Research Article, J Otol Rhinol Vol: 7 Issue: 1
May Hyperbaric Oxygen Therapy Play a Role in the Treatment of Allergic Rhinitis? A Double- Blind Experimental Study in Rat Model Hyperbaric Oxygen Therapy on Allergic Rhinitis
Erkan Vuralkan1*, Hatice Bengu Cobanoglu1, Bengusu Mirasoglu2, Sevdegul Mungan3, Abdullah Arslan4, Bekir Selim Bagli3, Akın Savas Toklu3 and Inan Utku Türkmen5
1Department of Otorhinolaryngology, University of Health Sciences, Faculty of Medicine, Trabzon Kanuni Research and Training Hospital, Trabzon, Turkey
2Department of Underwater and Hyperbaric Medicine, Istanbul University, Faculty of Medicine, Istanbul, Turkey
3Department of Pathology, Karadeniz Technical University, Faculty of Medicine, Trabzon, Turkey
4Department of Underwater and Hyperbaric Medicine, Necmettin Erbakan University, Faculty of Medicine, Konya, Turkey
5Basic Sciences Unit, TED University, Ankara, Turkey
*Corresponding Author : Erkan Vuralkan, MD
Department of Otorhinolaryngology, University of Health Sciences, Faculty of Medicine, Trabzon Kanuni Research and Training Hospital, Trabzon, Turkey
Tel: +905053783847
Fax: +904622302307
E-mail: erkanvuralkan@hotmail.com
Received: October 10, 2017 Accepted: January 06, 2018 Published: January 15, 2018
Citation: Vuralkan E, Cobanoglu HB, Mirasoglu B, Mungan S, Arslan A, et al. (2018) May Hyperbaric Oxygen Therapy Play a Role in the Treatment of Allergic Rhinitis? A Double- Blind Experimental Study in Rat Model Hyperbaric Oxygen Therapy on Allergic Rhinitis. J Otol Rhinol 7:1. doi: 10.4172/2324-8785.1000335
Abstract
Objectives: Medical treatments may be inadequate for the management of allergic rhinitis. Therefore, it requires different treatment options. We aimed to evaluate the effects of nasal steroid and hyperbaric oxygen (HBO) treatments on the nasal mucosa of an allergic rhinitis of a (AR) rat model.
Methods: Group 1 (control group); Group 2 toluene diisocyanate (TDI) group; Group 3 was exposed to TDI and treated with HBO; Group 4 was exposed to TDI and treated with intranasal
mometasone furoate. After the sacrification, the sections were then evaluated semi-quantitatively and parameters of histopathological inflammation, fibrosis and eosinophilia were assessed.
Results: The level of inflammation in the control group was significantly less severe than Group 2. There were statistically significant differences between Groups 2, 3 and 4. Evaluation of the
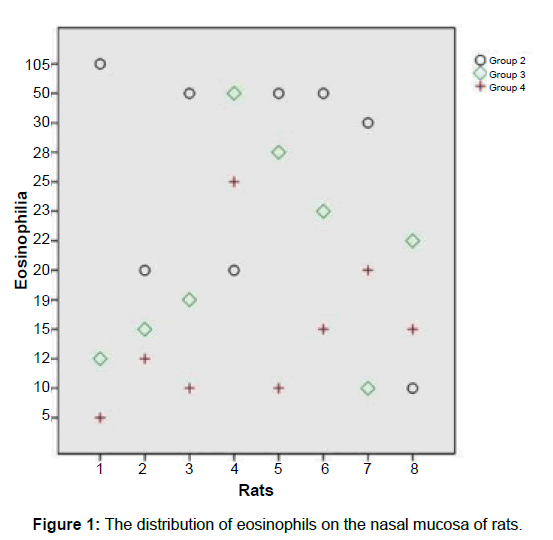
fibrosis scores showed that the scores of Group 4 were significantly higher. Group 1 has significantly lower eosinophil counts than the other groups. Group 2 has significantly higher eosinophil counts than group 4.
Conclusion: Anti-inflammatory effects of both nasal steroid and HBO treatments were found to be similar in our study. Nasal steroid therapy combined with HBO therapy may be used on treatmentresistant patients with persistent AR.
Keywords: Hyperbaric oxygen; Nasal mucosa; Allergic rhinitis
Introduction
Allergic rhinitis (AR) is the most common chronic immunological disease experienced by humans. AR has physical, social and psychological effects on quality of life. Recent studies have reported the roles of some mediators in the development of nasal allergy. Histamine, the major chemical mediator, leukotrienes, plateletactivating factor, prostaglandins and substance P are some of these mediators [1]. Nasal mucosa also contains many inflammatory cells that may contribute cytokines which trigger an allergic response. Sinonasal mucosal inflammation, eosinophilia, edema and increased mucus production play a key role in the pathophysiology of AR. European ENT specialists use “The Allergic Rhinitis and its Impact on Asthma (ARIA)” guideline for the treatment of allergy. Persistent allergic rhinitis is defined by the persistence of symptoms for more than 4 days per week and more than 4 weeks per year [2].
Hyperbaric oxygen (HBO) therapy has been used successfully in humans to treat various diseases. The patients are kept in a hyperbaric chamber, where 100% oxygen is delivered to the patient at pressures 2-3 times higher than sea level atmospheric pressure, and the increased pressure is intended to increase the amount of dissolved oxygen in the plasma of patients [3]. Recent studies have shown that high concentrations of oxygen might be immunosuppressive in animals. Hansbrough et al. [4] have shown the decrease in circulating levels of total leukocytes and lymphocytes. Several studies performed with specimens of brain and muscles or flaps have demonstrated the decreased leukocyte adhesion with using hyperbaric oxygen [5-7]. Fibroblast proliferation is similarly affected by the oxygen concentration [8].
There are different treatment options for allergic rhinitis. Disodium cromoglycate, ipratropium bromide, antihistaminics, nasal corticosteroids, systemic corticosteroids, antileukotrienes, specific immunotherapy can be used in the treatment of allergic rhinitis. However, these treatment modalities can frequently cause side effects such as nasal irritation, dryness, epistaxis and somnolence. Specific immunotherapy may also have serious side effects. Despite using combination therapy with oral antihistaminics and intranasal drugs, symptoms may not be adequately controlled. Side effects and drug interactions may be seen in patients with allergic rhinitis. Therefore, we need different treatment options for patients with allergic rhinitis. HBO treatment that demonstrated immunomodulatory effects may play a role in the mechanism of allergic rhinitis. The present study aimed to evaluate the effects of nasal steroid and HBO treatments on the nasal mucosa of an AR rat model.
Methods
Animal care and treatment (Compliance with Ethical Standards)
The present study had the approval of the ethics committee and complied with the guidelines for the care and use of experimental animals (2014/36). The study conformed to all regulations of the Helsinki Declaration on experimental studies.
A total of 30 healthy adult male Albino-Wistar rats each weighing 250-270 gr obtained from Department of Biology and Biomedical Application Techniques of Experimental Animals were included in the study. Since menstrual cycle can affect the hormonal changes of the nasal mucosa in female rats, male rats were used in our study. Before HBO treatment, the rats were taken to the Department of Underwater and Hyperbaric Medicine and kept at an appropriate temperature and humidity for 48 hours, then at 22 ± 2°C and 65-70% humidity and ad libitum conditions in a 12-hour light-dark cycle in an experimental room.
A total of 30 adult Albino-Wistar male rats were randomized into four groups. Group 1 (control group) (n=6) [this group of rats was not exposed to toluene diisocyanate (TDI) and hyperbaric conditions or enhanced oxygen concentrations]; Group 2 (TDI group) (n=8) was exposed to TDI; Group 3 (n=8) was exposed to TDI and treated with HBO; Group 4 (n=8) was exposed to TDI and treated with intranasal mometasone furoate (10 μl/d) [9]. As a steroid therapy, intranasal mometasone furoate (10 μl/d) was applied with micropipettes without any anesthetic premedication.
Toluene diisocyanate sensitization and provocation
Sensitization to TDI was achieved by the method described by Kitamura et al. [10] 10 ml of a 10% solution of TDI (Merck Millipore®, Darmstadt, Germany) in ethyl acetate was painted bilaterally on the nasal vestibule once a day for five consecutive days. Following an interval of 2 days, sensitization procedure was repeated. After an interval of 1-week, solution of 10% TDI was applied to the nasal vestibule to provoke nasal allergy-like symptoms in the TDIsensitized rats (for provocation).
HBO experimental procedure
The HBO treatment was administered in a small hyperbaric cabin that was designed for small animals with the capacity for 10 rats. Before the start of the HBO sessions, the hyperbaric cabin was ventilated with oxygen for 10 minutes to maintain 100% oxygen concentration within the cabin. The group of rats exposed to HBO underwent 30 sessions of HBO therapy at 2.5 ATA (1-atmosphere absolute-ATA) for 90 minutes including decompression and compression per session six days in a week. HBO treatment was started on the fifth day of sensitization and applied for 35 days in Group 3. At the end of the HBO therapy which lasted for 30 days, the rats were sacrificed after being anesthetized with ketamine hydrochloride (100 mg/kg, IP; Ketalar®, Eczacibasi Parke-Davis, Istanbul, Turkey) and Xylazine hydrochloride (10 mg/kg, IP; Alfazyne®, Alfasan International B.V., Woerden, The Netherlands).
Histopathological findings
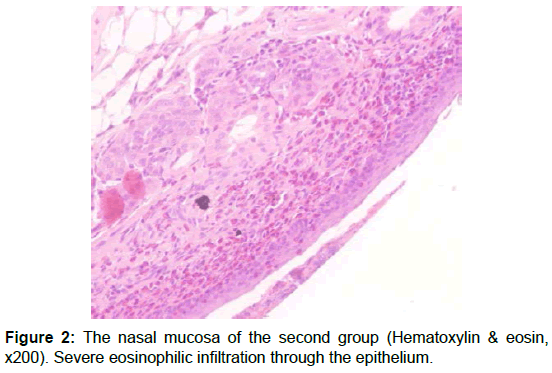
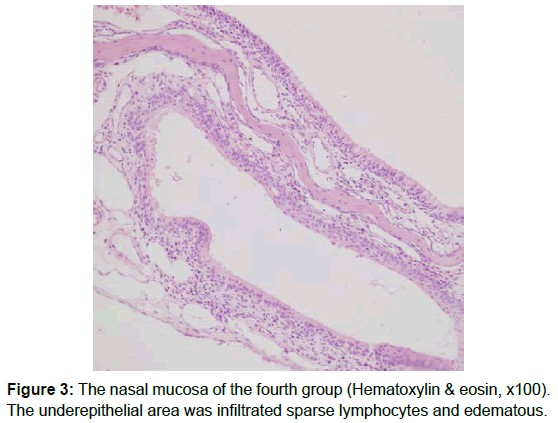
After the scarification, the rat heads were dissected along the nasal septum to expose the nasal cavity and the maxillary sinus. The mucosa of maxillary sinus and turbinates was carefully dissected from the bone. These mucosal specimens were fıxed in 10% formaldehyde solution in order of which was only known by the director of the study. Specimens were evaluated by a pathologist who was blinded to the allocation of the groups. Specimens were prepared as follows: after fixation in 10% neutral formalin, they were decalcified. Following routine paraffin processing, 5 μm sections were taken and stained with hematoxylin - eosin. The sections were then evaluated semi-quantitatively, using an Olympus BH-52 light microscope, and parameters of histopathological inflammation, fibrosis and eosinophilia were assessed. Fibrosis was classified based on fibroblastic cell count. Average of fibroblastic cell counts were calculated after examination of three different high-power fields under a light microscope. Fibroblastic cell count was scored as follows: 1-3 fibroblasts (+), 4-6 fibroblasts (++), 7-10 fibroblasts (+++), and >10 (++++). The degree of inflammation was categorized according to the intensity of the inflammation, and the number of inflammatory cells. Inflammatory activity was indicated as follows: (+) inflammatory cells dispersed as single cells (+) or clusters (++) throughout the stroma; (+++): inflammatory cells infiltrating the epithelium (Table 1) [11]. Average of eosinophil counts was calculated after examination of three different high-power fields under a light microscope.
Statistical analyses
Data was analyzed using the Statistical Package for the Social Sciences (SPSS) software program (22.0 for Windows) (SPSS Inc., Chicago, IL, USA). All differences associated with a chance probability of 0.05 or less were considered statistically significant. Statistical significance between groups for inflammation and fibrosis are tested with Fisher’s exact test. Statistical significance between groups for eosinophilia are tested with independent samples t-test.
Results
Intranasal application of TDI has induced nasal allergy-like behavior, such as sneezing and watery rhinorrhea, in TDI-sensitized rats. However, control rats showed no nasal allergy-like behavior during the study. Histopathological examinations were evaluated semi-quantitatively for inflammation and fibrosis (Table 1).
| Control group (n) | TDI group (n) | HBO group (n) | Steroid group (n) | ||
|---|---|---|---|---|---|
| Fibrosis | + | 7 | 3 | 7 | 8 |
| ++ | 1 | 5 | 1 | 0 | |
| +++ | 0 | 0 | 0 | 0 | |
| ++++ | 0 | 0 | 0 | 0 | |
| Inflammation | + | 5 | 0 | 4 | 3 |
| ++ | 1 | 1 | 3 | 4 | |
| +++ | 2 | 7 | 1 | 1 | |
Table 1: The distribution of inflammation and fibrosis scores in groups.
The level of inflammation in the control group was significantly less severe than Group 2 (p<0.05). Group 2 has significantly higher inflammation values than the Groups 3 and 4 (p= 0.010) (Table 2). There was no statistically significant difference between Groups 3 and 4 (p=1.00). Evaluation of the fibrosis scores showed that the scores of Group 4 were significantly higher (p<0.05). Group 1 has significantly lower eosinophil counts than the other groups (p<0.05) (Table 3). Groups 2 has significantly higher eosinophil counts than group 4 (p=0.034) (Figures 1-3).
| Group 1 | Group 2 | Group 3 | Group 4 | |
|---|---|---|---|---|
| Group 1 | - | 0.013* | 0.648* | 0.413* |
| Group 2 | 0.013* | - | 0.010* | 0.010* |
| Group 3 | 0.648* | 0.112* | - | 1.000* |
| Group 4 | 0.413* | 0.010* | 1.000* | - |
p<0.05
Table 2: Statistical analysis for inflammation between groups.
| Group 1 | Group 2 | Group 3 | Group 4 | |
|---|---|---|---|---|
| Group 1 | - | 0.01* | 0.003* | 0.006* |
| Group 2 | 0.01* | - | 0.112* | 0.034* |
| Group 3 | 0.003* | 0.112* | - | 0.116* |
| Group 4 | 0.006* | 0.034* | 0.116* | - |
p<0.05
Table 3: Statistical analysis for eosinophilia between groups.
Discussion
Some important functions are achieved by the nose and nasal cavities. The nasal cavity and mucosa humidify the air before it enters into the lungs. Nasal mucosal inflammation, edema, and increased mucus production occur in allergic rhinitis. Genetic predisposition to allergies leads to the development of AR. Repeated exposures to allergens lead to activation and maturation of B cells to plasma cells producing specific IgE antibodies. The basophils and mast cells introduce specific receptors to IgE. The activated mast cells release the preformed histamine and produce new mediators. Symptoms such as rhinorrhea, blockage, sneezing, and itching can occur in patients with AR.
Treatment options that were offered by the guideline have been shown to improve disease control [12]. ARIA includes qualityof- life measures to be used in the evaluation and treatment of AR and provides an evidence-based approach to the treatment of AR. The guideline has recently been updated using GRADE (Grading of Recommendations, Assessment, Development, and Evaluations) methodology [2,13]. Oral antihistamines, decongestants are often used to treat the patients with AR. However, patients with chronic symptoms can use an intranasal steroid spray regularly. Intranasal topical steroids are found to be effective in AR. Glucocorticoids show clinical efficacy with their ability to reduce eosinophil infiltration into the airway, viability and activation of inflammatory cells [14]. The other indications for intranasal steroids include acute rhinosinusitis, chronic rhinosinusitis with or without nasal polyps, postoperative treatment of nasal polyps and/or chronic rhinosinusitis and AR [15]. Systemic bioavailability of intranasal steroids depends on the type of steroid used. Steroids have some side effects on growth, eyes, bone and the hypothalamic- pituitary- adrenal axis. Steroids such as mometasone furoate and fluticasone furoate have low systemic bioavailability. Thus, these intranasal steroids are considered safer than the others [15]. Hyperbaric oxygen therapy provides 100% oxygen for inhalation at greater than one atmosphere absolute (ATA) in a chamber. HBO therapy increases the oxygen content of plasma. Some of the studies have reported that HBO treatment leads to changes in respiratory tracts. Long-term HBO therapy caused mild inflammation on the nasal mucosa [11,16]. Several cell-mediated reactions were suppressed, including allergic encephalomyelitis [17]. Recent studies have reported that HBO also has potent antiinflammatory effects [18,19]. Recent studies have shown that with HBO therapy tissue edema and histopathological changes occur and also levels of IL-6, IL-1 and TNF-alpha decrease [20].
There are many studies on mechanisms of AR. However, there is not enough information about the effects of HBO therapy on AR. In this study, we created AR model and observed the effects of HBO therapy on the allergic nasal mucosa. We applied TDI solution to the nasal vestibule to provoke nasal allergy-like symptoms. TDI is a chemical compound used in the plastics industry. TDI may cause asthma and nasal allergy in industrial workers. TDI induces nasal allergy-like behavior, such as watery rhinorrhea and sneezing. In the present study, inflammatory reaction in the nasal mucosa was more prominent in the TDI-sensitized rats. However, there was no statistically significant difference between nasal steroid and HBO therapy groups. Eosinophilia is the most common finding of AR on the nasal mucosa. There was significant eosinophilia in the TDIsensitized rats. Eosinophilia scores were similar in the nasal steroid and HBO therapy groups. Fibrosis scores in the nasal mucosa were more prominent in the nasal steroid group.
Edema is an abnormal accumulation of fluid in the intersticium as a result of changes in hydrostatic and oncotic pressures. Cellular activity in the inflamed tissue is more intense relative to normal tissue. Glucocorticoids reduce airway eosinophil infiltration, viability, and activation of inflammatory cells. Hansbrough et al. [4] have shown that hyperbaric treatment increases endogenous glucocorticoid levels. HBO therapy may supply too much adenosine triphosphate for cellular activation. HBO therapy can increase the amount of oxygen in tissue and blood with resultant reduction of vascular permeability [17].
Conclusion
Our study has shown that HBO treatment has anti-inflammatory effects on the allergic nasal mucosa. Anti-inflammatory effects of both nasal steroids and HBO treatments were found to be similar. These results have led us to believe that nasal steroid therapy combined with HBO therapy might be used for the treatment-resistant patients with persistent AR. Further studies are required for explaining the mechanisms HBO treatment of AR.
Acknowledgements
The English in this document has been checked by at least two professional editors, both native speakers of English.
References
- White MV, Kaliner MA (1992) Mediators of allergic rhinitis. J Allergy Clin Immunol 90: 699-704.
- Scadding GK, Durham SR, Mirakian R, Jones NS, Leech SC, et al. (2008) BSACI guidelines for the management of allergic and non-allergic rhinitis. Clin Exp Allergy 38: 19-42.
- Narozny W, Sicko Z, Stankiewicz CZ, Przewoźny T, Pegiel-Sićko E (2002) The effects of hyperbaric oxygen on nasal mucociliary transport. Clin Otolaryngol Allied Sci 27: 140-146.
- Hansbrough JF, Piacentine JG, Eiseman B (1980) Immunosuppression by hyperbaric oxygen. Surgery 87: 662-667.
- Zamboni WA, Roth AC, Russell RC, Graham B, Suchy H, et al. (1993) Morphologic analysis of the microcirculation during reperfusion of ischemic skeletal muscle and the effect of hyperbaric oxygen. Plast Reconstr Surg 91: 1110-1123.
- Tjärnström J, Wikström T, Bagge U, Risberg B, Braide M (1999) Effects of hyperbaric oxygen treatment on neutrophil activation and pulmonary sequestration in intestinal ischemia-reperfusion in rats. Eur Surg Res 31: 147-154.
- Thom SR (2004) Effects of hyperoxia on neutrophil adhesion. Undersea Hyperb Med 31: 123-131.
- Niinikoski J (1969) Effect of oxygen supply on wound healing and formation of experimental granulation tissue. Acta Physiol Scand Suppl 334: 1-72.
- Tsumuro T, Ogawa M, Minami K, Takubo M, Rahman A, et al. (2005) Effects of mometasone furoate on a rat allergic rhinitis model. Eur J Pharmacol 524: 155-158.
- Kıtamura Y, Miyoshi A, Murata Y, Kalubi B, Fukui H, et al. (2004) Effect of Glucocorticoid on Upregulation of Histamine H1Receptor mRNA in Nasal Mucosa of Rats Sensitized by Exposure to Toluene Diisocyanate. Acta Otolaryngol 124: 1053-1058.
- Vera-Cruz P, Ferreira M, Zagalo C, dos Santos JM, Aguas AP (2010) Structure of the rat nasal mucosa after acute and chronic hyperbaric oxygen therapy. Undersea Hyperb Med 37: 125-132.
- Rondón C, Canto G, Blanca M (2010) Local allergic rhinitis: a new entity, characterization and further studies. Curr Opin Allergy Clin Immunol 10: 1-7.
- Bousquet J, Lund VJ, van Cauwenberge P, Bremard-Oury C, Mounedji N, et al. (2003) Implementation of guidelines for seasonal allergic rhinitis: a randomized controlled trial. Allergy 58: 733-741.
- Zhang X, Moilanen E, Adcock IM, Lindsay MA, Kankaanranta H (2002) Divergent effect of mometasone on human eosinophil and neutrophil apoptosis. Life Sci 71: 1523-1534.
- Potter PC, Pawankar R (2012) Indications, efficacy, and safety of intranasal corticosteriods in rhinosinusitis. World Allergy Organ J 5: 14-17.
- Vuralkan E, Cobanoglu HB, Arslan A, Arslan S, Mungan S, et al. (2014) Effects of topical nasal steroids and diclofenac on the nasal mucosa during hyperbaric oxygen therapy: a double-blind experimental study. Eur Arch Otorhinolaryngol 271: 2213-2217.
- Warren J, Sacksteder MR, Thuning CA (1978) Oxygen immunosuppression: modification of experimental allergic encephalomyelitis in rodents. J Immunol 121: 315-320.
- Benson RM, Minter LM, Osborne BA, Granowitz EV (2003) Hyperbaric oxygen inhibits stimulus-induced proinflammatory cytokine synthesis by human blood-derived monocyte-macrophages. Clin Exp Immunol 134: 57-62.
- Al-Waili NS, Butler GJ (2006) Effects of hyperbaric oxygen on inflammatory response to wound and trauma: possible mechanism of action. ScientificWorldJournal 6: 425-441.
- Weisz G, Lavy A, Adir Y, Melamed Y, Rubin D, et al. (1997) Modification of in vivo and in vitro TNF-alpha, IL-1, and IL-6 secretion by circulating monocytes during hyperbaric oxygen treatment in patients with perianal Crohn's disease. J Clin Immunol 17: 154-159.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi