Research Article, J Pharm Drug Deliv Res Vol: 5 Issue: 2
Multifunctional Bile Acid Derivatives as Efficient RNA Transporters (Carriers)
| Liudmila Vasina1,2, Bhupendra C Reddy1, Elina Sievänen3 Erkki Kolehmainen3 and Vladimir Král1,2* | |
| 1University of Chemistry and Technology, Technická 5, 16628 Prague 6, Czech Republic | |
| 2First Faculty of Medicine, Charles University in Prague, Kateřinská 32, 12108 Praha, Czech Republic | |
| 3Department of Chemistry, P.O. Box 35, FI-40014 University of Jyväskylä, Finland | |
| Corresponding author : Vladimir Král
University of Chemistry and Technology, Technická 5, 16628 Prague 6, Czech Republic Tel: +420 220 444 298 Fax: +420 220 444 352 E-mail: vladimir .kral@vscht.cz |
|
| Received: December 31, 2016 Accepted: May 31, 2016 Published: June 02, 2016 | |
| Citation: Vasina L, Reddy BC, Sievänen E, Kolehmainen E, Král V (2016) Multifunctional Bile Acid Derivatives as Efficient RNA Transporters (Carriers). J Pharm Drug Deliv Res 5:2. doi:10.4172/2325-9604.1000147 |
Abstract
Multifunctional Bile Acid Derivatives as Efficient RNA Transporters (Carriers)
Abstract
RNA interference (RNAi) is an emerging technology that is revolutionizing many strategic approaches to biochemical pathway analysis, drug discovery, and therapy. The siRNA can be used to suppress a specific target. Even though these molecules may have potential and strong utility function, many limitations make their clinical application difficult, including delivery problems, side effects due to off-target actions, disturbance of physiological functions of the cellular machinery involved in gene silencing, and induction of the innate immune response. This paper focus on efficient RNA transports using designed carriers, guanidine steroids. It presents the synthesis of guanidine substituted bile acids (cholic acid, deoxycholic acid and lithocholic acid) by a simple method with moderate yield. The guanidine substituted compounds are shown to be very efficient tools in generating cellular delivery vehicles as determined by in vitro studies using PAMPA (parallel artificial membrane permeability) assay.
Keywords: Bile acids; Guanidine; RNA transport; Parallel artificial membrane permeability; siRNA
Keywords |
|
| Bile acids; Guanidine; RNA transport; Parallel artificial membrane permeability; siRNA | |
Introduction |
|
| RNAi is a sequence-specific endogenous gene-silencing mechanism conserved in many organisms to regulate gene expression [1-10]. Two types of small ribonucleic acid (RNA) molecules – microRNA (miRNA) and small interfering RNA (siRNA) – are central to RNA interference. RNAs are the direct products of genes, and these small RNAs can bind to other specific messenger RNA (mRNA) molecules and either increase or decrease their activity, for example by preventing an mRNA from producing a target protein. The specificity of RNAi also allows the inhibition of previously undruggable targets. The ubiquity of the RNAi pathway within the body and the ease with which siRNA can be used to suppress a specific target of interest make siRNAs a promising class of molecules for the treatment of cancer, viral infections, ocular disorders, and genetic diseases [5,11,12]. However, one serious limitation for clinical application is a delivery problem. For an active distribution of a drug, is necessary to develop its carrier loading, targeting, and transport using foolproof drug delivery. Despite common properties delivery of siRNA faces specific challenges due to apparent differences in size, stability of the formed nucleic acid complexes, the location and mechanism of action [13-28]. The ability of a drug or probe to cross a biological barrier has historically been viewed to be a function of its intrinsic physical properties. This view has largely restricted drug design and selection of agents within a narrow logP range. Molecular transporters offer a strategy to circumvent these restrictions. For example guanidinium-rich transporters (GRTs), typically highly water-soluble conjugates are able to readily pass through the nonpolar membrane of a cell and even cross tissue barriers. For many applications, the transporter-drug conjugate can be designed as a prodrug, with a linker that is chemically or biochemically cleaved after passage through a barrier, allowing for release of the free drug in targeted cells or tissue. Guanidinium groups are well-suited for adherence to cell surfaces bearing negatively-charged groups. The association would allow for increased residence time and therefore favor internalization. The association, though relatively strong, must be reversible to allow release after entry. Though there are many possible mechanisms for GRT uptake, implication of endocytosis is pivotal. To be therapeutically active, GRT conjugate has to escape from endosomes. Such attempt has been made including the cargo release from transporter. Once endosomal escape has been achieved in many cases it is still necessary to release the cargo from the transporter [13]. The tissue uptake of GRTs is of critical importance; therefore the potential of polyguanidinium transporters to enhance bioavailability of biotinylated oligoarginine in mice has been investigated [14]. GRTs have been shown to penetrate many types of tissue, calling attention to their selectivity. Such can be achieved by local administration or by targeted delivery system. One strategy to achieve selectivity is the use of local administration. This has potentially wide application as lung, buccal, ocular, skin and many other sites are readily reached by direct administration of a drug. GRTs show uptake in lung tissue when inhaled, as well as penetration of both the epidermal and dermal layers when applied topically. In addition to local delivery, a targeted delivery system can also be utilized to achieve specificity. This Targeted delivery approach has been demonstrated by using a prodrug strategy in which a transporter is transiently disabled by intramolecular interaction with a polycarboxylate connected to the transporter through a peptide link. The guanidinium-rich molecular transporters facilitate or enhance the delivery of a variety of cargos, including small molecules, metals, imaging agents, peptides, plasmids, and proteins across biological barrier [15,16]. Therefore, we investigated whether guanidinium-rich side chains can also bind siRNA through electrostatic and hydrogen-bonding associations and hydrophobic side chains to facilitate packing and cellular entry as effective siRNA complexation and delivery vehicles. Similarly, steroids represent an important class of natural products due to their high ability to penetrate cells and bind to nuclear and membrane receptors. Bile acids are steroidal acids found predominantly in the bile of mammals and other vertebrates. The cleavage of the peptide bond is presumably carried out in nature by an enzyme. In the laboratory, it is accomplished by an alkaline hydrolysis. The fact that minor changes in steroid structures can cause extensive changes in biological activity intrigued medicinal chemists. Naturally occurring steroid nuclei have been modified in several ways with the aim to find more active compounds, free from undesirable or without harmful side effects,and of recognizing the and recognize structural and stereo-chemical features required for the display of specific and selective physiological activity. The replacement of one or more functional groups in bile acid molecule brings notable modifications of its biological activity. Local delivery of siRNA is advantageous for tissues that are external and/or locally restricted including ocular, epidermal, pulmonary, colonic, and pancreatic tissue. Additionally, local delivery may be suitable for non-invasive therapies that require patient administration, such as eye drops and nasal sprays [17]. Focus of this work is on combination of guanidine moiety (specific for phosphodiester binding) with hydrophobic steroid skeleton. Such structural fragments combined in carrier design should provide, not only stabilization of RNA, but also facilitate transmembrane transport. | |
Result and Discussion |
|
| Chemistry | |
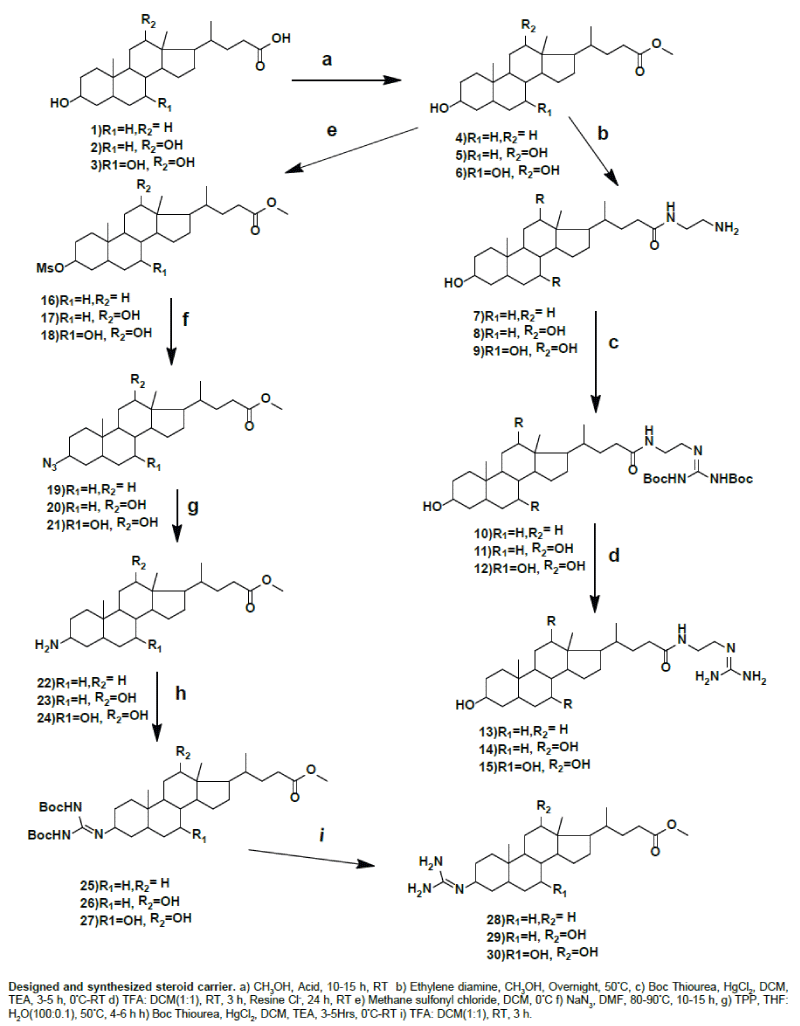
| We prepared guanidine substituted bile acids at C-3 position of methyl lithocholate, methyl deoxycholate and methyl cholate by using thiourea as reagent. In the preparation of guanidine substituted bile acids we follow several steps and used lithocholic acid, deoxycholic acid and cholic acid as starting material. We stabilize the reaction by using lithocholic acid. We esterified lithocholic acid and then C-3 hydroxy group is converted into leaving group by using methane sulfonyl chloride, which was confirmed by 1H NMR as a corresponding peak at 2.98 for OMS. The OMS substituted lithocholate reacted with sodium azide and then reduced to amine by using triphenyl phosphine. | |
| C-3 Amino methyl lithocholate reacted with Boc protected thiourea in the presence of HgCl2 from 0°C to RT for 3 h. Then the product is purified by using column chromatography and its structure was confirmed by using 1H NMR and 13CNMR and mass spectroscopy. In this Boc protons appear at 1.5 ppm and NH protons at 8.9 and 11.48 ppm. In 13CNMR C=N of Boc protected guanidine appear at 157.0 ppm. The guanidine was prepared by using TFA for deprotection of Boc. Guanidine compounds are confirmed by 1H NMR and 13C NMR and mass spectroscopy, in 13C NMR guanidine C=N appears at 157.7 ppm and the same procedure followed to methyl deoxycholate and methyl cholate, they are also confirmed by spectroscopic analysis. We also synthesized the C-26 guanidine substituted of lithocholyl ethyl amide, deoxycholyl ethyl amide and cholyl ethyl amide from bile acid esters of lithocholic acid, deoxycholic acid and cholic acid. Methyl lithocholate react with ethylene diamine and formed amido amine, which was reacted with Boc protected thiourea in presence of HgCl2 from 0°C to RT for 4 h. Then the product is purified by using column chromatography and its structure was confirmed by using 1H NMR and 13C NMR. In this Boc protons appear at 1.5 ppm and NH protons at 8.9.25 and 11.48 ppm. In 13C NMR C=N of guanidine, Boc C=O and NHC=O appear at 152.75, 156.96, and 174.12 ppm. The guanidine was prepared by using TFA for deprotection of Boc. The structures of guanidine compounds are confirmed by 1H NMR and 13C NMR and mass spectrometry, in 13C NMR guanidine C=N appears at 157 ppm and the same procedure followed to deoxycholyl ethyl amide and cholyl ethyl amide, they are also confirmed by spectroscopic analysis. | |
| Bioactivity | |
| In our experiment we use guanidine substituted bile acids in 5% methanol solution. In our experiment we use donor phase 300 μL solution, in this 275 μL PBS and 25 μL RNA. In acceptor, we use 200 μL PBS for control experiment. We incubated the plates for 6 h, and then we determined acceptor volume by UV-Vis spectroscopy for control experiment. In guanidine experiment, change the volume of PBS as 250 μL, 25 μL as guanidine bile acids and 25 μL RNA then incubated the plates for 6 h, then we calculated acceptor volume by UV-Vis spectroscopy, binding constant (K) were calculated from changes in absorbance of steroids (A) by nonlinear regression using the program letagroup spefo 2005 the values are given in the table below. We find good results for guanidine at C-26 of deoxycholyl ethyl amide and cholyl ethyl amide. Permeation of our membrane layer is strongly dependent on the pH, which used in the flux measurements. This indicates the importance of different pH values in permeation assays used in human bioavailability prediction [25]. | |
| Conclusion | |
| We synthesize guanidine substituted cholic acid, chenodeoxycholic acid, deoxycholic acid, and lithocholic acid. We studied RNA transport by using PAMPA assay with guanidine substituted bile acids, RNA from torula yeast and phosphate buffered saline (PBS). Our guanidine substituted compounds are proven to be a very efficient strategy to generate cellular delivery vehicles by studying PAMPA assay as in-vitro studies. | |
Experimental Section |
|
| Chemicals were from Sigma Aldrich and used without further purification. All solvents used for the spectroscopic and other physical studies were reagent grade and were further purified by literature methods (24). 1H and 13C NMR spectra were recorded in CDCl3 and MeOD-solutions on a Bruker Avance DRX 500 MHz (also 300 MHz 1H NMR data are given?) spectrometer equipped with a 5 mm diameter, broad band inverse detection probehead operating at 500.13 MHz in 1H and 125.77 MHz in 13C. The 1H and 13C NMR chemical shifts were expressed in parts per million (ppm) with reference to tetramethylsilane (TMS) as an internal standard. LC mass spectra were recorded on a Jeol SX 102 DA/600 mass spectrometer. Elemental analysis was performed on Thermo Finnigan Insturment. | |
| Synthesis | |
| Synthesis of methyl 3α, -hydroxy-5βb-cholane- 24-oate (4), methyl 3α, and 12α -dihydroxy-5b-cholane- 24-oate (5) and methyl 3α, 7α, 12α -trihydroxy 5βcholane- 24-oate (6): Compounds 4, 5 and 6 were synthesized in overall good yield starting from lithocholic acid, deoxycholic acid and cholic acid using the literature procedure [17]. | |
| Methyl 3α, -hydroxy-5β-cholane- 24-oate (4): White solid; yield=98%; 1H NMR (CDCl3, 300 MHz): δ 0.64 (s, 3H, CH3-18), 0.92 (s, 3H, CH3-19), 0.98 (d, J=6.0 Hz, 3H, CH3-21), 3.66 (s, 3H), 3.98 (br s, 1H, CH-3). | |
| Methyl 3α,12α -dihydroxy-5b-cholane- 24-oate (5): White solid; yield=95%; 1H NMR (CDCl3, 300 MHz): δ 0.68 (s, 3H, CH3-18), 0.91 (s, 3H, CH3-19), 0.98 (d, J=6.0 Hz, 3H, CH3-21), 3.62 (m, 1H, CH-12), 3.67 (s, 3H), 3.98 (br s, 1H, CH-3). | |
| Methyl 3α,7α,12α -trihydroxy-5βcholane- 24-oate (6): White solid; yield=97%; 1H NMR (CDCl3, 300 MHz): δ 0.68 (s, 3H, CH3- 18), 0.89 (s, 3H, CH3-19), 0.98 (d, J=6.0 Hz, 3H, CH3-21), 3.57 (m, 1H, CH-7), 3.67 (s, 3H), 3.89 (br s, 1H, CH-12), 4.01 (br s, 1H, CH-3). | |
| Synthesis of methyl-3α -mesyloxy-5β-cholane-24-oate (16), methyl-3α -mesyloxy-12α -hydroxy-5β-cholane-24-oate (17), methyl-3α -mesyloxy-7α -12α -dihydroxy-5β-cholane-24-oate [18,19,22]: To a solution of 4,5,6 (1 mmol) in dry CH2Cl2 (20 mL) was added triethylamine (mL, 0.1 mmol) at °C. Methane sulfonyl chloride (1.5 mmol in 10 mL CH2Cl2) was added dropwise in 10 min at 0°C, and ice was added to the reaction mixture immediately after addition was complete. The reaction mixture was extracted with CH2Cl2. The organic layer was washed with NaHCO3, water, and brine. The solvent was evaporated under reduced pressure. The crude product was purified by column chromatography (0.5% MeOH/CH2Cl2) to obtain pure products 16, 17 and 18. | |
| Synthesis of methyl-3α-mesyloxy-5β-cholane-24-oate (16): White solid; yield=85%; 1H NMR (CDCl3, 300 MHz): 0.65(s, 3H, CH3-18), 0.90 (s, 3H, CH3-19), 0.91 (d, J=6.1 Hz, 3H, CH3-21), 2.98(s, 3H, OMS), 3.65 (s, 3H), 4.56 (m, 1H, CH-3). | |
| Methyl-3α-mesyloxy-12α-hydroxy-5β-cholane-24-oate [17]: White solid; yield=83%; 1H NMR (CDCl3, 300 MHz): 0.65(s, 3H, CH3-18), 0.90 (s, 3H, CH3-19), 0.93 (d, J=6.0 Hz, 3H, CH3-21), 3.00(s, 3H, OMS), 3.66 (s, 3H), 3.94(s,1H, C-12) 4.60 (m, 1H, CH-3). | |
| Methyl-3α-mesyloxy-7α, 12α-dihydroxy-5β-cholane-24-oate [18]. White solid; yield=82%; 1H NMR (CDCl3, 300 MHz): 0.69(s, 3H, CH3-18), 0.91 (s, 3H, CH3-19), 0.98 (d, J=6.2 Hz, 3H, CH3-21), 2.98(s, 3H, OMS), 3.66 (s, 3H), 3.87 (m, 1H, C-12), 4.00 (m,1H, C-7) 4.46 (m, 1H, CH-3). | |
| Synthesis of methyl-3-azido-5β-cholane-24-oate [19], methyl- 3-azido-12α-hydroxy-5β cholane-24-oate (20), methyl-3-azido- 7α,12α dihydroxy-5β -cholane-24-oate (21)[19,22]: To a solution of 16,17,18 (1 mmol) in dry DMF (10 mL), sodium azide (5 mmol) was added and stirring was continued at 60–65 °C for 3–5 h. The reaction mixture was allowed to cool to room temperature. It was then poured into ice-cold water (30 mL) and extracted with EtOAc. The organic extract was washed with cold water and brine. Solvent was evaporated under reduced pressure to afford crude products 19, 20 and 21 which was purified by column chromatography on silica gel (10% EtOAc/ hexane) to produce pure compounds 19, 20 and 21. | |
| Synthesis of methyl-3-azido-5β-cholane-24-oate (19): White solid; yield=90%; 1H NMR (CDCl3, 300 MHz): 0.65(s, 3H, CH3-18), 0.91 (s, 3H, CH3-19), 0.93 (d, J=6.0 Hz, 3H, CH3-21), 3.66 (s, 3H), 3.95 (m, 1H, CH-3). | |
| Methyl-3-azido-12α-hydroxy-5β-cholane-24-oate (20): White solid; yield=89%; 1H NMR (CDCl3, 300 MHz): 0.65(s, 3H, CH3-18), 0.91 (s, 3H, CH3-19), 0.94 (d, J=6.2 Hz, 3H, CH3-21), 3.66 (s, 3H), 3.90(s,1H, C-12) 3.95 (m, 1H, CH-3). | |
| Methyl-3-azido-7α,12α dihydroxy-5β-cholane-24-oate (21): White solid; yield=87%; 1H NMR (CDCl3, 300 MHz): 0.69(s, 3H, CH3-18), 0.93 (s, 3H, CH3-19), 0.98 (d, J=6.3 Hz, 3H, CH3-21), 3.66 (s, 3H),3.87 (m, 1H, C-12), 3.98(bs,1H, C-7) 3.96 (m, 1H, CH-3). | |
| General procedure for the preparation of methyl-3-amino-5β- cholane-24-oate (22), methyl-3-amino-12α hydroxy-5β-cholane-24-oate (23), methyl-3-amino-7α, 12α dihydroxy-5β-cholane-24-oate (24) [20]: The azide (1 mmol) and PPh3 (1.5 mmol) were stirred in THF (5 mL) and H2O (0.1 mL) at 50°C. The reaction was monitored by TLC and was complete in 10–24 h. Solvents were removed by rotary evaporation. The residue was purified by column chromatography over silica gel. | |
| Methyl-3-amino-5β-cholane-24-oate (22). White solid; yield=83%; 1H NMR (MeOD, 300 MHz): 0.69 (s, 3H, CH3-18), 0.93 (d, J=6.6 Hz, 3H, CH3-21), 0.99 (s, 3H, CH3-19), 3.64 (s, 3H), 3.2 (s, 1H, CH-3). | |
| Methyl-3-amino-12α-hydroxy-5β-cholane-24-oate (23). White solid; yield=87%; 1H NMR (MeOD, 300 MHz): 0.66(s, 3H, CH3-18), 0.95 (m, 6H, CH3-19, CH3-21), 3.35(s,1H, C-12), 3.64 (s, 3H), 3.95 (m, 1H, CH-3); | |
| Methyl-3-amino-7α, 12α-dihydroxy-5β-cholane-24-oate (24). White solid; yield=85%; 1H NMR (MeOD, 300 MHz): 0.69 (s, 3H, CH3-18), 0.97 (s, 3H, CH3-19), 0.98 (d, J=6.0 Hz, 3H, CH3-21), 3.65 (s, 3H),3.86 (m, 1H, C-12), 3.98(bs,1H, C-7) 3.99 (s, 1H, CH-3); | |
| N, N′--Bis-tert-butoxycarbonylthiourea (23) | |
| To a stirred solution of thiourea (571 mg, 7.50 mmol) in THF (150 mL) under argon at 0 °C were added hexane and sodium hydride (1.35 g, 33.8 m mol, 60% in mineral oil). After 5 min, the ice bath was removed, and the reaction mixture was stirred at room temperature for 10 min. The mixture was cooled to 0 °C again, ditert- butyl dicarbonate (3.60 g, 16.5 mmol) was added, and the ice bath was removed after 30 min of stirring at that temperature. The resulting slurry was stirred for another 2 h at room temperature. Then the reaction was quenched with an aqueous solution of saturated NaHCO3 (10 mL). The reaction mixture was poured into water (250 mL) and extracted with EtOAc (3 ×70 mL). The combined organic layer was dried over Na2SO4, filtered, and concentrated in vacuo to give N, N′--di-tert-(butoxycarbonyl)thiourea as a white solid (86% yield). Mp 127-129 °C, 1H NMR (CDCl3) 1.50 (brs, 18 H) (Figure 1). | |
| Figure 1: PAMPA conditions study of transport: carrier concentration 0.1 mmol/l, RNA concentration 0.05 mmol/l, λ 256 nm | |
| General Procedure for the Synthesis of Methyl-3-Boc-protected guanidine-5β-cholane-24-oate (25), methyl-3- Boc-protected guanidine-12α-hydroxy-5β-cholane-24-oate (26), methyl-3- Bocprotected guanidine-7α,12α-dihydroxy-5β-cholane-24-oate (27) [21]: Each of the corresponding anilines was treated in DCM at 0 °C with 1.1 equiv of mercury (II) chloride, 1.0 equiv of N,N′-di-(tertbutoxycarbonyl) thiourea and 3.1 equiv of TEA. The resulting mixture was stirred at 0°C for 1 h and for the appropriate duration at room temperature. Then, the reaction mixture was diluted with EtOAc and filtered through a pad of celite to get rid of the mercury sulfide formed. The filter cake was rinsed with EtOAc. The organic phase was washed first with water (2 × 30 mL) and then with brine (1 × 30 mL), dried over anhydrous Na2SO4, and concentrated under vacuum to give a residue that was purified by silica gel, eluting with the appropriate DCM: MeOH mixture. | |
| Methyl-3-Boc-protected guanidine-5β-cholane-24-oate (25). White solid; yield=83%; 1H NMR (CDCl3, 500 MHz): 0.65(s, 3H, CH3-18), 0.91 (d, J=10.5 Hz, 3H, CH3-21), 0.99 (s, 3H, CH3-19), 1.50(s, 18H, Boc), 3.66 (s, 3H),4.54 (s, 1H, CH-3); 8.9 (s, 1H, BocNH), 11.48(s, 1H, BocNH).; 13C NMR (126 MHz; CDCl3) : 12.04, 18.27, 21.06, 24.16, 24.96, 26.22, 26.76, 28.08, 28.09, 28.32, 30.67, 31.04, 31.24, 35.05, 35.35, 35.68, 37.86, 39.90, 40.10, 42.75, 51.43, 55.93, 56.43, 153.28, 157.07, 174.73. | |
| Methyl-3- Boc-protected guanidine-12α-hydroxy-5β-cholane- 24-oate (26): White solid; yield=80%; 1H NMR (CDCl3, 500 MHz): 0.68(s, 3H, CH3-18), 0.97 (m, 6H, CH3-19, CH3-21), 1.50 (s, 18H, Boc), 3.66 (s, 3H), 3.98(s,1H, C-12), 4.49 (br s, 1H, CH-3); 8.9(s, 1H, BocNH), 11.48(s, 1H, BocNH).; 13C NMR (126 MHz; CDCl3) : 12.74, 17.34, 23.59, 23.86, 24.88, 25.91, 26.67, 27.40, 28.08, 28.29, 28.97, 30.62, 30.91, 31.06, 33.12, 34.60, 35.88, 37.85, 46.51, 47.36, 48.24, 51.47, 73.13, 152.63, 156.96, 174.67. | |
 |
|
| Methyl-3- Boc-protected guanidine-7α, 12α-dihydroxy-5β- cholane-24-oate (27). White solid; yield=76%; 1H NMR (CDCl3, 500 MHz): 0.70(s, 3H, CH3-18), 0.99 (m, 6H, CH3-19, CH3-21), 1.49 (d, J=4Hz, 18H, Boc), 3.66 (s, 3H), 3.86 (s, 1H, C-12), 3.98(br s,1H, C-7) 4.41 (br s, 1H, CH-3); 8.89(s, 1H, BocNH), 11.48(s, 1H, BocNH).; 13C NMR (126 MHz; CDCl3) : 13.09, 17.83, 23.62, 23.70, 25.23, 26.62, 27.84, 28.50, 28.71, 29.09, 31.27, 31.43, 33.97, 34.62, 35.49, 37.66, 39.87, 42.31, 46.80, 47.42, 51.68, 68.33, 72.91, 152.65, 157.74, 173.87. | |
| General Procedure for the Synthesis of the methyl-3-guanidine- 5β-cholane-24-oate (28), methyl-3- guanidine -12α hydroxy-5β- cholane-24-oate (29), and methyl-3- guanidine-7α, 12α- dihydroxy- 5β-cholane-24-oate (30): Each of the corresponding Boc-protected precursors (0.5 mmol) were treated with 15 mL of a 50 % solution of trifluoroacetic acid in DCM for 3 h. Then neutralize with ammonia solution and extracted with chloroform. The organic phase was washed first with water (2 × 30 mL) and then with brine (1 × 30 mL), dried over anhydrous Na2SO4, and concentrated under vacuum to give a residue that was purified by silica gel, eluting with the appropriate DCM: MeOH mixture. | |
| Methyl-3-guanidine-5β-cholane-24-oate (28). White solid, yield=67%; 1H NMR (MeOD, 500 MHz): 0.69 (s, 3H, CH3-18), 0.92 (d, J=5.0 Hz, 3H, CH3-21), 1.02 (s, 3H, CH3-19), 3.65 (s, 3H),3.83 (s, 1H, CH- 3).; 13C NMR (126 MHz; MeOD) : 12.44, 18.71, 22.12, 24.32, 25.20, 25.75, 27.26, 27.72, 29.19, 31.56, 31.85, 32.20, 36.06, 36.68, 37.01, 38.56, 41.14, 41.41, 43.90, 48.99, 49.62, 52.00, 57.41, 57.79, 157.77, 176.43. | |
| Methyl-3-guanidine-12α-hydroxy-5β-cholane-24-oate (29). White solid, yield=70%; 1H NMR (MeOD, 500 MHz): 0.74(s, 3H, CH3-18), 1.01 (s, 3H, CH3-19), 1.02 (d, J=5.0 Hz, 3H, CH3-21), 3.67 (s, 3H), 3.84 (s,1H, C-12), 3.99 (br s, 1H, CH-3).; 13C NMR (126 MHz; MeOD) :11.73, 16.12, 22.62, 23.39, 24.19, 25.67, 26.31, 27.22, 28.59, 30.09, 30.16, 30.46, 30.79, 32.72, 34.25, 35.31, 35.76, 37.21, 46.20, 46.51, 46.68, 47.50, 50.58, 72.58, 156.36, 175.07. | |
| Methyl-3-guanidine-7α, 12α-dihydroxy-5β-cholane-24-oate (30). White solid, yield=65%; 1H NMR (MeOD, 500 MHz): 0.69 (s, 3H, CH3-18), 0.96 (s, 3H, CH3-19), 0.97-0.98 (d, J=5.0 Hz, 3H, CH3- 21), 3.66 (s, 3H),3.86 (s, 1H, C-12), 3.98 (bs,1H, C-7) 4.41 (br s, 1H, CH-3).; 13C NMR (126 MHz; MeOD) :11.73, 16.12, 22.62, 23.39, 24.19, 25.67, 26.31, 27.22, 28.59, 30.09, 30.16, 30.46, 30.79, 32.72, 34.25, 35.31, 35.76, 37.21, 46.20, 46.51, 47.50, 50.58, 68.32, 72.58, 156.36, 175.07. | |
| Synthesis of N-lithocholyl ethylenediamine (7), N-deoxycholyl ethylenediamine (8), N-cholyl ethylenedimine (9): Compounds 4, 5 and 6 (10 g, 23.66 mmol) were treated with an excess of ethylenediamine (15 ml) in methanol (50 ml). The reaction mixture was stirred at room temperature for 48 h, then poured over ice-cold water (400 ml); the solid obtained was filtered off, dried, and purified by recrystallization from chloroform–methanol to give the pure products 7, 8 and 9. | |
| Lithocholylethylenediamine (7): White solid; yield=86%; 1H NMR (300 MHz; MeOD) ): 0.71(s, 3H, CH3-18), 0.98 (s, 3H, CH3-19), 1.01 (d, J=6.0 Hz, 3H, CH3-21), 2.72 (t, J=6.3Hz 2H, NH2CH2),3.24 (t, J=6.3Hz 2H, NHCH2),) 3.97 (br s, 1H, CH-3). | |
| Deoxycholylethylenediamine (8). White solid; yield=85%; 1H NMR (300 MHz; MeOD) ): 0.70(s, 3H, CH3-18), 0.93 (s, 3H, CH3-19), 1.02 (d, J=6.3 Hz, 3H, CH3-21), 2.70(t, J=6.3Hz 2H, NH2CH2), 3.22(t, J=6.3Hz 2H, NHCH2),3.57 (m, 1H, C-12) 3.95 (br s, 1H, CH-3). | |
| Cholylethylenedimine (9): White solid; yield=85%; 1H NMR (300 MHz; MeOD) 0.71(s, 3H, CH3-18), 0.91(s, 3H, CH3-19), 1.03 (d, J=6.3Hz, 3H, CH3-21), 2.70(t, J=6.3Hz 2H, NH2CH2),3.24-3.20(t, J=3.6Hz 2H, NHCH2),3.31 (m, 1H, C-12) 3.78(bs,1H, C-7) 3.95 (br s, 1H, CH-3). | |
| General Procedure for the synthesis of Boc protected guanidine substituted at C-26 of lithocholyl ethyl amide (10), Boc protected guanidine substituted at C-26 of deoxycholyl ethyl amide (11), Boc protected guanidine substituted at C-26 of cholyl ethyl amide (12) [21]: Each of the corresponding anilines was treated in DCM at 0 °C with 1.1 equiv of mercury (II) chloride, 1.0 equiv of N,N′-di-(tert butoxycarbonyl)imidazolidine-2-thione (for the 2-aminoimidazoline precursors), or N,N′-di-(tert-butoxycarbonyl)thiourea (for the guanidine precursors) and 3.1 equiv of TEA. The resulting mixture was stirred at 0°C for 1 h and for the appropriate duration at room temperature. Then, the reaction mixture was diluted with EtOAc and filtered through a pad of celite to get rid of the mercury sulfide formed. The filter cake was rinsed with EtOAc. The organic phase was washed first with water (2 × 30 mL) and then with brine (1 × 30 mL), dried over anhydrous Na2SO4, and concentrated under vacuum to give a residue that was purified by neutral alumina column flash chromatography, eluting with the appropriate hexane: EtOAc mixture. | |
| Boc protected guanidine substituted at C-26 of lithocholyl ethyl amide (10): White solid; yield=82%; 1H NMR (500 MHz; CDCl3) ): 0.62(s, 3H, CH3-18), 0.91 (br s, 6H, CH3-19, CH3-21), 1.50(s, 18H, Boc) 3.42 (m, 2H, NH2CH2), 3.61 (m, 3H, NHCH2, CH-3), 7.29(s, 1H, CH2-NH), 8.76(s, 1H, Boc-NH), 11.43(s, 1H, Boc-NH).; 13C NMR (126 MHz; CDCl3): 12.04, 18.40, 20.83, 23.38, 24.22, 26.43, 27.21, 28.03, 28.25, 30.56, 31.71, 33.56, 34.59, 35.36, 35.56, 35.87, 36.49, 40.19, 40.44, 40.88, 42.12, 42.74, 56.14, 56.16, 71.86, 84.21, 152.88, 157.06 174.03. | |
| Boc protected guanidine substituted at C-26 of deoxycholyl ethyl amide (11): White solid; yield=78%; 1H NMR (500 MHz; CDCl3): 0.66(s, 3H, CH3-18), 0.90 (s, 3H, CH3-19), 0.98(d, J=6.0 3H, CH3-21), 1.50(s, 18H, Boc) 3.42 (m, 2H, NH2CH2), 3.63(m, 3H, NHCH2, CH- 12), 3.95-3.97(m, 1H, C-3 ), 7.41(s, 1H, CH2-NH), 8.84(s, 1H, Boc- NH), 11.42(s, 1H, Boc-NH).; 13C NMR (126 MHz; CDCl3): 12.73, 17.45, 23.12, 23.65, 26.12, 27.12, 27.48, 28.00, 28.21, 28.55, 30.46, 31.571, 33.45, 33.62, 34.12, 35.26, 36.02, 36.42, 40.71, 42.08, 46.52, 47.33, 48.21, 71.79, 73.15, 84.16, 152.75, 156.96, 174.12. | |
| Boc protected guanidine substituted at C-26 of cholyl ethyl amide (12): White solid; yield=77%; 1H NMR (500 MHz; CDCl3) ): 0.65 (s, 3H, CH3-18), 0.87 (s, 3H, CH3-19), 0.98(d, J=5.0 3H, CH3-21), 1.50(s, 18H,Boc) 3.38 (m, 3H, NH2CH2, CH-12), 3.53 (m, 2H, NHCH2,), 3.82(br s, 1H, C-7), 3.95 (m, 1H, C-3 ), 7.48(s, 1H, CH2-NH), 8.59(s, 1H, Boc-NH), 11.42(s, 1H, Boc-NH).; 13C NMR (126 MHz; CDCl3): 12.88, 17.78, 22.05, 23.65, 26.12, 27.12, 27.48, 28.00, 28.21, 28.85, 30.46, 29.14, 29.81, 33.22, 34.72, 35.77, 40.19.02, 41.16, 41.63, 46.38, 46.52, 47.33, 48.21, 68.41, 71.79, 73.03, 79.45, 83.46, 152.98, 157.24, 174.43. | |
| General Procedure for the Synthesis of Guanidine substituted at C-26 of lithocholyl ethyl amide (13), Guanidine substituted at C-26 of deoxycholyl ethyl amide (14), Guanidine substituted at C-26 of cholyl ethyl amide (15): Each of the corresponding Boc-protected precursors (0.5 m mol) was treated with 15 mL of a 50% solution of trifluoroacetic acid in DCM for 3 h. Then neutralize with ammonia solution and extracted with chloroform. The organic phase was washed first with water (2 × 30 mL) and then with brine (1 × 30 mL), dried over anhydrous Na2SO4, and concentrated under vacuum to give a residue that was purified by silica gel, eluting with the appropriate DCM: MeOH mixture. | |
| Guanidine substituted at C-26 of lithocholyl ethyl amide (13): White solid; yield=65%; δH (500 MHz; CDCl3) ): 0.71(s, 3H, CH3- 18), 0.96 (br s, 3H, CH3-19), 1.02 (d, J=5.0, 3H, CH3-21), 3.1(t, 2H, NH2CH2), 3.2 (t, 2H, NHCH2), 4.45 (m, CH-3).; 13C NMR (126 MHz; MeOD) :11.06, 17.44, 20.53, 22.51, 23.85, 26.24, 26.94, 27.88, 28.65, 29.77, 31.87, 32.66, 34.26, 35.06, 35.46, 35.83, 35.95, 38.55, 40.12, 40.48, 42.12, 42.51, 56.01, 56.53, 70.99, 157.30, 175.79. | |
| Guanidine substituted at C-26 of deoxycholyl ethyl amide (14): White solid; yield=63%; δH (500 MHz; CDCl3) ): 0.71(s, 3H, CH3- 18), 0.96 (s, 3H, CH3-19), 0.98 (d, J=5.0 3H, CH3-21), 3.02(m, 4H, NH2CH2), 3.56(m, 1H, C-12); 4.97(m, 1H, C-3 ).; 13C NMR (126 MHz; MeOD) :11.06, 17.43, 20.52, 22.22, 23.83, 25.72, 26.06, 27.88, 29.77, 31.35, 31.72, 32.53, 34.26, 35.05, 35.83, 37.99, 39.97, 40.48, 41.80, 42.11, 42.49, 56.301, 56.53, 70.98, 79.34, 157.51, 176.41. | |
| Guanidine substituted at C-26 of cholyl ethyl amide (15): White solid; yield=60%; 1H (500 MHz; CDCl3) ): 0.64(s, 3H, CH3-18), 0.85 (s, 3H, CH3-19), 0.91(d, J=6.5 3H, CH3-21), 3.33 (m, 3H, NH2CH2, CH- 12), 3.53 (m, 2H, NHCH2,), 3.82(br s, 1H, C-7), 4.97(m, 1H, C-3 ).; 13C NMR (126 MHz; MeOD) :12.58, 18.07, 23.09, 24.22, 26.35, 26.89, 27.59, 28.22, 31.35, 31.00, 32.54, 32.86, 33.67, 34.94, 35.59, 35.80, 39.39, 42.08, 42.84, 43.32, 46.39, 50.87, 72.14, 80.12, 82.82, 158.93, 177.47. Sulfonyl chloride, DCM, 0°C f) NaN3, DMF, 80-90°C, 10-15 h, g) TPP, THF: H2O (100:0.1), 50°C, 4-6 h) Boc Thiourea, HgCl2, DCM, TEA, 3-5Hrs, 0°C-RT i) TFA: DCM (1:1), RT, 3 hrs. | |
| Bioactivity | |
| siRNA: siRNA studied through parallel artificial membrane permeability assay (PAMPA) is a method which determines the permeability of substances from a donor compartment, through a lipid- infused artificial membrane into an acceptor compartment. PAMPA provides a high throughput, non-cell based method for predicting passive, transcellular intestinal absorption, the process by which the majority drugs enter circulation. PAMPA model (Table 1), phosphate buffered saline (PBPAMPA Assays) and ribonucleic acid from torula yeast were procured from Sigma Aldrich. The PAMPA evolution instrument from pION Inc. was used in this study. In PAMPA, a “sandwich” structure was formed by a 96-well microtiter plate and a 96-well filter plate from Millipore (IPVH, 125 μm thick filter, 0.45 μm pore).The stock solutions of the carriers samples were prepared at 50 mM concentrations in methanol and stored at 0°C before use. Before being added to a 96-well filter plate, the stock solution was diluted first with buffer to achieve a final sample concentration of 10∼50 μM and to reduce the methanol concentration below 5% (v/v). The PAMPA plate was filled with a 300 μl diluted carriers (Ccarrier = 100 m mol/l) + RNA (CRNA=50 m mol/l) solution to prepare the “donor” wells, and the 96-well filter plate containing a synthetic phospholipid membrane was then placed on the donor wells. The “acceptor” wells were filled with 200 μl of buffer solution and placed on top of the “sandwich”, and the PAMPA instruments were incubated at 25°C in a sealed and saturated humidity container for a specified time. After reaching the permeation time, the PAMPA plate sandwich was separated, and the amount of carriers in both the donor and acceptor compartments were measured by comparing the experimental spectra with the UV spectrum (220∼400 nm) obtained from reference standards. A transport guanidine steroid (carriers) with RNA was studied by UV-VIS spectrometry (Figure 2). Binding constants (K) were calculated from changes in absorbance of guanidine steroids (A, nm) by nonlinear regression using the program Letagroup spefo 2005. The PAMPA sandwich (BD Gentest!, USA) was incubated for 5h without stirring [22-30]. | |
| Table 1: PAMPA conditions study of transport. | |
| Figure 2: Carrier and pH dependency of transport efficacy. | |
| Influence of protein binding on carrier-siRNA nanoparticulate complexes: Binding of drugs, specifically hydrophobic drugs to plasma proteins dramatically influence drug availability (fraction of free drug available for interaction for biological target is limited). Novel feature is connected with drug delivery using nanoparticles, where interaction of nanoparticles with plasma proteins has been for long time neglected. Recent findings gave sufficient evidence, that drug delivery using nanoparticles often exhibit reduced bioavailability by formation protein complexes on nanoparticle surfaces. In biological fluids, proteins bind to the surface of nanoparticles to form a coating known as the protein corona, which can critically affect the interaction of the nanoparticles with living systems. Addition of a protein corona (PC) or protein adsorption layer on the surface of nanomaterials following their introduction into physiological environments may modify their activity, bio-distribution, cellular uptake, clearance, and toxicity. Observed discrepancy between the in vitro and in vivo results for many nanoparticle based drug delivery systems is due, in part, to the adsorption of proteins and other biomolecules to the NP’s surface upon exposure to the biological medium in vivo. The protein corona may also compromise the targeting efficiency of NPs that are functionalized with targeting ligands. In the other hand, protein corona for RNA nanoparticulate delivery system plays positive role, as was reported in literature. Our study describes initial formation of carrier-RNA complexes for stabilization of RNA and facilitated model transmembrane delivery with novel non-toxic steroid guanidine carriers. We formed initially carrier-RNA complex which upon addition of HSA forms ternary complex. This dynamic complex changes over the time over the time as we do expect that following mechanism: model plasma protein (HAS) actually helps to release RNA from carrier-RNA complexes, as liphophilic carrier is transferred to hydrophobic domain of HSA and free RNA is slowly released over period of several hours as indicated from UV-Vis binding study. Our data are consistent with findings reported in recent literature for positive effects of RNA delivery influenced by plasma proteins [30,31]. | |
| Transport efficiency and cytotoxicity of novel steroid carriers | |
| Primary leukemic cells (PLC) - 4T1 200 μm were incubated for 2h with our novel transport agents C (0,1-1-10 mmol/l). Preliminary toxicity dates performed on several cell lines (PLC- 4T1) showed no effect on the cell, which means our careers are not toxic and ready for in vitro and in vivo. The most efficient intracellular transport was detected with steroid carriers (Figures 3-5). RNA carrier application with will be tested in our laboratory. | |
| Figure 3: Transport efficiency using steroid guanidine carriers using PAMPA | |
| Figure 4: Study of ternary complex formation: comparison of absorption at 256 nm for HSA, RNA, Carrier15-RNA complex and ternary complex Carrier15-RNA-HSA. Concetration RNA 5 micrmolar, HSA form 5 to 50 micromolar, carrier15 15 micromolar in phosphate buffer | |
| Figure 5: Cytotoxicity of novel steroid carriers | |
Conclusion |
|
| While facilitated transport of DNA, RNA in vivo is based on selective protein complexes, application of alternative ways is of great interest considering pharmaceutical potential of DNA/ RNA fragment, mainly siRNA. We provide here synthetic carriers, designed such way that selective charge complementarity (guanidinephosphate) and lipophilicity enhancement provided by steroid skeleton will synergically function as efficient polyphosphate carriers. | |
| We present here series of steroid guanidine synthesis carries and testing of transport efficacy. In summary, we found that our novel synthetic carries indeed provide efficient transmembrane transport of RNA as was assessed by extensive PAMPA study. | |
| In vivo transmembrane study along these lines are in progress in our laboratory. | |
Acknowledgments |
|
| This work was supported by the Ministry of Education, Youth and Sports of Czech Republic within the LQ1604 National Sustainability Program II (Project BIOCEV-FAR) and by the project „BIOCEV“ (CZ.1.05/1.1.00/02.0109). This work was supported by the Ministry of Education Youth and Sports of the Czech Republic Grant No. LH14008 (Kontakt II). | |
References |
|
|
|
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi 
