Research Article, J Otol Rhinol Vol: 8 Issue: 1
Prevalence of Allergic Rhinitis among Secondary School Students in Kinondoni District, Dar es Salaam, Tanzania
Zephania Saitabau Abraham1*, Sweety Bindiya Aubroo2, Victor Mashamba2, Daudi Ntunaguzi3 Kassim Babu Mapondella3 and Enica Richard Massawe3
1Department of Surgery-University of Dodoma, College of Health and Allied Sciences, Tanzania, Box 259, Dodoma,Tanzania
2Department of Otorhinolaryngology, Muhimbili National Hospital, Box 65001, Dar es Salaam,Tanzania
3Department of Otorhinolaryngology, Muhimbili University of Health and Allied Sciences, Box 65001, Dar es Salaam, Tanzania
*Corresponding Author : Zephania Saitabau Abraham
Department of Surgery, University of Dodoma, College of Health and Allied Sciences, Tanzania
E-mail: zsaitabau@yahoo.com
Received: January 21, 2019 Accepted: March 01, 2019 Published: March 09, 2019
Citation: Abraham ZS, Aubroo SB, Mashamba V, Ntunaguzi D, Mapondella KB, et al. (2019) Prevalence of Allergic Rhinitis among Secondary School Students in Kinondoni District, Dar es Salaam, Tanzania. J Otol Rhinol 8:1. doi: 10.4172/2324-8785.1000363
Abstract
Background: An estimation of about 1.4 billion people are suffering from allergic rhinitis worldwide. Allergic rhinitis (AR) is a common debilitating disorder that can adversely affect the quality of life and the academic performance of school-age children and its prevalence has steadily increased during the last decades. Data on the prevalence of allergic rhinitis among secondary school students is scarce despite its reported impact on academic performance of students. The aim of this study was thus to determine the prevalence of allergic rhinitis and its clinical presentation among secondary school students in Kinondoni District which is one of the largest districts in Dar es Salaam Region. Methods: A community based, cross-sectional prospective study was conducted in selected secondary schools in Kinondoni district, Dar-es-salaam Region from August 2016 to January 2017 using random sampling technique. Data was collected using pre-tested questionnaires and analyzed using SPSS program. Results: A point prevalence rate of 34.7% was reported with female predominance and 15-16 age group predilections were found in this study. Sneezing (65.9%) was the commonest nasal symptom and hypertrophied inferior turbinate (61.8%) the commonest physical finding. The commonest aeroallergen was the house dust mite (77.3%) and the least was dog fur (5.9%). Conclusion: Allergic rhinitis affects a significant proportion of the adolescent population with symptoms which have a negative impact on the lifestyle of these participants. The common aeroallergens are found within our immediate surroundings e.g. house dust mite, which can be controlled if participants are educated and proper clean environmental control measures are instituted.
Keywords: Prevalence; Allergic rhinitis; Secondary school; Students; Kinondoni; Tanzania
Background
Allergic rhinitis (AR) is an atopic disease with growing impact on everyday medical practice as its prevalence has steadily increased during the last decades [1]. The WHO (World Health Organization) classifies allergic rhinitis as the fourth most important chronic disease in the world. It is associated with significant morbidity and impact the quality of life of millions of individuals. Lost productivity at work or school and total healthcare expenditure exceeds several billion dollars annually in United States.
Allergic rhinitis is frequently ignored, under diagnosed, misdiagnosed, and mistreated, which not only is detrimental to health but also has social costs [2] with an estimation of >$6 billion spent on prescription medications alone [2]. The total burden of allergic rhinitis lies on impaired physical and social functioning and also in a financial burden for treatment of its co-morbid diseases [3].
AR symptoms should be a red flag for a potentially serious lifelong effect, as the younger the child, the greater the likelihood of suffering severe and persistent disease and its related co-morbidities later in life [4]. The negative effect of early symptoms should not be downplayed. Allergic Rhinitis is one of the most common as well synonyms of hay fever, which commonly arise as a response to allergens such as pollen, dust, animal dander (old skin) and mold. Classic symptoms of Allergic Rhinitis manifests as nasal congestion, nasal itch, rhinorrhea (runny nose), sneezing, and itchy eyes.
Allergic rhinitis is divided into seasonal and perennial. Seasonal rhinitis is a disease particularly of teenagers and young adults and appears to be less common in primary and preschool age children. In seasonal rhinitis, with relevant grass pollen sensitization, the link between allergen exposure and rhinitis is clear cut. However, in other situations such as perennial rhinitis and house dust mite allergen sensitization, the link between symptoms and allergen exposure is less certain [5].
Allergic Rhinitis (AR) has major co-morbidities in patients includes asthma, nasal polyps (NP), adenoid hypertrophy, tonsillitis, hypertrophied turbinate, conjunctivitis, rhinosinusitis (RS), otitis media with effusion (OME) and sleep apnea [6-8]. Patients suffer from insomnia snoring, and obstructive sleep apnea syndrome, which result in daytime somnolence, fatigue, irritability, depression, reduced productivity and memory and learning deficit. There is a strong correlation between severity of rhinitis symptoms, particularly nasal obstruction, and sleep impairment. Treatment of AR results in better sleep, reduced daytime somnolence, and improved quality of life [9].
Methods
Study design and participants
This study was a community based cross-sectional study and it was carried out between August 2016 to January 2017 and included all secondary day school students in the selected schools during the period of study in Kinondoni district, Dar es Salaam, Tanzania.
Study population
The study population included all secondary day school students in the selected schools during the period of study aged between 13 to 19 years.
Sampling method
Simple random sampling technique was used in which selection based on sample. The study population included secondary day school students aged between 13 to 19 years of age in randomly selected schools. A list of all private and government owned secondary day schools was obtained from the Kinondoni education officers in Dar-es-salaam and was divided into two groups, government or private. Using simple random sampling 7 secondary schools were selected from each group for the first stage. Then 2 classes were selected from each shcool from the frist stage using simple random technique and consent and assent forms were given to selected classes students.
Inclusion and exclusion criteria
All students aged between 13-19 years old (adolescents group) who consented to participate in the study. Students who were not present in school on the day of screening, refusal to participate in SPT, nasal infection or with skin diseases e.g. eczema
Sample size calculation
The sample size, n was calculated from the following formula;
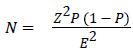
Whereby:
N= an estimated sample size
Z= Standard normal deviation (1.96) which correspond to 95% confidence interval
P= % (P is the prevalence of Allergic Rhinitis among secondary school students taken to be 38.6% from a study which was conducted in Kenya)
E= Margin Error (4 %)
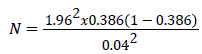
N = 569
Therefore, from above formula the required minimum sample size was 569.
Adjusting for non-response rate which was 10%, the required sample size was 626.
Data collection methods
An introduction letter by the education officer at the Kinondoni District education office was obtained after receiving relevant research clearance. The introduction letter was given to principals/ head teachers in those secondary day schools which were selected. At selected secondary day schools, the primary researcher and research assistants introduced themselves and explain clearly the aim and purpose of the study that was conducted.
Consent forms were distributed to 2 randomly selected classes and were collected by respective teachers.
Students whose parents/guardian/caretaker gave the informed consent were interviewed using the structured questionnaire/clinical examination form. The interview was conducted only by the primary researcher and two trained assistants. A study number was assigned to each student whose parent/guardian/caretaker had consented and data was recorded according to the structured questionnaire.
During screening of students, they were grouped into small groups until they were all screened in an available quite classroom where privacy of each student was maintained.
Data was administered in questionnaire inclusive of student’s age, sex, nasal symptoms, eye symptoms, age of onset of symptoms, triggering factors, presence or absence of family history of allergic rhinitis, results of skin prick allergy test(if conducted). Rhinoscopy findings were recorded in structured questionnaire. During rhinoscopy, aseptic techniques were applied for each pupil.
Using a questionnaire 626 students were interviewed by primary investigator and two trained assistants, students were divided into two groups; the symptomatic and asymptomatic.
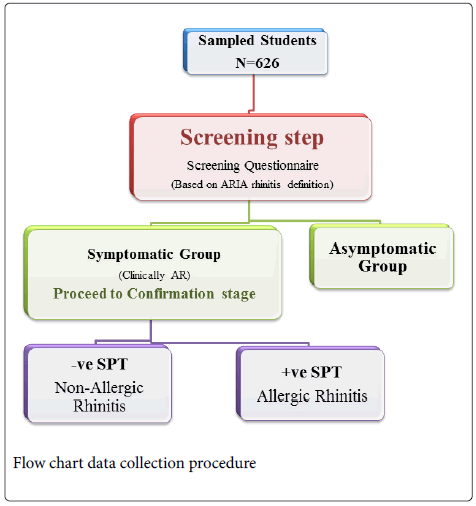
Students with watery runny nose with one or more of the other symptoms or with nasal congestion alone was also recruited as ARIA suggest that AR patient could only present with nasal congestion alone were considered to have symptoms of AR.
Students who were symptomatic for allergic rhinitis were proceeded for Confirmation of AR which were physical examination and SPT. The important physical findings included; a pale edematous nasal mucosa, hypertrophy of inferior turbinates, watery / mucoid rhinorrhoea, transverse nasal crease, allergic shiners and/or conjunctiva injection.
Skin allergy test was done by primary researcher and two trained assistants to all students who showed allergic signs and symptoms for the aim of determining the commonest allergen causing allergic rhinitis and this was done before the students start antihistamines or steroids. Those who were constantly on steroids and antihistamines were excluded from the study and if had already started but use of antihistamines or steroids intermediately were recruited in the study.
Data analysis
Data analysis was done using SPSS version 20 as per specific research questions using frequencies and percentages for statistical association between age, sex and allergic rhinitis. Chi-square test was used to compare proportions. P value of <0.05 was considered statistically significant.
Ethical considerations
Students were provided with an informed consent and then asked to provide written consent to participate in the study. For students younger than 18 years, informed consent was obtained from their parents or guardians. Ethical approval was provided by Research and Publication Committee of the Muhimbili University of Health and Allied Sciences (MUHAS).
Results
Demographic characteristics of the study population
Between August 2016 and January 2017, a total of 626 students were recruited from all secondary day school students in the selected schools Kinondoni District in Dar-es-salaam. Majority were females (54.1%, n=383) than males (46.9%, n=243), giving a male to female ratio of 1.6:1. The overall mean age of the participants was 15.3 years (SD ± 1.39). The mean age for male was 15.5 years (SD ± 1.4) while for female were 15.1 years (SD ± 1.36). Most of the participants belonged to the age group 15-16 years (46.18%, n=289).were recruited city. (Table 1)
| Frequency (n) | Percentage (%) | ||
|---|---|---|---|
| Gender | |||
| Males | 243 | 38.8 | |
| Females | 383 | 61.2 | |
| Total | 626 | 100 | |
| Age group | |||
| 13-14 years | 219 | 35 | |
| 15-16 years | 289 | 46.2 | |
| 17+ | 118 | 18.8 | |
| Total | 626 | 100 |
Table 1: Distribution of study population according to Gender and Age.
Out of 626 school children, 243 (38.8%) were males and 383 (61.2%) were females, where majority 289 (46.2%) were of age group 15-16 followed by other groups as shown in table 1 above.
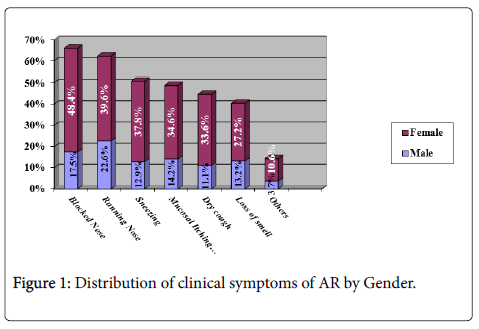
Out of 626 students, 217 (34.7%) had clinical features of allergic rhinitis where by majority (15.3%) were in the age group 15-16, (P= 0.421). Females had higher proportion 146 (23.3%) as compared to males 71 (11.3%), (P= 0.023). (Figure 1)
Nasal congestion was the most common symptom reported by majority of students (65.9%) and the most affected age group was 15-16 (29.5%).
Turbinate hypertrophy was the most common physical finding by majority of students (61.8%) and females were most affected than males (42.4%), (p>0.05). (Figure 2)
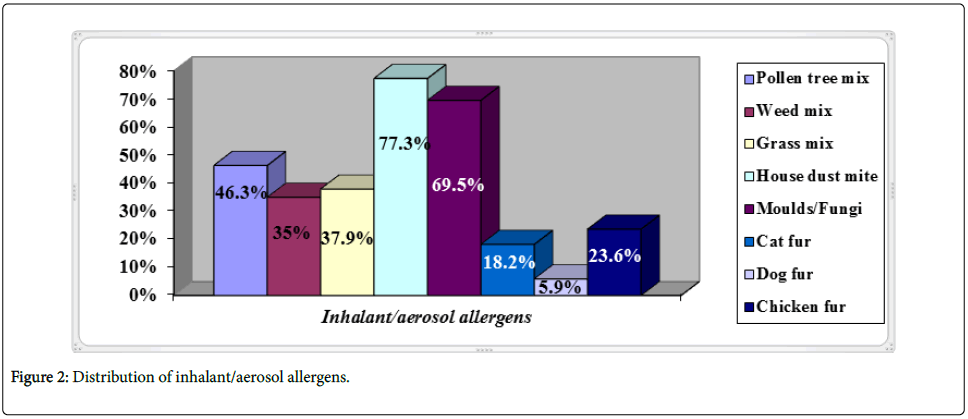
Majority of students were allergic to house dust mite (77.3%) followed by other allergens as shown on the histogram above.
Discussion
The present study was designed to determine the prevalence and clinical presentation of allergic rhinitis among adolescents aged 13-19 years in a community set up. Allergic rhinitis negatively affects the quality of life, sleep and impairs the process of learning [10-14]. This study correlated prevalence of AR to age, sex, determine the presentation and pattern of symptomology and common aeroallergens involved.
The worldwide prevalence of childhood allergic rhinitis varies from 8.5% - 14.6% [14,15]. In developed countries prevalence of allergic rhinitis in adolescents has been noted to be higher as compared to less developed countries. There is little information regarding trends of AR in Africa, as few studies have been done. Some studies done in African countries have shown prevalence to range 13.9% to 45.2% [16,17].
Prevalence of allergic rhinitis
In this study, the prevalence of AR was 34.7% based on clinical features using a questionnaire; this was further confirmed by using SPT which gave a prevalence of 32.4%. The results of this study are analogous to the findings of follow up studies done in South Africa and Kenya which involved children aged 13-14 years [18,19]. In the study done in South Africa prevalence of AR was noted to increase from 30.4% to 38.5% over a period of seven years [18]. Whereas, in the Kenyan study is significantly high increment of 38.6% from 14.9% was noted in the prevalence of AR over a six year period [19].
Contrary to the findings of this study, Said et al, in a study done at Bugando, Mwanza reported a lower prevalence of AR, which was 14.7% [20], similarly Nyembue et al, in their study from DRC reported a prevalence of 13.9% [16]. However the study population in these groups included participants aged from 5 to 83 years, whereas in this study the participants were aged 13-19 years. In comparison to this study, the study conducted in DRC was also community based, whereas the study done in Bugando medical centre was a hospital based study.
Geographical factors
Considerable variations have been noted in prevalence of AR both between countries and centres in the same country [21]. This has been postulated to be due to various factors such as climatic changes, dietary changes, and environmental factors. In this study a high prevalence of AR was noted, which may be attributable to continuing changes in the region over the last few decades, such as stepping up industrialization, changes in lifestyle, vehicle pollution, modernization and urbanization. Adaption of the western lifestyle has vastly contributed to this increment in AR. According to the “hygiene hypothesis”, the cleaner the environment the higher the prevalence of AR, therefore it is expected that the more urbanized the area the higher the prevalence of AR [11,22,23]. Since this study was conducted in Dar-es-Salaam which is a highly industrialized and polluted area a high prevalence was noted. These findings are comparable to studies conducted in similar environments, high prevalence of 38.5% in south Africa and 45.2% in both Lebanese and Nigerian adolescents were noted [17,18,23]. Higher prevalence have also been noted in western countries which are more industrialized and urbanized than the African countries [25,26].
| Allergic rhinitis | |||
|---|---|---|---|
| Non allergic (n %) | Allergic (n %) | Total (N %) | |
| Age Group | |||
| 13-14 | 145 (23.2) | 74 (11.8) | 219 (35.0) |
| 15-16 | 193 (30.8) | 96 (15.3) | 289 (46.2) |
| 17+ | 71 (11.3) | 47 (7.5) | 118 (18.8) |
| Total | 409 (65.3) | 217 (34.7) | 626 (100) |
| Gender | |||
| Male | 172 (27.5) | 71 (11.3) | 243 (38.8) |
| Female | 237 (37.9) | 146(23.3) | 383 (61.2) |
| Total | 409 (65.3) | 217 (34.7) | 626 (100) |
Table 2: Distribution of Patients with Allergic rhinitis who tested positive upon Skin Prick Test and those with Non allergic symptoms.
Prevalence in relation to age group
Although AR may have its onset at any age, the incidence of onset is greatest in children at adolescence with a decrease in incidence seen in advancing age. A prevalence of 11.8% was seen in age group of 13-14 years, 15.3% in age group of 15-16 years and a prevalence of 7.5% in participants above 17 years of age, however this difference in occurrence of AR among age groups was not statistically significant (p=0.421). In comparison to this study, several other studies have shown a three to four fold increment in prevalence of AR in the 13-14 age group [17,18,23]. A recent study done in Angola in 2017 also showed a 2 fold increase in prevalence of AR in children in this age group [27]. Although AR has been reported in infants, in most cases an individual requires two or more seasons of exposure to a new antigen before exhibiting clinical manifestations of AR [28]. Older children have been noted to have a higher prevalence than younger ones with a peak occurring in children aged 13-14 years. A study done in Bugando medical centre showed an overall prevalence of AR to be 14.7 % among which 68.9% of participants were aged below 20 years. Approximately 80% of individuals diagnosed with AR develop symptoms before age of 20 years [22]. The reason for this increased prevalence of AR in adolescents maybe due to increased exposure to allergens, irritants when they play around and are exacerbated by viral upper respiratory tract infections.
Prevalence in relation to sex
Females had a higher preponderance to AR as compared to males in this study (P=0.023). This finding was in agreement with several other studies which have shown a higher prevalence among girls than boys [18,29-32]. Other studies have so no difference in prevalence of AR in either sex [20,25,33]. However some studies have also shown predominance towards males [34]. The sex predilection of AR still remains a controversial issue with most literature not agreeing on sex predominance. It was found in some studies that “Hormonal rhinitis” and a higher rate of cosmetic use in females may account for increased prevalence [35,36]. In general, natural history of the disease shows initially higher prevalence rates among boys during childhood, shifting later to be higher among females during adolescence and adulthood.
Presentation and pattern of symptomatology
Allergic rhinitis is clinically defined as a symptomatic disorder of the nose induced after allergen exposure by an IgE-mediated inflammation [22].
Allergic rhinitis can also be defined as allergen induced inflammation of the nasal membrane and surrounding tissues that results in sneezing, rhinorrhoea, conjunctivitis, nasal congestion, and pruritis of the nose, palate, throat and ears [37].
Symptoms of allergic rhinitis
Nasal symptoms consisted of blocked nose, running nose and sneezing of which were 65.9%, 62.2%, and 50.7% respectively. Other symptoms included mucosal itch, dry cough, and loss of sense of smell. This finding is in agreement with several other studies which found nasal symptoms to be more predominant, of which blocked nose was the most common and troublesome symptom [20,38,39]. Blocked nose was found to reduce sleep time, resulting in decreased day time activity, particularly sport involvement which is more popular among adolescents [40].
Other symptoms included ocular symptoms which were watering, itching and redness of eyes. Similar finding were obtained in the Bugando study where ocular symptoms were reported by few participants and considered as trivial [20]. High prevalence of ocular symptoms in patients with AR has also been noted, 61.2% and 52.0% in Korea and France respectively [41,42]. Patients with moderatesevere persistent AR showed highest prevalence of ocular symptoms [41]. These ocular symptoms have been thought to be due to release of histamine by allergens which initiate a naso-ocular reflex causing above mentioned symptoms [43].
Signs of allergic rhinitis
This study showed inferior turbinate hypertrophy (61.8%) to be the most common sign followed by mucosal edema (30.4%) and lastly nasal crease (18.4%). Inferior turbinate hypertrophy was found to be the most common sign in several other studies. The Bugando study also showed inferior turbinate hypertrophy to be among one of the common signs of allergic rhinitis, with 40.5% [20].
In this study 47% children with allergic rhinitis had pale mucosa, 22.9% had pink, 17.5 had cyanotic and 12.9 had hyperemic nasal mucosa. In a study done in Iran 52% of children with allergic rhinitis had pale mucosa, 36% had violaceous and 12% had normal mucosal color [44].
Allergen sensitization by SPT
Skin prick testing (SPT) is generally considered to be the gold standard for diagnosis of AR, it is safe and effective and used as a routine diagnostic test. It is used frequently to assess specific sensitization and to verify the diagnosis of allergic (IgE mediated) disease in symptomatic subject [45].
Allergens in our study were identified by analyzing skin prick test (SPT) of 217 participants with positive clinical symptoms out of which, 203 tested positive for the SPT and 14 participants tested negative. This negative reaction could probably be due to participants being sensitized to other allergens not included in the allergic kit for tests, or not yet identified or some non-allergic forms of rhinitis among our cases.
This study reported a high sensitization to house dust mite (77.2%) followed by moulds/fungi (69.8%), then pollen tree (46.5%), grass (38.1%), weed (35.1%), and least to animal danders like chicken feather, cat fur, then dog dander in decreasing frequencies.
Dermatophagoides pteronyssinus (HDMs) was the predominant allergen in this study, and it is considered as a major source of allergens in house dust. Several studies have reported house dust mite to be the most common allergen [47-51].
Our study recorded mould/fungi (69.8%), as the second important source of allergens, which has also been observed in a study done in Iraq and Iran [50,52].
| Physical features | Male (n %) | Female (n %) | Total (N %) |
|---|---|---|---|
| Nasal Crease | 17 (7.8) | 23 (10.6) | 40 (18.4) |
| Allergic Shiner | 8 (3.67) | 6 (2.8) | 14 (6.5) |
| Hypertrophied Turbinate | 42 (19.4) | 92 (42.4) | 134 (61.8) |
| Nasal Mucosa Edema | 20 (9.2) | 46 (21.2) | 66 (30.4) |
| Others (Nasal Polyps, Denne’s lines) | 2 (0.9) | 3 (1.4) | 5 (2.3) |
Table 3: Distribution of physical features of AR by Gender.
Human skin scale is the food source of HDMs and the feces of HDM is the antigenic product [50]. The growth and proliferation of HDM depends on specific environmental factor such as temperature and humidity. As for mould/fungi, most allergenic moulds grow on nonliving organic matter, while a few require a living host. Both groups need moisture, oxygen, preformed carbohydrate and occasionally additional growth factors. Many familiar moulds grow actively at 20 °C and may flourish well above or below this temperature. Although fungal components may be ingestant allergens, inhaled spores are the major source of exposure. Dar-es- salaam has a relatively high humidity ratio and temperature which is compatible with both mite and mould/fungi growth hence an increased prevalence of the two allergens was noted in our study.
Sensitization to other allergens such as grass, weed and animal dander had a relatively low prevalence in this study. Conversely in European and North American countries, sensitization to cat and dog dander have been noted to be the most common aeroallergens [53,54]. This could probably be due to the increased number of cats and dogs that are present in the homes as pets in these countries in comparison to Tanzania.
Polysensitization by SPT
Most participants (84.1%) in this study were reactive to 2 or more allergens. Several other studies have shown similar results [55,56]. This polysensitization might be the result of various factors such as interactions of genetic or environmental factors which favor growth and vegetation of specific plant species that cause allergic reactions. It could also be due to cross-reactivity among allergens belonging to close reservoirs which reflects the presence of common genetic component [56-58].
Conclusion
Allergic rhinitis was found to be quite prevalent (34.7%) among secondary school students in Kinondoni District. Nasal obstruction was the most common symptom and turbinate hypertrophy was found to be the most common sign of AR. Dust mite was the most common allergen responsible for the symptomatology of AR in this study.
Ethics approval and consent to participate
The approval to conduct the study was granted by Ethics and Research Committee for Muhimbili University of Health and Allied Sciences
Availability of data and material
The detailed reported information can be obtained from the corresponding authors when needed and from archives of the department of otorhinolaryngology-MUHAS
References
- Hellings PW, Fokkens WJ (2006) Allergic rhinitis and its impact on otorhinolaryngology. Allergy Eur J Allergy Clin Immunol 61: 656-664.
- Greiner AN, Hellings PW, Rotiroti G, Scadding GK (2011) Allergic rhinitis. Lancet. 378: 2112-2222.
- Nathan RA (2007) The burden of allergic rhinitis. Allergy Asthma Proc 28: 3-9.
- Westman M, Stjärne P, Asarnoj A, Kull I, Van Hage M, et al. (2012) Natural course and comorbidities of allergic and nonallergic rhinitis in children. J Allergy Clin Immunol 129: 403-408.
- Kemp AS (2009) Allergic rhinitis. Paediatr Respir Rev 10: 63-68.
- Han DH, Rhee C (1997) Comorbidities of Allergic Rhinitis. Comorbidities Allerg Rhinitis
- Hardjojo A, Hadjojo A, Shek LP, van Bever HP, Lee BW (2011) Rhinitis in children less than 6 years of age: current knowledge and challenges. Asia Pac Allergy 1: 115-122
- Bousquet J (2003) Allergic Rhinitis and its Impact on Asthma (ARIA). Clin Exp Allergy Rev 3: 43-45.
- Seedat RY (2013) Allergic rhinitis-More than just a nuisance. Curr Allergy Clin Immunol. 26: 26-27.
- Craig TJ, McCann JL, Gurevich F, Davies MJ (2004) The correlation between allergic rhinitis and sleep disturbance. J Allergy Clin Immunol 114: S139-S145.
- Zivkovic Z, Cerovic S, Djuric-Filipovic I, Vukasinovic Z, Jocic-Stojanovic J, et al. (2012) Clinical Implications and Facts About Allergic Rhinitis (AR) in Children. Allerg Rhinitis.
- Walker S, Khan-Wasti S, Fletcher M, Cullinan P, Harris J, et al. (2007) Seasonal allergic rhinitis is associated with a detrimental effect on examination performance in United Kingdom teenagers: case-control study. J Allergy Clin Immunol 120: 381-387.
- Valls-Mateus M, Marino-Sanchez F, Ruiz-Echevarría K, Cardenas-Escalante P, Jiménez-Feijoo R, et al. (2017) Nasal obstructive disorders impair health-related quality of life in adolescents with persistent allergic rhinitis: A real-life study. Pediatr Allergy Immunol 28: 438-445.
- Mir E, Panjabi C, Shah A (2012) Impact of allergic rhinitis in school going children. Asia Pac Allergy 2: 93-100.
- Mallol J, Crane J, von Mutius E, Odhiambo J, Keil U, et al. (2013) The International study of asthma and allergies in childhood (ISAAC) phase three: A global synthesis. Allergol Immunopathol (Madr) 41: 73-85.
- Nyembue TD, Jorissen M, Hellings PW, Muyunga C, Kayembe JM (2012) Prevalence and determinants of allergic diseases in a Congolese population. Int Forum Allergy Rhinol 2: 285-293
- Olawuyi F, Osinusi K, Onadeko BO, Falade AG (1998) Prevalence and severity of symptoms of asthma, allergic rhino-conjunctivitis and atopic eczema in secondary school children in Ibadan, Nigeria. East Afr Med J 75: 695-698.
- Zar HJ, Ehrlich RI, Workman L, Weinberg EG (2007) The changing prevalence of asthma, allergic rhinitis and atopic eczema in African adolescents from 1995 to 2002. Pediatr Allergy Immunol 18: 560-565.
- Esamai FO, Ayaya S, Nyandiko W (2002) Prevalence of asthma, allergic rhinitis and dermatitis in primary school children in uasin gishu district, Kenya. East African Med J 79: 514-518.
- Said S, McHembe MD, Chalya PL, Rambau P, Gilyoma JM (2012) Allergic rhinitis and its associated co-morbidities at Bugando Medical Centre in Northwestern Tanzania; A prospective review of 190 cases. BMC Ear Nose Throat Disord 12: 13.
- Ait-Khaled N, Odhiambo J, Pearce N, Adjoh KS, Maesano IA, et al. (2007) Prevalence of symptoms of asthma, rhinitis and eczema in 13- to 14-year-old children in Africa: The international study of asthma and allergies in childhood phase III. Allergy Eur J Allergy Clin Immunol 62: 247-258.
- Bousquet J, Khaltaev N, Cruz AA, Denburg J, Fokkens WJ, et al. (2008) Allergic rhinitis and its impact on asthma (ARIA) 2008 Update. Allergy 63: 8-160.
- Reid CE, Gamble JL (2009) Aeroallergens, allergic disease, and climate change: Impacts and adaptation. Ecohealth 6: 458-470.
- Musharrafieh U, Al-Sahab B, Zaitoun F, El-Hajj MA, Ramadan F, et al. (2009) Prevalence of asthma, allergic rhinitis and eczema among Lebanese adolescents. J Asthma 46: 382-387.
- Peñaranda A, Aristizabal G, Garcia E, Vasquez C, Rodriguez-Martinez CE, et al. (2012) Allergic rhinitis and associated factors in schoolchildren from Bogotá, Colombia. Rhinol 50: 122-128.
- Blaiss MS (2008) Pediatric allergic rhinitis: Physical and mental complications. Allergy asthma Proc 29: 1-6
- Arrais M, Lulua O, Quifica F, Rosado-Pinto J, Gama JMR, et al. (2017) Prevalence of asthma and allergies in 13-14-year-old adolescents from Luanda, Angola. Int J Tuberc Lung Dis 21: 705-712.
- Kulig M, Klettke U, Wahn V, Forster J, Bauer CP (2000) Development of seasonal allergic rhinitis during the first 7 years of life. J Allergy Clin Immunol 106: 832-839.
- Borges WG, Burns DAR, Felizola MLBM, Oliveira BA (2006) Prevalence of allergic rhinitis among adolescents from Distrito Federal, Brazil: Comparison between ISAAC phases I and III. J Pediatr (Rio J) 82: 137-143.
- Mercer MJ, Joubert G, Ehrlich RI, Nelson H, Poyser MA, et al. (2004) Socioeconomic status and prevalence of allergic rhinitis and atopic eczema symptoms in young adolescents. Pediatr Allergy Immunol 15: 234-241.
- Kurt E, Metintas S, Basyigit I, Bulut I, Coskun E, et al. (2009) Prevalence and risk factors of allergies in turkey (PARFAIT): Results of a multicentre cross-sectional study in adults. Eur Respir J 33: 724-733
- Al-Ghobain, Al-Moamary, Al-Hajjaj, Al- Fayez B (2012) Prevalence of rhinitis symptoms among 16 to 18 years old. Indian J Chest Dis Allied Sci 55: 11-14.
- Josephine E, Eniola A, Yemisi A, Clement N (2014) The prevalence of nasal diseases in Nigerian school children. J Med Sci 5: 71-77
- Sofranac M (2008) correlation between allergic rhinitis, asthma and atopic dermatitis in children. Pediatrics 121: S91-S91.
- Wallace D, Dykewiz M, Berstein D, Blessingmoore J, Cox L, et al. (2008) The diagnosis and management of rhinitis: An updated practice parameter. J Allergy Clin Immunol 122: S1-84.
- Philpott CM, El-Alami M, Murty GE (2004) The effect of the steroid sex hormones on the nasal airway during the normal menstrual cycle. Clin Otolaryngol Allied Sci 29: 138-142.
- Gelfand EW (2005) Pediatric allergic rhinitis: Factors affecting treatment choice. Ear Nose Throat J 84: 163-168.
- Juniper EF, Guyatt GH, Dolovich J (1994) Assessment of quality of life in adolescents with allergic rhinoconjunctivitis: Development and testing of a questionnaire for clinical trials. J Allergy Clin Immuno 93: 413-423.
- Shedden A (2005) Impact of nasal congestion on quality of life and work productivity in allergic rhinitis: Findings from a large online survey. Treat Respir Med 4: 439-446.
- Sundberg R, Torén K, Höglund D, Aberg N, Brisman J (2007) Nasal symptoms are associated with school performance in adolescents. J Adolesc Health 40: 581-583
- Lee JE, Kim KR, Rha KS, Dhong HJ, Roh HJ, et al. (2013) Prevalence of ocular symptoms in patients with allergic rhinitis: Korean multicenter study. Am J Rhinol Allergy 27: 135-139
- Klossek JM, Annesi-Maesano I, Pribil C, Didier A (2012) The Burden associated with ocular symptoms in allergic rhinitis. Int Arch Allergy Immunol 158: 411-417.
- Baroody FM, Foster KA, Markaryan A, DeTineo M, Naclerio RM (2008) Nasal ocular reflexes and eye symptoms in patients with allergic rhinitis. Ann Allergy Asthma Immunol 100: 194-199
- Akbari H, Farid-hosseini R, Miri S, Amin R (2006) The prevalence of allergic rhinitis among 11-15 years-old children in Shiraz. Pediatr Allergy Immunol 1: 133-137.
- Bousquet J, Khaltaev N, Cruz AA, Denburg J, Fokkens WJ, et al (2008) ARIA: Allergic Rhinitis and its Impact on Asthma 2008 Update. 1-196.
- Edizer DT, Canakcioglu S (2013) Epidemiologic Features of House Dust Mite and Pollen Sensitizations in Patients with Allergic Rhinitis in Istanbul (1993-2006). Istanbul Med J 14: 29-34
- Ngahane BHM, Noah D, Motto MN, Njankouo YM, Njock LR (2016) Sensitization to common aeroallergens in a population of young adults in a sub-Saharan Africa setting: A cross-sectional study. Allergy Asthma Clin Immunol 12: 1.
- Farrokhi S, Gheybi MK, Movahed A, Tahmasebi R, Iranpour D, et al. (2015) Common aeroallergens in patients with asthma and allergic rhinitis living in southwestern part of Iran: Based on skin prick test reactivity. Iran J Allergy Asthma Immunol 14: 133-138.
- Moitra S, Sen S, Datta A, Das S, Prasanta SBD, et al. (2014) Study of Allergenicity Spectrum to Aero Allergens by Skin Prick Testing. Austin J Allergy 1: 1-4.
- Benzarti M, Mezghani S, Jarray M, Garrouche A, Khirouni S, et al. (2002) Skin test reactivity to seven aeroallergens in a Sousse area population sample. Tunis Med 80: 450-454.
- Leung R, Ho P, Lam CK, Lai CW (1997) Sensitization to inhaled allergens as a risk factor for asthma and allergic diseases in Chinese population. J Allergy Clin Immunol 99: 594-599.
- Taha B, Abd AI, Saleh RI, Mohammed T, Aljubory H (2011) A study of Skin prick test reactivity to common aeroallergens among patients with allergic rhinitis in salahelden governorate. Tikrit J Pure Sci 16: 40-44.
- R Jaramillo, A Calatroni, CH Weir MS (2014) Prevalence of allergic sensitization in the United States: Results from the National Health and Nutrition Examination Survey (NHANES) 2005-2006. J Allergy
- Bousquet PJ, Chinn S, Janson C, Kogevinas M, Burney P, et al. (2007) Geographical variation in the prevalence of positive skin tests to environmental aeroallergens in the European Community Respiratory Health Survey I. Allergy 62: 301-309.
- Aburuz S, Bulatova N, Tawalbeh M (2011) Skin prick test reactivity to aeroallergens in Jordanian allergic rhinitis patients. East Mediterr Health J 17: 604-610.
- Dowaisan A, Al-Ali S, Khan M, Hijazi Z, Thomson MS, et al. (2000) Sensitization to aeroallergens among patients with allergic rhinitis in a desert environment. Ann Allergy Asthma Immunol 84: 433-438.
- Weber RW (2003) Patterns of pollen cross-allergenicity. J Allergy Clin Immunol 112: 229-239.
- Fereidouni M, Hossini RF, Azad FJ, Assarehzadegan MA, Varasteh A (2009) Skin prick test reactivity to common aeroallergens among allergic rhinitis patients in Iran. Allergol Immunopathol (Madr) 37: 73-79.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi