Research Article, Endocrinol Diabetes Res Vol: 9 Issue: 1
Persistent Hypophosphatemic Osteomalacia Secondary to a Skull Base Mesenchymal Tumor: A Case Report and Review of Literature
Sibhi Ganapathy*, Adesh Jagadeesh, Rajesh Raykarand Shailesh AV
Department of Neurosurgery, St. Johns medical college and Hospital, Bangalore, India
*Corresponding Author: Dr Sibhi Ganapathy
Department of Neurosurgery, St. Johns medical college and Hospital, Bangalore, India
E-mail: sibhig09@gmail.com
Received date: 09 December, 2022, Manuscript No. ECDR-22-79425;
Editor assigned date: 14 December, 2022, Pre QC No. ECDR-22-79425 (PQ);
Reviewed date: 29 December, 2022, QC No. ECDR-22-79425;
Revised date: 09 January, 2023, Manuscript No: ECDR-22-79425 (R);
Published date: 16 January, 2023, DOI: 10.4172/2470-7570.1000322.
Citation: Ganapathy S, Jagadeesh A, Raykar R, Shailesh AV (2022) Persistent Hypophosphatemic Osteomalacia Secondary to a Skull Base Mesenchymal Tumor: A Case Report and Review of Literature. Endocrinol Diabetes Res 9:1.
Abstract
Oncogenic Osteomalacia is rare yet a recognised set of disorders that mimic rickets but are resistant to medical management. They are seen in a variety of tumors of the CNS including hemangiopericytomas, bony tumors, and meningiomas. The most common cause however is a Mesenchymal secretory tumor resembling a fibroma sitting in the base of the skull near the nasal sinuses. These tumors are seen to secrete Fibroblast Growth Factor 23 (FGF-23) which mimics parathyroid hormone leading to a Hypophosphatemic hypercalcaemic picture characterised by body aches, recurrent fractures, growth retardation, deformities, and proximal myopathy.
We report a case of tumor-induced osteomalacia caused by an infratemporal skull base tumor. The tumor was successfully resected via a subtemporal epidural approach. Phosphate level recovered immediately after resection. A review of relevant literature over the past 10 years as well as newer methods to diagnose and treat these rare entities are mentioned.
Keywords: Skull base Mesenchymal tumor; Fibroblast Growth Factor (FGF); Oncogenes; Osteomalacia; Hypoparathyroidism
Introduction
Tumor-induced Osteomalacia is a rare, acquired disorder. The patients typically present with inexplicable body pains and recurrent fractures, often incapacitating the patients. Lab analyses often reveal a Hypophosphatemic hypocalcaemia with increased renal excretion. Thus, the problem then points to a secretory lesion, which needs to be found and managed. Such tumors are usually small mesenchymal cell rests which typically inhabit the skull base. They characteristically secrete FGF-23 (fibroblast growth factor 23) as a tumor marker which aids in diagnosis as well as a marker of response to surgery, or radiation. The difficulty in diagnosis, detection of lesion, surgical technique coupled with a rare and surprising presentation make this a unique report. We also present a 10 year review of literature of the same to highlight the rarity and complexity of the disease process involved.
Case Presentation
A 36-year-old female patient presented to the outpatient clinic with a history of generalized body pains over the last 2 years which was gradually progressive from initially mild to unbearable now. The pain although generalized was predominantly over the hip and knee regions. The pain was associated for the past year with an inability to get up from squatting position. The weakness progressed to an inability to walk over the course of 1 year, now leaving the patient completely bedridden. There was no associated difficulty in holding objects or in fine movements of the hand. There was a difficulty in combing hair as well as reaching for objects on the top shelf. This weakness manifested over the last 6 months and has been progressively worsening as well [1-5].
There has been no paresthesia or numbness, associated with the weakness nor have there been any bowel and bladder disturbances. No headache, vomiting, seizures, excessive urination (polyuria), frequent urination (frequency), excessive thirst (polydipsia), abdominal pain radiating from the loin to the groin (indicating renal or ureteric calculi), fractures or deformity. The patient is a recently diagnosed hypertensive for 4 months. No history of diabetes, thyroid malfunction, previous surgeries, or hospitalizations. She is one of three children born of a no consanguineous marriage. Her mother died at 55 years of age due to spontaneous intracranial bleed. (The cause of this was not investigated at the time.) The patient is married and has 2 children as well as an early neonatal death secondary to neonatal pneumonia. No menstrual irregularities are present till date. She consumes a mixed diet and is devoid of vitamin and mineral deficiencies [6].
On examination, there was no thyroid enlargement or abdominal mass/organomegaly. Severe bony tenderness existed across all bony points to such an extent that movement and the assessment of power and tone was impossible. No sensory or other neurological deficit was present. Investigations revealed an elevated alkaline phosphatase with corresponding demineralization of bones as demonstrated on DEXA scans. Her spine in the anteroposterior view was -3.7 while the femur was -3.6 (0 being normal bone density). Her serum calcium was 8.4 gm/dl, and her serum phosphate was a mere 2.16 mg/dl which further reduced to 2.02 mg/dl. (Normal phosphate serum values are between 3-4.5 mg/dl) A 24 hour urinary phosphate assay showed a value of 0.05 (normal values range between 0.4-1.3 gm/24 hours) The ratio of Tubular maximum absorption of Phosphate (TmP) to Glomerular Filtration Rate (GFR) was low at 0.52 (normal value for the corresponding age group is 2.6-3.8) Vitamin D levels were adequate, parathormone levels were significantly elevated at 164.4 pg/ml (normal values range from 10-55 pg/ml) The other pertinent investigations are listed in Table 1.
| Sr.no | Investigation | Value (normal values in brackets) |
|---|---|---|
| 1 | Alkaline Phosphatase | 681 IU/L (44-147 IU/L) |
| 2 | ESR | 34 mm/hr (0-22 mm/hr) |
| 3 | Serum Calcium | 8.1 mg/dl (9-11 mg/dl) |
| 4 | Serum Phosphate | 2.16 (3-4.5 mg/dl) |
| 5 | 24-hour Urinary Phosphate excretion | 0.05 (2.6-3.8 mg/dl) |
| 6 | TmP/GFR | 0.52 (2.6-3.8) |
| 7 | Parathyroid hormone | 164.4 pg/ml (10-55 pg/dl) |
| 8 | Vitamin D | 43.3 ng/ml (30-50 ng/ml is adequate) |
| 9 | TSH | 0.45 mIU/L (0.5-5 mIU/L) |
| 10 | Serum Albumin | 4.5 gm/dl (3.4-5.4 bm/dl) |
| 11 | Hemoglobin | 12.10 gm/dl (10-15 gm/dl) |
| 12 | Bone Marrow Density | -3.7 AP spine and -3.6 Femur (0 normal) |
| 13 | Serum Magnesium | 1.8 mg/dl (1.7-2.2 mg/dl) |
| 14 | Serum Sodium | 139 mEq/L (135-145 mEq/L) |
| 15 | Serum Potassium | 3.4 mmol/L (3.6-5.2 mmol/L) |
| 16 | Serum Chloride | 106 mEq/L (96-106 mEq/L) |
Table 1: Relevant blood investigations.
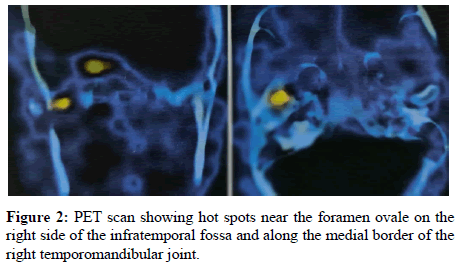
Thus, a diagnosis of a proximal myopathy with severe osteopenia and hypophosphatemia with renal phosphate wasting was made. The high levels of parathyroid hormone, lead to an investigation of the thyroid gland which was normal. Therefore, tumor induced osteopenia and hypophosphatemia was suspected. It remained to detect the location of the offending lesion. Based on the assumption that it would be a highly metabolically active lesion, a whole-body Positron emission Tomography (PET) scan was performed to detect a hotspot. The scan showed two metabolically active lesions, situated in the skull base (Figure 1). The smaller and less active lesion was seen anterior wall of the right glenoid fossa of the right Temporo Mandibular Joint (TMJ). It measured 0.9 × 0.7 cm in size. It was associated with another lesion on the lateral border of the right foramen ovale. This lesion was brighter in radio-isotope uptake and showed a centrally lucent metabolically active core with a peripheral sclerotic rim. The lesion was entirely extracranial inside the infratemporal fossa on the right side. No other abnormalities were noted elsewhere in the body. Both thyroid gland and pituitary glands were normal [7-13].
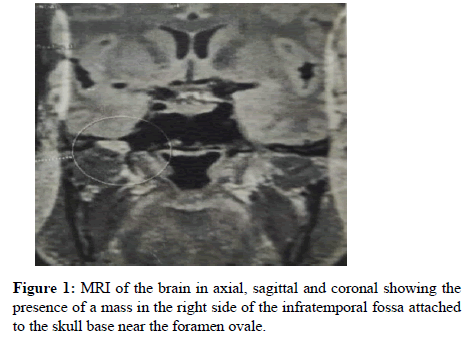
The corresponding MRI of the skull base and infratemporal fossa showed an enhancing lesion measuring 1.1 × 1.0 × 0.8 cm involving the greater wing of sphenoid. The lesion appeared hypo intense on T1, hyper intense on T2 with no evidence of diffusion restriction. There was in addition an irregular enhancement in the right malleolar region along the pterygoid fossa near the glenoid capsule of the right temporomandibular joint (Figure 2).
Once the offending lesion was located therapy was debated with a multidisciplinary meeting between neurosurgery, ENT (Skull base surgery), plastic surgery, nuclear medicine, neuroradiology, endocrinology, and intervention radiology. Options for ablation ranged from open neurosurgical excision of the lesion to radiofrequency ablation of the lesion through transcutaneous foramen ovale approach (used frequently for trigeminal neuralgia patients) the options were also extensively discussed with the patients’ relatives in a language and parlance understandable to them. It was decided to offer a micro neurosurgical excision of the lesion under neuro-navigation guidance [14-17].
Preoperatively the patient was load with intravenous phosphate and her serum electrolyte profiles were buffered to allow seamless anesthesia. The patient was positioned on the side with the neck extended and rotated to expose the temporal aspect of the skull base. A Sugita head fixation system was used to keep the head in position. After the induction of anaesthesia, entropy was used to confirm the depth of anesthesia. Medtronic Neuronavigation was set up with an accuracy of 2 mm to locate the miniscule lesion. A small U-shaped flap was employed to expose the temporal base just posterior to the temporomandibular joint as well as the intracranial temporal fossa. A small craniotomy was made to expose the temporal skull base. The distance to the foramen ovale was noted. Then the inferior margin of the bone was exposed under navigation guidance, and the foramen ovale was reached. The lateral aspect of the foramen was intensely vascular with a small lesion made out in the sub temporal soft tissue. Once confirmed by navigation, the lesion was devascularized and excised in to. The margins of the glenoid fossa were then inspected and cauterized to deal with the second less active lesion as well. The bone was then replaced and secured using titanium miniplates and self-tunnelling screws [18].
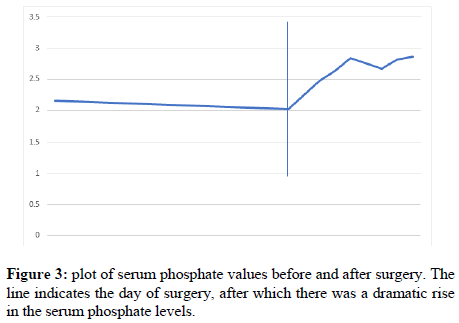
After the procedure, reversal of anaesthesia was swift and seamless. The patient was observed post operatively for 48 hours in the High Dependency Post-Operative unit (HDU), and then shifted to the ward. Her phosphate levels miraculously improved to normal in 4 days after surgery indirectly indicating a successful excision of the offending lesion (Figure 3). A post-operative CT scan was done to confirm no complications of the procedure had occurred such as hematomas or pneumocephalus [19].
The patient was discharged on post op day 7 after a near normal return of her phosphate levels and a moderate improvement of her pain and weakness. She was reviewed in the outpatient clinic of both endocrinology and neurosurgery and continues to make an impressive neurological and endocrinological recovery [20].
Results and Discussion
The most common group of tumors that are responsible for the peculiar set of symptoms exhibited by the patient in question, are called PMT or phosphatouric Mesenchymal Tumors. These are mesenchymal cell rests that become activated at various stages in life due to unknown reasons. The lesion secretes Fibroblast Growth Factor 23 (FGF-23) which leads to renal tubular loss of phosphates and subsequently initiates a biochemical cascade precipitating calcium loss. The resultant biochemical distortion leads to weak bones and muscle dysfunction which is responsible for the clinical profile. FGF-23 is a marker for these tumors and can easily be done by a simple blood ELISA assay which can confirm the diagnosis. The detection of location is much more difficult. The entrance of PET tomography has made metabolic hot spot detection much easier provided vital yet indirect evidence of the existence of these lesions. Newer targeted molecular PET studies such as DOTOTEC PET assays are more specific as they are the precursor molecules required by the PMTs and are therefore more specific to these mesenchymal entities [21].
The commonest locations of these entities are in the paranasal sinuses. Many are located either in the ethmoidal sinuses, or in the maxillary sinuses. Rarer sites include long bones, spine, intracranial lesions, orbits and skull base lesions. The skull base is unique as it may represent a confluence of 2 or more sinonasal sites which extend cranially involving the skull base of the middle or anterior cranial fossa. Intracranial extension is minimal and is usually extradural. However there are reports of intracranial lesions mimicking meningiomas which have eventually been identified as PMTs on histopathology. Apart from PMTS there are a few other lesions which can give rise to hypocalcaemia and osteomalacia. The pathological processes are different though and thus they may be differentiated based on the biochemical profile they present with. A rare tumor of the skull base is the Glomangioma which occasionally is enhanced with endocrine secretory cells which can present with this paraneoplastic syndrome. Glomangiomas are benign but a locally invasive lesion which cause local symptoms and enlarge to significant seizes necessitating attention and eventual surgery. Surgery is curative and rectifies the biochemical syndrome as well.
Rarely Brown tumors are seen in the skull base and are associated with hypocalcaemia and osteomalacia as well. However, they do not have hypophosphatemia and as associated with extremely high levels of parathyroid hormones making them quite different from the hypophosphaturia osteomalacia seen in the previous 2 cases. Brown tumors are a manifestation of a systemic disease and the management is therefore different to PMTs and Glomangiomas.
Different therapies have been tried for PMTs. Surgery remains the most effective and almost no recurrence of symptoms after a complete excision. The introduction of Neuronavigation, endoscopy and operative microscopy have significantly aided the resection accuracy of surgery making recurrence a rare and surprising entity. The benign nature of the lesion and slow growth potential of the mesenchymal cells makes even a recurrence slow to manifest and easy to detect and treat. In the last 10 years a review of literature has been attached showing only 1 case series of 2 patients were recurrence was experienced. No major morbidity was experienced in repeat surgery and the end result was complete tumor excision with relief of symptoms.
Apart from surgery targeted chemotherapy with bleomycin has been considered with almost no success. Radiotherapy was tried for 3 case reports due to incomplete resection. No additional benefit in delaying recurrence or increasing oncological clearance has been demonstrated by the addition of radiation to surgery. Radiation alone has also not shown the degree of symptom relief that surgery has shown.
Embolization of vascular feeders was done in 1 case which was identified to be inside the cavernous sinus making surgery difficult. No significant improvement was noted after the procedure and eventually surgery had to be performed to remove the lesion.
Lastly radiofrequency probe insertion and thermo destruction of the lesion was suggested by some authors where access to the lesion in the posterior and middle cranial fossas was challenging. The results of such procedures weren’t tabulated and hence it is difficult to predict outcome in this case. More documentation and analysis of the results, both short and long term, are required to define the usefulness of this procedure viz-a-viz PMT management.
Review of Literature
A review of papers published between 2010 and 2021 about skull base Phosphatouric mesenchymal tumors causing oncogenic osteomalacia and phosphaturia was done using PubMed and Google scholar (Table 2). It yielded 27 case reports and case series where a large number of cases were examined. A vast majority were managed surgically leading to complete resolution of the clinical and biochemical syndrome as in the case report above. Patients received radiation for incomplete resection, and 1 patient received embolization of feeding capillaries for a lesion situated inside the cavernous sinus [22-26].
| Study | Year of publication | Location of tumor | Clinical presentation | Procedure employed |
|---|---|---|---|---|
| Chokyu I, et al.[1] | 2012 | Middle cranial fossa base | Recurrent fractures with delayed bony fusion. Myalgia |
Extradural middle cranial fossa approach |
| Uno T, et al. [2] | 2011 | 2 cases 1 in the middle cranial fossa. 1 in the anterior cranial fossa invading the frontal lobe. | Myopathy Myalgia |
Extradural middle cranial fossa approach. Combined subtotal resection with bone removal for the anterior cranial fossa lesion |
| Kobayashi K, et al.[3] | 2011 | Temporal bone | Myopathy Myalgia | En bloc resection through an extradural middle cranial fossa approach |
| Hana T, et al. [4] | 2017 | Ethmoid sinus invading the anterior skull base | Severe myalgia | Functional endoscopic sinus surgery with en bloc resection of the lesion |
| Shah R, et al. [5] | 2018 (the study includes 5 more cases of TIO which originate from other sources (not skull base lesions) | Posterior fossa near the mastoid | Myalgia and tinnitus | Retromastoid craniotomy and excision of the lesion |
| Zhu Z, et al. [6] | 2021 | 12 patients had lesions in the middle cranial fossa and infra temporal fossa | Headache, myalgia, and weakness | Extradural craniotomy, and en bloc excision |
| Ellis MB, et al. [7] | 2016 | Cerebellar hemisphere extending onto the posterior fossa skull base | Ataxia and myalgia | Retromastoid craniotomy and tumor excision |
| Gresham MS, et al. [8] | 2017 | Ethmoid sinus and anterior skull base glomangioma induced TIO | epistaxis | Endoscopic endonasal approach and tumor excision |
| Massaccesi M, et al. [9] | 2021 | Right frontal sinus invading the anterior cranial fossa (histopathology was a ossifying fibro myxoid tumor) | Recurrent fractures and delayed bone healing | Endoscopic resection of the lesion with post-operative adjuvant radiotherapy |
| Gunawat PV, et al.[10] | 2019 | Middle cranial fossa and posterior fossa | Recurrent fractures | Infratemporal craniotomy and Retromastoid craniotomy |
| Tang R, et al. [11] | 2020 | Anterior ethmoidal sinus extending into the anterior cranial fossa | Nasal bleeds with myalgia | Tumor embolisation with endoscopic resection followed by radiation therapy |
| Kawthalkar AS, et al. [12] | 2020 | Middle cranial fossa and infratemporal fossa | Myalgia and myopathy | En bloc resection with trans orbital decompression employed in 2 cases |
| Savage CR, et al. [13] | 2009 | Pterygopalatine fossa extending into the middle cranial fossa | Recurrent Pathological fractures | Endoscopic resection followed by radiation therapy |
| Mishra T, et al. [14] | 2019 | Temporal bone | myopathy | En bloc resection |
| Tarasova VD. [15] | 2013 | Middle cranial fossa | Muscle pains | Stereotactic radiosurgery |
| Argersinger DP. [16] | 2020 | Jugular foramen | Rhinorrhea with myopathy | Far lateral approach with en bloc resection |
| Pal R, et al. [17] | 2019 | Report of 300 cases ranging from middle to anterior cranial fossa | Weakness, debilitation, myopathy, and severe myalgia | En bloc resections 18 |
| Deep NL, et al. [18] | 2014 | Posterior aspect of the nasal septum eroding into the anterior skull base | Epistaxis, rhinorrhea and myalgia | Endoscopic excision with skull base repair |
| Fathalla H, et al. [19] | 2015 | Anterior cranial fossa | Multiple occult fractures | Frontal craniotomy and resection of the lesion |
| Zhang L, et al. [20] | 2020 | Occipito-cervical junction | Spinal collapse with myelopathy and quadriplegia | Decompression with spinal stabilization |
| Ashish G, et al. [21] | 2014 | Temporal bone | Myalgia | resection |
| Ashish G, et al. [21] | 2018 | Anterior cranial fossa base, olfactory groove and frontal sinus | Myalgia, recurrent non healing fractures | Complete resection and drilling of the affected bone |
| Riminucci M, et al. [22] | 2020 | Ethmoid sinus and anterior skull base | Back pain | Ethmoidectomy with skull base repair |
| Mathis DA, et al. [23] | 2013 | Cavernous sinus | myalgia | embolization |
| Kurien R, et al. [24] | 2019 | Ethmoidal sinus eroding the anterior skull base | fractures | Ethmoidectomy with skull base repair |
| McCutcheon IE . [25] | 2011 | Anterior skull base | Myalgia Back pain fractures | Subtotal resection followed by radiotherapy |
| Politi D, et al. [26] | 2018 | Ethmoids extending into the anterior cranial fossa | Myalgia | Endoscopic resection with skull base repair |
Table 2: Case reports of skull base tumors of oncogenic Osteomalacia with hypophosphatemia over the last 10 years.
Results were best with surgery. Only 2 patients reported a recurrence which was also managed surgically and successfully resolved after 3 surgeries. Most lesions were located either in the anterior or middle cranial fossa floors extending into the infratemporal fossa. Rare locations included the craniovertebral junction, posterior fossa and frontal sinus. There were 3 reports of intracranial lesions resembling meningiomas or hemangiopericytomas which compressed the brain leading to midline shift and intracranial hematomas which needed emergency decompressions and tumor resections.
Usually the lesions were small (0.5 cm-0.9 cm) with a rich blood supply which when resected presented with a almost magical resolution of the biochemical profile. Suga, et al. have plotted the levels of FGF-23 before and after surgery in their case report and have shown along with the phosphate and calcium levels, a marked change in the FGF-23 levels which drops after resection dovetailing with the corresponding increase in phosphate and calcium levels in the blood.
Conclusion
Although PMTs are rare and an often undiagnosed lesion causing osteomalacia, they’re the main offender in phosphatouric osteomalacia syndrome and need to be investigated thoroughly. Involvement of a multidisciplinary team in the diagnosis and management of this entity will lead to complete resolution of the clinical and biochemical syndromes of the patients presenting excellent results in the short and long term. A high index of suspicion is warranted to detect these lesions.
References
- Chokyu I, Ishibashi K, Goto T, Ohata K (2012) Oncogenic osteomalacia associated with mesenchymal tumor in the middle cranial fossa: A case report. J Med Case Rep 6:1-5.
[Crossref] [Google scholar] [Indexed]
- Uno T, Kawai K, Kunii N, Fukumoto S, Shibahara J, et al. (2011) Osteomalacia caused by skull base tumors: Report of 2 cases. Neurosurgery 69:E239-E244.
[Crossref] [Google scholar] [Indexed]
- Kobayashi K, Nakao K, Kawai K, Ito K, Hukumoto S, et al. (2011) Tumor‐induced osteomalacia originating from the temporal bone: A case report. Head Neck 33: 1072-1075.
[Crossref] [Google scholar] [Indexed]
- Hana T, Tanaka S, Nakatomi H, Shojima M, Fukumoto S, et al. (2017) Definitive surgical treatment of osteomalacia induced by skull base tumor and determination of the half-life of serum fibroblast growth factor 23. Endocr J 64:1033-1039.
[Crossref] [Google scholar] [Indexed]
- Shah R, Lila AR, Jadhav RS, Patil V, Mahajan A, et al. (2019) Tumor induced osteomalacia in head and neck region: Single center experience and systematic review. Endocrine connections 8:1330-1353.
[Crossref] [Google scholar] [Indexed]
- Zhu Z, Xia W, Qi F, Wang W, Wang X, et al. (2021) Clinical characteristics and surgical outcomes of sinonasal lesions associated with tumor-induced Osteomalacia. Otolaryngol Head Neck Surg 165:223-231.
[Crossref] [Google scholar] [Indexed]
- Ellis MB, Gridley D, Lal S, Nair GR, Feiz-Erfan I, et al. (2016) Phosphaturic mesenchymal tumor of the brain without tumor-induced Osteomalacia in an 8-year-old girl: Case report. Journal of Neurosurgery: Pediatrics 17:573-577.
[Crossref] [Google scholar] [Indexed]
- Gresham MS, Shen S, Zhang YJ, Gallagher K (2017) Anterior skull base glomangioma-induced osteomalacia. J Neurol Surg Rep 78:e9-e11.
[Crossref] [Google scholar] [Indexed]
- Massaccesi M, Miccichè F, Rigante M, Petrone G, Lepre E, et al. (2021) Successful treatment of tumor-induced Osteomalacia by multidisciplinary therapy with radiation to intracranial fibromyxoid tumor. Case Rep Endocrinol 2021:8841259.
[Crossref] [Google scholar] [Indexed]
- Gunawat PV, Shaikh ST, Deopujari CE (2019) Tumour-induced Osteomalacia Secondary to Intracranial Tumours–Report of 2 Cases. J Assoc Physicians India 67:85-86.
- Tang R, Mao S, Lin H, Ye HB, Li DW, et al. (2020) Surgical treatment and outcomes for sinonasal and skull base phosphaturic mesenchymal tumors. Otolaryngol Head Neck Surg 162:674-682.
[Crossref] [Google scholar] [Indexed]
- Kawthalkar AS, Janu AK, Deshpande MS, Gala KB, Gulia A, et al. (2020) Phosphaturic mesenchymal tumors from head to toe: Imaging findings and role of the radiologist in diagnosing tumor-induced Osteomalacia. Indian J Orthop 54: 215-223.
[Crossref] [Google scholar] [Indexed]
- Savage CR, Zimmer LA (2009) Oncogenic osteomalacia from pterygopalatine fossa mass. J Laryngol Otol 123:1052-1054.
[Crossref] [Google scholar] [Indexed]
- Mishra T, Desouza MA, Patel K, Mazumdar GA (2019) Phosphaturic mesenchymal tumors involving skull bones: Report of two rare cases. Asian J Neurosurg 14:253-55.
[Crossref] [Google scholar] [Indexed]
- Tarasova VD, Trepp-Carrasco AG, Thompson R, Recker RR, Chong WH, et al. (2013) Successful treatment of tumor-induced Osteomalacia due to an intracranial tumor by fractionated stereotactic radiotherapy. J Clin Endocrinol Metab 98:4267-4272.
[Crossref] [Google scholar] [Indexed]
- Argersinger DP, Haring CT, Hanks JE, Kovatch KJ, Ali SA, et al. (2021) Phosphaturic mesenchymal tumors of the sinonasal area and skull base: Experience at a single institution. Ann Otol Rhinol Laryngol 131:647-654.
[Crossref] [Google scholar] [Indexed]
- Pal R, Bhadada SK, Singhare A, Bhansali A, Kamalanathan S, et al. (2019) Tumor-induced osteomalacia: Experience from three tertiary care centers in India. Endocrine connections 8:266-276.
[Crossref] [Google scholar] [Indexed]
- Deep NL, Cain RB, McCullough AE, Hoxworth JM, Lal D (2014) Sinonasal phosphaturic mesenchymal tumor: Case report and systematic review. Allergy Rhinol 5:2014.
[Crossref] [Google scholar] [Indexed]
- Fathalla H, Cusimano M, Di Ieva A, Karamchandani J, Fung R, et al. (2015) Osteomalacia-inducing tumors of the brain: A case report, review and a hypothesis. World neurosurgery 84:189-e1.
[Crossref] [Google scholar] [Indexed]
- Zhang L, Hu Y, Yan J, Yang J, Bai G, et al. (2020) Phosphaturic mesenchymal tumor of the occipitocervical region: Report of two rare cases and literature review.
- Ashish G, Mathew J, Thomas N, Kapoor N, Elanthenral S (2014) Phosphaturic mesenchymal tumour in the temporal bone: A rare presentation. Egyptian Journal of Ear, Nose, Throat and Allied Sciences 15:149-153.
- Riminucci M, Colangelo L, Ungari C, Cassoni A, Minisola S, et al. (2020) Naso-ethmoidal phosphaturic mesenchymal tumor: A rare tumor site for an uncommon paraneoplastic syndrome. Ear Nose Throat J 101:289-291.
[Crossref] [Google scholar] [Indexed]
- Mathis DA, Stehel EJ, Beshay JE, Mickey BE, Folpe AL (2013) Intracranial phosphaturic mesenchymal tumors: Report of 2 cases. Journal of neurosurgery 118:903-907.
[Crossref] [Google scholar] [Indexed]
- Kurien R, Rupa V, Thomas M (2019) Varied presentation of sinonasal phosphaturic mesenchymal tumour: Report of a case series with follow-up. Eur Arch Otorhinolaryngol 276:1677-1684.
[Crossref] [Google scholar] [Indexed]
- McCutcheon IE (2011) Osteomalacia caused by skull base tumors: Report of 2 cases-Commentary. Neurosurgery 69:E244.
- Politi D, Spinato G, Sulfaro S, Mauro P, Salviato T, et al. (2018) Ethmoid-nasal phosphaturic mesenchymal tumour. Austin J Infect Dis 5:1031.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi