Research Article, J Surg Clin Pract Vol: 2 Issue: 2
Similar 48-H Analgesic Effects of Pre Vs. Post-Operative Pregabalin in Orthopaedic Oncological Patients: A Randomised Double-Blind Placebo-Controlled Trial
Uri Amit1, Yehuda Kollender2, Solomon Dadia2, Shoshana Chazam1, Aviram Hochstadt1, Ron Flaishon3 and Avi A Weinbroum1*
1Department of Research and Development, Tel Aviv Sourasky Medical Centre, Tel Aviv, Israel
2Orthopaedic Oncology Unit, Tel Aviv Sourasky Medical Centre, Tel Aviv, Israel
3Post-Anaesthesia Care and Day Care Surgery Units, , Tel Aviv Sourasky Medical Centre, Tel Aviv, Israel
*Corresponding Author : Avi A. Weinbroum
Department of Research & Development, Tel Aviv Sourasky Medical Centre, 6 Weizmann Street; Tel Aviv 6423906, Israel
Tel: +972-3-6973237
Fax: +972-3-6925749
E-mail: draviw@tasmc.heatlh.gov.il
Received: February 27, 2018 Accepted: April 04, 2018 Published: April 11, 2018
Citation: Amit U, Kollender Y, Dadia S, Chazam S, Hochstadt A, et al. (2018) Similar 48-H Analgesic Effects of Pre Vs. Post-Operative Pregabalin in Orthopaedic Oncological Patients: A Randomised Double-Blind Placebo-Controlled Trial. J Surg Clin Pract 2:2.
Abstract
Background: Pain after the excision of orthopaedic-oncologic lesions is intense, and its suppression requires unusual measures. Multimodal analgesia, the currently preferred analgesic methodology, consists of two or more drugs administered concurrently, an approach that induces better analgesia than if using a single agent. Pregabalin is an analgesia adjuvant that is increasingly administered perioperatively. Its 24-h pre- vs. 24-h post-incisional analgesic effects have never been compared in orthopaedic-oncologic patients.
Methods: We compared prospectively, double-blindly and randomly the 48-h postoperative analgesic requirements and numerical pain scores following the administration of pregabalin 150 mg twice daily before surgery and placebo after surgery (the PRE group) versus placebo before surgery and pregabalin after surgery (the POST group), in 50 patients undergoing small bone and soft tissue tumour resection.
Results: Both groups had similar American Society of Anaesthesiologists class, age and weight values. Both protocols resulted in similar patient-rated pain scales. The immediate (48-h) total consumption of opioids and non-opioid analgesics, as well as their late (3 and 6 months) usage were lower in the POST group. Neither group reported significant adverse events.
Conclusion: Either 24-h pre- or postoperative pregabalin (150 mg bid) provides similar 48-h analgesia after minor orthopaediconcologic lesion resections. However, pregabalin postoperative coadjuvation spares opioids or non-steroidal anti-inflammatory drug usage.
Keywords: Orthopaedics; Oncology; Pregabalin; Preoperative; Postoperative; Analgesia
Introduction
Satisfactory pain relief is an essential component in the complex management of the surgical patient. Optimal postoperative pain control correlates with better clinical outcomes and patient’s overall satisfaction rate. Despite new techniques and drug protocols for postoperative pain management, many patients still experience pain following surgery [1-3]. Multimodal analgesia [4] is a pharmacological approach where opioids, the main stay of postoperative analgesia, are coadjuvated by drugs that are of lower analgesic potency by themselves. Their co-administration improves pain scores, thus sparing opioid consumption and, therefore, diminished opioids’ side effects [5,6]. Pregabalin is an antiepileptic that is used in chronic pain [7,8]. Pregabalin (S+-3-isobutyl GABA) binds selectively to the alpha(2)-delta site of presynaptic voltage-gated calcium channels, which inhibit the release of pro-nociceptive neuropeptides [9]. This prevents the release of excitatory neurotransmitters that activate pain pathways, thus reducing intensification of neural response to incoming pain stimulus. Reviews and meta-analyses of pregabalin have concluded that, compared to placebo, its perioperative use permits an opioid-sparing effect and improves postoperative pain scores [10,11]. It has been tested in various orthopaedic procedures, with encouraging results [12,13]. The results of preoperative usage of analgesic drugs as opposed to their postoperative administration have been inconclusive [14]. We herein compared the pre-emptive 24-h pre-incisional administration of pregabalin vs. the 24-h postoperative (preventive) ones on pain grades, and on analgesics requirements, during the immediate and delayed periods following surgery.
Materials and Methods
All patients were enrolled in Tel-Aviv Sourasky Medical Centre; this study was approved by the institutional human research and ethics committee and was registered on http://www.clinicaltrials.gov (NCT01359059). After receiving full explanation of the aims of the study and the protocols, the study drug, and the numerical rating scale (NRS, 0 = no pain at all, 10 = unbearable pain), consenting participants signed a Helsinki-approved informed consent. Consenting American Society of Anaesthesiologists (ASA) physical status I-III patients, who were scheduled to undergo surgery for bone and soft tissue open cancer diagnosis and removal in orthopaedic-oncological department between November 2012 and October 2013, were eligible for enrolment. There were zero participants who suffered from neuropathic pain, and pain scores were all ≤ 4/10 in the numerical rating score (NRS), which indicates non-pain conditions (data not shown). Exclusion criteria were as follows: known allergy to opioids, pregabalin, various intraoperative anaesthetics, or non-steroidal antiinflammatory drugs (NSAIDs), a history of chronic pain unrelated to the current disease, present/past psychiatric disorders, and recent use of opioids or centrally acting drugs. Patients who were to undergo regional anaesthesia, those of age <18 or >80 years, pregnant women, or those of a history of severe liver or renal failure, were also excluded. Postoperative collected data were excluded at time points when SpO2 measured <92% under 40% oxygen by facemask, or changes in heart rate or blood pressures >20% compared to preoperative values, postoperative need for controlled or assisted ventilation, or clinically apparent emergence delirium. Data pertaining to patients who were not discharged within 72 h after surgery, or who necessitated postoperative intensive care surveillance, were also discarded. These clinical limitations follow previous documentations indicating the non-reliability of drug-related effects on pain or analgesic requirements under such conditions. Fifty patients were prospectively, double-blindly and randomly (using the RANDOM. com software, RANDOM.ORG. Randomness and Integrity Services Ltd., Dublin, Ireland; www.random.org) allocated into two groups. The medical centre’s pharmacist handled randomization and blinding; the signor researcher only (A. A. W.) was acquainted with these records, whereas all others were blinded as to the drug assignment. Patients who received 150 mg pregabalin at 20:00 the evening before surgery and again 1.5 h before surgery, comprised the PRE group; patients who were given identical capsules containing 500 mg starch (placebo) at the same preoperative times, comprised the POST group. No other premedication was administered. Postoperatively, the patients in the PRE group were given placebo, and the patients in the POST group received doses of pregabalin 150 mg. Either group was given the blinded capsule 2 h after termination of the surgery, and then twice for the first 24 h postoperatively. The 2-h-postoperative dose was added to compensate for surgery time lasting >3 h and the need for perioperative blood and fluid replacements, which would distort the pharmacodynamics and kinetics of the tested drug. All patients were given general anaesthesia (GA). The same surgical and anaesthesia teams performed all the procedures. GA consisted of intravenous (IV) propofol and fentanyl given for induction, and during maintenance, and muscle relaxants, as deemed appropriate by the attending anaesthetist, whose choices and drug doses took into account the intraoperative hemodynamic and surgical circumstances. Nitrous oxide was omitted from the anaesthetic protocol. Extubation was performed in the operating theatre and the patient was transferred to the post-anaesthesia care unit (PACU) for recovery follow-up. The first vital signs and pain scores were rated at the time of arrival. Analgesia consisted of IV or oral opioids, NSAIDs, or their combinations. Upon the first request of the patient for pain relief in the PACU, and when the pain NRS was >4/10, the first analgesic dose was granted by the attending anaesthetist, who was unaware of the patient’s group assignment and who attended to all the patients in the PACU. Additional demands for antinociceptives were satisfied with IV or oral opioids, dipyrone or paracetamol, or intramuscular diclofenac, as customary in our institution. The patients were transferred to the department of orthopaedic oncology when pain control, as evidenced by two consecutive pain scores (by NRS) were ≤4/10, minimal time of stay has passed (≥ 1 h), and Aldrete Discharge Scoring resulted ≥9/10. The main outcome measures included (1) subjectively rated pain intensity, using the 0 to10 NRS; (2) consumption of various analgesics, opioids being expressed as morphine equivalents (see below); and (3) late (1, 3, and 6 months) self-rated NRS and analgesic consumption (reported by telephone). Morphine (MO) milligram equivalent (MME) doses were calculated based on conversion formulae of opioid tables to morphine [15].
Statistics
The analyses were performed using the SPSS Release for Windows Version 24 (IBM, Armonk, NY, USA, 2016). Based on our previous studies [16,17], a power table consisting of pain NRS delta of 2, alpha = 0.05, and a power of 0.8, resulted in 18 patients/group. The demographic data (age, weight, ASA physical class), intraoperative parameters, length of stay (LOS), and the amounts of the perioperative administered drugs, were all compared using the twotailed t-test. Analyses of large variance parametric records used the Mann-Whitney U-Test. The pain scores and the overall satisfaction levels were similarly analysed. Gender distributions, as well as rates of side effects, were analysed using the Fisher exact test. Demographics, surgery and anaesthesia data of eventual dropped-outs, were analysed using the “intent-to-treat” method. All values are expressed as mean ± standard deviation (SD) from the mean, percentage of the cohorts, or number of cases, with the statistical significance being defined as P ≤ 0.05.
Results
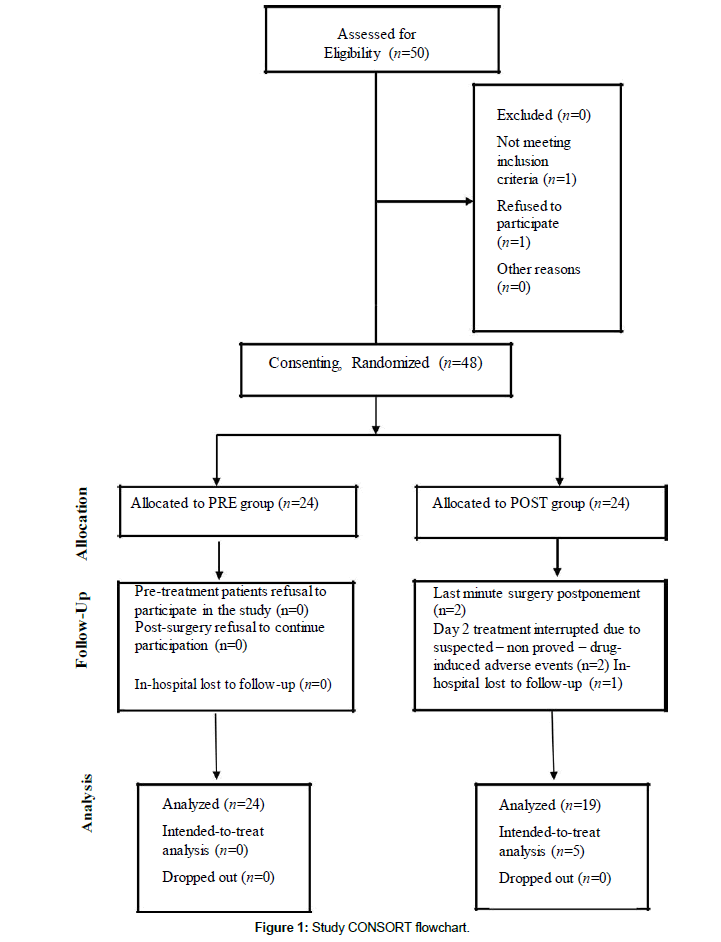
Of the 50 patients enrolled, 24 PRE and 19 POST patients completed the study (Figure 1). There were no significant differences between the groups with regard to demographics, intraoperative data (Table 1), and surgery-related complications (none, data not shown). The main goal of this investigation was the comparable non-inferiority of pre- vs. post-incisional pregabalin administration in terms of postoperative pain scores and analgesic requirements. Importantly, both the on-admission and the preoperative pain scores were similar between the two study groups (Table 2); the preoperative usage of NSAIDs was also similar between the groups (data not shown).
| Parameter (n of patients) | PRE groupa (24) | POST groupa (19) | P value |
|---|---|---|---|
| Age (years) | 42 ± 13.5 | 46.1 ± 15.2 | 0.316 (U) |
| Gender (males, %) | 42 | 53 | 0.547 (F) |
| Weight (kg) | 78 ± 17.4 | 74.8 ± 15.8 | 0.47 (U) |
| ASA (class) | 1.38 ± 0.5 | 1.44 ± 0.51 | 0.78 (U) |
| Total Intraoperative MME (mg/patient) | 16.38 ± 4.48 | 15.82 ± 7.55 | 0.951 (U) |
| LOS (days) | 2 ± 1 | 1.5 ± 0.9 | 0.037 (U) |
aSee text for group definitions.
ASA, American Society of Anesthesiologists; MME, morphine milligram equivalent; LOS, Length of stay; U, Mann Whitney U-Test; F, Fisher Exact Test.
Table 1: Demographic and administrative data (including drop-outs).
| Time point | Parameter | PRE groupa | POST groupa | P value |
|---|---|---|---|---|
| Admission | Pain (NRS) | 0.73 ± 1.67 | 0.76 ± 1.86 | 0.905 (U) |
| Preoperative | Pain (NRS) | 0.1 ± 0.45 | 0 ± 0 | 0.823 (U) |
| Intra-Op | Epidural Block (n users) | 2 (8%) | 3 (16%) | 0.640 (F) |
| Fentanyl (mg/patient) | 0.15 ± 0.04 | 0.15 ± 0.05 | 1.0 (U) | |
| In PACU | Pain (NRS) | 1.19 ± 1.16 | 0.92 ± 0.94 | 0.549 (U) |
| MME (mg/user) | 27.1 ± 14 | 20 ± 11.6 | 0.123 (U) | |
| Morphine (mg/user) | 6.69 ± 4.4 | 4.44 ± 2.92 | 0 .209 (U) | |
| Morphine (n users) | 13 (54%) | 9 (47%) | 0.757 (F) | |
| All opioids (n users) | 17 (71%) | 13 (68%) | 0.864 (C) | |
| Non-opioids (n users)c | 9 (38%) | 14 (74%) | 0.031 (F) | |
| All analgesics (n user) | 18 (75%) | 16 (84%) | 0.461 (F) | |
| POD0b | Pain (NRS) | 1.70 ± 2.17 | 1.16 ± 1.14 | 0.763 (U) |
| MME (mg/user) | 14.89 ± 7.07 | 8.25 ± 1.06 | 0.400 (U) | |
| MME (mg/user), PCAs excludedc | 15.08 ± 3.9 | 7.5 ± 0 | 0.286 (U) | |
| All opioids (n users) | 9 (38%) | 2 (11%) | 0.145 (F) | |
| Non-opioids (doses/user) | 1.21 ± 0.43 | 1.38 ± 0.74 | 0.815 (U) | |
| Non-opioids (n users) | 14 (58%) | 8 (42%) | 0.364 (F) | |
| All analgesics (n users) | 17 (71%) | 8 (42%) | 0.071 (F) 0.058 (C) | |
| POD1 | Pain (NRS) | 1.86 ± 2.31 | 1.71 ± 1.58 | 0.716 (U) |
| MME (mg/user) | 38 ± 36.39 | 0 ± 0 | 0.001 | |
| All opioids (n users) | 2 (8.3%) | 0 (0%) | 0.495 (F) | |
| Non-opioids (n users) | 14 (58%) | 8 (42%) | 0.364 (F) | |
| All analgesics (n users) | 14 (58%) | 8 (42%) | 0.364 (F) | |
| POD2 | Pain (NRS) | 1.81 ± 2.84 | 0.57 ± 1.51 | 0.413 (U) |
| All opioids (n users) | 0 | 0 | 0 | |
| Non-opioids (dose/user)c | 1.33 ± 0.52 | 0 ± 0 | 0.021 (U) | |
| Non-opioids (n users)c | 6 (25%) | 0 | 0.027 (F) | |
| All analgesics (n users) | 6 (25%) | 0 | 0.027 (F) | |
| Discharge time | Pain (NRS) | 1.82±3.26 | 1.59±2.44 | 0.827 (U) |
| Requests for home medications (n) | 20 | 13 | 0.295 (F) | |
| Side effects during stay (n events) | 6 (25%) | 2 (11%) | 0 .269 (F) | |
| Overall satisfaction rate (NRS) | 9.4 ± 0.95 | 9.27 ± 1.22 | 0.821 (U) |
aSee text for group definitions.
bAfter discharge from PACU.
NRS, numerically rating scale; PCA, patient-controlled analgesia; POD, postoperative day; PACU, post-anaesthesia care unit; MME, morphine milligram equivalent:
all types of opioids, given by any route (see text for calculations); F, Fisher Exact Test; U, Mann-Whitney U-Test.
cDypirone users (1 gr/dose/patient).
Table 2: In-hospital pain and analgesics consumption.
The NRS pain scores during the patients’ stay in the PACU were similar between the two groups; these continued to differ only slightly between them during the remainder of the study duration, without reaching a level of significance (Table 2). Overall, patients of both groups made use of statistically similar amounts of drugs, both opioids and NSAIDs. Nevertheless, fewer POST patients demanded NSAIDs (mostly dipyrone), during the 48 h of the immediate followup, compared to the PRE patients (Table 2). The incidence of PONV did not differ between the groups (Table 2), nor did the consumption of antiemetics (data not shown); the overall satisfaction rates of the patients were also similar (Table 2). The late (out-of-hospital) pain scores, in both the PRE and the POST patients, were reportedly similar during the first 2 weeks and 1 month after surgery (Table 3). At 3 and 6 months after surgery, NRS was lower by a magnitude of 3-5 times in the POST compared to the PRE patients (Table 3). The same figures depicted the rate of use of analgesics, where none of the POST patients made use of such drugs 3 and 6 months postoperatively (Table 3).
| Parameter/time point | PRE groupa (n=13) | POST groupa (n=15) | P value |
|---|---|---|---|
| Pain (NRS) 2 weeks | 4.35 ± 3.27 | 4.33 ± 3.24 | 0.964 (U) |
| Pain (NRS) 1 month | 2.58 ± 2.71 | 2.73 ± 2.84 | 0.964 (U) |
| Pain (NRS) 3 months | 2.08 ± 3.09 | 0.73 ± 1.79 | 0.217 (U) |
| Pain (NRS) 6 months | 2.27 ± 2.8 | 0.38 ± 1.39 | 0.063 (U) |
| Percent drugb users 1 month | 38% | 40% | 1 (F) |
| Percent drugb users 3 months | 23% | 0% | 0.087 (F) |
| Percent drugb users 6 months | 23% | 0% | 0.087 (F) |
aSee text for group definitions.
bAny type of oral analgesic.
NRS, numerical rating scale; U, Mann Whitney U-Test; F, Fisher Exact Test.
Table 3: Out of Hospital Postoperative Follow-Up.
Discussion
Gabapentinoids are increasingly used as part of multimodal analgesia protocols. Comparisons between pre- and postoperative pregabalin in orthopaedic-oncologic patients, are very rare [18], mainly due to difficulties in setting-up reliable RTCs and the oral use of the drugs. The present study indicates that there were statistically significant differences between the two groups regarding the drug’s effects on the number of opioid users, from admission to discharge, including 3 and 6 months after discharge. In addition, there were lower rates of NSAIDs consumptions and hospital LOS between the PRE (mean [SD] = 2 [1]) and POST (1.5 [0.9]) and the POST group (p=0.037). The latter decreased LOS could have positive economic and social impacts on both the health-care system (smaller budget spent on brief stay), and patient’s rehabilitation and well-being (restoration of familial relations, faster return to work, better quality of life), all requiring further exploration among the orthopaedic oncological cohort in future research. This report limited the periods of pregabalin administrations to 24 h before and a 48 h follow-up period after surgery, in part to exclude post-surgery influences. Fentanyl was used during the operations in both the PRE and POST groups; its mean administered doses resulted identical. One has to deduce that differences or similarities in MME consumptions, and in other variables, could not be attributed to flexibility in fentanyl dosing during surgery, as determined by patients’ needs, extension of surgery, or of anaesthetist’s considerations, but simply to the two different pregabalin protocols. Two phenomena, the multimodal analgesia and the bone-originated pain in oncological patients, are relevant to the discussion of the effects of pregabalin, and have been considered in the past; therefore, they are mentioned herein briefly. The former is a strategy that combines analgesics and non-analgesics with different neuro-pharmacological mechanisms of action, to contend to the complexity of perioperative pain. This engenders multifactorial expressions, partly emanated from continuously modified pain neurotransmitter sites, and their pathways. This approach optimizes pain relief, while a lower incidence of adverse events occurs, as compared to those encountered when using singledrug protocols [19-21], especially opioids. The second factor that was at the basis of this study was the intense pain that accompanies orthopaedic-oncologic lesions. In our group’s earlier studies [6,22,23], where adjuvants (such as ketamine) were omitted, the orthopaediconcologic patients consumed multi-fold amounts of opioids and rescue analgesics, and pain intensity was much higher. Moreover, in the absence of such an adjuvant, side effects were reported by 12-15 cases in groups of 25 to 30 patients, i.e., 2-5 times the rate recorded in the current study or where ketamine was administered. These data support our conclusion that the herein results are the beneficial results of the use of oral pregabalin in orthopaedic-oncologic patients. The possible superiority of pre- or postoperative administration of an analgesic is still unclear. Our group documented higher levels of postoperative pain, and increased morphine consumption, following a pre-incisional single-dose of epidural morphine deposition compared to its post-incisional administration [14]. Indeterminate results were reported when two pre-incisional doses of pregabalin were compared to six post-incisional doses in a cohort similar to the present one [18]. Our current results indicate that pregabalin, at the clinically accepted dose and rates of administration [21,24], affected pain similarly whether is provided before or after surgery, with the exception that the latter spared more appreciably NSAIDs’ usage. These results would require further investigations in order to better elucidate the apparent pharmacological additive effects arising from the co-administration of drugs of the two groups. Importantly, none experienced excessive sedation, nor did the drug cause patients to drop out from the study after its ingestion, nor did it increase side effects, including PONV.
Several studies showed that a single high pregabalin dose (≥300 mg), administered multi-modally or alone, when given before an intervention, would potentiate pain reduction [25,26]. A review of >1100 patients found that a single preoperative gabapentin (1200 mg) resulted in the reduction of both postoperative pain intensity and opioid consumption [27]. Contrarily, a comparison of 75 mg of pregabalin, given either one h before molar extraction under local anaesthesia or shortly after it (34 patients) [28], resulted superior in reducing pain if given post-interventionally. These studies are part of the reason for the present investigation, especially since the authors of the latter report [28] did not explain their rather unusual results. The fact that pre- and postoperative pregabalin can similarly attenuate post-surgical pain, sustains the principle of proof of the present results, where gabapentinoids were administered perioperatively. Being this one of the first studies where pregabalin was tested in orthopaedic oncological patients, adverse events were worth recording. All patients woke up calm, were haemodynamically stable, coherent, and capable of responding to commands. Neither of dizziness, ataxia, blurred vision, and diplopia, had been attributed to pregabalin dose-dependently [21,24,29], was noted in any of our patients, nor was there any drop out of the study due to excessive sedation. Finally, similar to gabapentin [30], pregabalin was reported to decrease PONV [31]. In the current work, the use of antiemetics and the number of cases of overt PONV were negligible in both the PRE and POST protocols. These figures are 2-3 times lower than those reported in our previous studies [6,22,23], where orthopaedic-oncologic patients, who underwent interventions by the same teams, were treated with opioids and/ or NSAIDs alone. Our earlier data [18] support the current results, although more doses of pregabalin were administered postoperatively compared to the only 24-h herein. It has been suggested that even if administered for ≥5 days postoperatively, pregabalin would improve analgesia during the first 24-48 h of administration, i.e., independently of the duration of its intake [12,13,29,31]. This was a methodological reason that was behind the present limited protocol, in addition to the principal goal of the study, i.e., comparing the effects of identical PRE to POST pregabalin doses. The lower rate of drug consumption that was recorded 3 and 6 months after surgery in the POST individuals is unexplainable by our data. Confirmatory data regarding late effects of gabapentinoids in modulating postoperative pain [32], hyperalgesia, and central sensitization [33], have been reported. Since late central sensitization is an important part in the evolvement of acute into chronic pain, and since mechanisms of peripheral nociceptive impulses integrate central neuroplasticity, the effects of gabapentinoids seem effective in these study models [34]. They support both the above theory, as well as our data, in being involved in late postoperative pain; these, however, still require additional large-scale study proofs.
Study Limitations
Several limitations to this study need to be addressed. One is the preoperative 20% dropouts in the POST group, due to surgical and administrative reasons. This may have hampered the reach for significant statistical differences between the tested pain NRS values. In addition, there was no true placebo group, which would receive no pregabalin, either before or after surgery. This was due to some objections raised by the IRB, which argued that based on the proven benefits of gabapentinoids and those of multimodal analgesia protocols, patients who are expected to experience intense postoperative pain, would be withheld from a common analgesic regimen. We therefore refer to similar interventions undertaken by the same surgical and anaesthesia teams for such purposes of comparison. Finally, the pharmacodynamics and kinetics of pregabalin could not be delineated in the study participants, since blood samples were not obtained from them.
Conclusions
Despite the above limitations, this work adds to current knowledge by articulating the importance of LOS in orthopaedic oncological patients treated with pregabalin postoperatively. This apparently first of its kind report also attests that pregabalin provides similar analgesic postoperative potentials, whether administered before or after orthopaedic-oncological medium-size surgery. The immediate and the unexpected 3-6 month (late) postoperative pain reduction and analgesics sparing effects that accompanied the drug’s postoperative administration, as well as the associated shorter hospital LOS, are of great consequences, and warrant each further clinical clarifications.
References
- Vadivelu N, Mitra S, Narayan D (2010) Recent advances in postoperative pain management. Yale J Biol Med 83:11-25.
- Breivik H, Stubhaug A (2008) Management of acute postoperative pain: still a long way to go! Pain 137: 233-234.
- Apfelbaum J, Chen C, Mehta S, Gan T (2003) Postoperative pain experience: results from a national survey suggest postoperative pain continues to be undermanaged. Anesth Analg 97: 534-540.
- Joshi GP (2005) Multimodal analgesia techniques and postoperative rehabilitation. Anesthesiol Clin North Am 23:185-202.
- Buvanendran A, Kroin J (2007) Useful adjuvants for postoperative pain management. Best Pract Res Clin Anaesthesiol 21: 31-49.
- Kollender Y, Bickels J, Stocki D, Maruoani N, Chazan S, et al. (2008) Subanaesthetic ketamine spares postoperative morphine and controls pain better than standard morphine does alone in orthopaedic-oncological patients. Eur J Cancer 44: 954-962.
- Dworkin RH, Corbin AE, Young JP Jr, Sharma U, LaMoreaux L, et al. (2003) Pregabalin for the treatment of postherpetic neuralgia: a randomized, placebo-controlled trial. Neurology 60: 1274-1283
- Lesser H, Sharma U, LaMoreaux L, Poole RM (2004) Pregabalin relieves symptoms of painful diabetic neuropathy: a randomized controlled trial. Neurology 63: 2104-2110.
- Tuchman M, Barrett JA, Donevan S, Hedberg TG, Taylor CP (2010) Central sensitization and Ca(V)alpha(2)delta ligands in chronic pain syndromes: pathologic processes and pharmacologic effect. J Pain 11:1241-1249.
- Dauri M, Faria S, Gatti A, Celidonio L, Carpenedo R (2009) Gabapentin and pregabalin for the acute post-operative pain management. A systematic-narrative review of the recent clinical evidences. Curr Drug Targets 10: 716-733.
- Engelman E, Cateloy F (2011) Efficacy and safety of perioperative pregabalin for post-operative pain: a meta-analysis of randomized-controlled trials. Acta Anaesthesiol Scand 55: 927-943.
- Burke SM, Shorten GD (2010) Perioperative pregabalin improves pain and functional outcomes 3 months after lumbar discectomy. Anesth Analg 110: 1180-1185.
- Buvanendran A, Kroin JS, Della CJ, Kari M, Moric M (2010) Perioperative oral pregabalin reduces chronic pain after total knee arthroplasty: a prospective, randomized, controlled trial. Anesth Analg 110: 199-207.
- Bronstein I, White I, Ekstein MP, Brill S, Chazan S, et al. (2011) Higher postoperative pain and increased morphine consumption follow pre- rather than post-incisional single dose epidural morphine. Minerva Anestesiol 77: 408-417.
- Mercadante S, Caraceni A (2011) Conversion ratios for opioid switching in the treatment of cancer pain: a systematic review. Palliat Med 25: 504-515.
- Ekstein MP, Weinbroum AA (2011) Immediate postoperative pain in orthopedic patients is more intense and requires more analgesia than in post-laparotomy patients. Pain Med 12: 308-313.
- Chazan S, Buda I, Nesher N, Paz J, Weinbroum AA (2010) Low-dose ketamine via intravenous patient-controlled analgesia device after various transthoracic procedures improves analgesia and patient and family satisfaction. Pain Manag Nurs 11: 169-176.
- Amit A, Kollender Y, Dadia S, Dany MA, Bickels J, et al. (2015) Pre- versus Post-Incisional Pregabalin in Orthopaedic-Oncologic Patients: A Comparative, Randomized, Double-Blind, Controlled Study. J Adv Clin Pharmacol 2: 27-38.
- Woolf CJ (2004) American College of Physicians; American Physiological Society. Pain: moving from symptom control toward mechanism-specific pharmacologic management. Ann Intern Med 40: 441-451.
- Weinbroum AA (2003) A single small dose of postoperative ketamine provides rapid and sustained improvement in morphine analgesia in the presence of morphine-resistant pain. Anesth Analg 96: 789-795.
- Weinbroum AA (2012) Non-opioid IV adjuvants in the perioperative period: pharmacological and clinical aspects of ketamine and gabapentinoids. Pharmacol Res 65: 411-429.
- Weinbroum AA, Bender B, Bickels J, Nirkin A, Marouani N, et al. (2003) Preoperative and postoperative dextromethorphan provides sustained reduction in postoperative pain and patient-controlled epidural analgesia requirement: a randomized, placebo-controlled, double-blind study in lower-body bone malignancy-operated patients. Cancer 97: 2334-2340.
- Weinbroum AA, Gorodetzky A, Nirkin A, Kollender Y, Bickels J, et al. (2002) Dextromethorphan for the reduction of immediate and late postoperative pain and morphine consumption in orthopedic oncology patients: a randomized, placebo-controlled, double-blind study. Cancer 95: 1164-1170.
- Eipe N, Penning J, Yazdi F, Mallick R, Turner L, et al. (2015) Perioperative use of pregabalin for acute pain—a systematic review and meta-analysis. Pain 156: 1284-1300.
- Alimian M, Imani F, Hassani V, Rahimzadeh P, Sharifian M (2012) Effects of single-dose pregabalin on postoperative pain in dacryocystorhinostomy surgery. Anesth Pain 2: 72-76.
- Mathiesen O, Jacobsen LS, Holm HE, Randall S, Adamiec-Malmstroem L, et al. (2008) Pregabalin and dexamethasone for postoperative pain control: a randomized controlled study in hip arthroplasty. Br J Anaesth 101: 535-541.
- Ho KY, Gan TJ, Habib AS (2006) Gabapentin and postoperative pain – a systematic review of randomized controlled trials. Pain 126: 91-101.
- Cheung CW, Choi WS, Leung YY, Lui F, Ng JK, et al. (2012) A double-blind randomized crossover study to evaluate the timing of pregabalin for third molar surgery under local anesthesia. J Oral Maxillofac Surg 70: 25-30.
- Fassoulaki A, Melemeni A, Tsaroucha A, Paraskeva A (2012) Perioperative pregabalin for acute and chronic pain after abdominal hysterectomy or myomectomy: a randomised controlled trial. Eur J Anaesthesiol 29: 531-536.
- Srivastava U, Kumar A, Saxena S, Mishra AR, Saraswat N (2010) Effect of preoperative gabapentin on postoperative pain and tramadol consumption after minilap open cholecystectomy: a randomized double-blind, placebo-controlled trial. Eur J Anaesthesiol 27: 331-335.
- Mishriky BM, Waldron NH, Habib AS (2015) Impact of pregabalin on acute and persistent postoperative pain: a systematic review and meta-analysis. Br J Anaesth 114: 10-31.
- Dahl JB, Mathiesen O, Moniche S (2004) Protective premedication: an option with gabapentin and related drugs? A review of gabapentin and pregabalin in the treatment of post-operative pain. Acta Anaesthesiol Scand 48: 1130-1136.
- Fassoulaki A, Melemeni A, Stamatakis E, Petropoulos G, Sarantopoulos C (2007) A combination of gabapentin and local anaesthetics attenuates acute and late pain after abdominal hysterectomy. Eur J Anaesthesiol 24: 521-528.
- Tuchman M, Barrett JA, Donevan S, Hedberg TG, Taylor CP (2010) Central sensitization and Ca(V)α2δ ligands in chronic pain syndromes: pathologic processes and pharmacologic effect. J Pain 11: 1241-1249.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi