Research Article, J Nephrol Ren Dis Vol: 1 Issue: 2
Fish Oil Supplementation Ameliorates Long Term Cisplatin Treatment Induced Toxicity and Oxidative Damage in Rat Kidney
Naqshbandi A, Farooqui Z, Khan MW, Rizwan S, Khan SA, Khan F and Yusufi ANK*
Department of Biochemistry, Aligarh Muslim University, Aligarh, UP, India
*Corresponding Author : Yusufi ANK
Department of Biochemistry, Faculty of Life Sciences, Aligarh Muslim University, Aligarh, UP, India
E-mail: yusufi@lycos.com, ayusufi@gmail.com
Received: July 28, 2017 Accepted: September 13, 2017 Published: September 20, 2017
Citation: Naqshbandi A, Farooqui Z, Khan MW, Rizwan S, Khan SA, et al. (2017) Fish oil supplementation ameliorates long term cisplatin treatment induced toxicity and oxidative damage in rat kidney. J Nephrol Ren Dis 1:2.
Abstract
Background: Cisplatin (CP) is an effective chemotherapeutic agent widely used against various malignancies. However, CP exerts several side effects through oxidative stress by generating reactive oxygen species (ROS). The major side effect of CP chemotherapy is dose dependent nephrotoxicity that limits its clinical usage. We have previously shown that fish oil (FO) supplementation ameliorated gentamicin induced nephrotoxicity as evident by increase in the activities of brush border membrane (BBM) and free radical scavenging enzymes in rat kidney. In the present study we aimed to investigate whether fish oil (FO) can prevent the long term CP-induced nephrotoxic effects.
Methods: Rats were divided into four groups viz. control, CP, CPF and FO alone group. Rats in the groups CPF and FO were fed experimental diet prior to and simultaneously with or without CP treatment (3 mg/kg bwt i.p) respectively.
Results: FO supplementation to CP treated significantly mitigated the CP induced increase in serum creatinine (Scr) and blood urea nitrogen (BUN) and decrease in the activities of brush border membrane (BBM) enzymes in renal cortical and medullary homogenates as well as in isolated BBM vesicles (BBMV). FO consumption also reduced CP induced alterations in the activities of carbohydrate metabolism enzymes and in the enzymatic and non-enzymatic antioxidant parameters.
Conclusion: These results suggest that FO have potential to abrogate CP induced nephrotoxicity and can be a useful adjuvant in long term CP chemotherapy without any side effects. Moreover, the inherent anti-cancer properties of FO with CP treatment may exhibits synergistic beneficial effects.
Keywords: Cisplatin, Nephrotoxicity, Fish oil, Brush border membrane, Carbohydrate metabolism
Abbreviations
ACPase: Acid Phosphatase; ALP: Alkaline Phosphatase; ATP: Adenosine Triphosphate; BBM: Brush Border; Membrane; BBMV: Brush Border Membrane Vesicle; BUN: Blood Urea Nitrogen; CAT: Catalase; CP: Cisplatin; Fbpase: Fructose 1,6-Bisphosphatase; FO: Fish Oil; G6PDH: Glucose-6-Phosphate Dehydrogenase; GGTase: ɤ-Glutamyl Transferase; HK: Hexokinase; HMP: Hexose Monophosphate; LAP: Leucine Aminopeptidase; LDH: Lactate Dehydrogenase; LPO: Lipid Peroxidation; MDH: Malate Dehydrogenase; ME: Malic Enzyme; PUFA: Polyunsaturated Fatty Acids; ROS: Reactive Oxygen Species; SOD: Superoxide Dismutase
Introduction
Kidney is a complex organ which performs a variety of essential functions in the body including clearance of endogenous waste, reabsorption of nutrients and elimination of xenobiotics and/or their metabolites. However, certain drugs and environmental toxicants accumulate in the kidney and induce deleterious nephrotoxic effects [1-3]. Cisplatin (cis-diamminedichloroplatinum II, CP) is one of the most prominent antineoplastic drug. The administration of CP at high doses in repeated cycles results in irreversible renal failure [4], thus, preventing full advantage of its chemotherapeutic efficacy. Previous studies in our lab have shown that administration of a single dose of CP led to declined renal functions as evident by increased serum creatinine and blood urea nitrogen accompanied by decreased creatinine clearance [5,6]. The toxic effects of CP are generally associated with necrosis, apoptosis and its ability to induce reactive oxygen species (ROS) generation [5,7-9]. These ROS directly act on cellular components viz. proteins, lipids and nucleic acids and cause injury and loss of function of the kidney and other tissues. Therefore, it is important to develop efficient strategy to attenuate CP induced side effects. Numerous approaches have been attempted to curtail the toxic effects of CP chemotherapy, however, the use of dietary supplements are gaining great attention in mitigating CP induced nephrotoxicity due to their potential safety, nutritive and therapeutic values.
Fish oil (FO) enriched in ω-3 PUFA provide one such dietary source of biologically active components that has been shown to be co-preventative and co-therapeutic in a wide variety of pathologies including cancers, cardiovascular disorders, diabetes, depression, arthritis and disorders of the kidney and intestine [10-12]. FO has also been shown to prevent drug induced apoptosis and inflammation [13]. As an essential nutrient, omega-3 polyunsaturated fatty acids (ω-3 PUFA) are known to incorporate in cellular membranes, alter their properties and enhance the expression of antioxidant enzymes [14,15]. The strong antioxidant potential of ω-3 PUFA in protecting against multiple organ toxicities induced by a variety of free radicals generating agents including drugs and toxicants is well documented [3,13,16-18]. Interestingly, there are also evidences that FO supplementation increases the efficacy of CP and doxorubicin in inhibiting tumor growth [19,20]. Recently, we have observed that dietary FO prevented alterations in the kidney structure and excretory/metabolic functions induced by a single nephrotoxic dose of CP [5]. Since cancer chemotherapy requires administration of CP in multiple doses over a long duration, therefore, the present study was undertaken to investigate whether the protective efficacy of FO extends for the long term multiple dose CP treatment induced nephrotoxicity. The results of the present study indicate that FO supplementation offered protection against multiple dose CP treatment induced nephrotoxicity, alterations in the enzymes of brush border membrane (BBM), carbohydrate metabolism and enzymatic and non-enzymatic antioxidant parameters. We conclude that FO consumption prior to and along with CP treatment resulted in improved renal functional capabilities and structural integrity by reducing CP induced oxidative damage to the rat kidney.
Materials and Methods
Chemicals and drugs
Fish oil (FO) was purchased from Menhaden, Sigma Chemical Co., USA. Cisplatin was obtained from Sigma–Aldrich Chemical Co., St. Louis, MO, USA. All other chemicals used were of analytical grade and were purchased either from Sigma Chemical Corp. or SRL (Mumbai, India). CP solution (2 mg/ml) was freshly prepared in 0.9% normal saline by continuous stirring at room temperature for 10 min.
Diet
A nutritionally adequate laboratory pellet diet was obtained from Aashirwaad Industries, Chandigarh (India). The diet was crushed finely and mixed with 15% FO and stored in airtight containers. Vitamin E as DL-α-tocopherol (270 mg/kg chow) was also added to the modified rat chows in order to meet the increased metabolic requirement for antioxidants on a diet high in PUFAs.
Experimental design
The animal experiments were conducted according to the guidelines of the committee for Purpose of Control and Supervision of Experiments on Animals (CPCSEA), Ministry of Environment and Forests, Government of India. Adult male Wistar rats (10 rats/group), weighing 150-200 g were acclimatized to the animal facility for one week on a standard rat diet (Aashirwad Industries, Chandigarh, India) and water ad libitum. Multiple injections of CP (3 mg/kg body weight/day), were administered to control and FO (15%, w/v) consuming rats to induce nephrotoxicity every fifth day to control and FO-feeding rats over a period of 25 days. The control rats received an equivalent amount of normal saline in the same manner. Body weight of the rats was recorded at the start and completion of the experimental procedure. The rats were sacrificed under light ether anesthesia. Blood samples were collected and the kidneys were removed and processed for the preparation of homogenates and BBM vesicles (BBMVs) as described below. All the preparations and analyses of various parameters were carried out simultaneously under similar experimental conditions to avoid any day-to-day variations.
Preparation of homogenates
After the completion of the experiment, the kidneys were removed, decapsulated and kept in ice-cold buffered saline (154 mM NaCl, 5 mM Tris-HEPES, pH 7.5). The cortical and medullary regions were carefully separated as described earlier [21]. A 15% (w/v) homogenate was prepared in 0.1 M Tris-HCl buffer pH 7.5 using Potter-Elvehejem homogenizer (Remi motors, Mumbai, India) with five complete strokes. The homogenate was centrifuged at 3000g at 4˚C for 15 min to remove cell debris and the supernatant was saved in aliquots and stored at -20˚C for assaying the enzymes of carbohydrate metabolism, free-radical scavenging enzymes and for estimation of total-SH and lipid peroxidation (LPO).
Preparation of brush border membrane vesicles (BBMV)
BBMV were prepared from whole cortex using the MgCl2 precipitation method as previously described [22]. Briefly, freshly minced cortical slices were homogenized in 50 mM mannitol and 5 mM Tris-HEPES buffer pH 7.0 (20 ml/g), in a glass Teflon homogenizer with 4 complete strokes. The homogenate was then subjected to high speed homogenization in an Ultra Turex homogenizer (Type T-25, Janke & Kunkel GMBH & Co. KG. Staufen) for three strokes of 15 s each with an interval of 15 s. MgCl2 was added to the homogenate to a final concentration of 10 mM and the mixture stirred for 20 min on ice. The homogenate was centrifuged at 2000g for 10 min in a Beckman centrifuge (J2 MI, Beckman instruments Inc, Palo Alto, C.A. USA) using JA-17 rotor and the supernatant was then recentrifuged at 35,000g for 30 min. The pellet was resuspended in 300 mM mannitol and 5mM Tris-HEPES, pH 7.4, with four passes by a loose fitting Dounce homogenizer (Wheaton IL, USA) and centrifuged at 35,000g for 20 min in a 15 ml corex tube. The outer white fluffy pellet of BBMV was resuspended in small volume of buffered 300 mM mannitol. Aliquots of homogenates and BBMV were saved and stored at -20˚C for BBM enzyme analyses. Each sample of BBMV was prepared by pooling tissues from two rats.
Serum chemistries
Blood samples were centrifuged at 2000g for 10 mins and sera were separated. Samples then deproteinated with 3% Trichloroacetic acid in a ratio 1: 3, left for 10 minutes and then centrifuged at 2000g for 10 mins. The protein free supernatant was used to determine inorganic phosphate (Pi) and creatinine (Scr). The precipitate was used to quantitate total phospholipids (PLs). Blood Urea Nitrogen (BUN) and cholesterol (Chl) levels were determined directly in serum samples. Glucose (Glc) was estimated by o-toluidene method using kit from Span diagnostics (Mumbai, India). These parameters were determined by standard procedures as mentioned in a previous study [23].
Enzyme assays
The activities of BBM biomarkers enzymes, alkaline phosphatase (ALP), leucine amino peptidase (LAP), γ- glutammyl transferase (GGTase) in the cortical and medullary homogenates and in BBMVs and lysosomal enzyme, acid phosphatase (ACPase) in the homogenates were assayed as described by Farooq et al. [24]. The enzymes of carbohydrate metabolism, e.g., lactate dehydrogenase (LDH), malate dehydrogenase (MDH), glucose-6-phosphate (G6PDH) dehydrogenase and NADP-malic enzyme (ME), involved in oxidation of NADH or reduction of NADP were determined by measuring the extinction changes at 340 nm in a spectrophotometer (Cintra 5; GBC Scientific Equipment, Pty., Victoria Australia) as described previously [21,23]. The other enzymes, glucose-6- phosphatase (G6Pase), fructose-1, 6-bisphospatase (FBPase) and hexokinase (HK) were determined as described in our previous studies Salim et al. [23]. LPO and total SH levels and the activities of superoxide dismutase (SOD), catalase (CAT) and glutathione peroxidase (GSH-Px) were determined as described earlier by Priyamvada et al. [16]. Protein concentration was determined by the modified method of Lowry et al. [25] as described by Yusufi et al. [22].
Statistical analyses
All data are expressed as Mean ± SEM for at least five different preparations. Statistical evaluation was conducted by one-way ANOVA and by unpaired student’s t test using SPSS 7.5 software. A probability level of p < 0.05 was selected as indicating statistical significance.
Result
The effect of long-term fish oil (FO) consumption and multiple cisplatin (CP) injections was determined on various parameters in rat serum and kidney. In general, the rats remained clinically well throughout the study. There was no significant difference in daily food and fluid intake between control and other experimental rats (data not shown). However, CP caused a slight decrease in the body weights of rats (Table 1).
| Groups | Before Treatment | After Treatment | % change |
|---|---|---|---|
| Control | 130 ± 8.30 | 133.33 ± 10.60 | +3 |
| CP | 131.25 ± 6.10 | 118.75 ± 9.20 | -10 |
| CPF | 131.25 ± 10.60 | 126.25 ± 4.50 | -3 |
| FO | 130 ± 7.50 | 128.11 ± 6.90 | -1 |
Table 1: Effect of dietary fish oil (FO) on body weight (in gms) of rats with / without CP treatment.
Effect of dietary fish oil (FO) on multiple dose CP treatment induced alterations in serum parameter
The renal dysfunction induced by multiple CP injections was characterized by elevated levels of Scr (+60%), BUN (+96%), Chl (+37%) and PLs (+50%), accompanied by a significant decrease in serum Glc (-10%) and Pi (-26%) (Table 2). Prolonged FO-diet consumption by CP-treated rats prevented CP induced increase in Scr, Chl and BUN indicating attenuation in CP-induced nephropathy. Serum Glc and Pi, however, profoundly increased in CPF co treated rats (Table 2). Dietary supplementation of FO alone decreased BUN, Scr, PLs and cholesterol whereas glucose and Pi were further increased in FO as compared to control group.
| Enzyme | Creatinine (mg/dl) | BUN (mg/dl) | Cholesterol (mg/dl) | Phospholipids (mg/dl) | Phosphate (μmol/ml) | Glucose (mg/dl) |
|---|---|---|---|---|---|---|
| Groups | ||||||
| Control | 1.00 ± 0.03 | 10.04 ± 0.90 | 89.53 ± 2.02 | 125.6 ± 3.46 | 2.56 ± 0.07 | 103.4 ± 6.71 |
| CP | 1.60 ± 0.06* (+60%) | 19.71 ± 1.64* (+96%) | 122.95 ± 5.42* (+37%) | 188 ± 18.53* (+50%) | 1.90 ± 0.06* (-26%) | 92.69 ± 3.27* (-10%) |
| CPF | 1.21 ± 0.03*† (+21%) | 14.97 ± 1.36*† (+49%) | 101.82 ± 2.98*† (+14%) | 140.25 ± 3.91* (+12%) | 2.21 ± 0.04*† (+14%) | 125.7 ± 7.96*† (+22%) |
| FO | 0.978 ± 0.05 (-2%) | 9.7 ± 0.36 (-3%) | 84.62 ± 0.67 (-5%) | 111.34 ± 1.98* (-11%) | 3.21 ± 0.078* (+25%) | 144.82 ± 2.98* (+40%) |
Table 2: Effect of dietary fish oil (FO) with and without multiple dose CP treatment on serum parameters.
Effect of dietary fish oil (FO) on multiple dose CP treatment induced alterations in BBM and lysosomal enzymes in the renal homogenates and in isolated BBMV
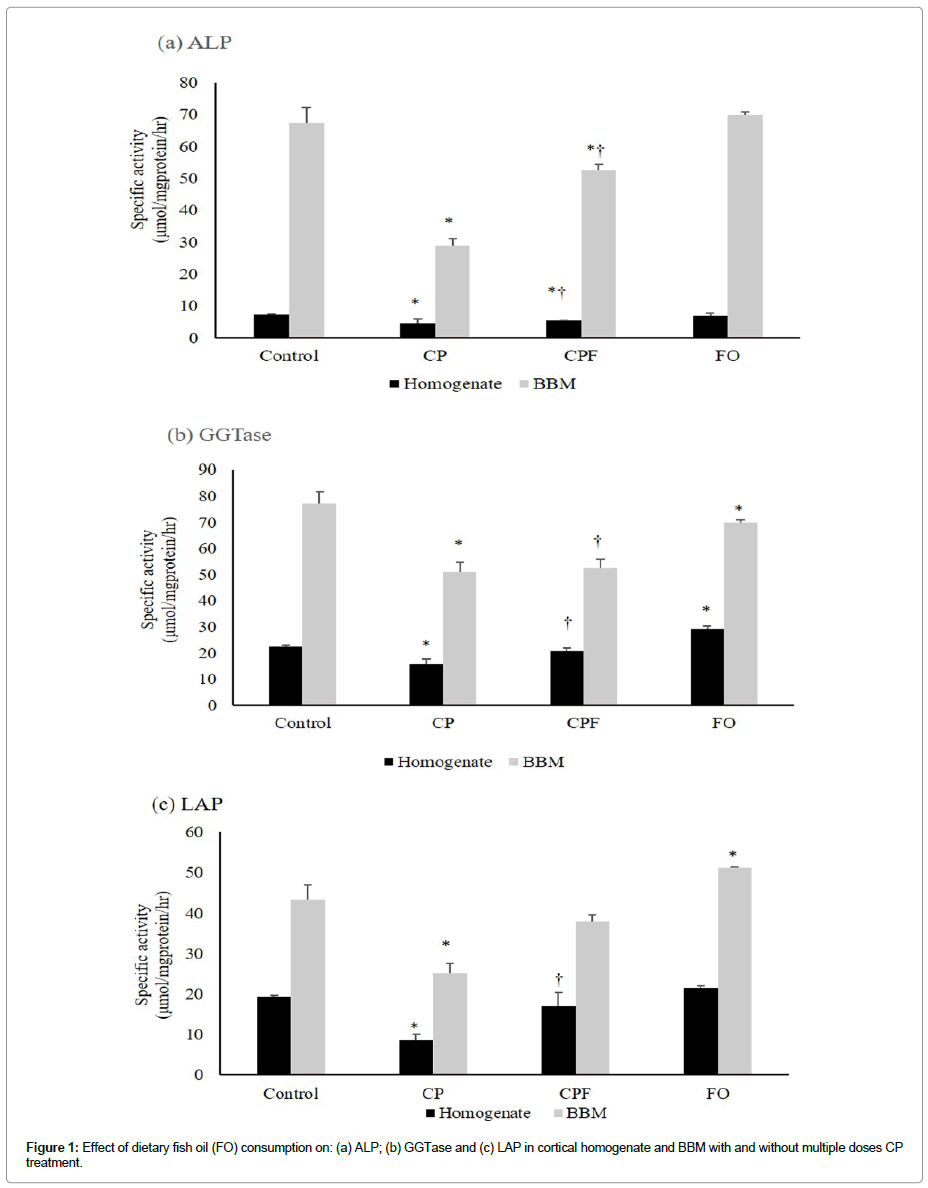
The renal proximal tubular BBM has been shown as the major site of CP-induced renal injury [1]. Since BBM contains a number of hydrolytic enzymes and transport systems, the structural and functional integrity was determined by the status of specific biomarker enzymes in the homogenates and isolated BBMVs under various experimental conditions (Tables 3 and 4). CP treatment caused marked decrease in specific activities of ALP (-37%), GGTase (-30%), and LAP (-56%) in renal cortical homogenates (Table 3a). Similar to the cortex, the activities of BBM enzymes were also lowered but to a greater extent in the medulla by CP (Table 3b). Consumption of FO diet alone, however, caused increase in GGTase and LAP activities while ALP activity decreased slightly compared to control rats. However, FO consumption prior to and along with CP treatment markedly prevented CP induced decrease in various BBM enzyme activities both in the cortex and medulla (Table 3). Furthermore, the activity of ACPase, a lysosomal enzyme significantly increased by CP treatment both in the cortical and medullary homogenates (Table 3a and 3b). However, the CP-induced effect on ACPase activity appeared to be mitigated by FO administration.
| Enzyme | ALP (mmol/mg protein/h) | GGTase (mmol/mg protein/h) | LAP (mmol/mg protein/h) | ACPase (mmol/mg protein/h) |
|---|---|---|---|---|
| Groups | ||||
| a) Cortex | ||||
| Control | 7.38 ± 0.14 | 22.36 ± 0.76 | 19.23 ± 0.29 | 4.36 ± 0.20 |
| CP | 4.63 ± 1.24* (-37%) | 15.6 ± 2.04* (-30%) | 8.4 ± 1.59* (-56%) | 5.38 ± 0.12* (+23%) |
| CPF | 5.4 ± 0.22*† (-27%) | 20.64 ± 1.28† (-8%) | 16.94 ± 3.42† (-12%) | 4.63 ± 0.13† (+6%) |
| FO | 6.89± 0.86 (-7%) | 29.13±1.09* (+30%) | 21.48± 0.54 (+12%) | 3.89 ± 0.12 (-11%) |
| b) Medulla | ||||
| Control | 52.53 ± 2.28 | 30.95 ± 2.09 | 19.16 ± 0.92 | 1.41 ± 0.08 |
| CP | 23.88 ± 2.22* (-55%) | 20.51 ± 0.86* (-34%) | 6.78 ± 0.33* (-65%) | 2.3 ± 0.12* (+63%) |
| CPF | 38.28 ± 0.81*† (-27%) | 24.9 ± 0.07*† (-20%) | 17.54 ± 0.94† (-8%) | 1.89 ± 0.08 (+34%) |
| FO | 45.61 ± 1.23 (-13%) | 38.46 ± 0.067* (+24%) | 20.12 ± 0.76 (+5%) | 1.44 ± 0.03 (+2%) |
Table 3: Effect of dietary fish oil (FO) with and without multiple dose CP treatment on biomarker enzymes of BBM and lysosomes in homogenates in a) cortex and b) medulla.
The effect of CP, FO alone and their combination (CPF) on BBM marker enzymes was further analyzed in isolated BBMV preparations. A similar activity pattern of the enzymes was observed as in cortical homogenates, however, the magnitude of the effects of CP and CPF were much more prominent in BBMV than in the respective homogenate values (Table 4). The result suggests that CP caused severe damage to the structural architecture of BBM and FO administration was able to maintain BBM integrity as in short term CP treatment either by lessening the damage caused or by increasing the regeneration processes.
| Enzyme Groups |
ALP (mmol/mg protein/h) | GGTase (mmol/mg protein/h) | LAP (mmol/mg protein/h) |
|---|---|---|---|
| Control | 67.36 ± 4.64 | 77.07 ± 4.65 | 43.21 ± 3.83 |
| CP | 28.89 ± 2.29* (-57%) | 50.98 ± 3.76* (-34%) | 25.2 ± 2.28* (-42%) |
| CPF | 52.47 ± 1.87*† (-22%) | 63.55 ± 3.37† (-17%) | 37.87 ± 1.71 (-12%) |
| FO | 69.86 ± 0.98 (+4%) | 86.78 ± 1.21* (+13%) | 51.12 ± 0.25* (+18%) |
Table 4: Effect of dietary fish oil (FO) with and without multiple dose CP treatment on activities of biomarker enzymes of BBM in cortical BBMV.
Effect of dietary fish oil (FO) on multiple dose CP treatment induced alterations on metabolic enzymes in renal cortex and medulla
The renal transports and reabsorptive function depends on structural integrity of BBM and available energy as ATP, which is generated by various metabolic pathways including glycolysis and oxidative metabolism. Hence, it is important to determine whether FO diet would prevent/reduced CP induced alterations in the metabolic pathways that affects kidney functions.
The effect of CP and its combination with FO (CPF) was assessed on the activities of various enzymes of carbohydrate metabolism involved in glycolysis, TCA cycle, gluconeogenesis and HMP shunt pathway in renal cortex and medulla (Table 5).
| Enzymes Group | HK (mmol/mg protein/h) | LDH (mmol/mg protein/h) | MDH (mmol/mg protein/h) | G6Pase (mmol/mg protein/h) | FBPase (mmol/mg protein/h) | ME (mmol/mg protein/h) | G6PDH (mmol/mg protein/h) |
|---|---|---|---|---|---|---|---|
| a) Cortex | |||||||
| Control | 2.77 ± 0.11 | 2.23 ± 0.2 | 17.17 ± 0.8 | 1.21 ± 0.09 | 3.01 ± 0.259 | 0.51 ± 0.02 | 0.07 ± 0.01 |
| CP | 3.37 ± 0.09* (+22%) | 3.8 ± 0.48* (+70%) | 9.62 ± 0.73* (-44%) | 1.3 ± 0.11* (+7%) | 3.11 ± 0.15 (+3%) | 0.69 ± 0.08* (+35%) | 00.04 ± 0.01* (-43%) |
| CPF | 3.19 ± 0.19 (+15%) | 2.57 ± 0.11† (+15%) | 15.84 ± 0.32† (-8%) | 1.27 ± 0.1 (+5%) | 3.05 ± 0.14* (+1%) | 0.44 ± 0.03*† (-14%) | 0.07 ± 0.002*† (0%) |
| FO | 2.72 ± 0.235 (-2%) | 2.13 ± 0.16 (-4%) | 20.35 ± 0.54* (+19%) | 1.83 ± 0.10* (+51%) | 3.28 ± 0.201 (+9%) | 0.388 ± 0.11* (-24%) | 0.089 ± 0.02* (+27%) |
| b) Medulla | |||||||
| Control | 3.05 ± 0.46 | 3.07 ± 0.33 | 23.7 ± 0.76 | 2.73 ± 0.16 | 3.75 ± 0.22 | 0.17 ± 0.02 | 0.932 ± 0.011 |
| CP | 3.83 ± 0.22* (+26%) | 6.21 ± 0.28* (+102%) | 13.64 ± 1.14* (-42%) | 2.06 ± 0.05* (-25%) | 3.29 ± 0.1* (-12%) | 0.27 ± 0.017* (+59%) | 0.687 ± 0.017* (-26%) |
| CPF | 3.12 ± 0.5† (+2%) | 3.87 ± 0.25† (+26%) | 19.03 ± 0.98*† (-20%) | 2.4 ± 0.29† (-12%) | 3.49 ± 0.12 (+7%) | 0.22 ± 0.013*† (+29%) | 0.796 ± 0.013*† (-15%) |
| FO | 2.71 ± 0.17 (-11%) | 3.22 ± 0.10 (-5%) | 26.45 ± 0.55 (+12%) | 2.98 ± 0.027 (+9%) | 4.25 ± 0.35* (+13%) | 0.141 ± 0.02* (-17%) | 0.956 ± 0.002 (+3%) |
Table 5: Effect of dietary fish oil (FO) with and without multiple dose CP treatment on carbohydrate metabolic enzymes in homogenates of a) cortex and b) medulla.
Effect on carbohydrate metabolism in renal cortex:
CP treatment to control rats significantly increased the activities of LDH, a marker of anaerobic glycolysis (+70%), HK (glycolysis) (+22%), G6Pase (+7%) and FBPase (+3%) (Gluconeogenesis) whereas decreased MDH activity, an enzyme of TCA cycle (-44%) (Table 5a). However, when CP treatment was extended to FO-fed rats, CP-induced alterations in the activities above mentioned metabolic enzymes were mitigated. CP elicited decrease in MDH activity was normalized to near control value by FO diet consumption. Moreover, CP treatment to significantly decreased G6PDH (-43%) but increased ME (+35%) activities (Table 5a). CP elicited decrease in G6PDH activity was completely prevented and the value was normalized back to the control value. CP induced ME activity increase was arrested by dietary FO supplementation.
Effect on carbohydrate metabolism in renal medulla
The activities of LDH and MDH in medullary homogenates were similarly affected by CP treatment albeit to different extent than in the cortical homogenates (Table 4b). Similar to cortex, G6PDH activity decreased whereas ME and HK activity increased by CP in the medulla. Feeding of FO enriched diet to CP treated rats prevented CP induced alterations in various enzyme activities. FO consumption to CP treated rats reduced/prevented the increase in LDH, HK and ME activities and decrease in MDH, G6PDH, G6Pase and FBPase activities (Table 5b).
Effect of dietary fish oil (FO) on multiple dose CP treatment alterations in antioxidant defense parameters in renal cortex and medulla
In agreement with previous studies, CP significantly decreased the activities of SOD, CAT and GSH-Px and increased LPO (measured in terms of malondialdehyde) both in the renal cortex and medulla (Table 6). CP treatment significantly enhanced LPO and decline in total-SH in both the renal tissues. However, CP induced cellular damage was much more apparent in renal cortex than in medulla as reflected by greater increase in LPO and decrease in total- SH. CP treatment caused marked decrease in SOD (-36%), CAT (-25%) and GSH-Px (-33%) activities in the renal cortex. In medulla, the activity of SOD (-19%), CAT (-33%) and GSH-Px (-47%) also showed significant decline by CP administration. However, CP administration to FO fed rats prevented the decline in SOD, CAT and GSH-Px activities. Dietary FO supplementation to CP treated rats also prevented CP elicited increase in LPO and decrease in total- SH groups. Dietary FO consumption alone significantly enhanced the activities of antioxidant enzymes in both the renal cortex and medulla and the activities of these enzymes in FO group were found to be higher than in the CP or control groups. The results indicate marked protection by FO diet against CP induced oxidative damage to renal tissues.
| Parameters Groups | Lipid peroxidation (nmol/gm tissue) | Total SH (µmol/gm tissue) | SOD (Units/mg protein) | CAT (µmol/mg protein/ min) | GSH-Px (µmol/mg protein/ min) |
|---|---|---|---|---|---|
| a) Cortex | |||||
| Control | 47.11 ± 1.67 | 3.49 ± 0.55 | 60 ± 4.24 | 237 ± 7.95 | 0.15 ± 0.03 |
| CP | 148.48 ± 4.09 * (+215%) | 2.45 ± 0.77 * (-30%) | 38.14 ± 2.0 * (-36%) | 178.05 ± 7.85* (-25%) | 0.1 ± 0.005 * (-33%) |
| CPF | 73.07 ± 5.62*† (+55%) | 2.88 ± 0.28 *† (-17%) | 53.05 ± 0.87 † (-12%) | 215.12 ± 7.68 † (-9%) | 0.107 ± 0.01 (-29%) |
| FO | 55.12 ± 1.90* (+17%) | 3.56 ± 0.93 (+2%) | 79.98 ± 0.93* (+33%) | 265.14 ± 4.12 (+12%) | 0.181 ± 0.016* (+21%) |
| b) Medulla | |||||
| Control | 26.53 ± 0.81 | 1.44 ± 0.07 | 109.5 ± 6.43 | 198.95 ± 7.19 | 0.17 ± 0.01 |
| CP | 48.21 ± 1.07 * (+99.81%) | 1.13 ± 0.16 * (-22%) | 88.34 ± 4.31 * (-19%) | 133.15 ± 11.04 * (-47.13%) | 0.09 ± 0.01 * (-47%) |
| CPF | 38.38 ± 0.86*† (+82.71%) | 1.82 ± 0.3 *† (+26%) | 118.07 ± 2.04 † (+8%) | 193.58 ± 13.71 † (-26.59%) | 0.12 ± 0.01*† (-29%) |
| FO | 29.63 ± 0.54* (+12%) | 1.59 ± 0.93 (+10%) | 161.13 ± 4.14* (+47%) | 231.19 ± 5.46* (+16%) | 0.20 ± 0.016* (+18%) |
Table 6: Effect of dietary fish oil (FO) with multiple doses CP on non-enzymatic and enzymatic antioxidant parameters in homogenates of (a) cortex and (b) medulla.
Discussion
The consumption of dietary fats is essential for life; however they also affect our health in many different ways. The large intake of saturated fat, Trans fat and refined oils are the primary cause of modern sufferings such as cancer, diabetes, depression, hypertension and cardiovascular diseases [26]. However, the consumption of unsaturated fats, especially PUFA, was shown to have numerous health benefits [27,28]. This stems from the large body of evidences pointing to cardio-protective properties and prevention of other chronic diseases including bronchial asthma, diabetes mellitus and Alzheimer [29]. Therefore, the role of FO has been extensively investigated as a therapeutic option due to the high proportion of constituent ω-3 fatty acids. In addition, FO supplementation has been shown to enhance, the endogenous antioxidant defense system and provide resistance against free radical attacks [30]. In the present study we have tested our hypothesis that FO administration would ameliorate multiple doses CP treatment induced nephrotoxic and other adverse effects in the kidney (Figure 1).
Multiple injections of CP were administered to control and FO consuming rats and the possible protective effect of FO was investigated against CP induced nephrotoxic alterations. In agreement with our previous studies, CP administration in multiple dose resulted in marked increase in Scr and BUN, accompanied by significant decrease in serum glucose and Pi with increase in serum cholesterol and phospholipids (Table 2) [1,8] . However, FO administration prior to and along with CP treatment significantly mitigated CP induced alterations in serum parameters and led to a significant recovery in the levels of Scr and BUN. Serum glucose and cholesterol were also improved and serum phospholipids were profoundly enhanced in FO consuming CP treated rats.
As a step towards understanding the biochemical mechanisms of CP induced toxicity as well as its effect on the structural and functional integrity of the plasma membrane, the activities of BBM marker enzymes were determined in cortical and medullary homogenates, as well as in isolated BBMVs preparations. A significant decline in the activities of ALP, GGTase and LAP were observed after CP treatment, both in the cortical homogenates and in isolated BBMVs, however, the extent of decline was more in BBMVs than in the respective cortical homogenates (Table 3) and (Table 4). Similar decrease in the activities of BBM enzymes was observed in medulla. The reduction in the activities of BBM enzymes in the kidney suggest a severe damage to the structural architecture of the membrane as suggested by excessive lipid peroxidation and confirmed by earlier morphological studies [6]. The alterations in the structural architecture of BBM could have caused loss of BBM/BBM enzymes into the tubular lumen followed by excessive excretion of these enzymes in the urine, as reported earlier for other toxicants [2]. The decrease in BBM enzyme activities could also be due to CP generated ROS that could have caused oxidative modification and further inactivation of BBM enzymes. However, FO consumption in combination with CP treatment resulted in the attenuation of CP induced alterations in the BBM enzyme activities both in the cortical and medullary homogenates as well as in isolated BBMVs. The results convincingly demonstrate that FO consumption prevented the CP elicited decrease in the activities of BBM enzymes. The observed ameliorative effect of FO could be due to its ROS scavenging and/or antioxidant properties that could have neutralized the CP generated ROS and thus, protects the structural integrity of BBM [5,15,31]. Moreover, FO could have accelerated the repair and/ or regeneration process of damaged membrane by incorporating the constituent PUFAs into the membrane thereby causing changes in the lipid composition which in turn affect the biophysical characteristics of the membrane and the activities of membrane associated enzymes [5,32]. The activity of lysosomal enzyme, ACPase was significantly increased both in the renal cortex and medulla in CP treated rats. However, FO consumption prior to and along with CP treatment, mitigated the CP induced increase in ACPase activity.
Reabsorption of important ions and molecules is the major function of kidney that in turn depends on the available energy (ATP) through various metabolic pathways. Therefore, any alterations to these metabolic pathways caused by toxic insult would affect the functional capabilities of the kidney. Hence, to assess the functional aspects, the activities of various enzymes of carbohydrate metabolism were determined in the renal tissue homogenates prepared from animals of different experimental groups. In agreement with the previous studies, the activities of various enzymes involved in glycolysis, TCA cycle, gluconeogenesis and HMP shunt pathway were altered both in the renal cortical and medullary homogenate by CP treatment (Table 5). CP treatment caused increase in glycolysis as evident by increased HK activity with a simultaneous increase LDH activity, both in the renal cortex and medulla. As LDH catalyzes the formation of lactate from pyruvate under anaerobic condition, this suggests a shift in the glucose metabolism to an anaerobic mode. Further, multiple dose CP treatment also caused decrease in MDH activity in both the renal tissues, thereby pointing again to the shift in energy dependence from aerobic to anaerobic glycolysis and that could be due to CP-induced mitochondrial dysfunction [7]. An impaired oxidative metabolism of glucose/fatty acid due to mitochondrial dysfunction would result in lower ATP production by oxidative metabolism and hence lower renal transport function. This was further accompanied by reduced activities of key gluconeogenic enzymes viz. G6Pase and FBPase, both in the renal cortex and medulla (Table 5). The activity of G6PDH, an enzyme of HMP-shunt pathway, however profoundly declined while the activity of NADP-ME was increased in the renal tissues by multiple dose CP treatment. However, FO supplementation to CP-treated rats significantly mitigated the CP induced alterations in carbohydrate metabolism as evident by higher activities of LDH, MDH and gluconeogenic enzymes in CPF group as compared to CP treated rats. These protective effect of FO could be attributed to the fact that the constituent ω-3 PUFA might have upregulated the genes encoding these enzymes and thus the activities involved in carbohydrate metabolism were enhanced [33,34]. Furthermore, FO consumption by incorporating fatty acids in mitochondrial membrane could have lowered the number of damaged mitochondria or affected macromolecules or increased number of normally active organelles or macromolecules.
Several studies have reported that CP treatment enhances the generation of free radicals and ROS that overcome the antioxidant capabilities of target tissues, resulting in oxidative damage [5,8,9]. In agreement with previous observations, multiple dose CP treatment significantly enhanced lipid per oxidation (LPO) and depleted total thiol (-SH) levels indicating induction of oxidative stress [8]. CP treatment also perturbed endogenous antioxidant defense mechanism as indicated by decrease in the activities of SOD, CAT and GSHPx. On the other hand, numerous studies have demonstrated the therapeutic benefits of FO against oxidative stress induced pathologies [35,36]. FO, by virtue of the antioxidant properties attributed to ω-3 PUFA constituents, significantly enhanced the renal antioxidant defense mechanism in FO consuming rats. The SOD activity was not only improved in the renal cortex after FO consumption in CP treated rats (CPF group) but was significantly higher in renal medulla in comparison to the control group. However, GSH-Px activity showed insignificant improvement after FO consumption. These alterations were associated with lowering of LPO in renal tissues. The results suggest that FO provides protection against CP nephropathy or oxidative damage either by SOD-mediated or by CAT mediated mechanism or involving both the mechanisms simultaneously.
In conclusion, the results of present study show that long term CP treatment induces generation of free radicals that causes oxidative damage to cellular organelles, especially mitochondria and plasma membrane. In contrast, FO consumption prior to and simultaneously with CP treatment considerably mitigates CP induced nephrotoxicity. The alleviation of oxidative stress induced cellular damage is the underlying mechanism of FO mediated renoprotective effect. Owing to its free radical scavenging and/or antioxidant properties, FO improves the structural integrity of various organelles/macromolecules and eventually enhances renal functional capabilities. Moreover, the inherent anticancer properties of FO together with CP may exhibit a synergistic beneficial effect. Thus, FO may have potential for clinical application as an adjuvant in long term CP chemotherapy, to abrogate the nephrotoxic and other deleterious effect of CP without compromising its therapeutic efficacy.
Acknowledgements
University Grants Commission (UGC) for the award of scholarship to A.N. and S.R. and financial support to the department from University Grants Commission (UGC-DRF) is also gratefully acknowledged. Indian Council of Medical Research (ICMR), New Delhi, India is acknowledged for the award of SRF (Senior Research Fellowship) to W.K.
References
- Fatima S, Yusufi AN, Mahmood R (2004) Effect of cisplatin on renal brush border membrane enzymes and phosphate transport. Hum Exp Toxicol 12: 547-54.
- Banday AA, Priyamvada S, Farooq N, Yusufi AN, Khan F (2008) Effect of uranyl nitrate on enzymes of carbohydrate metabolism and brush border membrane in different kidney tissues. Food Chem Toxicol 46: 2080-2088.
- Khan MW, Priyamvada S, Khan SA, Khan S, Yusufi AN, et al. (2012) Protective effect of ω-3 polyunsaturated fatty acids (PUFAs) on sodium nitroprusside–induced nephrotoxicity and oxidative damage in rat kidney. Hum Exp Toxicol 31: 1035-1049.
- Karie S, Launay-Vacher V, Deray G, Isnard-Bagnis C (2010) Drugs renal toxicity. Nephrol Ther 6: 58–74.
- Naqshbandi A, Khan MW, Rizwan S, Rehman S, Khan F (2012) Studies on the protective effect of dietary fish oil on cisplatin induced nephrotoxicity in rats. Food Chem Toxicol 50: 265-273.
- Farooqui Z, Ahmed F, Rizwan S, Shahid F, Khan AA, et al. (2017) Protective effect of Nigella sativa oil on cisplatin induced nephrotoxicity and oxidative damage in rat kidney. Biomed Pharmacother 85: 7-15.
- Santos NA, Bezerra CC, Martins NM, Curti C, Bianchi ML, et al. (2008) Hydroxyl radical scavenger ameliorates cisplatin-induced nephrotoxicity by preventing oxidative stress, redox state unbalance, impairment of energetic metabolism and apoptosis in rat kidney mitochondria. Cancer Chemother Pharmacol 61: 145-155.
- Khan SA, Priyamvada S, Khan W, Khan S, Yusufi AN, et al. (2009) Studies on the protective effect of green tea against cisplatin induced nephrotoxicity. Pharmacol Res 60: 382-391.
- Hagar H, El Medany A, Salam R, El Medany G, Nayal OA (2015) Betaine supplementation mitigates cisplatin-induced nephrotoxicity by abrogation of oxidative/nitrosative stress and suppression of inflammation and apoptosis in rats. Exp Toxicol Pathol 67: 133-141.
- Bhattacharya A, Banu J, Rahman M, Causey J, Fernandes G (2006) Biological effects of conjugated linoleic acids in health and disease. J Nutr Biochem 17: 789-810.
- Berquin IM, Edwards IJ, Chen YQ (2008) Multi-targeted therapy of cancer by omega-3 fatty acids. Cancer Lett 269: 363-377.
- Weitz D, Weintraub H, Fisher E, Schwartzbard AZ (2010) Fish oil for the treatment of cardiovascular disease. Cardiol Rev 18: 258-263.
- Khan MW, Priyamvada S, Khan SA, Khan S, Yusufi AN (2015) Fish/flaxseed oil protect against nitric oxide-induced hepatotoxicity and cell death in the rat liver. Hum Exp Toxicol 35: 302–311.
- Bhattacharya A, Lawrence RA, Krishnan A, Zaman K, Fernandes G (2003) Effect of dietary n-3 and n-6 oils with and without food restriction on activity of antioxidant enzymes and lipid peroxidation in livers of cyclophosphamide treated autoimmune-prone NZB/W female mice. J Am Coll Nutr 22: 388-399.
- Carrillo-Tripp M, Feller SE (2005) Evidence for a mechanism by which ω-3 polyunsaturated lipids may affect membrane protein function. Biochemistry 44: 10164-10169.
- Priyamvada S, Priyadarshini M, Arivarasu NA, Farooq N, Yusufi AN, et al. (2008) Studies on the protective effect of dietary fish oil on gentamicin-induced nephrotoxicity and oxidative damage in rat kidney. Prostaglandins Leukot Essent Fatty Acids 78: 369-381.
- Naqshbandi A, Khan MW, Rizwan S, Yusufi AN, Khan F (2011) Studies on the protective effect of fish oil against cisplatin induced hepatotoxicity. Biol Med 3: 86-97.
- Uygur R, Aktas C, Tulubas F, Uygur E, Kanter M, et al. (2014) Protective effects of fish omega 3 fatty acids on doxorubicin induced testicular apoptosis and oxidative damage in rats. Andrologia 46: 917-926.
- Yam D, Shinitzky M (2001) Suppression of tumor growth and metastasis by dietary fish oil combined with vitamins E and C and cisplatin. Cancer Chemother Pharmacol 47: 34-40.
- Hardman WE, Avula CR, Fernandes G, Cameron IL (2001) Three percent dietary fish oil concentrate increased efficacy of doxorubicin against MDA-MB 231 breast cancer xenografts. Clin Cancer Res 7: 2041-2049.
- Khundmiri SJ, Asghar M, Khan F, Salim S, Yusufi AN (2004) Effect of ischemia and reperfusion on enzymes of carbohydrate metabolism in rat kidney. J Nephrol 17: 377-383.
- Yusufi AN, Murayama N, Gapstur SM, Szczepanska-Konkel M, Dousa TP (1994) Differential properties of brush-border membrane vesicles from early and late proximal tubules of rat kidney. Biochimica et Biophysica Acta (BBA)-Biomembranes 1191: 117-132.
- Salim S, Farooq N, Priyamvada S, Asghar M, Yusufi AN (2007) Influence of Ramadan-type fasting on carbohydrate metabolism, brush border membrane enzymes and phosphate transport in rat kidney used as a model. Brit J Nutr 98: 984-990.
- Farooq N, Yusufi AN, Mahmood R (2004) Effect of fasting on enzymes of carbohydrate metabolism and brush border membrane in rat intestine. Nutr Res 24: 407-416.
- Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193: 265-275.
- Tokunaga M, Takahashi T, Singh RB, Rupini D, Toda E, et al. (2012) Diet, nutrients and noncommunicable diseases. Open Nutra J 5: 146-159.
- Simopoulos AP (1991) Omega-3 fatty acids in health and disease and in growth and development. Am J Clin Nutr 54: 438-463.
- Doughman SD, Krupanidhi S, Sanjeevi CB (2007) Omega-3 fatty acids for nutrition and medicine: considering microalgae oil as a vegetarian source of EPA and DHA. Curr Dia Rev 3: 198-203.
- Harris WS (2007) Omega-3 fatty acids and cardiovascular disease: a case for omega-3 index as a new risk factor. Pharmacol Res 55: 217-223.
- Erdogan H, Fadillioglu E, Ozgocmen S, Sogut S, Ozyurt B, et al. (2004) Effect of fish oil supplementation on plasma oxidant/antioxidant status in rats. Prostaglandin Leukot Essent Fatty Acids 71: 149-152.
- Priyamvada S, Khan SA, Khan MW, Khan S, Yusufi AN, et al. (2010) Studies on the protective effect of dietary fish oil on uranyl-nitrate-induced nephrotoxicity and oxidative damage in rat kidney. Prostaglandins Leukot Essent Fatty Acids 82: 35-44.
- Chakrabarti KB, Hopewell JW, Wilding D, Plowman PN (2001) Modification of doxorubicin-induced cardiotoxicity: effect of essential fatty acids and ICRF-187 (dexrazoxane). Eur J Cancer 37: 1435-1442.
- Yoshida H, Ikeda I, Tomooka M, Mawatari M, Imaizumi K, et al. (2001) Effect of dietary seal and fish oils on lipid metabolism in hamsters. J Nutr Sci Vitaminol 47: 242-247.
- Yilmaz HR, Songur A, Özyurt B, Zararsiz I, Sarsilmaz M (2004) The effects of n-3 polyunsaturated fatty acids by gavage on some metabolic enzymes of rat liver. Prostaglandins Leukot Essent Fatty Acids 71: 131-135.
- Saada HN, Said UZ, Mahdy EM, Elmezayen HE, Shedid SM (2014) Fish oil omega-3 fatty acids reduce the severity of radiation-induced oxidative stress in the rat brain. Int J Radiat Biol 90: 1179-1183.
- Jangale NM, Devarshi PP, Bansode SB, Kulkarni MJ, Harsulkar AM (2016) Dietary flaxseed oil and fish oil ameliorates renal oxidative stress, protein glycation, and inflammation in streptozotocin–nicotinamide-induced diabetic rats. J Physiol Biochem 72: 327-336.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi