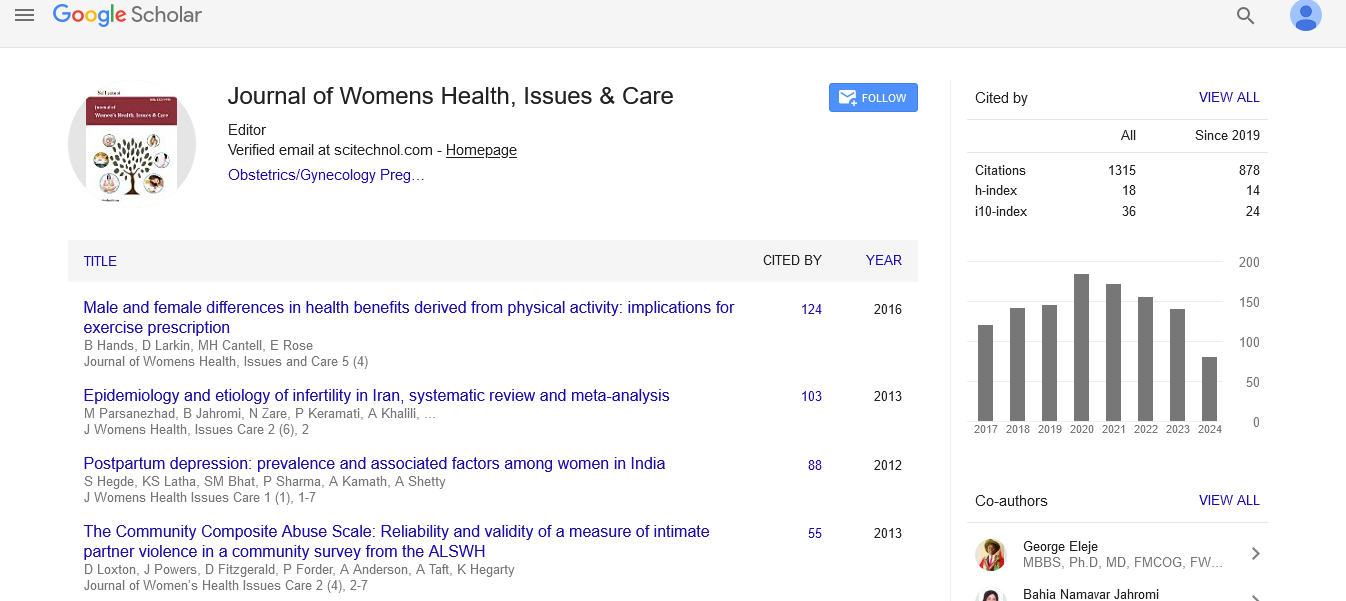
Review Article, J Womens Health Issues Care Vol: 5 Issue: 3
Long-Term Effects of Maternal Nutrition and Childhood Growth on Later Health
| Mohammadreza Vafa1,2 and Salma Mahmoodianfard3* | |
| 1Department of Nutrition, School of Public Health, Iran University of Medical Sciences, Tehran, Iran | |
| 2Endocrine Research Center (Firouzgar), Institute of Endocrinology and Metabolism (Hemmat Campus), Iran University of Medical Sciences, Iran | |
| 3Department of Clinical Nutrition, School of Nutritional Sciences & Dietetics, Tehran University of Medical Sciences, Iran | |
| Corresponding author :Salma Mahmoodianfard Department of Clinical Nutrition, School of Nutritional Sciences & Dietetics, Tehran University of Medical Sciences, Iran E-mail: s.mahmoodianfard@gmail.com |
|
| Received: October 02, 2015 Accepted: April 14, 2016 Published: April 18, 2016 | |
| Citation: Vafa M, Mahmoodianfard S (2016) Long-Term Effects of Maternal Nutrition and Childhood Growth on Later Health. J Womens Health, Issues Care 5:3. doi:10.4172/2325-9795.1000230 |
Abstract
Objectives: This paper reviews the importance of maternal nutrition and weight gain with birthweigh and neonatal/childhood growth, and highlights the r isk of chronic diseases in later life.
Methods: The data was sourced based on the result of original and review articles relating to the life exposures, pregnancy weight gain, birth weight, childhood growth and the risk of chronic diseases in adult life.
Findings: Experimental studies have suggested that both maternal undernutrition and overnutrition are involved in later disease risk. Maternal macronutrient deficiency leads to LBW and subsequently insulin resistance and adiposity in later life. It seems that micronutrient deficiencies contribute to long-term negative effects such as metabolic syndrome and related disorders. As well as fetal life, early infancy, the adiposity rebound period and puberty also account as critical periods for the development of obesity in adulthood.
Conclusion: It is now widely accepted that the risks of adult chronic diseases may have their developmental origins in fetal life. Maternal under-nutrition or over-nutrition affect the infant’s health . Both macro- and micro-nutrients are critical for appropriate pregnancy outcomes. Understanding their precise patho-physiological mechanism are critical to apply new strategies to prevent the adverse effects of maternal dietary restriction and environmental factors in early stages of life.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi