Case Report, Med Microbiol Rep Vol: 6 Issue: 1
Mycobacterial Spindle Cell Pseudotumor Presenting as Peritoneal Masses in a Suspected Case of Human Immunodeficiency Virus
Adeel Zafar1*, Nausheen Yaqoob2, Hania Naveed2, Rabeea Shah3 and Saba Jamal4
1Department of Microbiology, The Indus Hospital and Health Network, Karachi, Pakistan
2Department of Histopathology, The Indus Hospital and Health Network, Karachi, Pakistan
3Department of Infectious Diseases, The Indus Hospital and Health Network, Karachi, Pakistan
4Department of Anatomic and Clinical Pathology, The Indus Hospital and Health Network, Karachi, Pakistan
*Corresponding Author: Adeel Zafar Department of Microbiology, TheIndus Hospital and Health Network, Karachi, Pakistan, Tel: 923215171090; E-mail: adeel.zafar@tih.org.pk
Received date: 01 December, 2022, Manuscript No. MMR-22-48857; Editor assigned date: 03 December, 2022, PreQC No. MMR-22-48857(PQ); Reviewed date: 17 December 2022, QC No. MMR-22-48857; Revised date: 01 February 2022, Manuscript No. MMR-22-48857(R); Published date: 04 March 2022, DOI: 10.4172/mmr.100039
Citation: Zafar A, Yaqoob N, Naveed H, Shah R, Jamal S (2022) Mycobacterial Spindle Cell Pseudotumor Presenting as Peritoneal Masses in a Suspected Case of Human Immunodeficiency Virus. Med Microbiol Rep 6:3.
Abstract
Mycobacterial spindle cell pseudotumor is a rare entity occurring in immuno-compromised patients mostly with acquired immunodeficiency syndrome, organ transplant and immuno-suppressive therapy. It occurs in various body sites and most commonly non-tuberculous mycobacteria are the causative agent followed by Mycobacterium tuberculosis. It is characterized by proliferation of spindle shaped histiocytes with acid fast bacilli. It histologically mimics spindle cell neoplasms including kaposi’s sarcoma. We present a case of Mycobacterial spindle cell pseudotumor in an immunocompromised patient with peritoneal mass and discuss the characteristic findings.
Keywords: Mycobacterial Spindle Cell Pseudotumor Presenting as Peritoneal Masses in a Suspected Case of Human Immunodeficiency Virus
Introduction
Mycobacterial Spindle cell Pseudotumor (MSP) is characterized by localized proliferation of spindle shaped histiocytes colonized by acid fast mycobacteria. It most commonly involves lymph nodes, but, extranodal sites including skin, lungs, brain and other visceral organs are on record. It occurs in immunocompromised states mostly in Human Immunodeficiency Virus (HIV) infection but non-HIV related immunocompromised states also predispose to this condition. MSP clinically and pathologically resembles mesenchymal tumors especially Kaposi Sarcoma. Both Non Tuberculous Mycobacteria (NTM) and tuberculous mycobacteria are responsibles for this condition. Most common pathogen is Mycobacterium avium intracellulare complex followed by tuberculous mycobacteria. We present a case of MSP in peritoneum due to tuberculous mycobacteria in an immunocompromised patient. Histopathologically the lesions were composed of spindle and epithelioid histiocytes, small foci of necrosis, and numerous Acid-Fast Bacilli (AFB) consistent with mycobacterial spindle cell tumor.
Ethical review
The individual case report does not require approval from institutional review board of ethical committee hence ethical review was not obtained. The consent from the patient’s guardian was obtained for the complete anonymity of his identity.
Case Report
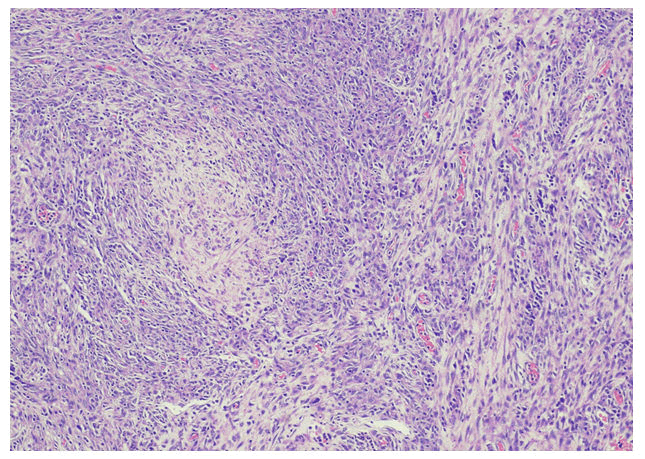
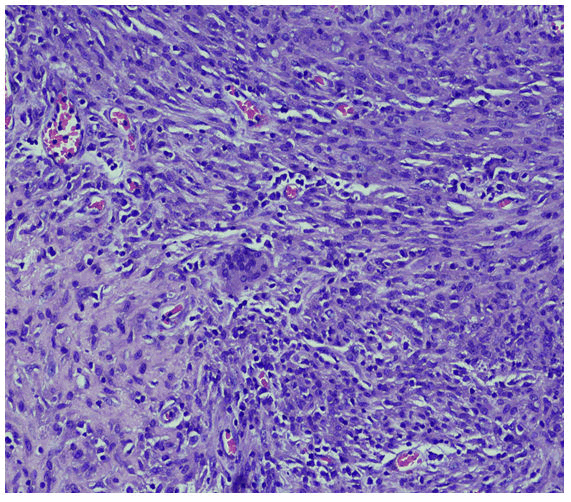
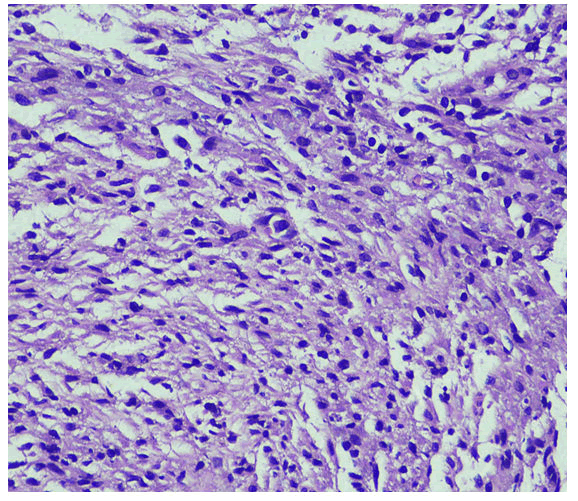
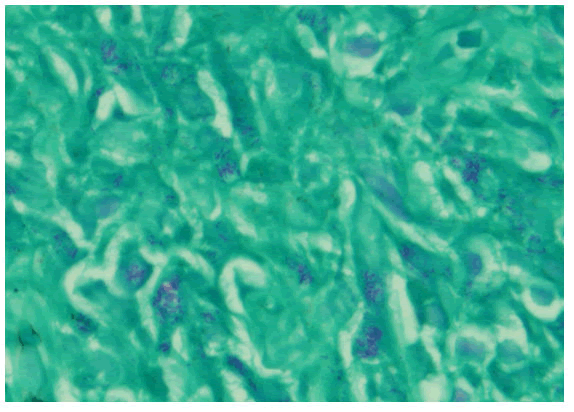
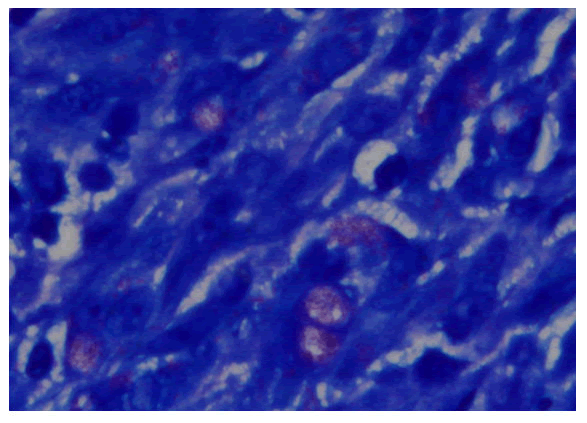
with complaints of generalized weakness, lethargy, vomiting, weight loss about 25-35 Kilograms, irritability and aggressive behavior for last four months [1]. He was diagnosed on presumed grounds from other hospital as abdominal Tuberculosis two months ago before presenting to our hospital. Anti-Tuberculous Therapy (ATT) 3 drug regimen was started. He took ATT for two months. Later the patient came with drug induced toxicity as shown on deranged LFTs (Total bilirubin 5.10 mg/dl, direct bilirubin 4 mg/dl, GGT 90 U/L, SGPT 62 U/L). He was unable to take orally and was drowsy so he was admitted in medical ICU. On examination he was cachectic and severely malnourished [2]. Vitally he was stable and blood pressure was 100/68 mmHg, temperature was 37°C, pulse was 98/minute and respiratory rate was 24/minute. At the time of admission his complete blood count showed hemoglobin 9.2 gm/dl, HCT: 26.6%, MCV: 84 fL, WBC: 4.3 × 103 /μl. Differential leukocyte count showed neutrophils 83.1%, lymphocytes 6.3%, monocytes 4.9%, eosinophils 4.1% and basophils 1.6%. His CD4 count was 153 cells/μL (normal 430-1740 cells/μL). Serum potassium was 1.5 mEq/L. He was started on second line ATT and was managed conservatively and remained admitted in hospital for ten days [3]. After 12 days he again came to ER with severe abdominal pain, altered sensorium and lethargy. Examination was suggestive of tender distended abdomen. General surgery team was taken on board and he underwent complex laparotomy. There were multiple and complex adhesions between grossly diseased bowel loops and omentum. Tissue from peritoneal nodule was sent for histopathological examination [4]. Samples from peritoneal pus were also sent for GeneXpert and microscopy. GeneXpert showed Mycobacterium tuberculosis and AFB smear from peritoneal pus showed 20 AFB on fluorescent staining. Patient also tested positive for COVID 19 and later the patient died on next day of admission. Histopathologically the lesions were composed of spindle and epithelioid histiocytes admixed with lymphoplasmacytic infiltrate (Figure 1) ill-defined granulomas, (Figure 2) small foci of necrosis, (Figure 3). Immunohistochemical stains ASMA and desmin were focally positive and CD68 highlighted sheets of histiocytes. Ziehl Neelson (ZN) stain and modified Ziehl Neelson (ZN) stain revealed numerous acid fast bacilli, (Figure 4 and Figure 5). It was diagnosed as Mycobacterial spindle cell pseudotumor [5].
Discussion
MSP is a rare condition and benign reactive lesion caused by both tuberculous mycobacetria and Non-Tuberculous Mycobacteria (NTM). Different case reports have been reported in literature in various sites of the body which includes both nodal and extranodal sites. Among the reported sites for MSP includes skin, lymph nodes, spleen, appendix, bone marrow and brain [6]. It mostly occurs in immunocompromised states which include HIV, diabetes mellitus, solid organ transplants, bone marrow transplant and immunosuppressive therapy. The first patient of MSP reported in literature presented with progressive swelling in hand in 1985 that underwent a cardiac transplant. Mycobacterium avium intracellulare complex was the causative pathogen identified in this case [7].
We present a case of a young male, who was suspected of having immunocompromised disease so HIV screening and CD4 cell count was done. HIV was non-reactive on screening serological test but CD4 cell count was significantly low. He also had persistently low lymphocyte count. In current case report, the causative agent identified was Mycobacterium tuberculosis which is a rare cause of MSP. Previously tubercular MSP has been reported in an Acquired Immunodeficiency Syndrome (AIDS) patient who presented with generalized lymphadenopathy. Biopsy of the lymph node showed spindle shaped histiocytes with numerous acid fast bacilli4. Another case of MSP caused by Mycobacyterium tuberculosis has been reported in renal transplant patient who was on immunosuppressive therapy and cutaneous MSP was diagnosed in this patient [8]. Xiaoyan et al reported MSP caused by Myobacterium tuberculosis in lymph node of 45-year-old male patient newly diagnosed with AIDS. It is mainly characterized by spindle shaped proliferation of histiocytes.
MSP closely resembles mesenchymal tumors particularly Kaposi Sarcoma (KS), other differential diagnoses include nodular fasciitis, benign fibrous histiocytoma, inflammatory myofibroblastic tumor, inflammatory pseudotumor, follicular dendritic cell sarcoma, spindle cell melanoma, myogenic or neural proliferations and neuroendocrine tumors. Therefore it has to be distinguished as all these pathologic conditions have different clinical courses, treatment and outcomes. Acid fast stain can reliably distinguish between these conditions and MSP [9]. The main features present in KS include more compact and larger fascicles which have slit like spaces present in between the cells, spindle cell proliferation, nuclear pleomorphism and increased number of mitosis. Extravasated RBCs are also associated with KS. In contrast, MSP is characterized by loose storiform pattern of plump spindle shaped histiocytes having eosinophilic to granular cytoplasm. Granulomatous inflammation may or may not be present. Immunohistochemical stains can also help to distinguish between these conditions. CD68 and S100 are positive in histiocytes in MSP, however S100 has variable staining and it can be negative. Spindle cells in MSP also express smooth muscle actin and variable desmin. KS cells stain positive with CD34 and CD31 markers [10].
Douglas et al reported a case of MSP in abdominal wall where patient was on continuous steroid therapy due to stem cell transplant and causative agent was Mycobacterium abscessus. Another case of MSP in appendix has been reported where patient was immunocompromised due to AIDS and the exact species of mycobacteria could not be determined in this case as the patient died of AIDS complications however numerous AFB were seen on ZN staining.
Conclusion
In conclusion the MSP should not be missed and one should list this in differential diagnosis along with other tumors of mesenchymal cells in patients who have the conditions which affect their immune status. MSP should be differentiated from other histologic mimics by special stains and immunohistochemical studies on tissue biopsy. MSP is a treatable non-neoplastic entity, it should be included in the differential diagnosis of spindle cell tumors especially in immunosuppressed patients.
Conflict of Interest
The Authors declare that there is no conflict of interest.
References
- Philip J, Beasley MB, Dua S (2012) Mycobacterial spindle cell pseudotumor of the lung. Chest 142: 783-784.
[Crossref] [Google Scholar] [PubMed]
- Basilio-de-Oliveira CA, Eyer-Silva WA, Valle HDA, Rodrigues AL, Pimentel ALP, et al. (2001) Mycobacterial spindle cell pseudotumor of the appendix vermiformis in a patient with AIDS. Brazil J Infect Dis 5: 98-100.
- Kirchner A, Sergeyenko A, Keane J, Belinski K, Liu W (2021) Diagnosis of cutaneous mycobacterial spindle cell pseudotumor in the absence of typical inflammatory cells: A case report. J Cutan Pathol 48: 1178-1181.
[Crossref] [Google Scholar] [PubMed]
- Dhibar DP, Sahu KK, Singh S, Bal A, Chougale A, et al. (2018) Tubercular mycobacterial spindle cell pseudotumour: A case report. Iran J Med Sci 43: 94.
- Yeh I, George E, Jokinen CH (2011) Cutaneous mycobacterial spindle cell pseudotumor: A potential mimic of soft tissue neoplasms. Am J Dermatopathol 33: e66-e69.
- Krause JR, Findeis SK (2020) Mycobacterial spindle cell pseudotumor in a woman with HIV. Baylor Univers Med Center Proceed 33: 438-439.
- Singh B, Kaur P, Majachani N, Guragai N, Gupta S, et al. (2021) Mycobacterial spindle cell pseudotumor of the lymph nodes. J Investigat Med High Impact Case Rep 9: 23247096211013190.
- Liao X, Xu X (2015) Spindle cell pseudotumor of lymph node caused by Mycobacterium tuberculosis infection. Am J Clin Pathol 144: A306-A306.
- Lim DD, Bushra A, Zhang H (2021) Abdominal wall mycobacterial spindle cell pseudotumor lesion. Baylor Univers Med Center Proceed 34: 505-506.
- Sfeir MM, Schuetz A, Van Besien K, Borczuk AC, Soave R, et al. (2018) Mycobacterial spindle cell pseudotumour: Epidemiology and clinical outcomes. J Clin Pathol 71: 626-630.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi