Research Article, J Nephrol Ren Dis Vol: 1 Issue: 1
Placement of Central Venous Dialysis Catheters without X-Ray: Safety and Feasibility
Claudius Küchle*, Yana Suttmann and Ming Wen
Department of Nephrology, Klinikum rechts der Isar, Technical University of Munich, Ismaninger Street. 22, 81675, München, Germany
*Corresponding author: Claudius Küchle
Department of Nephrology, Klinikum rechts der Isar, Technical University of Munich, Munich, Germany
Tel: 0049 89 4140 5052
Fax: 0049 89 4140 4817
E-mail: claudius.kuechle@mri.tum.de
Received date: February 15, 2017 Accepted date: February 17, 2017 Published date: February 24, 2017
Citation: Küchle C, Suttmann Y, Wen M (2017) Placement of Central Venous Dialysis Catheters without X-Ray: Safety and Feasibility. J Nephrol Ren Dis 1:1.
Abstract
Objective: After a placement of central venous catheters chest x-ray is the standard procedure to exclude malpositions and pneumothoraces. In this study we combined ultrasonographic techniques to obviate the routine use of x-ray. Setting: Dialysis Center of the Department of Nephrology, Klinikum Rechts der Isar, Technical University of Munich, Munich, Germany.
Patients: 298 consecutive adult patients undergoing central venous catheter placement for hemodialysis. Interventions: None Measurements and Main
Results: 298 consecutive patients received a central venous catheter for dialysis by using chest ultrasound to localize the target vessel, guide the puncture, confirm the tip position using agitated saline as a contrast media and to rule out pneumothoraces. The ultrasound results were proven by a chest x-ray after the first dialysis session. During the two years observation period, two catheter malpositions were readily observed with ultrasound and directly corrected in the operation room. Ultrasound of the chest revealed four pneumothoraces, three of them could be confirmed by chest x-ray.
Conclusion: The prospective data presented illustrate that the combination of bedside ultrasonographic techniques is not inferior to x-ray. Our method is less time consuming, without radiation exposure and less costly than chest x-ray investigation after central venous catheter placement. As the localization of the tip is based on agitated saline as a contrast media, it adds no infrastructural demands. It is now our well founded believe that the study does challenge the mandatory use of a post insertion x-ray.
Keywords: Central venous dialysis catheter; Catheter tip location; X-ray control; Pulmonary sonography; Echocardiography; Pneumothorax
Abbreviations
ASBTE: Agitated Saline Bubble Enhanced Transthoracic Echocardiogram; CVC: Central Venous Catheter
Introduction
Since central venous catheters (CVC) were introduced into daily routine by Sven-Ivar Seldinger in 1953 [1], the procedure is hampered by the necessity to scrutinize the placement of the catheter tip and the need to rule out possible complications of the puncture. Clinical judgment does neither predict the position of CVC nor the presence of complications. Thus, chest x-ray is as yet the standard procedure to exclude malpositions and pneumothoraces [2,3]. Nonetheless, the necessity of this routine chest radiograph is questionable, as it inevitably leads to a troublesome delay in further therapy. It furthermore contributes to radiation exposure and adds costs [4-6]. 2014 we described a method to verify the position of the CVC tip by using agitated saline bubble enhanced transthoracic echocardiography (ASBTE) [6]. Combining this technique with a routine chest ultrasound after every insertion to rule out pneumothoraces, we established a routine ultrasound control protocol with the aim to place central venous catheters without x-ray control.
Methods and Results
The study was approved by the ethical committee of the Klinikum Rechts der Isar, Technical University of Munich.
298 consecutive patients who received a dialysis catheter between January 1th 2014 and December 31th 2015 in the Dialysis Center of the Department of Nephrology, Klinikum Rechts der Isar, Technical University of Munich, were included. We placed 206 double lumen tunneled catheters and 92 double lumen Shaldon catheters via internal jugular vein or angulus venosus (266 right, 32 left). Ten patients after femoral vein catheterization were excluded since there is no need for a chest x-ray, but tip localization was invariably proven by agitated saline bubble enhanced ultrasound (ASBTE) of the inferior v. cava anyhow. Data were collected routinely using a SAP system (SAP GmbH, Germany). The data included indication, catheter type, time of catheterization, location, complications and time of chest x-ray control. The physicians experience was variable, spanning less than one to more than ten years. Yet, all operators acquired basic skills on ultrasound of thoracic organs. The patients’ blood pressure, ECG and oxygen saturation were monitored continuously throughout the procedure. Different lengths of dialysis catheters were chosen according patients size and location of the catheter.
• All punctures of target vessels were performed using a 7.5 MHz linear ultrasound transducer guided Seldinger technique with a standard introducer needle.
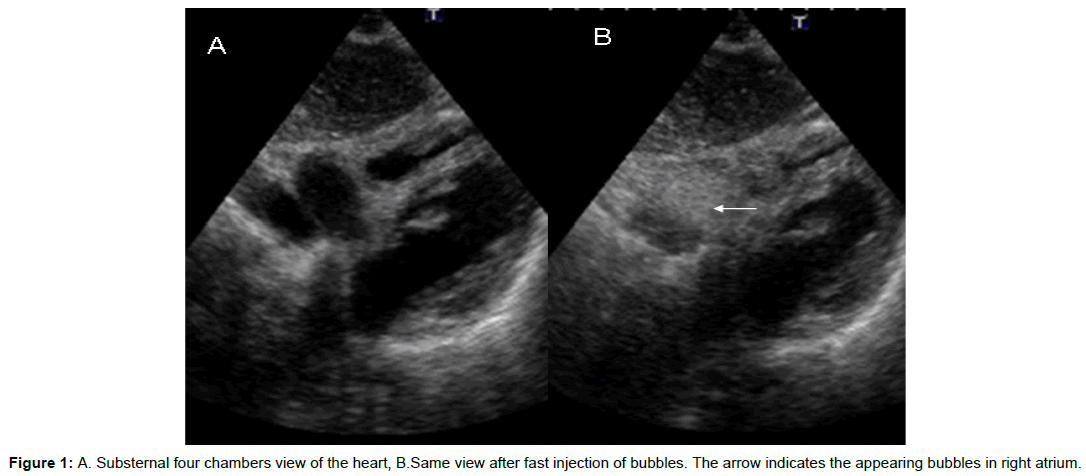
• Each catheter placement was than controlled by saline bubble enhanced transthoracic echocardiogram (ASBTE). As previously reported, using this technique any misplacement of the central venous catheter (CVC) is indicated by a delayed arrival and a decreased echogenicity of applied bubbles when judging an ultrasound view of the right atrium. The place where the bubbles first appear within the right atrium even allows the localization of the tip of the catheter (Figure 1) The bubbles are produced by fervidly shaking a 20-ml syringe filled with 19 ml of saline solution (NaCl 0.9%) and 1 ml air, then all visible air from syringe is removed and the bubble containing fluid rapidly injected through the distal lumen of the catheter. A substernal ultrasound view of the right atrium is obtained with a 3.75 MHz sector transducer, using a Toshiba Nemio SSA 550 A system [6].
| Patients: | |
|---|---|
| Number | 298 |
| Age (years) mean ± standard deviation | 64.55 ± 15.13 |
| Catheter characteristics | |
| Shaldon catheter, n (%) | 92 (30.87) |
| Permanent tunneled catheters, n (%) | 206 (69.13) |
| Catheter positions: | |
| Right jugular vein, n (%) | 266 (89.26) |
| Left jugular vein, n (%) | 32 (10.74) |
| Complications: | |
| Malpositions in the v.subclavia, n (%) | 2 (0.67) |
| Pneumothoraces seen in ultrasound, n (%) | 4 (1.34) |
| Pneumothoraces seen in x-ray, n (%) | 3 (1.01) |
| Pneumothoraces missed in ultrasound, n (%) | 0 (0) |
| Pneumothoraces missed in x-ray, n (%) | 1 (25) |
Table 1: Patients, catheter characteristics, positions and complications.
• Since ASBTE does not exclude pneumothorax, arterial injury with e.g. hemothorax or air embolism, we applied chest ultrasound as the final step after every puncture. Pulmonary ultrasound was done as described by Blaivas M in 2005 [7]. In brief, the parietal and visceral pleural surfaces are visualized sonographically as separated echogenic lines. With respiration, these two bright lines slide past each other, seen as moving vertical lines adjunct to the visceral pleura. If air intrudes in between the parietal and visceral pleura, the visceral pleural is covered by air, which is not penetrated by ultrasound. So, no sliding lung sign is observable. We searched for pneumothoraces using a 7.5 MHz linear transducer, depth settings were minimized to evaluate the pleural lines and some deeper tissue (~5– 7 cm). The sonographic views consisted of three locations on the relevant hemi thorax (anterior second intercostal space at the midclavicular line, fourth intercostal space at the anterior axillary line and sixth intercostal space at the mid axillary line) to assure for the presence of a sliding lung sign. This entire approach takes less than thirty seconds.
• Finally the proper position of CVC and exclusion of pneumothorax was reappraised by a chest x-ray after the dialysis session in all cases (medium 4h 58min after insertion). The radiologists were unaware of the ultrasound results.
As a result two catheter (0.67%) malpositions occurred. Both malpositioned catheters were readjusted readily in the operation room. No further catheter misplacement was discovered in post procedural x-ray that was performed after dialysis. Ultrasound of the chest revealed four pneumothoraces (1.34%), only three could be confirmed by chest x-ray. No unexpected pneumothorax was revealed by x-ray.
Discussion
The placement of a CVC for dialysis is associated with severe complications even in experienced hands, resulting in morbidity and mortality. The main function of a chest radiograph after placement of CVC is to rule out malposition or pneumothorax, and thus is regarded mandatory before using a catheter. In many cases x-ray requires the transport to the radiology department with its limitations of monitoring. Inevitably this leads to a troublesome delay in further therapy. If chest x-ray hence should be avoided, it must be replaced by a non inferior technique that overcomes these problems (as well as radiation and cost). Describing ASBTE to enable a clear visualization of the catheter tip we provided one component for such a technique [6]. In the actual study the idea to combine ASBTE with pleural ultrasound, has proven ultrasound to be a tool that renders x-ray dispensable. The idea was prospectively proven on 298 consecutive catheterizations. Two catheter malpositions (0.67%) were detected by ASBTE and instantly corrected. Decisively, no further misplacements were discovered in a post procedural x-ray and during further course of therapy. Using pulmonary ultrasound we detected four pneumothoraces (1.34%), one had to be treated with a chest tube immediately. Of the four pneumothoraces seen on ultrasound, one was missed by x-ray. This is not exceptionally surprising: as emphasized by Omar HR, the sensitivity of chest x-ray for identifying pneumothoraces in a supine patient is only around 50%, whereas ultrasound identifies a pneumothorax with a sensitivity ranging from 92%-100% in a supine patient [8]. Crucial for our argumentation once again is that no unforeseen pneumothorax was identified on x-ray. Therefore, our technique seems at least to be not inferior to x-ray. As a matter of cause the amount of time saved by ultrasound depends on the local settings: in our study the time between catheter insertion and chest x-ray averaged 28 min. The mean time using bedside ASBTE and pleural sonography was 3.2 minutes (minimum: 2 min, maximum: 5 min).
The low overall complication rate in our study is sufficiently explained by the fact that the majority of our catheter insertions were performed via the right internal jugular vein (89.3%), which is known to carry a low risk of pneumothorax and hematothorax. Particularly, the low complication rate is in good accordance with our conclusion that the complete setting is safe and feasible in daily routine.
Our technique allows the exclusion of a pneumothorax and the control of the catheter’s tip location directly after catheter placement. It is time- saving and avoids delay of the further therapy. Furthermore, the typical effective dose for a posterior anterior chest x-ray to adult patients of 0.02 mrem (mSv) [9] is eliminated. Besides it lessens costs. Without exposure to radiation, the combination of ASBTE and chest ultrasound offers further significant advantage over the routine use of a chest radiograph, e.g. in pregnant women and children. It addition, our data address an often heard refutation: that a pneumothorax will only be recovered two to four hours after insertion of a catheter. Our x-rays were done at least 3 hours after insertion of the CVC, nevertheless finding less pneumothoraces compared to the immediate pleural ultrasound, rendering non inferiority of immediate ultrasound obvious.
Conclusion
The ultrasound guided placement of CVC for dialysis in combination with agitated saline bubble enhanced transthoracic echocardiogram to reveal catheter misplacements and chest ultrasound to rule out pneumothoraces is safe and feasible in daily clinical routine. The presented ultrasound control protocol adverts the time consuming, radiation exposing and costly chest x-ray investigations after CVC placements with no detectable loss of safety.
Our study does challenge the mandatory use of a post insertion x-ray.
References
- Seldinger SI (1953) Catheter replacement of the needle in percutaneous arteriography; a new technique. Acta radiolo 39: 368-376.
- Eisen LA, Narasimhan M, Berger JS, Mayo PH, Rosen MJ, et al. (2006) Mechanical complications of central venous catheters. J Intensive Care Med 21: 40-46.
- Abood GJ, Davis KA, Esposito TJ, Luchette FA, Gamelli RL (2007) Comparison of routine chest radiograph versus clinician judgment to determine adequate central line placement in critically ill patients. J Trauma 63: 50- 56.
- Molgaard O, Nielsen MS, Handberg BB, Jensen JM, Kjaergaard J, et al. (2004) Routine X-ray control of upper central venous lines: Is it necessary?. Acta Anaesthesiol Scand 48: 685-689.
- Bailey SH1, Shapiro SB, Mone MC, Saffle JR, Morris SE, et al. (2000) Is immediate chest radiograph necessary after central venous catheter placement in a surgical intensive care unit?. Am J Surg 180: 517-521.
- Wen M, Stock K, Heemann U, Aussieker M, Küchle C (2014) Agitated saline bubble-enhanced transthoracic echocardiography: a novel method to visualize the position of central venous catheter. Crit Care Med 42: 231-233.
- Blaivas M, Lyon M, Duggal S (2005) A prospective comparison of supine chestradiography and bedside ultrasound for the diagnosis of traumatic pneumothorax. Acad Emerg Med 12: 844-849.
- Omar HR, Abdelmalak H, Mangar D, Rashad R, Helal E, et al. (2010) Occult pneumothorax, revisited. J Trauma Manag Outcomes 4: 4-12.
- Wall BF, Hart D (1997) Revised radiation doses for typical X-ray examinations. Report on a recent review of doses to patients from medical X-ray examinations in the UK by NRPB. National Radiological Protection Board. Br J Radiol 70: 437-439.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi