Case Report, J Nephrol Ren Dis Vol: 1 Issue: 2
EPS in a Patient Following a Four Week Exposure to Peritoneal Dialysis
Malik S1*, Rajamani K2, Lalagianni N2, Carrington C2 and Ilham A2
1Department of Nephrology, Leicester General Hospital, Leicester, UK
2Department of Nephrology, University Hospital of Wales, Cardiff, UK
*Corresponding Author : Shafi Malik
Consultant Nephrologist and Transplant Physician, Leicester General Hospital, Leicester, UK
Tel: +441162584132
E-mail: shafi.malik@sky.com
Received: October 20, 2017 Accepted: November 01, 2017 Published: November 07, 2017
Citation: Malik S, Rajamani K, Lalagianni N, Carrington C, Ilham A (2017) EPS in a Patient Following a Four Week Exposure to Peritoneal Dialysis. J Nephrol Ren Dis 1:2.
Abstract
Encapsulated Peritoneal Sclerosis (EPS) is a rare but fatal complication in patients on long-term peritoneal dialysis (PD). We report a patient who was established on peritoneal dialysis for four weeks and subsequently developed EPS. 60 year old lady with end stage renal failure (ESRF) secondary to presumed chronic glomerulonephritis presented with 3 months history of abdominal pain, diarrhoea, nausea and 25 kg weight loss over a 3 month period. The patient approached ESRF four months preceding this presentation and was initially established on PD; however, the patient requested a modality change to haemodialysis (HD) within 4 weeks purely due to choice. Multiple CT scans of abdomen and pelvis failed to account for her symptoms. Patient later developed spontaneous bacterial peritonitis and underwent a laparotomy. Biopsies of the peritoneum showed chronic inflammation with areas of mature scarring consistent with EPS.
Keywords: Peritoneal dialysis; Encapsulating peritoneal sclerosis; Fibrosis; Bowel; End stage renal failure
Introduction
Encapsulated peritoneal sclerosis is an uncommon but serious life-threatening complication in patients on long-term PD [1]. This clinical syndrome was first described by Ghandi in 1980 [2]. EPS is characterized by a progressive, inflammatory process resulting in sheets of fibrous tissue that cover, bind and constrict the viscera, thereby compromising the motility and function of the bowel. This process results in decreased ultrafiltration and eventually leads to bowel obstruction. The possible causes include spontaneous idiopathic process in young women, peritoneovenous shunting, practolol therapy, peritoneal dialysis, chemotherapy, IV drug abusers, in carcinoid syndrome or linked to beta-blockers [3]. The consistent risk factors in PD are: duration of therapy, glucose exposure, inflammation – peritonitis, chemical exposure, chlorhexidine, genetic factors.
Case Report
A 60 year old Caucasian lady presented to a tertiary centre with complaints of being general unwell, weight loss and abdominal distension associated with loss of appetite and diarrhoea of 4 months duration. The patient was established on continuous ambulatory peritoneal dialysis (CAPD) 4 months ago as this was her modality of choice; however, the patient remained on this therapy only for 4 week duration and requested a modality change to HD due to social circumstances rather than difficulties with PD. While on CAPD the patient’s treatment consisted of 4 exchanges daily of 1.36% strength bags, with glucose content of 13.6 gm., PD fluid osmolarity 344 mmol. The lady was established on haemodialysis via a left brachio-cephalic arterio venous fistula (AVF). Other past medical history included pulmonary embolism, Chronic Obstructive Pulmonary Disorder controlled on inhalers and diet controlled type 2 diabetes mellitus. On examination, the patient was severely cachectic, abdomen was distended; fluid thrill was present. Examination of cardiovascular and respiratory systems was normal.
Patient was investigated during this admission for her complaints. Computed Tomography (CT) colonography was reported as normal and an ultrasound of the abdomen confirmed ascites with normal appearances of liver, spleen and normal portal blood flow. CT abdomen and pelvis with contrast showed large volume of intraabdominal fluid, increased density and stranding with omental and peritoneal fat most likely due to chronic peritoneal inflammation and no evidence of intra-abdominal malignancy. Additionally, she was found to have low serum albumin level 15 g/L(35-50 g/l)normal coeliac immunology, serum copper level was 5.8 μmol/L (11-22 μmol/L) and a normal short synacthen test (0 min:321 nmol/L, 30 min:479 nmol/L),this was performed in view of her persistent low blood pressure (80/45 mm/hg). The ascites was sampled and reported as a transudate, with normal cell count and no organisms were seen on gram stain, cultures were negative. A transthoracic echocardiogram showed left ventricular ejection fraction of > 55% with no evidence of right heart failure. A gastroenterology opinion was sought and it was felt that the ascites was due to hypoalbuminemia related to severe malnutrition. Patient was started on naso-gastric feeding and was discharged and to be followed up as outpatient.
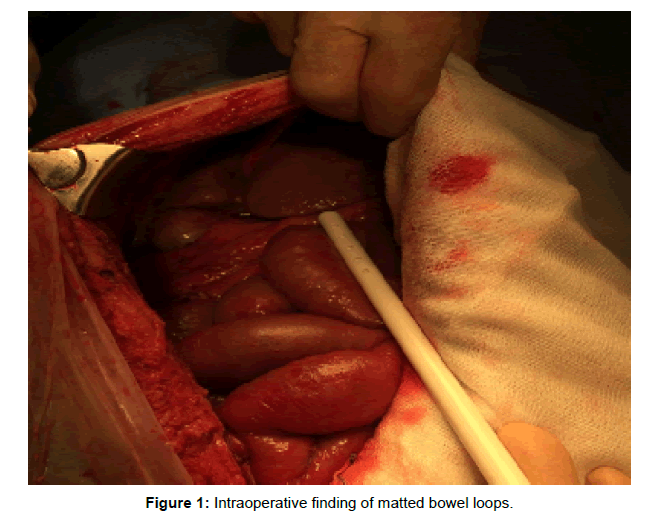
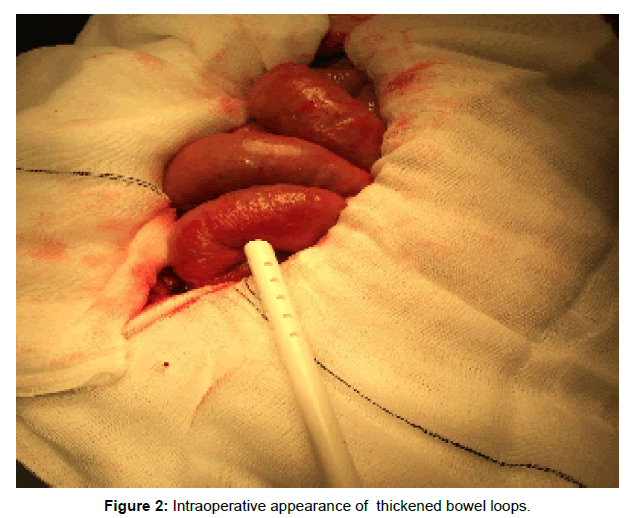
Two weeks later, she was admitted with worsening abdominal pain, raised inflammatory marker (CRP 252 mg/L, NR<5) and diagnosed with septic shock. Clinically the patient was peritonitic and an ascitic fluid tap revealed WBC >250 × 10*6 with 70% polymorphs and 30% lymphocytes, RBC 1805 × 106, gram stain was negative. She was started on once a day meropenem 1gm intravenously empirically awaiting further sensitivities, the ascitic fluid culture was later reported to have no growth. Inflammatory markers did not improve and the patient deteriorated clinically, therefore, decision was made to perform an exploratory laparotomy. This revealed thickened peritoneal membrane with tethering (Figures 1 and 2). PD catheter was removed during this surgery and omentum biopsy performed during the surgery showed:
Areas of focal haemorrhage with sections of the peritoneum show scarring with areas of granulation tissue, chronic inflammation and loose fibroblastic areas. No evidence of dysplasia or malignancy. In addition some sections showed areas of fat necrosis.
Following the laparotomy the patient was transferred to the intensive care unit and once hemodynamically stabilised she was transferred to the national referral centre for EPS decortication surgery, immediately following transfer to the national centre patient was diagnosed with fungal peritonitis and despite maximum medical care she subsequently died.
Discussion
Based on the above case, the suspicion of EPS was low as the duration of PD was only 4 weeks. The incidence of EPS reported by several observational studies show the incidence and prognosis is related to the duration of PD therapy duration. EPS incidences (and mortality rates) were 0%, 0.7% (0%), 2.1% (8.3%), 5.9% (28.6%), 5.8% (61.5%), and 17.2% (100%) in patients that had undergone PD for 3, 5, 8, 10, 15, and more than 15 years respectively [3-6]. Radiological findings played an important role in diverting the attention towards EPS in our case. CT imaging in diagnosis has been earlier described in Tarzi’s study were bowel tethering and peritoneal thickening and calcification were the most specific parameters and presence of loculations was the least specific parameter [7].
Abdominal imaging has been used to confirm the diagnosis of EPS; however until recently there was no consensus on its specific radiologic features. The exact pathogenesis for EPS is still obscure, a number of hypothesis have been proposed, one such hypothesis is the two hit theory of EPS pathogenesis -deterioration of the peritoneum as a result of the peritoneal dialysis (PD) procedure (the first “hit”) and superimposition of inflammatory stimuli such as infectious peritonitis (the second “hit”) are thought to play key roles in the pathogenesis of EPS [8].
The most characteristic feature of EPS is the formation of a peritoneal capsule by the deposition and organization of fibrin. Thus, fibrin deposition is likely to be a key event in the pathogenesis of EPS [8]. This is resulted from the exudation of plasma from the peritoneal vasculature. The exudation is chronic in the deteriorated peritoneum of long-term PD due to the vascular sclerosis involving the peritoneal vasculature (vasculopathy) [9]. EPS rarely occurs during PD treatment because the exuded fibrins are easily washed away during dialysate exchanges. However, after discontinuation of PD, the fibrin deposition results in acceleration and promotion of capsule formation around the intestine. The second important feature of EPS is progressive peritoneal fibrosis. The adhesion of the peritoneum causes serious intestinal immobility, resulting in ileus and poor absorption. This fibrosis is frequently observed in the peritonea of patients with peritoneal sclerosis and infectious peritonitis (especially during the late stage). A recent study has revealed that CCL18 ⁄ pulmonary and activation-regulated chemokine (PARC) are pro-fibrotic and stimulates collagen production independent of the effect of transforming growth factor beta but these have not been studied in peritoneal dialysis (PD) patients [10].
Until now, EPS has been known to be a fatal complication in patients who have had long-term peritoneal dialysis therapy; it should also be considered in patients with short exposure. To our knowledge, this is the first reported case to develop EPS with only a four-week exposure to PD. This case amplifies that pathogenesis is related to the content of the dialysate rather than the duration alone. Clinical suspicion with radiological findings is important for early diagnosis. The exact pathogenesis involved at the molecular level needs to be further studied.
References
- Hong KD, Bae JH, Jang YJ, Jung HY, Cho JH, et al. (2013) Encapsulating peritoneal sclerosis: case series from a university center. Korean J Intern Med 28: 587-593.
- Gandhi VC, Humayun HM, Ing TS, Daugirdas JT, Jablokow VR, et al. (1980) Sclerotic thickening of the peritoneal membrane in maintenance peritoneal dialysis patients. Arch Intern Med 140: 1201-1203.
- Akbulut S (2015) Accurate definition and management of idiopathic sclerosing encapsulating peritonitis. World J Gastroenterol 21: 675-687.
- Brown EA, Van Biesen W, Finkelstein FO, Hurst H, Johnson DW, et al. (2009) Length of time on peritoneal dialysis and encapsulating peritoneal sclerosis: position paper for ISPD. Perit Dial Int 29: 595-600.
- Brown MC, Simpson K, Kerssens JJ, Mactier RA (2009) Encapsulating peritoneal sclerosis in the new millennium: a national cohort study. Clin J Am Soc Nephrol 4: 1222-1229.
- Kawanishi H, Kawaguchi Y, Fukui H, Hara S, Imada A, et al. (2004) Encapsulating peritoneal sclerosis in Japan: a prospective, controlled, multicenter study. Am J Kidney Dis 44: 729-737.
- Tarzi RM, Lim A, Moser S, Ahmad S, George A, et al. (2008) Assessing the validity of an abdominal CT scoring system in the diagnosis of encapsulating peritoneal sclerosis. Clin J Am Soc Nephrol 3: 1702-1710.
- Schmidt DW, Flessner MF (2008) Pathogenesis and treatment of encapsulating peritoneal sclerosis: basic and translational research. Perit Dial Int 28: S10-S5.
- Honda K, Oda H (2005) Pathology of encapsulating peritoneal sclerosis. Perit Dial Int 25: S19-S29.
- Ahmad S, North BV, Qureshi A, Malik A, Bhangal G, et al. (2010) CCL18 in peritoneal dialysis patients and encapsulating peritoneal sclerosis. Eur J Clin Invest 40: 1067-1073.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi