrium luminal and glandular epithelium of a few animal groups including human [21,34,51,52]. The coarticulation of activin subunits, receptors and restricting protein shows that endometrial epithelial and stromal cells are equipped for producing and reacting to activin, and that there is a tight neighborhood direction of Activin-Activities inside the endometrium [53]. The declaration of Activin-A its related particles shifts amid the menstrual cycle in the human endometrium as the uterus redesigns and separates to shape the decidua [54,55]. Activin βAand βBsubunits mRNA and protein are communicated by glandular and surface epithelium in non-pregnant endometrium [55] and, dimeric activin-A is available in uterine liquid [53] and menstrual blood [56] of cycling ladies. The outflow of activin subunits is confined in the cytoplasm of endometrial epithelium and it essentially increments in the luminal and glandular epithelial cells amid the secretory stage also, remains moderately steady finished whatever remains of the cycle [53,55]. Moreover, activin-A deliberate in endometrial washes gathered from cycling ladies connected essentially with menstrual cycle days and the thickness of endometrium [53]. A comparable example of articulation has additionally been seen in the stromal cells amid the cycle what’s more, the articulation was most grounded in the late secretory stage where decidualisation and fetus implantation happen [54,55]. Further prove for the up-control of activin subunits blend with decidualisation was gotten from thinks about where the endometrium was widely decasualized by intrauterine conveyance of progestin [57] and in quality exhibit contemplates inspecting decidualisation-related qualities [58]. Recently decasualized cells are the primary wellspring of maternally inferred Activin-A amid pregnancy to encourage the spread of decidualisation all through the endometrium by advancing the decidualisation of neighboring cells [32-34,57,59]. Amid pregnancy, the placenta is a noteworthy wellspring of serum activin-A, which increments as the pregnancy advances [21,24,60,61]. Be that as it may, the articulation of activins by the cytotrophoblast is low amid early pregnancy proposing that trophoblast intrusion is actuated by maternally determined activins [32]. Activin-A directs trophoblastic cell glue properties by adjusting the statement of E-and N-cadherin [62,63] what’s more, integrins [35]. Activin-A likewise advances attack of first-trimester cytotrophoblasts until 10 weeks growth by expanding the articulation of grid metalloproteinases-2, 7 and 9 and its activities are hindered by follistatin [33].
Activins in the pathogenesis and finding of EP
Concentrates on the articulation example of activins, their sort II receptors what’s more, follistatin by fallopian tubes bearing an ectopic pregnancy have demonstrated a critical increment in βA-subunit, sort IIA and IIB receptors what’s more, follistatin contrasted with ordinary control [6,10]. Lucidly, an increment in the statement of those particles was additionally seen in tubal examples gathered from patients with EP and who were certain for Chlamydia trachomatis antibodies when contrasted with tubal tests gathered from EP patients and who had negative response for the antibodies [5]. In any case, a current report did not identify noteworthy distinction in the articulation example of these atoms amongst implantation and remote destinations gathered from chronicled tubal pregnancy examples. Henceforth, it has been proposed that the pathologic articulation of activins and their related atoms by the tubal epithelium may assume a critical part in the pathogenesis of EP however, not in the assurance of implantation site [10]. Serum levels of activin-A and follistatin increment fundamentally all through pregnancy what’s more, a few examinations have proposed a feto placental source for these proteins. Serum levels of activin-A additionally diminish within the sight of nonviable trophoblast [21,64,65]. Thus, activin-A has been examined as a potential marker for the conclusion and separation between ordinary intrauterine pregnancy (IUP), unsuccessful labor and ectopic pregnancy [37]. In any case, the revealed comes about the symptomatic estimation of Activin-A are as yet far from being obviously true. The underlying examination by Florio [36] showed that a solitary estimation of serum activin-A gave a very delicate and particular marker in 536 patients with pregnancy of obscure area (PUL) to separate between practical ordinary IUP, unnatural birth cycle and EP with an affectability of 100% and specificity of 99.6% at a cut-off estimation of 370 pg/mL. Afterward, a similar research group has revealed that serum centralizations of activin-A were fundamentally lower in 30 patients determined to have tubal EP when contrasted with control and, an affectability of 96.7% and a specificity of 100% for the finding of EP were accomplished at the cutoff level of 0.43 ng/mL [66]. These outcomes have additionally been affirmed by other research bunches in the next years yet with an alternate cut-off estimation of 504 pg/mL that accomplished an affectability of 97% and specificity of 93.5% [22,67-70]. Moreover, Daponte [67] too measured the affectability and specificity of follistatin in the analysis of EP. Their outcomes have demonstrated that both serum follistatin and activin-A/follistatin proportion had bring down execution contrasted with serum Activin-A in the finding of EP. Oppositely, another two examinations revealed that a solitary estimation of serum activin-A was not one or the other touchy nor particular for the conclusion of early pregnancy disappointment counting EP contrasted with β-hCG [71,72]. Their outcomes illustrated that serum activin-A levels gave a region under the bend (AUC) of 0.61 for falling flat PUL, 0.64 for IUP also, 0.51 for EP, and the model in light of serum hCG levels gave an AUC of 0.95 for fizzling PUL, 0.97 for IUP and 0.67 for EP [71]. The irregularities in the revealed cut-off qualities and the aftereffects of the diverse examinations could be because of changeability in trophoblastic exercises also, insufficient decidulisation amid ectopic implantation [67]. It has additionally been proposed that some trophoblastic cells may carry on as fizzling pregnancy amid EP while different EPs could have greater action furthermore, carry on more like IUPs [71], which could give a conceivable clarification for the detailed varieties in serum levels of activin-A in ladies with EP [67]. Another clarification for the previously mentioned varieties in the consequences of activin-A could be identified with the gestational age of the selected members in the distinctive examinations. The expansion in serum Activin-A and follistatin amid typical pregnancy begins at week 6 and achieves its top at week 38 of pregnancy. The first consider by Florio [36] included patients in the principal trimester yet the creators have not revealed the gestational age for control.
EP and miscarriage groups
In the accompanying investigation by Rausch [22] the mean gestational age for EP and IUP was 45.15 ± 18.95 and 48.8 ± 18.34 days, separately [22]. The latest examinations that have detailed a demonstrative esteem for serum activin-A included just patients between 6 to 8 [67] and 5 to 6 weeks gestational age [70]. On the other hand, the two examinations that demonstrated no an incentive for serum Activin-A in the finding of EP the gestational age for each gathering was not revealed by Kirk [71] and Warrick [72] connected a review plan that included pregnant females with ≤10 weeks development. The ectopic pregnancy bunch in ponder revealed by Warrick [72] had a middle of 5 weeks (range1– 10 weeks) incubation contrasted with 7 weeks (run 1-10 weeks) for both IUP and unnatural birth cycle. Along these lines, future examinations should classifications the patients with bombing early pregnancy as indicated by gestational age (e.g. ≤ and >6 weeks) and to contrast the outcomes and those acquired from gestational matured coordinated controls. The natural exercises of activins are firmly directed by follistatin as the authoritative of activin to follistatin is practically irreversible [23]. Serum activin is usually bound with the long shape follistatin (FS-315) [29], while the short frame (FS-288) has high proclivity for cell layer activins [73]. The as of now accessible ELISA packs for the identification of activin-A what’s more, follistatin can’t recognize the free and bound types of the two proteins. Moreover, the follistatin pack measures both the long and short structures. In this way, the detailed outcomes from the diverse examinations are appeared at the level of aggregate activin-A furthermore, follistatin and the improvement of ELISA units that measure the free type of the two proteins would uncover definitely their qualities in the determination of early pregnancy disappointment. Furthermore, the larger part of concentrates that exhibited the confinement of activins in the endometrium and placental tissues researched the articulation example of the βA-and βB-subunits and not the develop dimeric proteins. Subsequently, it could be hypothesized that the endometrium and trophoblast are link of incorporating the distinctive activin develop dimers (activin-A, - B and - AB). In addition, produced comes about because of quality thump out examinations have demonstrated that each activin subunit has unmistakable capacities and these subunits don’t practically cover in all settings in vivo [74,75]. Subsequently, the consideration of the other activin develop dimeric proteins (e.g. activin-B and - AB) could add to the execution of serum Activin-A in the determination of strange early pregnancy.
Conclusions
Ectopic pregnancy is a threaten health problem and it is the leading cause of maternal morbidity and/or mortality during the first trimester. Early diagnosis of EP would allow more conservative treatment approaches and preservation of prospective fertility. However, the currently used diagnostic modalities need to be improved and new biomarkers need to be found. Activins and their related proteins play an important role in the regulation of endometrial receptivity, trophoblast activity and embryo implantation. Pathological expression of these candidate proteins has been associated with abnormal implantation and failing early pregnancy at the tissue and serum levels. Although, the diagnostic value of a single measurement of serum activin-A in differentiating abnormal from normal pregnancy is controversial, it merits further research because this biomarker feels to be promising.
Future studies should classify the patients according to the gestational age and to include other activin mature dimer proteins. It could also be worthy to combine serum activins with other serum biomarkers (e.g. inhibins, vascular endothelial growth factor) for early and accurate diagnosis of EP. Additionally, the development of ELISA kits that measure the free form of activins could increase their diagnostic performances. Further studies are needed to develop biomarkers and diagnostic modalities for the early diagnosis of EP and prevention of its associated maternal complications.
Competing Interests
The author declares that he has no competing interests.
References
- Singh M, Chaudhry P, Asselin E (2011) Bridging endometrial receptivity and implantation: network of hormones, cytokines, and growth factors. J Endocrinol 210: 5-14.
- Dimitriadis E, White CA, Jones RL, Salamonsen LA (2005) Cytokines, chemokines and growth factors in endometrium related to implantation. Hum Reprod Update 11: 613-630.
- Coughlan C, Ledger W, Wang Q, Liu F, Demirol A, et al. (2014) Recurrent implantation failure: definition and management. Reprod Biomed Online 28: 14-38.
- Jia-Rong Z, Shuang-Di L, Xiao-Ping W (2009) Eutopic or ectopic pregnancy: a competition between signals derived from the endometrium and the fallopian tube for blastocyst implantation. Placenta 30: 835-839.
- Barnhart KT (2009) Clinical practice Ectopic pregnancy. N Engl J Med 361: 379-387.
- Refaat B, Amer S, Ola B, Chapman N, Ledger W (2008) The expression of activinbetaAand -betaB-subunits, follistatin, and activin type II receptors in fallopian tubes bearing an ectopic pregnancy. J Clin Endocrinol Metab 93: 293-299.
- Jurkovic D, Wilkinson H (2011) Diagnosis and management of ectopic pregnancy. BMJ 342: 3397.
- Sivalingam VN, Duncan WC, Kirk E, Shephard LA, Horne AW (2011) Diagnosis and management of ectopic pregnancy. J Fam Plann Reprod Health Care 37: 231-240.
- Khan KS, Wojdyla D, Say L, Gülmezoglu AM, Van Look PF (2006) WHO analysis of causes of maternal death: a systematic review. Lancet 367: 1066-1074.
- Refaat B, Simpson H, Britton E, Biswas J, Wells M, et al. (2012) Why does the fallopian tube fail in ectopic pregnancy? The role of activins, inducible nitric oxide synthase, and MUC1 in ectopic implantation. Fertil Steril 97: 1115-1123.
- Kao LY, Scheinfeld MH, Chernyak V, Rozenblit AM, Oh S, et al. (2014) Beyond ultrasound: CT and MRI of ectopic pregnancy. AJR Am J Roentgenol 202: 904-911.
- Kirk E, Bottomley C, Bourne T (2014) Diagnosing ectopic pregnancy and current concepts in the management of pregnancy of unknown location. Hum Reprod Update 20: 250-261.
- Hajenius PJ, Mol F, Mol BW, Bossuyt PM, Ankum WM, et al. (2007) Interventions for tubal ectopic pregnancy. Cochrane Database Syst Rev: CD000324
- Lipscomb GH (2007) Medical therapy for ectopic pregnancy. Semin Reprod Med 25: 93-98.
- Bachman EA, Barnhart K (2012) Medical management of ectopic pregnancy: a comparison of regimens. Clin Obstet Gynecol 55: 440-447.
- Dillon KE, Sioulas VD, Sammel MD, Chung K, Takacs P, et al. (2012) How and when human chorionic gonadotropin curves in women with an ectopic pregnancy mimic other outcomes: differences by race and ethnicity. Fertil Steril 98: 911-916.
- Morse CB, Sammel MD, Shaunik A, Allen-Taylor L, Oberfoell NL, et al. (2012) Performance of human chorionic gonadotropin curves in women at risk for ectopic pregnancy: exceptions to the rules. Fertil Steril 97: 101-106. e2.
- Cartwright J, Duncan WC, Critchley HO, Horne AW (2009) Serum biomarkers of tubal ectopic pregnancy: current candidates and future possibilities. Reproduction 138: 9-22.
- Cabar FR, Fettback PB, Pereira PP, Zugaib M (2008) Serum markers in the diagnosis of tubal pregnancy. Clinics (Sao Paulo) 63: 701-708.
- Barnhart K, Speicher DW (2011) Molecular diagnosis of ectopic pregnancy. Expert Rev Mol Diagn 11: 759-762.
- Florio P, Luisi S, Ciarmela P, Severi FM, Bocchi C, et al. (2004) Inhibins and activins in pregnancy. Mol Cell Endocrinol 225: 93-100.
- Rausch ME, Sammel MD, Takacs P, Chung K, Shaunik A, et al. (2011) Development of a multiple marker test for ectopic pregnancy. Obstet Gynecol 117: 573-582.
- Refaat BA, Bahathiq AO, Sockanathan S, Stewart RL, Wells M, et al. (2004) Production and localization of activins and activin type IIA and IIB receptors by the human endosalpinx. Reproduction 128: 249-255.
- Schneider-Kolsky M, D'Antona D, Evans LW, Taylor N, O'Connor A, et al. (2000) Maternal serum total activin A and follistatin in pregnancy and parturition. BJOG 107: 995-1000.
- Barton DE, Yang-Feng TL, Mason AJ, Seeburg PH, Francke U (1989) Mapping of genes for inhibin subunits alpha, beta A, and beta B on human and mouse chromosomes and studies of jsd mice. Genomics 5: 91-99.
- Mason AJ, Hayflick JS, Ling N, Esch F, Ueno N, et al. (1985) Complementary DNA sequences of ovarian follicular fluid inhibin show precursor structure and homology with transforming growth factor-beta. Nature 318: 659-663.
- Ebner R, Chen RH, Lawler S, Zioncheck T, Derynck R (1993) Determination of type I receptor specificity by the type II receptors for TGF-beta or activin. Science 262: 900-902.
- Wrana JL, Tran H, Attisano L, Arora K, Childs SR, et al. (1994) Two distinct transmembrane serine/threonine kinases from Drosophila melanogaster form an activin receptor complex. Mol Cell Biol: 944-950.
- Schneyer AL, Hall HA, Lambert-Messerlian G, Wang QF, Sluss P, et al. (1996) Follistatin-activin complexes in human serum and follicular fluid differ immunologically and biochemically. Endocrinology 137: 240-247.
- Thompson TB, Lerch TF, Cook RW, Woodruff TK, Jardetzky TS (2005) The structure of the follistatin:activin complex reveals antagonism of both type I and type II receptor binding. Dev Cell 9: 535-543.
- Bahathiq AO, Stewart RL, Wells M, Moore HD, Pacey AA, et al. (2002) Production of activins by the human endosalpinx. J Clin Endocrinol Metab 87: 5283-5289.
- Jones RL, Findlay JK, Farnworth PG, Robertson DM, Wallace E, et al. (2006) Activin A and inhibin A differentially regulate human uterine matrix metalloproteinases: potential interactions during decidualization and trophoblast invasion. Endocrinology 147: 724-732.
- Jones RL, Findlay JK, Salamonsen LA (2006) The role of activins during decidualisation of human endometrium. Aust N Z J Obstet Gynaecol 46: 245-249.
- Jones RL, Kaitu'u-Lino TJ, Nie G, Sanchez-Partida LG, Findlay JK, et al. (2006) Complex expression patterns support potential roles for maternally derived activins in the establishment of pregnancy in mouse. Reproduction 132: 799-810.
- Stoikos CJ, Salamonsen LA, Hannan NJ, O’Connor AE, Rombauts L, et al. (2010) Activin A regulates trophoblast cell adhesive properties: implications for implantation failure in women with endometriosis associated infertility. Hum Reprod 25: 1767-1774.
- Florio P, Severi FM, Bocchi C, Luisi S, Mazzini M, et al. (2007) Single serum activin a testing to predict ectopic pregnancy. J Clin Endocrinol Metab 92: 1748-1753.
- Prakash A, Laird S, Tuckerman E, Li TC, Ledger WL (2005) Inhibin A and activin A may be used to predict pregnancy outcome in women with recurrent miscarriage. Fertil Steril 83: 1758-1763.
- Refaat B, Ledger W (2011) The expression of activins, their type II receptors and follistatin in human Fallopian tube during the menstrual cycle and in pseudo-pregnancy. Hum Reprod 26: 3346-3354.
- Lyons RA, Saridogan E, Djahanbakhch O (2006) The effect of ovarian follicular fluid and peritoneal fluid on Fallopian tube ciliary beat frequency. Hum Reprod 21: 52-56.
- Kriebs JM, Fahey JO (2006) Ectopic pregnancy. J Midwifery Womens Health 51: 431-439.
- Lu RZ, Matsuyama S, Nishihara M, Takahashi M (1993) Developmental expression of activin/inhibin beta A, beta B, and alpha subunits, and activin receptor-IIB genes in preimplantation mouse embryos. Biol Reprod 49: 1163-1169.
- Gandolfi F, Modina S, Brevini TA, Passoni L, Artini P, et al. (1995) Activin beta A subunit is expressed in bovine oviduct. Mol Reprod Dev 40: 286-291.
- Sidis Y, Fujiwara T, Leykin L, Isaacson K, Toth T, et al. (1998) Characterization of inhibin/activin subunit, activin receptor, and follistatin messenger ribonucleic acid in human and mouse oocytes: evidence for activin’s paracrine signaling from granulosa cells to oocytes. Biol Reprod 59: 807-812.
- He ZY, Liu HC, Mele CA, Barmat L, Veeck LL, et al. (1999) Expression of inhibin/activin subunits and their receptors and binding proteins in human preimplantation embryos. J Assist Reprod Genet 16: 73-80.
- Nusing RM, Barsig J (1997) Inflammatory potency of activin A. Effect on prostanoid and nitric oxide formation. Adv Exp Med Biol 407: 243-248
- Lagadec P, Raynal S, Lieubeau B, Onier N, Arnould L, et al. (1999) Evidence for control of nitric oxide synthesis by intracellular transforming growth factor-beta1 in tumor cells. Implications for tumor development. Am J Pathol 154: 1867-1876.
- Nusing RM, Barsig J (1999) Induction of prostanoid, nitric oxide, and cytokine formation in rat bone marrow derived macrophages by activin A. Br J Pharmacol 127: 919-926.
- Ciarmela P, Wiater E, Vale W (2008) Activin-A in myometrium: characterization of the actions on myometrial cells. Endocrinology 149: 2506-2516.
- Horne AW, Critchley HO (2012) Mechanisms of disease: the endocrinology of ectopic pregnancy. Expert Rev Mol Med 14: e7.
- Shaw JL, Horne AW (2012) The paracrinology of tubal ectopic pregnancy. Mol Cell Endocrinol 358: 216-222.
- Sugawara K, Kizaki K, Herath CB, Hasegawa Y, Hashizume K (2010) Transforming growth factor beta family expression at the bovine feto-maternal interface. Reprod Biol Endocrinol 8: 120.
- Zhang H, Nagaoka K, Imakawa K, Nambo Y, Watanabe G, et al. (2013) Expression of inhibin/activin subunits in the equine uteri during the early pregnancy. Reprod Domest Anim 48: 423-428.
- Florio P, Severi FM, Luisi S, Ciarmela P, Calonaci G, et al. (2003) Endometrial expression and secretion of activin A, but not follistatin, increase in the secretory phase of the menstrual cycle. J Soc Gynecol Investig 10: 237-243.
- Mylonas I, Makovitzky J, Hoeing A, Richter DU, Vogl J, et al. (2006) Inhibin/activin subunits beta-A (-betaA) and beta-B (-betaB) are differentially localised in normal, hyperplastic and malignant human endometrial tissue. Acta Histochem 108: 1-11
- Mylonas I, Jeschke U, Wiest I, Hoeing A, Vogl J, et al. (2004)Inhibin/activin subunits alpha, beta-A and beta-B are differentially expressed in normal human endometrium throughout the menstrual cycle. Histochem Cell Biol 122: 461-471.
- Reis FM, Nascimento LL, Tsigkou A, Ferreira MC, Luisi S, et al. (2007) Activin A and follistatin in menstrual blood: low concentrations in women with dysfunctional uterine bleeding. Reprod Sci 14: 383-389.
- Jones RL, Salamonsen LA, Zhao YC, Ethier JF, Drummond AE, et al. (2002) Expression of activin receptors, follistatin and betaglycan by human endometrial stromal cells; consistent with a role for activins during decidualization. Mol Hum Reprod 8: 363-374.
- Kao LC, Tulac S, Lobo S, Imani B, Yang JP, et al. (2002) Global gene profiling in human endometrium during the window of implantation. Endocrinology 143: 2119-2138.
- Jones RL, Salamonsen LA, Findlay JK (2002) Activin A promotes human endometrial stromal cell decidualization in vitro. J Clin Endocrinol Metab 87: 4001-4004.
- Fowler PA, Evans LW, Groome NP, Templeton A, Knight PG (1998)A longitudinal study of maternal serum inhibin-A, inhibin-B, activin-A, activin-AB, pro-alphaC and follistatin during pregnancy. Hum Reprod 13: 3530-3536.
- O’Connor AE, McFarlane JR, Hayward S, Yohkaichiya T, Groome NP, et al. (1999) Serum activin A and follistatin concentrations during human pregnancy: a cross-sectional and longitudinal study. Hum Reprod 14: 827-832.
- Ferreira MC, Witz CA, Hammes LS, Kirma N, Petraglia F, et al. (2008) Activin A increases invasiveness of endometrial cells in an in vitro model of human peritoneum. Mol Hum Reprod 14: 301-307.
- Li Y, Klausen C, Cheng JC, Zhu H, Leung PC (2014) Activin A, B, and AB increase human trophoblast cell invasion by up-regulating N-cadherin. J Clin Endocrinol Metab 99: E2216-2225.
- Luisi S, Florio P, D’Antona D, Severi FM, Sanseverino F, et al. (2003) Maternal serum inhibin A levels are a marker of a viable trophoblast in incomplete and complete miscarriage. Eur J Endocrinol 148: 233-236.
- Muttukrishna S, Jauniaux E, Greenwold N, McGarrigle H, Jivraj S, et al. (2002)Circulating levels of inhibin A, activin A and follistatin in missed and recurrent miscarriages. Hum Reprod 17: 3072-3078.
- Florio P, Reis FM, Battista R, Luisi S, Moncini I, et al. (2011) Serum activin A levels are lower in tubal than intrauterine spontaneously conceived pregnancies. Gynecol Endocrinol 27: 391-395.
- Daponte A, Deligeoroglou E, Garas A, Pournaras S, Hadjichristodoulou C, et al. (2013) Activin A and follistatin as biomarkers for ectopic pregnancy and missed abortion. Dis Markers 35: 497-503.
- Rausch ME, Barnhart KT (2012) Serum biomarkers for detecting ectopic pregnancy. Clin Obstet Gynecol 55: 418-423.
- Roghaei MA, Sabet F, Mohamadi K (2012) Diagnostic accuracy of serum activin A in detection of ectopic pregnancy. J Res Med Sci 17: 378-381.
- Junior EJ, Oliveira GL Silva M, Araujo E, Camano L (2014) Serum activin A levels and tubal ectopic pregnancy. Iran J Reprod Med 12: 227-228.
- Kirk E, Papageorghiou AT, Van Calster B, Condous G, Cowans N, et al. (2009)The use of serum inhibin A and activin A levels in predicting the outcome of ‘pregnancies of unknown location’. Hum Reprod 24: 2451-2456.
- Warrick J, Gronowski A, Moffett C, Zhao Q, Bishop E, et al. (2012) Serum activin A does not predict ectopic pregnancy as a single measurement test, alone or as part of a multi-marker panel including progesterone and hCG. Clin Chim Acta 413: 707-711.
- Nakamura T, Sugino K, Titani K, Sugino H (1991) Follistatin, an activin-binding protein, associates with heparan sulfate chains of proteoglycans on follicular granulosa cells. J Biol Chem 266: 19432-19437
- Brown CW, Houston-Hawkins DE, Woodruff TK, Matzuk MM (2000) Insertion of Inhbb into the Inhba locus rescues the Inhba-null phenotype and reveals new activin functions. Nat Genet 25: 453-457.
- Brown CW, Li L, Houston-Hawkins DE, Matzuk MM (2003) Activins are critical modulators of growth and survival. Mol Endocrinol 17: 2404-2417.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi