Research Article, Arch Transplant Vol: 4 Issue: 5
Renal Transplant for Patients on Long-term dialysis (20 or more years) at our Center
Koichi Kozaki1,2*, Naoki Adachi1, Nahoko Ohtani1, Akiho Okada1, Taketo Kato1, Kenji Yuzawa2 and Tohru Terashima1
1Department of Surgery, National Hospital Organization Mito Medical Center, Ibaraki, Japan
2Transplantation Surgery, National Hospital Organization Mito Medical Center, Ibaraki, Japan
*Corresponding Author : Koichi Kozaki
Department of Surgery and Transplantation Surgery, National Hospital Organization Mito Medical Center, 280 Sakuranosato, Ibaraki-machi, Higashiibaraki-gun, Ibaraki, ZIP 311-3193, Japan
E-mail: k.kozaki.d@mn.hosp.go.jp
Received: March 05, 2018 Accepted: April 20, 2018 Published: April 27, 2018
Citation: Kozaki K, Adachi N, Ohtani N, Okada A, Kato T, et al. (2018) Renal Transplant for Patients on Long-term dialysis (20 or more years) at our Center. Arch Transplant 2:1.
Abstract
Background: In Japan, patients undergoing dialysis (HD) are increasing every year, and the number has reached 329,609 at the end of 2016. Out of them, 26,313 patients were undergoing longterm HD (LHD; 20 or more years), which is 8.3% of the total number. The number of renal transplantation (RTx) was 1,648 in 2016, 1,471 for living donor RTx (LRTx) and 177 for deceased donor RTx (DRTx). Moreover, in Japan, DRTx did not increase, while the number of patients enrolled in the waiting list for DRTx reached approximately 13,000. Due to this discrepancy, the mean waiting period of DRTx is approximately 17 years, and many patients waiting for DRTx have no choice but to choose LRTx. LHD patients are at a risk of serious morbidity and mortality from the various complications of HD, which hamper outcomes of RTx. Patients and methods: This study was designed to investigate the results of RTx in LHD patients. Ninety patients underwent RTx between May 2006 and December 2017, of which 14 patients were in the LHD group. The most important surgical problem in RTx of the patients on LHD is a contracted disuse bladder (CDB). Its incidence was 42.9% (6 out of 14 patients) in LHD group and 5.3% (4 out of 76 patients) in non-LHD group respectively, significantly higher in non-LHD group than in non-LHD group(p=0.02). Results: All of patients with a CDB underwent ureteroureterostomy. Patients on LHD showed significantly lower graft survival and patient survival rate than those on non-LHD (LHD: 8/14, 57.1% vs. non-LHD: 62/76, 81.6%, p=0.02 and LHD: 12/14, 85.7% vs. non- LHD: 73/76, 96.1%, p=0.004). Conclusion: Preoperative evaluation and perioperative countermeasures are required in LHD patients.
Keywords: Renal transplant; Hemodialysis; Ureteroureterostomy
Introduction
In Japan, the hemodialysis patients are increasing year by year, and it was reported that the number rose to 329,609 according to the statistics at the end of 2016 [1]. Patients on a long-term dialysis (LHD; 20 or more years) were 26,313 which accounted for 8.3% of the total number. Due to the progress in the management of end-stage renal disease and the shortage of deceased donor kidneys, the number of patients undergoing LHD is increasing continuously. And, in our country, there are very few, deceased donor renal transplantation (DRTx), therefore the waiting time of enrolled patients is extremely prolonged, with the mean waiting time being approximately 17 years [2,3].
Most patients with LHD are at a risk of serious morbidity and mortality due to several complications, such as cardiovascular disease, cerebrovascular disease, malignant tumors, hepatitis C, and contracted disuse bladder. Because the QOL of patients with LHD after renal transplantation (RTx) may be remarkably worsened by these complications, the evaluation of complications before RTx is important. We analyzed RTx patients who underwent RTx at Mito Medical Center to determine the impact of LHD on RTx outcomes. RTx in patients on LHD is considered to be common in Japan and is rarely performed in Western Europe or the United States.
In this article, we described the results of RTx in LHD patients at our hospital suggesting the importance of comprehensive and close preoperative evaluation of the patient on LHD.
Patients: We analyzed 90 patients who underwent RTx between May 2006 and December 2017 in this retrospective observational study. Seventy-one patients underwent living donor RTx (LRTx) and 19 underwent DRTx. Fourteen patients were on LHD, while the remaining were on HD for less than 20 years. Of the 14 patients on LHD, 11 were male and three were female; 12 underwent DRTx; and the mean age was 55.3 (range: 44-62) years old. Of the 76 patients on non-LHD, 63 were male and 13 were female, with a mean age of 46.1 (range: 15-75) years old (Table 1).
| LHD group | Non-LHD group | |
|---|---|---|
| Number of patients | 14 | 76 |
| Type of RTx | Deceased donor : 12 | Deceased donor : 7 |
| Living donor : 2 | Living donor : 69 | |
| Mean age (year) | 55.3 (44~62) | 46.1 (15~75) |
| Gender | Male : 11 | Male : 63 |
| Female : 3 | Female : 13 | |
| Mean HD period (month) | 302.5 (242~385) | 61.5 (0~232) |
LHD: Long-term Dialysis; more than 20 years Non-LHD: less than 20 years on HD.
Table 1: Background of Patients.
Immunosuppressive method: All patients received quadruple immunosuppression, with cyclosporine or tacrolimus, mycophenolate mofetil, prednisolone and basiliximab.
Methods
We collected data on age, gender, serum creatinine level, HD period, the number of patients with cardiovascular disease, cerebrovascular disease, malignancy, infectious disease (pneumonia, cystitis, pyelonephritis, cytomegalovirus (CMV) infection etc.), acute cellular rejection (ACR), hepatitis C virus (HCV) antibodypositivity (HCV-positive) and patients who underwent urinary tract reconstruction with ureteroureterostomy in end-to-end fashion from donor-to-recipient ureter for a contracted disuse bladder (CDB) in addition to graft loss and death after RTx. All data were collected from medical records.
As important concern for RTx in LHD patients is an increased risk of urologic complications because of an atrophic bladder caused by an extended anuric period. Generally, the bladder volume of transplant recipients with a dialysis history of 10 or more years is decreased due to atrophy, and ureteroneocystostomy may cause such complications as urinary leakage and fistula when the bladder volume is less than 50 ml because of long-term anuria. In such cases, an anastomosis of graft ureter with the native ureter is advantageous. Therefore, we assumed the cut-off value of bladder volume less than 50 ml.
Statistical analysis
Results are presented as means values. Paired t-test was used for comparison of the LHD and non-LHD patients. All analyses were performed as two-tailed; p-values of <0.05 were considered to be statistically significant. Survival rates were estimated with the Kaplan-Meier method. StatView for Macintosh (version 5.0 Abacus Corporation, Baltimore, MD, USA) was used for statistical analysis.
Results
The mean HD period was 302.5 and 61.5 months in the LHD and non-LHD groups, respectively. Comparison between the LHD and the non-LHD groups revealed no statistically significant difference in the incidence of cardiovascular disease, and cerebrovascular disease, and the serum creatinine levels, respectively.
There were significantly more HCV positive patients in the LHD group than in the non-LHD group (LHD group: 5/14, 35.7% vs. non-LHD: 4/76, 5.3%, p=0.0008). Post-transplant liver dysfunction was detected in 8 of 9 HCV-positive patients (88.9%) and in 5 of 81 HCV-negative patients (6.2%). A high rate of post-transplant liver dysfunction was observed in HCV-positive patients. In addition, 6 of 9 HCV-positive patients were on LHD for 20 or more years.
Among patients who underwent urinary tract reconstruction using ureteroureterostomy for a CDB were 6 of 14 (42.9%) in the LHD group and 4 of 76 (5.3%) were in the non-LHD group (LHD: 42.9% vs. non-LHD: 5.3%, p=0.02) (Tables 2 and 3). Significantly many patients underwent ureteroureterostomy in the LHD group.
| LHD group | Non-LHD group | P value | |
|---|---|---|---|
| Number of HCV positive patients | 5 | 4 | 0.0008 |
| (5/14 ; 35.7%) | (4/76 ; 5.3%) | ||
| Number of CDB | 6 | 4 | 0.02 |
| (6/14 ; 42.9%) | (4/76 ; 5.3%) |
LHD: Long-term Dialysis; more than 20 years No-LHD: less than 20 years on HD
Table 2: Incidence of HCV-positivity and CDB.
| LHD group | Non-LHD group | P value | |
|---|---|---|---|
| Cardiovascular disease | 6 | 9 | NS |
| (6/14; 42.9%) | (9/76; 11.8%) | ||
| Cerebrovascular disease | 2 | 3 | NS |
| (2/14; 14.3%) | (3/76; 3.9%) | ||
| Malignancy | 0 | 6 | 0.008 |
| (6/76; 7.9%) | |||
| Infection | 8 | 23 | 0.008 |
| (8/14; 57.1%) | (23/76; 30.3%) | ||
| CMV Infection | 1 | 6 | 0.02 |
| (1/14; 7.1%) | (6/76; 7.9%) | ||
| ACR | 4 | 27 | <0.0001 |
| (4/14; 28.6%) | (27/76; 35.5%) |
ACR: Acute Cellular Rejection
Table 3: Incidence and types of Complications.
The incidence of cardiovascular and cerebrovascular disease showed no significant difference between both groups. The incidence of infectious diseases was higher, but the incidence of malignant tumors, CMV, and ACR was lower in the LHD group than in the non-LHD group (Table 3).
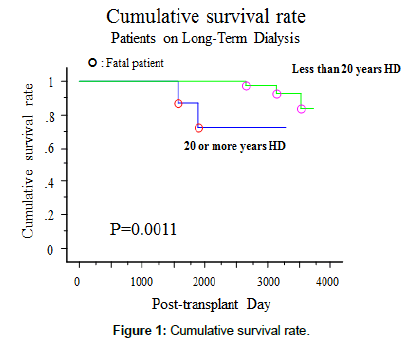
There were no significant differences in the post-RTx mean serum creatinine level between both groups. The incidence of graft loss was 42.9% in the LHD group and 18.4% in the non-LHD group, which was significantly higher in the LHD group than in the non- LHD group (p=0.02). The rate of patient death was 14.3% in the LHD group and 3.9% in the non-LHD group, which was significantly higher in the LHD group than in the non-LHD group (p=0.004), and the survival rate was significantly lower in the LHD group (p=0.0011, Table 4 and Figure 1).
| LHD group | Non-LHD group | P value | |
|---|---|---|---|
| Mean S-Cr (mg/dl) | 1.32 (0.76~1.7) (N=8) | 1.55 (0.77~2.68) (N=62) | N.S |
| Graft loss | 6 | 14 | 0.02 |
| (6/14; 42.9%) | (14/76; 18.4%) | ||
| Mortality | 2 | 3 | 0.004 |
| (2/14; 14.3%) | (3/76; 3.9%) |
LHD: Long-term Dialysis; more than 20 years Non-LHD: less than 20 years on HD
Table 4: Serum creatinine, graft loss and mortality after kidney transplantation.
The causes of graft loss are shown in (Table 5). Two patients in the LHD group died of infectious diseases and three patients in the non- LHD group died of an infection, malignant tumor and cardiovascular disease, respectively.
| LHD group (N=6) | Non-LHD group (N=14) | |
|---|---|---|
| Death with functioning graft | 1 | 0 |
| Acute rejection | 0 | 2 |
| Chronic rejection | 1 | 4 |
| Noncompliance | 0 | 1 |
| Nonfunctioning kidney | 2 | 1 |
| Nephrotoxity | 0 | 1 |
| Nephritis | 1 | 1 |
| Pyelonephritis | 1 | 0 |
| Other | 0 | 4 |
Table 5: Cause of graft loss after kidney transplantation.
Discussion
In Japan, approximately 1,500 cases per year of RTx are performed, and the number of patients on HD is increasing year by year. Furthermore, the number of patients on LHD is also increasing as the HD techniques are progressing [1,2]. RTx between spouses comprises approximately 40% of the total LRTx, because their child raising period is finally over or they have reached a retirement age after having played their social role and either of one is on dialysis for a long term waiting for RTx [2]. According to the Annual Report of Japan Society for Transplantation: Renal Transplant Registry in 2016, the average HD period was 2.9 ± 4.8 years for LRTx and 16.1 ± 8.4 years for DRTx. In DRTx, patients on HD for more than 10 years were 73.9%, and those on HD for over 20 years accounted for 34.2% of the total number [2]. In Japan, for these reasons, most of DRTx recipients are often on LHD. The LHD patients often develop various complications, including cardiovascular disorder, anti-HCV positivity and CDB [3].
As our series of results, there were many HCV antibody-positive and CDB patients in LHD group. The renal graft survival rate was low, and mortality was high in LHD group (Tables 2 and 3). These results are similar to those of reports from other institutions [4]. It was always thought that HCV-positive patients tended to have a long dialysis history. Moreover, when an HCV-positive patient on dialysis underwent RTx, renal graft survival and patient survival rates were decreased, compared to those in HCV-negative recipients. Therefore, antiviral treatment should be used before RTx for HCV-positive patients to prevent posttransplant viral proliferation, the development of liver disease and HCV-associated nephropathy resulting in poor graft survival and patient survival rates after RTx. Furthermore, as liver dysfunction may progress after RTx in HCV-positive patients, HCV eradication is important because hepatic fibrosis may progress even if liver function is normal.
Six of 9 HCV-positive patients had a dialysis history of 10 or more years, and 5 had a dialysis history of 20 or more years. In Japan, erythropoietin became available for clinic use in 1990, but dialysis induction began before 1990 in these 5 patients. All received blood transfusions to treat renal anemia. Therefore, blood transfusion was a probable cause of HCV infection in these 5 patients. Two other patients received blood transfusion during surgery before 1990, and one patient underwent RTx in China. These were the presumed causes of HCV infection. The source of infection was unknown in the remaining patients.
In HCV-positive patients with an eGFR ≤ 50 mL⋅min-1⋅1.73 m-2, including those on dialysis and many RTx recipients, ribavirin (Rib) is contraindicated, and, in principle, IFN monotherapy is indicated. While there are reports of IFN therapy exerting relatively significant effects on HCV in RTx patients, there are also reports of IFN therapy resulting in renal graft loss due to IFN-induced rejection. In Japan, daclatasvir/asunaprevir (DCV/ASV) combination therapy was introduced as IFN-free antiviral therapy in September 2015 [5]. The safety of this therapy in RTx patients or HD patients has not been established. If the efficacy and safety of DCV/ASV therapy in RTx patients and HD patients can be verified in the future, IFN therapy will probably no longer be used. However, IFN therapy, which is unrelated to drug resistance, may be given to HCV-infected patients with multiple-drug resistant viruses induced by the treatment with oral medications alone. In the future, HCV eradication therapy should be performed more actively in HCV-infected renal transplant recipients so as to protect and preserve hepatic function and improve long-term survival of renal grafts.
To repeat, DCV/ASV combination therapy was introduced in Japan as IFN-free antiviral therapy in September 2015. However, its safety in HD patients has not been established, and most of patients still require IFN therapy. It is required to cultivate a mutual and better understanding between transplant physicians/surgeon and doctors engaged in dialysis therapy to ensure that HCV patients who are planning RTx receive the treatment for HCV before RTx. However, HCV treatment has not yet been provided appropriately in HD facilities, and is a serious problem [6].
The incidence of ACR was lower in the LHD than in the non- LHD group. Patients with chronic renal failure and those on dialysis exhibit immunocompromised feature, with impaired neutrophil, monocytes, macrophage, and T and B lymphocyte function [7]. These are especially common in the elderly on long-term dialysis, and lead to impaired immunocompetence [8]. Prevention of rejection with use of immunosuppressive agents may further impair immunocompetence. This may have accounted for the deaths of 2 patients on LHD due to infection. A patient on LHD may be sensitized before RTx by pregnancy or blood transfusion; these high-risk patients must be monitored for antibody mediated rejection; it is essential to recognize that these patients are always in an immunocompromised state [7].
Most patients with LHD are at a risk of serious morbidity and mortality due to several complications, such as cardiovascular disease, cerebrovascular disease, malignant tumors, HCV, and CDB. Since the QOL of patients with LHD after RTx may be remarkably worsened by these complications, the close and comprehensive evaluation before RTx and perioperative countermeasures are important by the close cooperation with other specialty departments in these patients with various complications.
Conclusion
No significant difference between both groups was observed in the serum creatinine levels, but the prognosis was poor in patients on LHD. Our results suggest that HD treatment for 20 or more years causes adverse effects on graft and survival in post-transplant patients. Although the improvement in the complications induced by LHD are limited even after the RTx, most of the patients can achieve good ADL and QOL after RTx [9]. For further improvement in outcomes of RTx in LHD patients, preoperative, close and comprehensive evaluation and perioperative countermeasures are required.
Acknowledgement
The authors thank all physician, staff in Mito Medical Center.
References
- Masakane I (2016) 2016 Annual Dialysis Data Report, JSDT Renal Data Registry. J Jpn Soc Dial Ther 51: 1-51.
- Annual progress report from the Japanese Renal Transplant Registry: number of renal transplantations in 2016 and follow-up survey (2016) Jpn J transplant 52 : 113-133.
- Okamoto M, Suzuki T, Nobori S, Ushigome H, Yoshimura N (2009) A case of successful kidney transplantation after an extremely long-term maintenance haemodialysis of 38 years. NDT Plus 2: 398-400.
- Ushigome K, Sakai K, Suzuki T, Nobori S, Yoshimura N, et al. (2008) Kidney transplantation for patients on long-term hemodialysis. Transplant Proc 40: 2297-2298.
- Kohjima M, Motomura K, Koyanagi T, Tada S, Yamashita N, et al. (2016) Therapeutic safety and effectiveness of dual oral therapy with Daclatasvir and Asunaprevir for chronic HCV patients with renal dysfunction. 57: 142-145.
- Kozaki K, Adachi N, Okada A, Kato T, Yuzawa K, et al. (2017) The current status of renal transplant recipients with hepatitis C: A single center experience. Annals Clin Lab Res 5: 154-157.
- Nakao T, Matsumoto H, Okada T (2000) Infectious disease of chronic renal failure and hemodialysis patients. J Jpn Soc Int Med 89: 2304-2308.
- Sakamoto M, Kobayashi S, Ishii S, Mori T, Ooyama T (1986) Some nutritional parameters and immunocompetence in elderly people. J Jpn Soc Nutr Food Sci 39: 1-8.
- Tominaga Y, Uchida K, Haba T, Takagi H (1996) Renal transplantation for patients in long-term hemodialysis. J Jpn Soc Dial Ther 29: 351-358.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi