Research Article, J Spine Neurosurg Vol: 8 Issue: 2
One-Year NDI and VAS Outcomes from the Single-Level PEEK-on-Ceramic SimplifyTM Disc FDA IDE Trial
Fred H Geisler1*, David G Maislin2, Brendan T Keenan2 and Greg Maislin2
1Department of Medical Imaging, College of Medicine at the University of Saskatchewan, Saskatoon SK S7N 5C9, Canada
2Biomedical Statistical Consulting, Wynnewood PA, USA
*Corresponding Author : Fred H Geisler
Department of Medical Imaging, College of Medicine at the University of Saskatchewan
Saskatoon SK S7N 5C9 Canada/ Neurosurgeon as Medical Monitor for Single-Level Simplify Disc IDE study for Simplify Medical, Inc., 685 North Pastoria Avenue, Sunnyvale, CA 94085, USA
Tel: 312-813-0645
E-mail: fredgeisler@me.com
Received: March 15, 2019 Accepted: March 15, 2019 Published: March 29, 2019
Citation: Geisler FH, Maislin DG, Keenan BT, Maislin G (2019) One-Year NDI and VAS Outcomes from the Single-Level PEEK-on-Ceramic Simplify™ Disc FDA IDE Trial. J Spine Neurosurg 8:2. doi: 10.4172/2325-9701.1000320
Abstract
This study was performed to evaluate the early clinical results at one year for the Simplify™ Cervical Artificial Disc. We compared outcomes for 150 Simplify Disc subjects at one-year followup in a prospective, multicenter, FDA IDE clinical trial with 119 propensity score matched historical control subjects who received conventional anterior cervical discectomy and fusion (ACDF) for single-level cervical degenerative disc disease. The outcome measures included the change from preoperative baseline to one-year in Neck Disability Index (NDI) and visual analog scales (VAS) for neck and arm pain, with scores for the few missing oneyear follow-up implicitly imputed using mixed models for repeated measures (MMRM). The MMRM was used to estimate within group and between group differences controlling for propensity score subclass and the relevant baseline value. The adjusted mean changes (and standard errors) in NDI from baseline to one year were -46.7 (SE=1.7, p<0.001) and -38.1 (SE=1.9, p<0.001) for Simplify Disc subjects and ACDF control subjects, respectively. The adjusted Simplify Disc vs. control difference in mean NDI change at one year was -8.7 (SE=2.7) with p=0.002; the 95% confidence interval for the mean difference was -14.0 to -3.3. The adjusted mean changes in VAS neck and arm pain from baseline to one year were -62.4 (SE=2.0, p<0.001) and -55.2 (SE=2.3, p<0.001) for Simplify Disc and ACDF controls, respectively. The adjusted Simplify Disc vs. control difference in mean VAS neck and arm pain change at one year was -7.3 (SE=3.3) with p=0.029 (95% CI -13.8 to 0.8). Therefore, it can be concluded that the one-year clinical results of the Simplify Disc are superior to ACDF for both 1) improvement of NDI and 2) improvement in VAS neck and arm pain. Inspection of all eight prior FDA cervical total disc replacement studies indicates that these good results for the Simplify Disc can be expected to continue for five years and beyond, but longer term follow-up is necessary for verification.
Keywords: Cervical disc arthroplasty; PEEK (Polyetheretherketone); Propensity score; Magnetic resonance imaging
Introduction
Current FDA-approved total disc replacements (TDR) are all comprised of metallic endplates (cobalt-chromium-molybdenum, titanium, or metal/ceramic composite), with most having a polymer-based core comprised of ultrahigh molecular weight polyethylene or polyurethane. The clinical success demonstrated in multiple randomized prospective FDA monitored studies of the approved cervical TDRs [1,2] attests to their safety, effectiveness, and durability as a group of implants. However, post-surgical magnetic resonance (MR) imaging at the operative and adjacent levels of the cervical spine is often compromised, and in some patients a critical decision making imaging component is unreadable secondary to the artifact induced by the metallic plates of the current commercially available artificial discs [1-3]. This imaging artifact issue can lead to deferred diagnosis, inaccurate diagnosis, or an escalation of the imaging studies to an invasive computed tomography (CT)-myelogram to obtain imaging information otherwise routinely available on MR imaging if no metallic cervical TDR artifact were present.
Magnetic resonance is currently the preferred mode for diagnostic imaging prior to spine surgery, but CT use markedly increases after complex spine surgery involving implants [4], specifically because of the MR imaging artifacts from the implants and the subsequent poor visualization of the adjacent spinal structures. To minimize exposure to ionizing radiation [5] and concomitant risk of cancer [6], a cervical artificial disc that has minimal artifact and affords clear visualization of the operative and adjacent levels with MR would be preferable to the current metallic designs.
The use of polyetheretherketone (PEEK) for spinal implants continues to increase [7], primarily due to its mechanical properties and positive imaging properties (i.e. no metallic artifact on MRI or CT). The purpose of this study was to evaluate the function and pain scores at one-year follow-up from the single-level FDA Investigational Device Exemption (IDE) study for the PEEK-on-ceramic Simplify™ Artificial Cervical Disc.
Materials and Methods
Description of implant
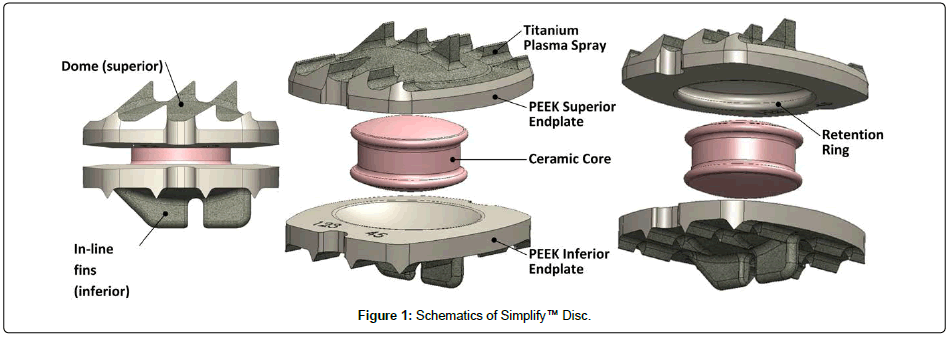
The Simplify™ Disc is a three-piece intervertebral prosthesis consisting of two PEEK endplates with titanium coating on the surface adjacent to the bony endplate (thin osteo-integration promoting surface treatment) and a mobile zirconia-toughened alumina (ZTA) core (Figure 1). The mechanical design and physiologic movement of the Simplify Disc were based on the Kineflex®|C Cervical Artificial Disc (SpinalMotion, Mountain View, CA), which consisted of two cobalt-chromium-molybdenum (CCM), titanium-coated endplates with a biconvex CCM core. An IDE clinical trial [8] of the Kineflex|C Disc with five-year follow-up demonstrated excellent clinical results and validated the clinical efficacy of this three-part geometric design with dual articulations.
In addition to providing motion and height restoration, the Simplify Disc is designed to permit subsequent visualization of cervical anatomy using MR imaging without the significant radiographic artifact present in other cervical TDRs which incorporate a substantial metal component. Along with changes in endplate and core materials, the Simplify Disc design included minor modifications to the geometry based on anatomical measurements taken during the Kineflex|C IDE. The main significant difference between the Kineflex|C Cervical Artificial Disc and the Simplify Disc is that the materials chosen for the Simplify Disc have a greatly reduced MR imaging artifact.
The Simplify Disc implant system is available in multiple configurations of footprint, height, lordosis and titanium coating thickness. All endplates feature smooth concave articulating surfaces to permit ±12° flexion-extension and lateral bending, unlimited axial rotation, and a limited amount (<1.6 mm) of translation in the horizontal plane.
The Simplify Disc is provided packaged and preassembled, and inserts as a single unit following a complete discectomy. A variation of the Simplify Disc (Kineflex Prime Disc) has been implanted in South Africa since 2013, and the Simplify Disc has been commercially available in the UK and Germany since 2016 [9].
Parent clinical study
The parent study for this one-year results report is a prospective, controlled, multicenter clinical trial (US FDA IDE #G140154, NCT02667067) intended to demonstrate that the Simplify Disc is at least as safe and effective as conventional anterior cervical discectomy and fusion (ACDF) when used to treat one disc level between C3 to C7 for cervical degenerative disc disease (DDD). DDD for this study was defined as intractable radiculopathy (arm pain and/or a neurological deficit) with or without neck pain or myelopathy due to a single-level abnormality localized to the level of the disc space in subjects who are unresponsive to conservative management.
The parent study was prospectively designed to compare the Simplify Disc to a non-concurrent historical control group with subject-level data in a parallel group design. The historical control group for both the parent study and for the current one-year study is formed from the randomized ACDF arm (N=133) of the completed multi-center, prospective, randomized clinical study of the Kineflex|C Disc trial [8].
The parent study confirmed control group comparability could be achieved using propensity score sub-classification [10], and prospectively enrolled 166 subjects from sixteen (16) sites between February 2016 and February 2018. The 166 total includes 150 for comparison to ACDF and 16 training cases (one per site). The parent study is prospectively utilizing a two-year composite clinical success (CCS) endpoint as the primary FDA effectiveness endpoint. Individual success with this CCS requires all of the following four criteria: 1) at least a 15-point improvement in the Neck Disability Index (NDI) at 24 months compared with baseline; 2) maintenance or improvement in neurologic status at 24 months compared with baseline; 3) no device failures or revision, reoperation, removal and/ or supplemental fixation within 24 months of index procedure; and 4) the absence of major adverse events within 24 months. NDI has strong and well-documented convergent and divergent validity with other instruments used in the evaluation of patients with neck pain [11].
Statistical analysis plan for current study
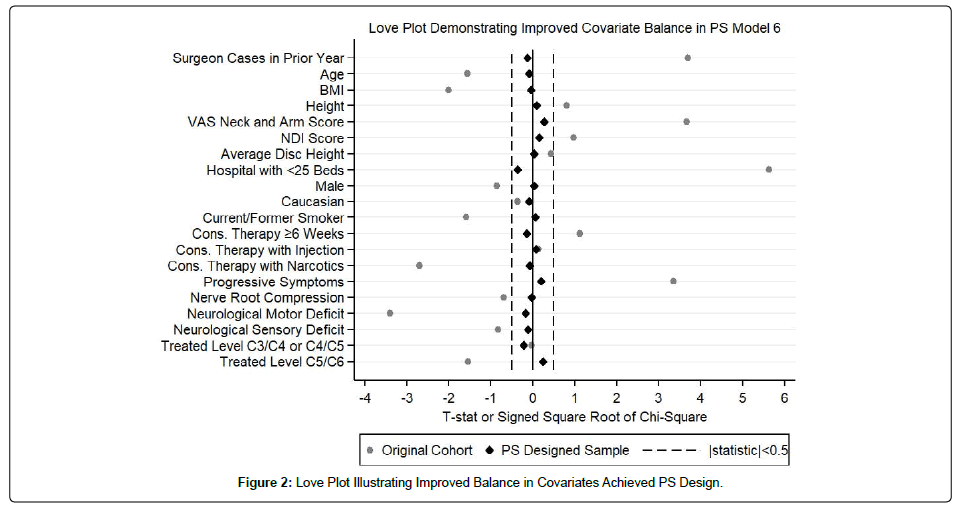
This study is a rigorously designed observational study using propensity score (PS) sub-classification [10,12,13] to simultaneously control for a set of 20 clinically relevant covariates (Figure 2) when making comparisons between device groups. The statistician conducting the PS modeling was and continues to be blinded without access to subject level outcome data. Balance between device groups was verified using a “Love plot” [14] that compares covariate balance between groups before and after the PS design (Figure 2).
The examined outcome variables in this one-year outcome report are changes from baseline to one-year in the NDI and visual analog scale (VAS) for neck and arm pain. For subjects experiencing a secondary surgical intervention (SSI), the last NDI prior to the SSI was used to determine change scores.
The null hypotheses for these analyses are that the mean improvement in NDI from baseline to one year and the mean improvement in VAS neck and arm pain from baseline to one year are the same for subjects implanted with the Simplify Disc and subjects receiving ACDF. The alternative hypotheses are that the mean changes differ between device groups. These hypotheses were tested using a Mixed Model for Repeated Measures (MMRM) [15]. The MMRM approach is a direct likelihood approach that implicitly imputes missing endpoint values using intermediate outcomes and baseline covariates. Baseline covariates included PS subclass and the baseline value of either NDI or VAS neck and arm pain. MMRM parameter estimates, standard errors, p-values and confidence intervals were determined using SAS PROC MIXED (SAS Institute, Cary NC, Version 9.4). The Hochberg approach [16] was used to account for multiple comparisons with overall type 1 error set to 0.05.
Results
PS Sub-classification analysis
Balance through sub-classification in the sample of all 150 Simplify™ Disc subjects and 119 (89.5%) of the original 133 ACDF controls was achieved. The remaining 14 controls were trimmed to achieve covariate balance within PS subclasses. Comparison of the characteristics between selected and trimmed ACDF subjects are shown in Table 1, along with data from the Simplify Disc subjects to illustrate the general improved similarity in baseline variables. In particular, compared to included subjects, we see that trimmed ACDF control subjects were generally older (mean 48.8 years vs. 43.0 years, p=0.074); had lower VAS (mean 59.9 vs. 77.6, p<0.0001) and lower NDI (52.9 vs. 62.9, p=0.008); were more likely to be male (78.6% vs. 40.3%, p=0.007); had a neurological motor deficit (78.6% vs. 63.0%, p=0.068); and were less likely to have progressive symptoms (14.3% vs. 67.2%, p=0.0001). In each instance, included ACDF controls demonstrate average values more similar to Simplify Disc than excluded subjects, demonstrating the utility of the PS sub-classification design to promote control group comparability. Figure 2 summarizes the success of the PS design in achieving covariate balance. This figure summarizes t-tests for continuous covariates and signed square root of chi-square statistics for categorical covariates before implementing the PS design and after adjusting for PS subclass. Covariate balance within PS subclass is comparable to that expected through randomization.
| Measure | Simplify™ Disc | ACDF Control | ||
|---|---|---|---|---|
| Included | Trimmed | p* | ||
| N | 150 | 119 | 14 | – |
| Surgeon Number of Cases in Prior Year† | 4.06 ± 0.91 | 3.72 ± 0.71 | 3.51 ± 0.59 | 0.385 |
| Age (years) | 43.0 ± 8.9 | 44.0 ± 7.1 | 48.8 ± 8.9 | 0.074 |
| Body Mass Index (kg/m2) | 27.5 ± 5.2 | 28.6 ± 5.6 | 30.2 ± 5.8 | 0.319 |
| Height (inches) | 67.7 ± 4.0 | 67.2 ± 4.1 | 68.3 ± 4.1 | 0.244 |
| VAS Neck and Arm Pain | 81.6 ± 12.4 | 77.6 ± 14.1 | 59.9 ± 10.7 | <0.0001 |
| Neck Disability Index (NDI) | 63.3 ± 12.5 | 62.9 ± 12.9 | 52.9 ± 11.3 | 0.008 |
| Average Disc Height | 3.32 ± 0.74 | 3.29 ± 0.80 | 3.14 ± 0.68 | 0.619 |
| Hospital with <25 Beds, % | 67.3% | 35.3% | 14.3% | 0.142 |
| Male, % | 39.3% | 40.3% | 78.6% | 0.007 |
| Caucasian, % | 87.3% | 89.1% | 85.7% | 0.659 |
| Current or Former Smoker, % | 38.0% | 46.2% | 57.1% | 0.439 |
| Conservative Therapy ≥6 Weeks, % | 91.3% | 86.6% | 92.9% | >0.999 |
| Conservative Therapy with Injection, % | 46.7% | 44.5% | 57.1% | 0.371 |
| Conservative Therapy with Narcotics, % | 44.0% | 58.8% | 71.4% | 0.362 |
| Progressive Symptoms, % | 80.0% | 67.2% | 14.3% | 0.0001 |
| Nerve Root Compression, % | 60.7% | 63.0% | 78.6% | 0.377 |
| Neurological Motor Deficit, % | 35.3% | 52.9% | 78.6% | 0.068 |
| Neurological Sensory Deficit, % | 44.0% | 48.7% | 50.0% | 0.929 |
| Treated Index Level‡, % | ||||
| C3/C4 | 2.0% | 2.5% | 0.0% | 0.658 |
| C4/C5 | 4.7% | 5.0% | 0.0% | |
| C5/C6 | 53.3% | 60.5% | 78.6% | |
| C6/C7 | 40.0% | 31.9% | 21.4% | |
| Notes: †Natural log transformed; ‡C3/C4 and C4/C5 collapsed into single group in PS model, due to low frequency; *p- value from Wilcoxon test (continuous data) and chi-squared or Fisher’s exact tests (categorical data) comparing included and excluded ACDF subjects. |
||||
Table 1: Comparison of ACDF Subjects Included and Excluded from the Final PS Subclassification Design.
| Visit | Simplify™ Disc (N= 148) | ACDF Control (N= 119) | ||||||
|---|---|---|---|---|---|---|---|---|
| N | Estimate | LB | UB | N | Estimate | LB | UB | |
| PreOp | 148 | 63.3 | 61.0 | 65.5 | 119 | 62.9 | 60.4 | 65.5 |
| Week 6 | 146 | 22.9 | 19.8 | 26.1 | 113 | 34.6 | 31.0 | 38.3 |
| Month 3 | 145 | 17.1 | 14.0 | 20.2 | 114 | 27.4 | 23.8 | 30.9 |
| Month 6 | 144 | 16.9 | 13.8 | 20.0 | 104 | 24.6 | 21.0 | 28.2 |
| Month 12 | 133 | 16.5 | 13.2 | 19.7 | 101 | 25.1 | 21.3 | 28.9 |
| Notes: * Results based on mixed model for repeated measures (MMRM) that included device group, baseline value, and propensity score subclass. |
||||||||
Table 2: Adjusted Mean NDI Over Time with 95% Confidence Intervals*.
Missing data and secondary surgical interventions at the index level
In the starting population of 150 Simplify Disc subjects, two experienced intra-operative protocol deviations and underwent fusion rather than receiving the Simplify Disc. The data from these two subjects were removed from the analysis, resulting in a baseline of 148 subjects who received the intended group treatment.
There was one case of secondary surgical intervention (SSI) in the Simplify Disc group within the 12 month follow-up period. A Simplify Disc explant occurred 49 days after the index surgery; this event was attributed to the surgical procedure itself and not related to the device. There was also one PS-selected ACDF subject who experienced an SSI prior to the one-year anniversary on day 168. Subjects with SSI generally had relatively poorer clinical status leading up to the SSI which is accounted for in the statistical modeling by including interim NDI or VAS in the MMRM.
As of the database lock on January 11, 2019, 133 of 148 (90%) Simplify Disc subjects had NDI observations at one year. Among 119 control subjects, 101 (85%) had NDI observations at one year. There were no missing baseline NDI values. However, one Simplify Disc subject and two ACDF control subjects had no follow-up NDI values and could not be included in the MMRM for NDI. The remaining subjects missing NDI at one year were included in the analysis by way of the MMRM.
Among Simplify Disc and control subjects, 132 of 148 (90%) and 101 of 119 (85%) subjects, respectively, had VAS neck and arm pain observations at one year. There were no missing baseline VAS values. Only one ACDF control subject had no follow-up VAS and could not be included in the MMRM. All Simplify Disc subjects had at least one follow-up visit and so could be included in the MMRM.
Early functional and pain results
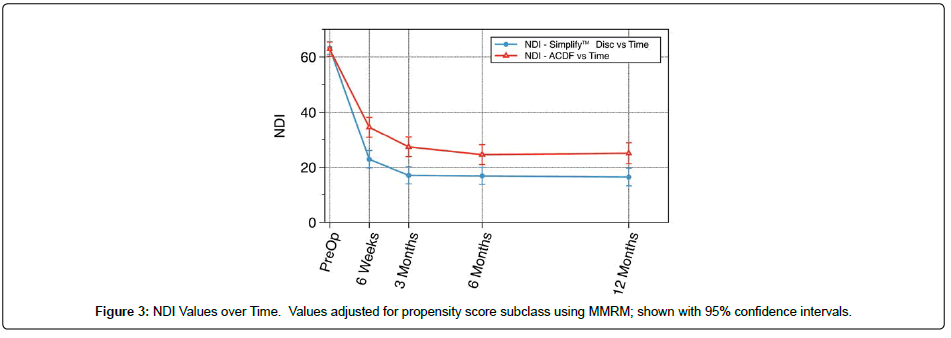
Figure 3 and Table 2 summarize the results from the MMRM for NDI values over time including PS subclass and baseline NDI adjusted mean values over time with 95% confidence intervals. At baseline, the adjusted mean values were nearly identical between subjects receiving the Simplify Disc and subjects undergoing ACDF, as expected through the PS design. At every follow-up timepoint, the covariate-adjusted 95% confidence intervals were non-overlapping between groups, with the lower and better outcome NDI in the Simplify Disc group.
In the MMRM for changes in NDI from baseline, the adjusted mean changes (standard errors) from baseline to one year were -46.7 (SE=1.7, p<0.001) and -38.1 (SE=1.9, p<0.001) for Simplify Disc and ACDF controls, respectively. The adjusted Simplify Disc vs. control difference in mean changes at one year was -8.7 (SE=2.7) with p=0.002. The 95% confidence interval for the mean difference is -14.0 to -3.3. These results indicate that the null hypothesis of equality in mean changes to one-year between groups can be rejected. Therefore, it can be concluded that the Simplify Disc is statistically superior to ACDF in terms of improvement in NDI from baseline to one-year.
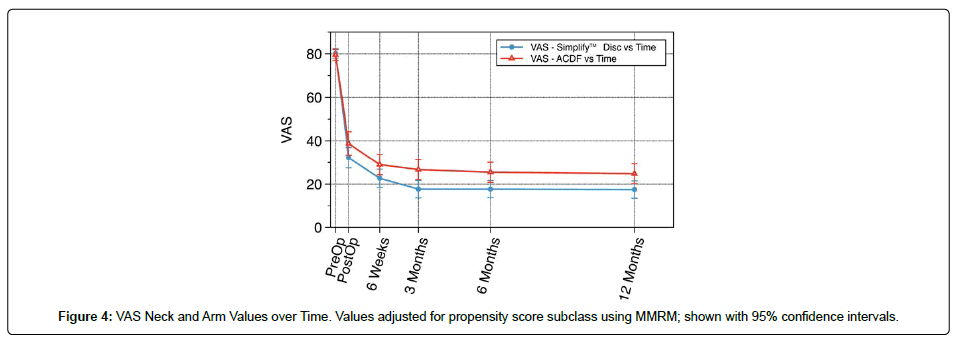
Figure 4 and Table 3 summarize the results from the MMRM for VAS neck and arm pain, along with comparable results at baseline from the PS-adjusted analysis of covariance. At baseline, the adjusted mean values were nearly identical between subjects receiving the Simplify Disc and subjects undergoing ACDF, as expected through PS modeling.
| Visit | Simplify™ Disc (N= 148) | ACDF Control (N= 119) | ||||||
|---|---|---|---|---|---|---|---|---|
| N | Estimate | LB | UB | N | Estimate | LB | UB | |
| PreOp | 148 | 80.2 | 78.0 | 82.4 | 119 | 79.5 | 77.0 | 82.1 |
| PostOp | 146 | 32.2 | 27.5 | 37.0 | 116 | 38.8 | 33.4 | 44.2 |
| Week 6 | 146 | 22.7 | 18.5 | 26.9 | 114 | 29.0 | 24.2 | 33.7 |
| Month 3 | 144 | 17.7 | 13.7 | 21.7 | 114 | 26.7 | 22.1 | 31.3 |
| Month 6 | 144 | 17.7 | 13.8 | 21.7 | 104 | 25.5 | 20.9 | 30.2 |
| Month 12 | 132 | 17.5 | 13.5 | 21.5 | 101 | 24.8 | 20.2 | 29.4 |
| Notes: * Results based on mixed model for repeated measures (MMRM) that included device group, baseline value, and propensity score subclass. |
||||||||
Table 3: Adjusted Mean VAS Neck & Arm Pain Over Time with 95% Confidence Intervals*.
In the MMRM for changes in VAS neck and arm pain from baseline, the adjusted mean changes (standard errors) from baseline to one year are -62.4 (SE=2.0, p<0.001) and -55.2 (SE=2.3, p<0.001) for Simplify Disc and ACDF controls, respectively. The adjusted device group difference in mean change from baseline at one year is -7.3 (SE=3.3) with p=0.029. The 95% confidence interval is -13.8 to 0.8.
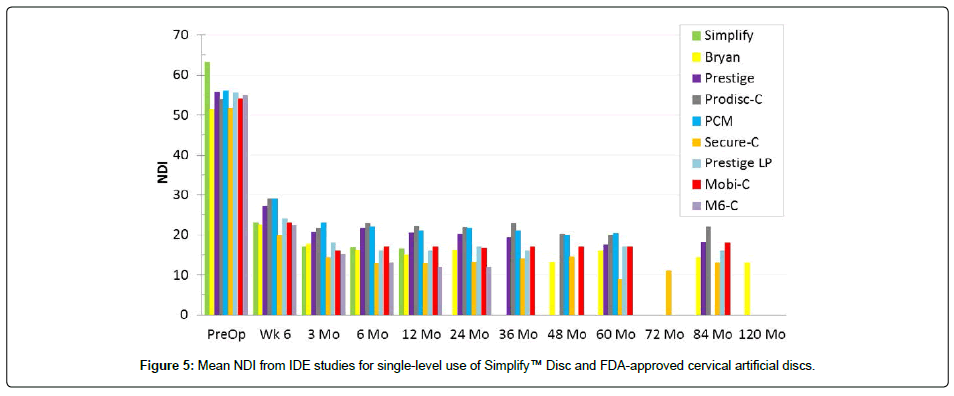
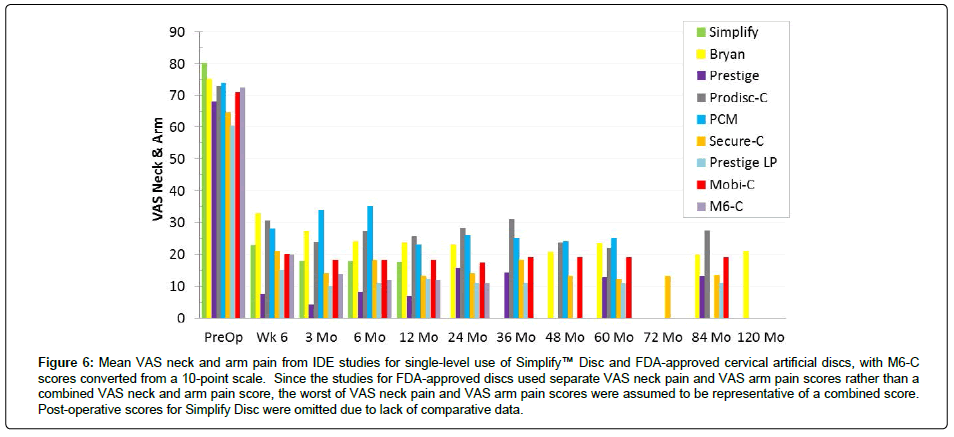
When applying the Hochberg method to account for multiple comparisons, since the maximum p-value is less than 0.05, the null hypotheses concerning NDI and VAS neck and arm pain are both rejected. Therefore, it can be concluded that the one-year clinical results of the Simplify Disc are superior to ACDF for both 1) improvement of NDI and 2) improvement in VAS neck and arm pain (Figures 5 and 6).
Figure 6: Mean VAS neck and arm pain from IDE studies for single-level use of Simplify™ Disc and FDA-approved cervical artificial discs, with M6-C scores converted from a 10-point scale. Since the studies for FDA-approved discs used separate VAS neck pain and VAS arm pain scores rather than a combined VAS neck and arm pain score, the worst of VAS neck pain and VAS arm pain scores were assumed to be representative of a combined score. Post-operative scores for Simplify Disc were omitted due to lack of comparative data.
Clinical imaging
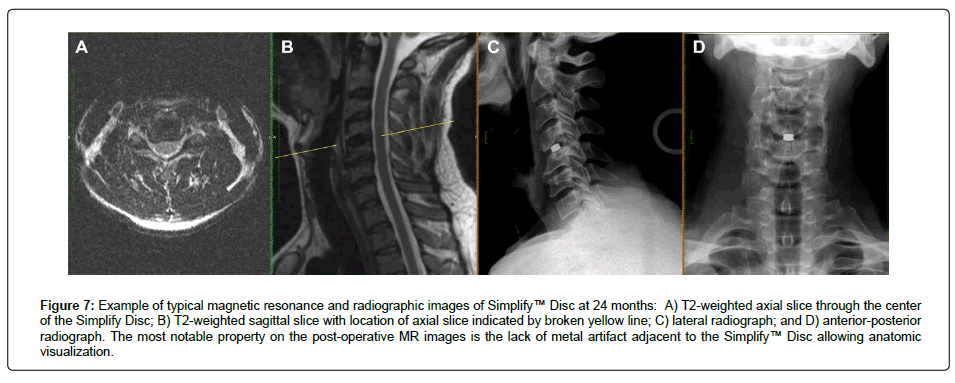
The Simplify Disc induced minimal MR image artifact on postoperative MR imaging and was was visible on plain X-ray images to assess TDR placement. Figure 7 is a typical example of clinical imaging of both the MR and plain X-rays images at the protocol specified 24 months timepoint. The most notable property on the MR images is the lack of metal artifact adjacent to the Simplify Disc. This lack of adjacent artifact is markedly different from all other FDA approved cervical TDRs which have a large adjacent metal artifact signature which often obscures important anatomic structures [1-3]. The excellent post-operative MR imaging of the Simplify™ Disc is a major differentiating feature which was routinely demonstrated in this study.
Figure 7: Example of typical magnetic resonance and radiographic images of Simplify™ Disc at 24 months: A) T2-weighted axial slice through the center of the Simplify Disc; B) T2-weighted sagittal slice with location of axial slice indicated by broken yellow line; C) lateral radiograph; and D) anterior-posterior radiograph. The most notable property on the post-operative MR images is the lack of metal artifact adjacent to the Simplify™ Disc allowing anatomic visualization.
Discussion
The early results from this study are consistent with the preliminary results published previously [17]. These one-year results can also be compared to those from other prospective, multicenter clinical trials for eight FDA-approved cervical TDRs, which have a longer reported follow-up period. The other studies are summarized in Table 4. These studies consistently report statistically and clinically significant improvements in NDI and VAS neck and arm pain from preoperative values at all post-operative timepoints. Note that all scores typically reach a minimal plateau by three months and then remain stable thereafter to 5 years and beyond the follow-up time period. Visual inspection of these graphs indicates that clinical course in all of these prior studies is a 3-month recovery period of increasing function and decreasing pain which then remains durable for 5 years and beyond (Figures 5 and 6). Thus, it is anticipated that the improvements in function and pain noted in the one-year Simplify™ Disc results will be stable through five years and beyond, but long-term follow-up is necessary to verify this anticipated stability of the one-year results with time.
| Disc | IDE initiation | Test Subjects* | Test Subjects at 2-Yr Follow-up | Subsequent Surgical Interventions at 2 Yr Follow-up** | FDA Approval | Longest Follow-up (yrs) | References |
|---|---|---|---|---|---|---|---|
| Bryan | May 2002 | 242 | 230 (95%) | 6 (2.5%) | May 2009 | 10 | Heller [18], Lavelle [19] |
| Prestige | Oct 2002 | 276 | 223 (80%) | 5 (2.2%) | Jul 2007 | 7 | Mummaneni [20], Burkus [21,22] |
| ProDisc-C | Aug 2003 | 103 | 101 (98%) | 2 (1.9%) | Dec 2007 | 7 | Murrey [23], Delamarter [24], Zigler [25], Janssen [26] |
| PCM | Jan 2005 | 214 | 195 (89%) | 11 (5.2%) | Oct 2012 | 5 | Phillips [27,28] |
| Secure-C | Jul 2005 | 148 | Not reported | 6 (2.5%) | Sep 2012 | 8 | Vaccaro [29,30] |
| Prestige LP | Nov 2005 | 280 | 272 (97.1%) | 16 (5.7%) | Jul 2014 | 7 | Gornet [31,32] |
| Mobi-C | Apr 2006 | 164 | 155 (94.3%) | 2 (1.2%) | Aug 2013 | 7 | Hisey [33], Radcliff [34] |
| M6-C | May 2014 | 160 | 136 (85%) | 3 (1.9%) | Feb 2019 | 2 | SSED [35] |
| Notes: * Number of subjects implanted with test device ** Includes removal, revision, supplemental fixation, and reoperation at the index level; percentages reflect those reported in source documents (adjusted for differences in follow-up in some but not all cases) or, if not reported, were crudely estimated as the number of SSIs divided by total number of test subjects. |
|||||||
Table 4: Prospective, Multicenter Clinical Studies for Cervical Artificial Disc (Single-Level Use).
The Simplify™ Disc demonstrated MRI compatibility and good imaging of clinically relevant structures such as the spinal cord, exiting rootlets, neural foramen, and posterior bony margins. Thus, we anticipate that the need for post-operative CT and CT myelograms will be minimized or eliminated due to the minimal MR artifact induced by this PEEK-on-ceramic Simplify™ Disc.
Conclusion
The one-year results of the Simplify™ Disc from a prospectively designed PS sub-classification analysis provide robust evidence that the Simplify Disc is superior to ACDF in terms of improvements in NDI from baseline to one year (-46.7 vs. -38.1, p=0.002). Similarly, the Simplify Disc is also superior to ACDF with regards to improvement in VAS neck and arm pain from baseline to one year (-62.4 vs. -55.2, p=0.029). Post-operative MR imaging has verified the excellent MR image quality with the Simplify Disc. Data collection for the parent study is planned to continue through 24-month follow-up to obtain the verifying longer term outcome data.
Acknowledgements
We would like to thank Beth Neil and Melissa K.C. Lui at Simplify Medical for their assistance with this manuscript.
References
- Sekhon LH, Duggal N, Lynch JJ, Haid RW, Heller JG, et al. (2007) Magnetic resonance imaging clarity of the Bryan, Prodisc-C, Prestige LP, and PCM cervical arthroplasty devices. Spine (Phila Pa 1976) 32: 673-680.
- Sundseth J, Jacobsen EA, Kolstad F, Nygaard OP, Zwart JA, et al. (2013) Magnetic resonance imaging evaluation after implantation of a titanium cervical disc prosthesis: a comparison of 1.5 and 3 Tesla magnet strength. Eur Spine J 22: 2296-2302.
- Fayyazi AH, Taormina J, Svach D, Stein J, Ordway NR (2015) Assessment of magnetic resonance imaging artifact following cervical total disc arthroplasty. Int J Spine Surg 9: 30.
- Patel VV, Andersson GBJ, Garfin SR, Resnick DL, Block JE (2017) Utilization of CT scanning associated with complex spine surgery. BMC Musculoskelet Disord 18: 52.
- Linet MS, Slovis TL, Miller DL, Kleinerman R, Lee C, et al. (2012) Cancer risks associated with external radiation from diagnostic imaging procedures. CA Cancer J Clin 62: 75-100.
- Shuryak I, Sachs RK, Brenner DJ (2010) Cancer risks after radiation exposure in middle age. J Natl Cancer Inst 102: 1628-1636.
- Kurtz SM (2012) Applications of Polyaryletheretherketone in Spinal Implants: Fusion and Motion Preservation. In: PEEK Biomaterials Handbook. SM Kurtz (ed) Elsevier: Waltham. pp: 201-220.
- Coric D, Guyer RD, Nunley PD, Musante D, Carmody C, et al. (2018) Prospective, randomized multicenter study of cervical arthroplasty versus anterior cervical discectomy and fusion: 5-year results with a metal-on-metal artificial disc. J Neurosurg Spine 28: 252-261.
- Simplify Disc Instructions for Use (2017) Simplify Medical.
- Rosenbaum PR, Rubin DB (1983) The central role of the propensity score in observational studies for causal effects. Biometrika 70: 41-55.
- Vernon H (2008) The Neck Disability Index: state-of-the-art, 1991-2008. J Manipulative Physiol Ther 31: 491-502.
- Rubin DB (2001) Using propensity scores to help design observational studies: application to the tobacco litigation. Health Services and Outcomes Research Methodology 2: 169-188.
- Maislin G, Rubin DB (2010) Design of non-randomized medical device trials based on sub-classification using propensity score quintiles, topic contributed session on medical devices. Proceedings of the Joint Statistical Meetings 2182-2196.
- Ahmed A, Husain A, Love TE, Gambassi G, Dell'Italia LJ, et al. (2006) Heart failure, chronic diuretic use, and increase in mortality and hospitalization: an observational study using propensity score methods. Eur Heart J 27: 1431-1439.
- Verbeke G, Molenberghs G (2000) Linear Mixed Models for Longitudinal Data. Springer-Verlag: New York.
- Hochberg Y (1988) A sharper Bonferroni procedure for multiple tests of significance. Biometrika 75: 800-802.
- Maislin G, Maislin DG, Keenan BT, Alvis MR (2018) Preliminary clinical outcomes from the polyetheretherketone on ceramic simplify™ Disc FDA IDE Trial. J Spine Neurosurg 7:4.
- Heller JG, Sasso RC, Papadopoulos SM, Anderson PA, Fessler RG, et al. (2009) Comparison of BRYAN cervical disc arthroplasty with anterior cervical decompression and fusion: clinical and radiographic results of a randomized, controlled, clinical trial. Spine (Phila Pa 1976), 34: 101-107.
- Lavelle WF (2018) 10-year Outcomes of Cervical Disc Replacement with the BRYAN(R) Cervical Disc: Results from a Prospective, Randomized, Controlled Clinical Trial. Spine (Phila Pa 1976), 2018.
- Mummaneni PV, Burkus JK, Haid RW, Traynelis VC, Zdeblick TA (2007) Clinical and radiographic analysis of cervical disc arthroplasty compared with allograft fusion: a randomized controlled clinical trial. J Neurosurg Spine 6: 198-209.
- Burkus JK, Haid RW, Traynelis VC, Mummaneni PV (2010) Long-term clinical and radiographic outcomes of cervical disc replacement with the Prestige disc: results from a prospective randomized controlled clinical trial. J Neurosurg Spine 13: 308-318.
- Mummaneni PV, Burkus JK, Haid RW, Traynelis VC, Zdeblick TA (2014) Clinical and radiographic analysis of an artificial cervical disc: 7-year follow-up from the Prestige prospective randomized controlled clinical trial: Clinical article. J Neurosurg Spine 21: 516-528.
- Murrey D, Janssen M, Delamarter R, Goldstein J, Zigler J, et al. (2009) Results of the prospective, randomized, controlled multicenter Food and Drug Administration investigational device exemption study of the ProDisc-C total disc replacement versus anterior discectomy and fusion for the treatment of 1-level symptomatic cervical disc disease. Spine J 9: 275-286.
- Delamarter RB, Murrey D, Janssen ME, Goldstein JA, Zigler J, et al. (2010) Results at 24 months from the prospective, randomized, multicentre Investigational Device Exemption trial of ProDisc-C versus anterior cervical discectomy and fusion with 4-year follow-up and continued access patients. SAS J 4: 122-128.
- Zigler JE, Delamarter R, Murrey D, Spivak J, Janssen M (2013) ProDisc-C and anterior cervical discectomy and fusion as surgical treatment for single-level cervical symptomatic degenerative disc disease: five-year results of a Food and Drug Administration study. Spine (Phila Pa 1976), 38: 203-209.
- Janssen ME, Zigler JE, Spivak JM, Delamarter RB, Darden BV II, et al. (2015) ProDisc-C Total Disc Replacement Versus Anterior Cervical Discectomy and Fusion for Single-Level Symptomatic Cervical Disc Disease: Seven-Year Follow-up of the Prospective Randomized U.S. Food and Drug Administration Investigational Device Exemption Study. J Bone Joint Surg Am 97: 1738-1747.
- Phillips FM, Lee JY, Geisler FH, Cappuccino A, Chaput CD, et al. (2013) A prospective, randomized, controlled clinical investigation comparing PCM cervical disc arthroplasty with anterior cervical discectomy and fusion. 2-year results from the US FDA IDE clinical trial. Spine (Phila Pa 1976) 38: E907-E918.
- Phillips FM, Geisler FH, Gilder KM, Reah C, Howell KM, et al. (2015) Long-term Outcomes of the US FDA IDE Prospective, Randomized Controlled Clinical Trial Comparing PCM Cervical Disc Arthroplasty With Anterior Cervical Discectomy and Fusion. Spine (Phila Pa 1976) 40: 674-683.
- Vaccaro A, Beutler W, Peppelman W, Marzluff JM, Highsmith J, et al. (2013) Clinical outcomes with selectively constrained SECURE-C cervical disc arthroplasty: two-year results from a prospective, randomized, controlled, multicenter investigational device exemption study. Spine (Phila Pa 1976) 38: 2227-2239.
- Vaccaro A, Beutler W, Peppelman W, Marzluff J, Mugglin A, et al. (2018) Long-Term Clinical Experience with Selectively Constrained SECURE-C Cervical Artificial Disc for 1-Level Cervical Disc Disease: Results from Seven-Year Follow-Up of a Prospective, Randomized, Controlled Investigational Device Exemption Clinical Trial. Int J Spine Surg 12: 377-387.
- Gornet MF, Burkus JK, Shaffrey ME, Argires PJ, Nian H, et al. (2015) Cervical disc arthroplasty with PRESTIGE LP disc versus anterior cervical discectomy and fusion: a prospective, multicenter investigational device exemption study. J Neurosurg Spine 23: 558-573.
- Gornet MF, Burkus JK, Shaffrey ME, Nian H, Harrell FE Jr (2016) Cervical disc arthroplasty with prestige LP disc versus anterior cervical discectomy and fusion: seven-year outcomes. Int J Spine Surg 10: 24.
- Hisey MS, Bae HW, Davis R, Gaede S, Hoffman G, et al. (2014) Multi-center, prospective, randomized, controlled investigational device exemption clinical trial comparing Mobi-C Cervical Artificial Disc to anterior discectomy and fusion in the treatment of symptomatic degenerative disc disease in the cervical spine. Int J Spine Surg 8.
- Radcliff K, Davis RJ, Hisey MS, Nunley PD, Hoffman GA (2017) Long-term evaluation of cervical disc arthroplasty with the mobi-C(c) cervical disc: A randomized, prospective, multicenter clinical trial with seven-year follow-up. Int J Spine Surg 11: 31.
- FDA (2019) Summary of Safety and Effectiveness Data (SSED)-M6-C Artificial Cervical Disc, PMA NumberP170036.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi