Review Article, Endocrinol Diabetes Res Vol: 3 Issue: 2
Statins Cause Lifestyle-Related Diseases-Biochemical Mechanism
Yoko Hashimoto1* and Harumi Okuyama2
1Department of Biochemistry, School of Dentistry, Aichi-Gakuin University, Nagoya, Japan
2Nagoya City University, and Institute for Consumer Science and Human Life, Kinjo Gakuin University, Nagoya, Japan
*Corresponding Author : Yoko Hashimoto, PhD
School of Dentistry, Aichi-Gakuin University, 464-8650 Nagoya, Japan
Tel: +81-52-751-2561
E-mail: yokuteku@dpc.agu.ac.jp
Received: June 09, 2017 Accepted: June 16, 2017 Published: June 22, 2017
Citation: Hashimoto Y, Okuyama H (2017) Statins Cause Lifestyle-Related Diseases-Biochemical Mechanism. Endocrinol Diabetes Res 3:2. doi: 10.4172/2470-7570.1000122
Abstract
While statins are being used worldwide for the prevention of cardiovascular diseases, statin users have an increased probability of developing diabetes, arteriosclerosis, and other lifestyle-related diseases. Here, we review the molecular mechanisms underlying the onset of osteoporosis, chronic kidney disease, type 2 diabetes, and arteriosclerosis, and lowered testosterone levels induced by statin use. Statins inhibit 3-hydroxy-3-methylglutaryl coenzyme A (HMG-CoA) reductase, which is a rate-limiting enzyme of the mevalonate pathway that generates geranylgeranyl diphosphate (GGPP), 7-dehydrocholesterol (7-DHC), and cholesterol. GGPP is essential for vitamin K2 (VK2) synthesis from ingested vitamin K1. 7-DHC is a precursor of vitamin D3 (VD3). VD3 and VK2 upregulate the mRNA expression of osteocalcin (OC) and matrix Gla protein (MGP), respectively, via activation of their respective nuclear receptors. VK is essential for γ-carboxylation-mediated activation of several proteins. γ-Carboxylated MGP inhibits arterial and kidney calcifications. Insulin synthesis is stimulated by OC and γ-carboxylated protein S. GGPP is essential for secretion of insulin and luteinizing hormone (LH), and insulin action. VK2, OC, and LH stimulate testosterone synthesis via activation of protein kinase A. Both VK2 and VD3 are essential for these processes, and their deficiency leads to the onset of diabetes, vascular calcifications/ arteriosclerosis, and disruption of testosterone synthesis. VD and VK2 supplementations ameliorate the symptoms of chronic kidney disease and osteoporosis; therefore, statin is contraindicated in those patients. Thus, caution must be exercised when prescribing statins for long-term use. Collectively, deficiencies of VD3 and VK2 induced by long-term statin use promote the onset of lifestylerelated diseases.
Keywords: Statin; Lifestyle-related disease; Type 2 diabetes mellitus; Vitamin D; Vitamin K; Insulin synthesis/secretion; Osteocalcin; Matrix Gla protein; Testosterone synthesis
List of Abbreviations
HMG-CoA: 3-Hydroxy-3-Methylglutaryl Coenzyme A; GGPP: Geranylgeranyl Diphosphate; 7-DHC: 7-Dehydrocholesterol; VK1: Vitamin K1; VK2: Vitamin K2; VD3: Vitamin D3; OC: Osteocalcin; MGP: Matrix Gla Protein; LH: Luteinizing Hormone; Isopentenyl- PP: Isopentenyl Diphosphate; Farnesyl-PP: Farnesyl Diphosphate; cOC: γ-Carboxylated OC; SXR: Steroid and Xenobiotic Receptor; PXR: Pregnane X receptor; VDR: Vitamin D Receptor; GGCX: γ-Glutamyl Carboxylase; cMGP: γ-Carboxylated MGP; PTH: Parathyroid Hormone; cProtein S: γ-Carboxylated Protein S; RabdiGG: Digeranylgeranylated Rab; MMPs: Matrix Metalloproteinases; ENPP: Ectonucleotide Pyrophosphatase/Phosphodiesterase; Ank: Ankylosis
Introduction
Statins are widely used to control blood cholesterol levels. Besides their cholesterol-lowering effect, statins are reported to exhibit pleiotropic effects, including the prevention of cardiovascular disease via decreasing aortic valve calcium accumulation [1-3]. However, several reports have suggested that statin use increases the risk of diabetes mellitus [4-6]; the pharmacological mechanisms underlying statin-induced diabetes have been partly clarified [7]. Here, we report the molecular mechanisms through which long-term statin therapy causes arteriosclerosis and diabetes, and disturbs testosterone synthesis. Statins inhibit 3-hydroxy-3-methylglutaryl coenzyme A (HMG-CoA) reductase, which catalyzes the rate-limiting step in the mevalonate pathway. This pathway provides various important substances such as 7-dehydrocholesterol (7-DHC) and geranylgeranyl diphosphate (GGPP); 7-DHC is a common precursor of vitamin D3 (VD3) and cholesterol, whereas GGPP is necessary for the production of vitamin K2 (VK2) from vitamin K1 (VK1) as the side-chain source [8,9], secretion of insulin and luteinizing hormone (LH) [10,11], and insulin action [12-14].
There are two forms of VD—VD2 and VD3—of plant and animal origin, respectively. VD plays a major role in bone mineral homeostasis by promoting the transport of calcium and phosphate to ensure that the blood levels of these ions are sufficient for normal mineralization of type I collagen matrix in the skeleton. In addition, VD serves to maintain calcium homeostasis in the small intestine, bones, and kidneys. VK is distributed abundantly in the brain, kidneys, pancreas, gonadal tissues, liver, and bones. VK consists of three forms, with different side chains bound to the naphthoquinone ring. VK1 and VK2 (menaquinone-4, MK-4) differ only in the side chains; VK1 possesses a phytyl group whereas VK2 possesses a repeating, unsaturated trans-polyisoprenyl group. MKs include a range of related forms generally designated as MK-n, where n is the number of isoprenyl units. VKs play a role in the bone-building process as well as in the classic blood coagulation pathway. Since the 1980s, statins are widely used for lowering cholesterol, and since then, VD, VK, and osteocalcin (OC) actions and membrane trafficking have been studied extensively, which revealed the following mechanisms:
VK2 is synthesized from dietary VK1, using GGPP as the source of the side chain [8,9,15].
GGPP is necessary for the secretion of peptide hormones (insulin, LH, etc.) and for insulin action [10-14].
VD3 and VK2 activate their respective nuclear receptors to modify the expression of various genes (especially, those encoding OC and matrix Gla protein [MGP]) related to bone homeostasis [16-19].
OC is crucial for not only glucose and bone metabolism, but also steroidogenesis [20,21].
In this review, we summarize the molecular mechanisms by which long-term statin use increases the risk of multiple types of lifestylerelated diseases, including diabetes mellitus and cardiovascular disease, through the inhibition of VD3 and VK2 biosynthesis.
Intermediates of cholesterol biosynthesis are necessary for the production of both VD3 and VK2
Cholesterol is synthesized from acetyl-CoA via the mevalonate pathway in various tissues, including the brain. Besides cholesterol, the pathway generates many important lipid metabolites such as isopentenyl, geranyl, and farnesyl diphosphates, GGPP, lanosterol, and 7-DHC (Figure 1). Statins suppress cholesterol biosynthesis dosedependently via the inhibition of HMG-CoA reductase and reduce the levels of various intermediates of cholesterol biosynthesis, among which GGPP and 7-DHC are essential for the synthesis of VK2 and VD3, respectively. Lanosterol is necessary for the prevention of lens protein aggregation and cataract development [22]. In general, more cholesterol is biosynthesized in the body than is absorbed from food sources. Therefore, the inhibition of HMG-CoA reductase by longterm administration of statins could cause serious adverse effects by reducing the supply of VK2 and VD3 to tissues. In addition, bile acids, which are made from cholesterol, are essential for the intestinal absorption of both VD and VK.
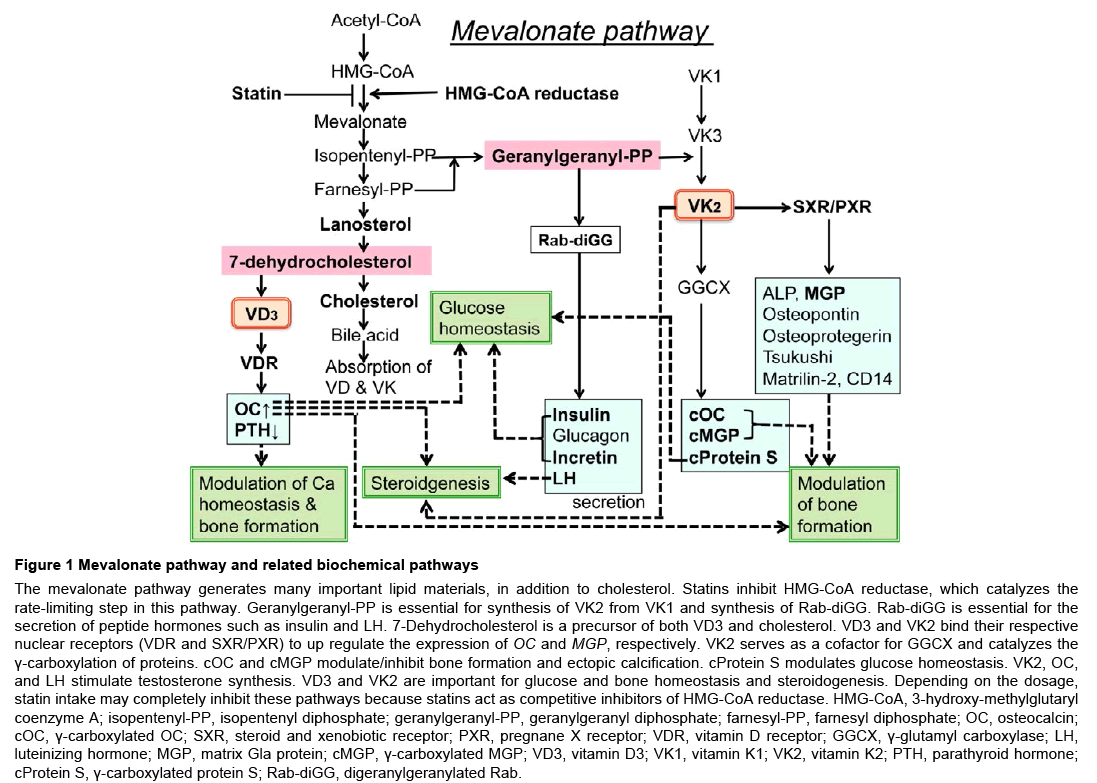
Figure 1: Mevalonate pathway and related biochemical pathways
The mevalonate pathway generates many important lipid materials, in addition to cholesterol. Statins inhibit HMG-CoA reductase, which catalyzes the rate-limiting step in this pathway. Geranylgeranyl-PP is essential for synthesis of VK2 from VK1 and synthesis of Rab-diGG. Rab-diGG is essential for the secretion of peptide hormones such as insulin and LH. 7-Dehydrocholesterol is a precursor of both VD3 and cholesterol. VD3 and VK2 bind their respective nuclear receptors (VDR and SXR/PXR) to up regulate the expression of OC and MGP, respectively. VK2 serves as a cofactor for GGCX and catalyzes the γ-carboxylation of proteins. cOC and cMGP modulate/inhibit bone formation and ectopic calcification. cProtein S modulates glucose homeostasis. VK2, OC, and LH stimulate testosterone synthesis. VD3 and VK2 are important for glucose and bone homeostasis and steroidogenesis. Depending on the dosage, statin intake may completely inhibit these pathways because statins act as competitive inhibitors of HMG-CoA reductase. HMG-CoA, 3-hydroxy-methylglutaryl coenzyme A; isopentenyl-PP, isopentenyl diphosphate; geranylgeranyl-PP, geranylgeranyl diphosphate; farnesyl-PP, farnesyl diphosphate; OC, osteocalcin; cOC, γ-carboxylated OC; SXR, steroid and xenobiotic receptor; PXR, pregnane X receptor; VDR, vitamin D receptor; GGCX, γ-glutamyl carboxylase; LH, luteinizing hormone; MGP, matrix Gla protein; cMGP, γ-carboxylated MGP; VD3, vitamin D3; VK1, vitamin K1; VK2, vitamin K2; PTH, parathyroid hormone; cProtein S, γ-carboxylated protein S; Rab-diGG, digeranylgeranylated Rab.
Vitamins D and K
VD is the only vitamin synthesized in the body. In general, 90–95% of our VD requirement is synthesized from casual exposure to sunlight [23]. 7-DHC, a precursor of VD3 (Figure 1), is activated in the skin by ultraviolet irradiation and is metabolized constitutively to 25-hydroxyvitamin D3 in the liver and then to 1α,25-dihydroxyvitamin D3 (VD3) in the kidneys [16]; the latter hydroxylation is regulated by feedback mechanisms that sense the blood levels of VD3, calcium, parathyroid hormone (PTH), and phosphate. VD3 induces termination of its own signal in target tissues [16]. Subsequently, it is secreted, and thereafter, VD3 exerts its biological functions by regulating gene transcription via binding to its nuclear receptor, vitamin D receptor (VDR), in target cells [16,17].
Dietary VK1 is endogenously converted via VK3 into VK2, which contains a side chain of four isoprene units from GGPP [8,9]; the phytyl side chain of VK1 is cleaved to form VK3, which is delivered through the mesenteric lymphatic system and blood circulation to tissues, where it is converted to VK2 by a prenyltransferase, such as UbiA prenyltransferase-containing protein 1 (UB1AD1), using GGPP as a substrate [8,9,24]. VK1 is the major form (> 90%) of dietary VK; however, the major form of VK in mammalian tissues is VK2 [8]. Both VD3 and VK2 function as ligands for their respective nuclear receptors, VDR and steroid and xenobiotic receptor (SXR)/pregnane X receptor (PXR), and upregulate the mRNA levels of several genes that participate in bone and glucose homeostasis and steroidogenesis. Ligand-bound VDR or SXR/PXR heterodimerizes with retinoid X receptor and then interacts with the VD- or VK-response element, respectively, at the promoter regions of target genes [16-19]. When VD3 binds to VDR, the mRNA expression of OC, osteopontin, and the receptor activator of NF-κB ligand (RANKL) is up regulated, while PTH mRNA expression is downregulated [16,25,26]. VK2 exerts various actions mediated through nuclear receptor-dependent transcriptional regulation and protein kinase-dependent pathways [18,19,27]. VK2 treatment of osteosarcoma cells increases the mRNA levels of osteoblast markers such as bone alkaline phosphatase, osteoprotegerin, osteopontin, and MGP through activating SXR/PXR [18]. Moreover, SXR/PXR mediates VK2-activated transcription of extracellular matrix-related genes (tsukushi [Tsku], matrilin-2 [MATN2], and CD14), resulting in the accumulation of collagen in osteoblasts [19].
Statins promote the onset of type 2 diabetes mellitus by disrupting energy metabolism
The mechanisms of type 2 diabetes induction via GGPP, VD3, and VK2 deficiencies by statin treatment are illustrated in Figure 2. The binding of two geranylgeranyl (GG) groups — supplied from GGPP and required to associate with the target membrane [28] — to Rab protein is essential for the secretion of peptide hormones such as insulin and LH [10,11,29]. Rab GG transferase or GG transferase- II catalyzes the transfer of the GG group, a 20-carbon-derivative of mevalonate, to two C-terminal cysteines of Rab GTPases to facilitate their membrane targeting for optimal interaction with the effector proteins and/or fusion of secretory vesicles with the plasma membrane [28,30]. Statins inhibit insulin secretion by generating GGPP deficiency (Figure 2).
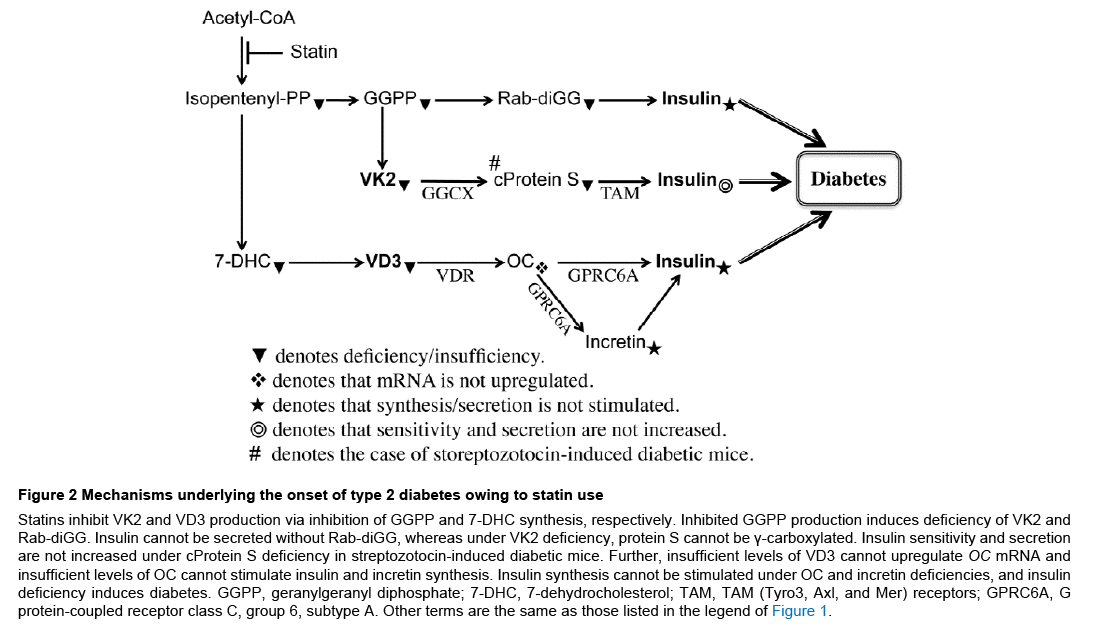
Figure 2: Mechanisms underlying the onset of type 2 diabetes owing to statin use
Statins inhibit VK2 and VD3 production via inhibition of GGPP and 7-DHC synthesis, respectively. Inhibited GGPP production induces deficiency of VK2 and Rab-diGG. Insulin cannot be secreted without Rab-diGG, whereas under VK2 deficiency, protein S cannot be γ-carboxylated. Insulin sensitivity and secretion are not increased under cProtein S deficiency in streptozotocin-induced diabetic mice. Further, insufficient levels of VD3 cannot upregulate OC mRNA and insufficient levels of OC cannot stimulate insulin and incretin synthesis. Insulin synthesis cannot be stimulated under OC and incretin deficiencies, and insulin deficiency induces diabetes. GGPP, geranylgeranyl diphosphate; 7-DHC, 7-dehydrocholesterol; TAM, TAM (Tyro3, Axl, and Mer) receptors; GPRC6A, G protein-coupled receptor class C, group 6, subtype A. Other terms are the same as those listed in the legend of Figure 1.
Moreover, GG is necessary for the binding of Rho and Rab proteins to enable the translocation of glucose transporter 4 (GLUT4) to the cell surface in response to insulin, thereby promoting sugar uptake [12-14]. Insulin cannot act in the absence of GGPP. Racdependent actin reorganization plays a crucial role in insulininduced recruitment of GLUT4 to the surface of muscle cells. Moreover, statins inhibit the biosynthesis of selenoproteins, which are known to be required for the trafficking of GLUT4 to the plasma membrane [7]. Dolicol, a polyisoprenoid synthesized from isoprenyl intermediates and inhibited by statins, carries the sugar moiety to the insulin receptor precursor; N-glycosylation is essential for the mature receptor to move to the plasma membrane [7]. Thus, statins inhibit both insulin secretion and action, leading to decreased insulin sensitivity, and consequently, the onset of diabetes. In addition, geranylgeranylated Rab protein is required for the secretion of proteinases [matrix metalloproteinases (MMPs) and cathepsins] and digestive enzymes (amylase) [31-37]. GGPP deficiency shows inhibitory effects on the lysosomal/endosomal secretion pathway. These secretions are also inhibited by statin use. VD3 upregulates OC mRNA levels by activating VDR (Figure 2). The osteoblast-derived hormone OC stimulates the synthesis/secretion of insulin through binding to its putative receptor G protein-coupled receptor class C, group 6, subtype A (GPRC6A) on the pancreatic β-cell surface, thereby increasing pancreatic β-cell proliferation and creating insulin sensitivity [38]. With statin use, OC mRNA is not up regulated because of VD3 deficiency, and therefore, insulin synthesis is not stimulated (Figure 2). In addition, OC stimulates incretin (glucagonlike peptide-1) synthesis/secretion via binding to GPRC6A in small intestinal cells, and subsequently, incretin stimulates the synthesis/ secretion of insulin [39,40]. Thus, OC increases insulin synthesis/ secretion both directly and indirectly. Furthermore, OC is involved in glucose metabolism by increasing adiponectin secretion from adipocytes [38]. OC is crucial for glucose homeostasis as well as bone homeostasis [41,42]. Statins induce insufficient OC production by generating VD3 deficiency, owing to deficient levels of 7-DHC, eventually leading to diabetes. One of the characteristic functions of VK is to serve as a cofactor for γ-glutamyl carboxylase (GGCX), catalyzing the post-translational modification of specific glutamate side chains to γ-carboxyglutamate (Gla) in several proteins. VK stores are limited in humans, but they can be recycled. VK hydroquinone functions as a cofactor for GGCX, which γ-carboxylates OC and MGP. VK-dependent protein S is a 75-kDa multimodular anticoagulant factor found in vertebrates that regulates inflammation and cell apoptosis. Recently, it was reported that exogenous protein S improves blood glucose level, glucose tolerance, insulin sensitivity, and insulin secretion significantly in protein S-overexpressing transgenic as compared to wild-type streptozotocin-induced diabetic mice [43]. Protein S attenuates diabetes by inhibiting the apoptosis of β-cells and the development of diabetic nephropathy. It regulates the inflammatory response and apoptosis pathways through TAM (Tyro3, Axl, and Mer) receptor tyrosine kinases [44]. γ-Carboxylation enables protein S to bind to TAM receptors [45], which belong to a family of VK-dependent receptor tyrosine kinases that have important effects on hemostasis, inflammation, and tumor cell regulation [46,47]. VK modulates glucose metabolism via γ-carboxylation of protein S, thus suppressing diabetes progression (Figure 2). Statin treatments induce VK2 deficiency and might cause the accumulation of VK3, which does not function as a cofactor for GGCX. Indeed, dietary VK intake reduces the risk of type 2 diabetes [48]. In the case of protein S, the VK-dependent carboxylated form is the active form that stimulates insulin synthesis/secretion in streptozotocin-induced diabetic mice. However, VK-independent, non-carboxylated OC retains hormonal activity to stimulate insulin synthesis/secretion. Insulin signaling in osteoblasts increases OC activity by lowering osteoprotegerin mRNA expression [49]. RANKL and osteoprotegerin are known as an osteoclastogenesis factor and an osteoclastogenesis inhibitory factor, respectively. An increased ratio of RANKL to osteoprotegerin stimulates bone resorption by increasing the number of osteoclasts. The acidic pH (4.5) in the resorption lacunae is sufficient to decarboxylate γ-carboxylated OC (cOC) to under-carboxylated OC (ucOC) [49]. Non-carboxylated OC and ucOC, but not cOC, promote glucose metabolism. VD3 upregulates OC and RANKL mRNA levels, and VK2 upregulates osteoprotegerin mRNA by activating their receptors [18,26]. Therefore, osteoclasts, VD3, and VK2 determine the carboxylation status and function of OC. Abnormal energy metabolism is a key feature in metabolic syndrome.
Insulin is the only hormone that can lower blood glucose levels. Insufficient insulin secretion is directly connected to failure to maintain blood glucose homeostasis, leading to the onset of type 2 diabetes. Postmenopausal women who use statins are 1.5 times more likely to develop type 2 diabetes than non-users [5]. In a previous study, participants on statin treatment (n = 2,142) were found to have a 46% increased risk of developing type 2 diabetes [6]. Statin treatment decreased insulin sensitivity by 24% and insulin secretion by 12% compared with non-statin users (p < 0.01). Simvastatin and atorvastatin treatments decreased both insulin sensitivity and insulin secretion in a dose-dependent manner [6]. Collectively, these findings suggest that statins inhibit the synthesis and secretion of insulin by inhibiting the production of VK2, VD3, and GGPP, ultimately leading to diabetes (Figure 2).
Statins cause atherosclerosis and cardiovascular diseases because of insufficient levels of VD3 and VK2
In statin users, the mRNA levels of OC and MGP are low because of VD3 and VK2 deficiencies. Expression of MGP is detected in the calcified atherosclerotic plaques as well as in normal blood vessels. MGP-deficient mice show excessive cartilage formation and growth plate mineralization [50], and develop extensive arterial calcification [51]. Serum MGP levels are inversely correlated with the severity of coronary artery calcification. Data from rodent studies have revealed that cMGP, but not non-carboxylated MGP, plays a key role in the inhibition of tissue calcification [50]. Thus, cMGP is a potent inhibitor of arterial calcification. Indeed, insufficient VK or inhibition of the VK recycling system, e.g., by warfarin, causes abnormal ectopic (vascular) calcification and excessive bone formation [52]. VK modulates the calcification of the soft and hard tissues through γ-carboxylation of MGP and OC. Thus, VK plays a significant role in bone homeostasis.
Geleijnse et al. [53] reported that dietary intake of VK2, but not VK1, reduced the risk of coronary heart disease in the populationbased Rotterdam Study. In their study, severe coronary artery calcification was inversely associated with VK2 intake, and this relationship persisted after adjustment for BMI, smoking, education, diabetes, and dietary factors (Figure 3). The results suggest that increased intake of VK2 suppresses coronary artery calcifications. In contrast, statins have been implicated in the calcification of vascular smooth muscle cells and mesenchymal cells [54,55]. Saremi et al. [56] examined the relationship between the frequency of statin use and the volume of coronary artery calcification in diabetics, and reported that frequent statin use (in > 50% of study visits, n = 161) accelerated the progression of vascular calcification in 4.6 years of follow-up in type 2 diabetes patients with advanced atherosclerosis as compared to less frequent statin use (in ≤ 50% of visits, n = 36; p < 0.01). The progression of artery calcification volume was 2-fold higher in highfrequency than in low-frequency statin users. Figure 4 explains this calcification mechanism. MGP mRNA expression is expected to be low because insufficient VK2 cannot activate SXR/PXR. Moreover, carboxylation of MGP is not processed adequately owing to VK2 deficiency. The deficient level of cMGP cannot inhibit ectopic bone formation.
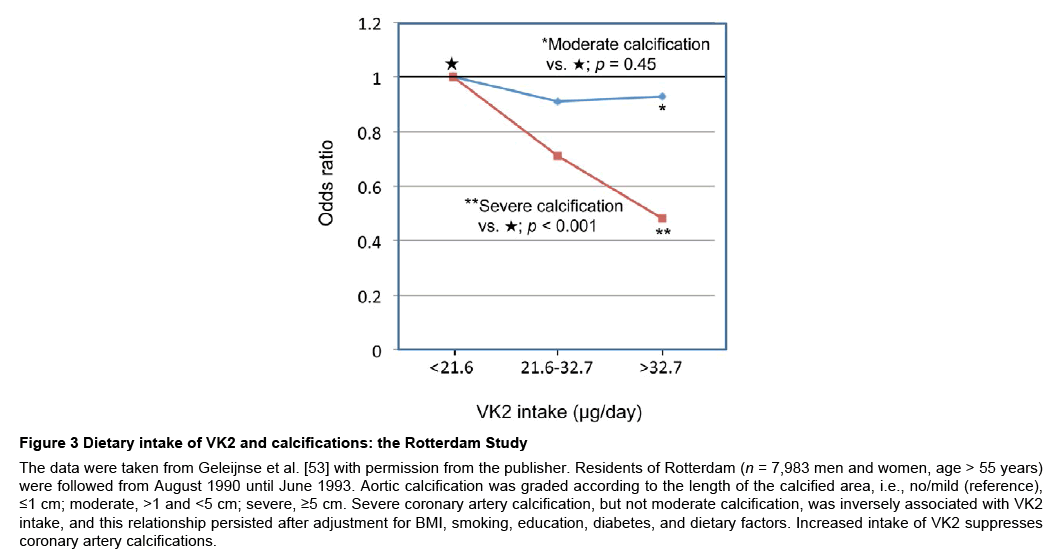
Figure 3: Dietary intake of VK2 and calcifications: the Rotterdam Study
The data were taken from Geleijnse et al. [53] with permission from the publisher. Residents of Rotterdam (n = 7,983 men and women, age > 55 years) were followed from August 1990 until June 1993. Aortic calcification was graded according to the length of the calcified area, i.e., no/mild (reference), ≤1 cm; moderate, >1 and <5 cm; severe, ≥5 cm. Severe coronary artery calcification, but not moderate calcification, was inversely associated with VK2 intake, and this relationship persisted after adjustment for BMI, smoking, education, diabetes, and dietary factors. Increased intake of VK2 suppresses coronary artery calcifications.
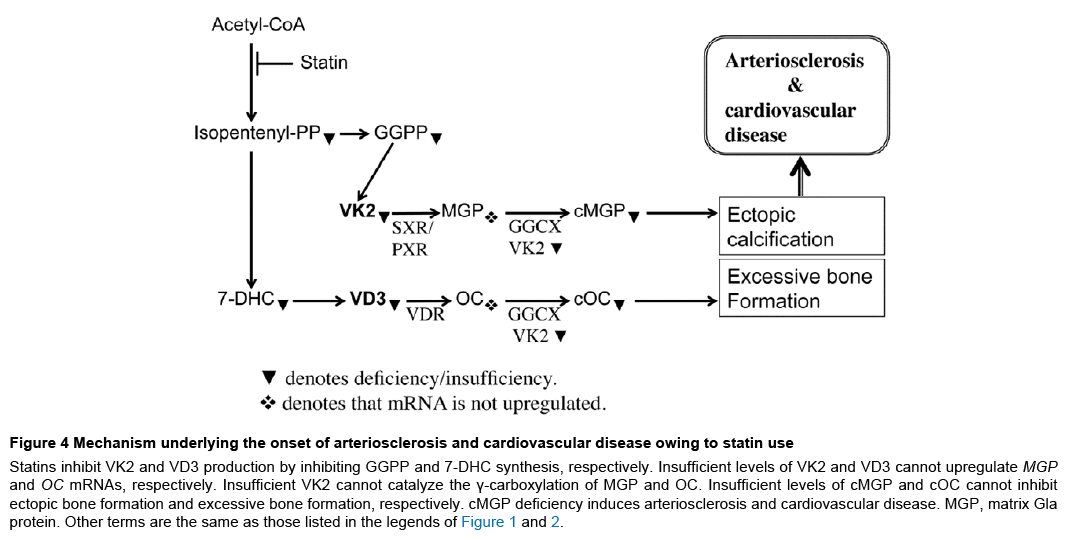
Figure 4: Mechanism underlying the onset of arteriosclerosis and cardiovascular disease owing to statin use
Statins inhibit VK2 and VD3 production by inhibiting GGPP and 7-DHC synthesis, respectively. Insufficient levels of VK2 and VD3 cannot upregulate MGP and OC mRNAs, respectively. Insufficient VK2 cannot catalyze the γ-carboxylation of MGP and OC. Insufficient levels of cMGP and cOC cannot inhibit ectopic bone formation and excessive bone formation, respectively. cMGP deficiency induces arteriosclerosis and cardiovascular disease. MGP, matrix Gla protein. Other terms are the same as those listed in the legends of Figure 1 and 2.
Moreover, VD3 suppresses bone mineralization in osteoblastic cells by up regulating progressive ankylosis (Ank) and ectonucleotide pyrophosphatase/phosphodiesterase (ENPP) mRNA levels, which inhibit the expansion of hydroxyapatite crystals by increasing extracellular pyrophosphoric acid levels through activating VDR [57]. Low VD levels are strongly associated with severe vascular calcifications [58]. Hence, VD plays a significant role in bone homeostasis as well. Therefore, long-term statin use causes excessive ectopic bone formation and leads to vascular calcifications and arteriosclerosis. Thus, although statins are used for the treatment of arteriosclerosis [1,2], they themselves are associated with a high risk of arteriosclerosis (Figure 4).
Statins disrupt endocrine function by inhibiting testosterone production
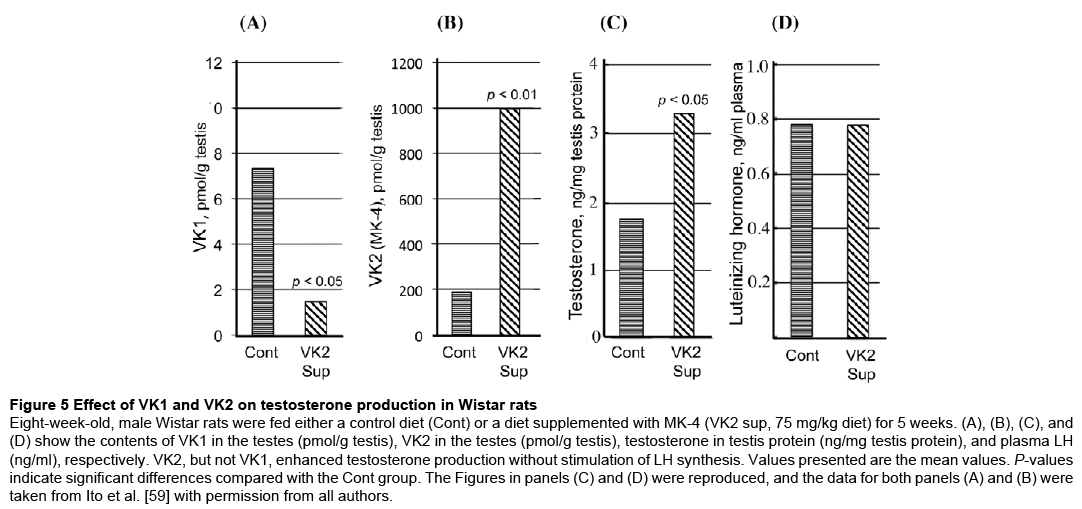
Ito et al. [59] showed that VK2, but not VK1, stimulates testosterone, but not LH, synthesis in 8-week-old, male Wistar rats (Figure 5). VK2 enhances testosterone production by upregulating CYP11A mRNA expression through the activation of protein kinase A (PKA), and this activation is crucial for testosterone production in Leydig cells. CYP11A catalyzes the production of pregnenolone from transported cholesterol, a rate-limiting step of testosterone production.
Figure 5: Effect of VK1 and VK2 on testosterone production in Wistar rats
Eight-week-old, male Wistar rats were fed either a control diet (Cont) or a diet supplemented with MK-4 (VK2 sup, 75 mg/kg diet) for 5 weeks. (A), (B), (C), and (D) show the contents of VK1 in the testes (pmol/g testis), VK2 in the testes (pmol/g testis), testosterone in testis protein (ng/mg testis protein), and plasma LH (ng/ml), respectively. VK2, but not VK1, enhanced testosterone production without stimulation of LH synthesis. Values presented are the mean values. P-values indicate significant differences compared with the Cont group. The Figures in panels (C) and (D) were reproduced, and the data for both panels (A) and (B) were taken from Ito et al. [59] with permission from all authors.
Long-term statin treatment reduces testosterone synthesis by inhibiting three different routes:
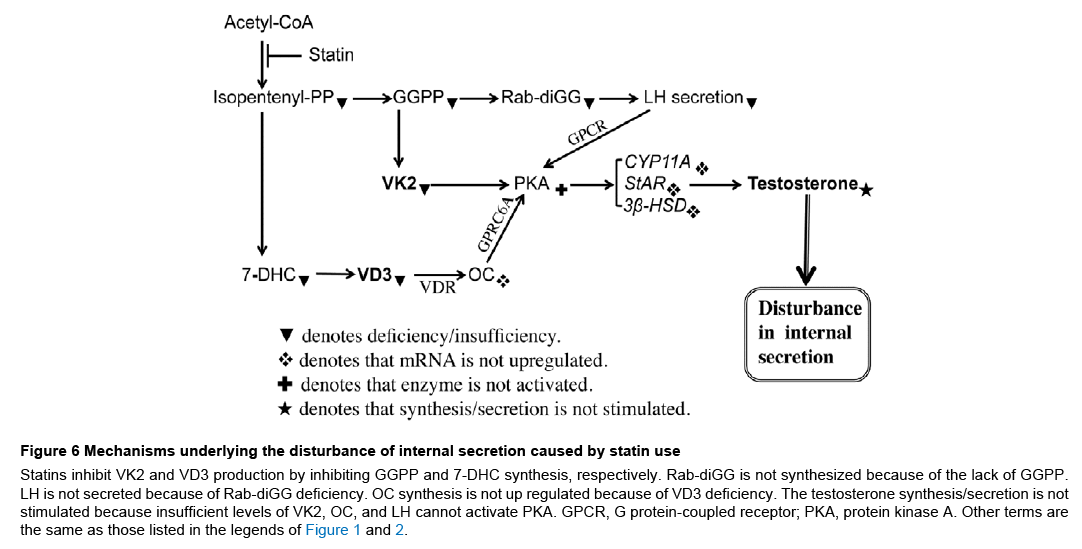
The VK2 route: VK2 stimulates testosterone production in rats via increasing the mRNA levels of CYP11A through activating PKA, not via LH synthesis [59,60]. VK2 deficiency due to statin-induced GGPP deficiency leads to lower testosterone production (Figure 6). VK2 does not influence plasma LH levels (Figure 5) [59].
Figure 6: Mechanisms underlying the disturbance of internal secretion caused by statin use
Statins inhibit VK2 and VD3 production by inhibiting GGPP and 7-DHC synthesis, respectively. Rab-diGG is not synthesized because of the lack of GGPP. LH is not secreted because of Rab-diGG deficiency. OC synthesis is not up regulated because of VD3 deficiency. The testosterone synthesis/secretion is not stimulated because insufficient levels of VK2, OC, and LH cannot activate PKA. GPCR, G protein-coupled receptor; PKA, protein kinase A. Other terms are the same as those listed in the legends of Figure 1 and 2.
The LH route: production of testosterone in Leydig cells is stimulated by LH, which is secreted from the pituitary gland [11,29]. When LH binds to its receptor, a G protein-coupled receptor (GPCR) located on the membrane surface of Leydig cells, PKA, and other steroidogenic proteins are activated [59]. Stimulation of testosterone production by LH is inhibited by H89, a specific inhibitor of PKA, but not by warfarin. Statins reduce LH secretion because of GGPP deficiency. Therefore, testosterone synthesis is not stimulated. Unlike insulin, testosterone does not require GG for its secretion, because it is hydrophobic; it is secreted from Leydig cells via diffusion [59].
The OC route: VD3, derived from 7-DHC, stimulates OC synthesis through binding to VDR [17]. OC has been shown to promote testosterone biosynthesis in mouse testes by binding to GPRC6A expressed in Leydig cells [20,21]. OC stimulates the expression of CYP11A, StAR, and 3β-HSD, which are required for testosterone synthesis, in a cAMP-response element-binding protein-dependent manner [20]. Cholesterol is the starting material for testosterone synthesis. StAR transports cholesterol into the inner membrane of the mitochondria, where CYP11A catalyzes the production of pregnenolone from the transported cholesterol. Statins induce insufficient OC production by creating VD3 deficiency, owing to deficient levels of 7-DHC, leading to reduced testosterone synthesis. OC signaling in Leydig cells does not require LH [21].
Moreover, insulin signaling in osteoblasts increases OC activity by activating bone resorption, and OC promotes testosterone synthesis. However, statins inhibit insulin synthesis and secretion, as mentioned above (Figure 2). Both VD3 and VK2 modulate steroidogenesis through the up regulation of CYP11A mRNA via PKA activation. Statin treatment inhibits all these testosterone production routes (Figure 6).
Long-term statin use promotes the onset of and worsens chronic kidney disease (CKD)
Epidemiological evidence suggests a possible link between bone disease and atherosclerosis in CKD. Therefore, renal osteodystrophy has been renamed CKD mineral bone disorder (CKD-MBD). VK deficiency is involved in the progression of CKD-MBD. CKD induces not only abnormal bone mineral metabolism but also vascular calcification, and in particular, arterial medial calcification. Fusaro et al. [61] reported that the major complications in CKD are vascular calcification and fractures, and the authors strongly discouraged the use of warfarin in CKD patients. We agree with these authors and believe that statins are as harmful to these patients as warfarin. The VK recycling system maintains sufficient VK levels for the activation of VK-dependent proteins through GGCX. However, warfarin inhibits the VK recycling system by inhibiting two of its important enzymes, quinone reductase and epoxide reductase, resulting in functional VK deficiency [61,62]. Moreover, low VD levels are strongly associated with severe vascular calcifications in hemodialysis patients with CKD [58]. In CKD patients, statin and warfarin use causes ectopic calcification and worsening of CKD symptoms, finally leading to arteriosclerosis and cardiovascular disease. Therefore, statin and warfarin are contraindicated in these patients. In CKD patients, kidney function is weak, and PTH is secreted excessively [16]. Therefore, it is unlikely that both activation of VD3 by hydroxylation and secretion of active VD3 from the kidneys occur simultaneously in CKD patients. Nuclear binding of VD3 down regulates the expression of PTH [16]. However, insufficient VD3 cannot activate VDR nor down regulate PTH mRNA expression. Statin use worsens the symptoms of CKD by causing excessive PTH synthesis. Insufficient VD3 and VK2 biosynthesis cannot up regulate OC and MGP mRNAs, respectively. Insufficient VK cannot serve as a cofactor for GGCX, which in turn cannot catalyze the γ-carboxylation of OC and MGP. This causes excessive aortic and kidney calcifications (Figure 4). Dialysis patients require VD and VK2 supplementation because of arterial calcification owing to impaired kidney function. Therefore, statin use is also contraindicated in dialysis patients.
Statin use is contraindicated in the treatment of osteoporosis
In 1999, Mundy et al. [63] reported that statins stimulate bone formation and increase bone volume in vitro and in vivo in rodents by up regulating the expression of bone morphogenetic protein-2 (BMP-2) in bone cells. Therefore, they suggested that statins have therapeutic applications for osteoporosis. Subsequently, Edward et al. [64] reported that statins increase bone formation by increasing bone mineral density in postmenopausal women. Moreover, Chan et al. [65] reported that statins protect against non-pathological fractures and decrease the risk of osteoporotic fractures in elder women, possibly owing to their bone mineral density-increasing effect. However, Wada et al. [66] reported that bone mineral density was significantly lower in statin users that in non-users in Japanese patients with type 2 diabetes. They suggested that statin might not increase bone mineral density, at least in Japanese subjects with type 2 diabetes. To date, the relationship between statin use and bone mineral density is still unclear. In 2014, we reported that mice with (functional) VK deficiency caused by dietary canola and hydrogenated soybean oils, formed ectopic bone with volumes four times larger than those in soybean oil-fed animals implanted with BMPs into a gap in the fascia of the right femoral muscle [52]. Thus, in the case of (functional) VK deficiency, BMPs induce excessive ectopic bone formation. Therefore, in the experiment of Mundy et al., BMP-2 might have induced excessive bone formation because statin treatment inhibits both the production and the γ-carboxylation of MGP and OC (Figure 4).
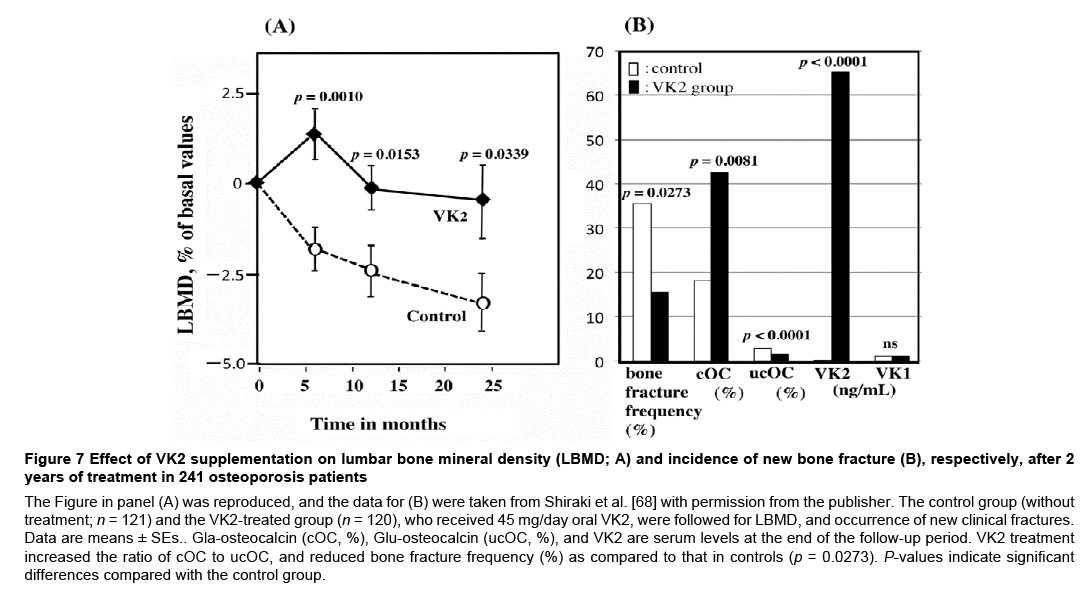
Clinical studies have demonstrated that supplementation of VK2, but not VK1, is effective for preventing osteoporosis fracture [67,68]. VK2 supplementation up regulates the expression of bone markers, increases bone density in vivo [18], and sustains lumbar bone mineral density [68] (Figure 7). Moreover, VK2 improves bone quality by up regulating Tsku, MATN2, and CD14 mRNA levels via activation of SXR/PXR [19]. In fact, both VD and VK2 are used for the treatment of osteoporosis to reduce the incidence of bone fracture in Japan. However, statins induce VD3 and VK2 deficiencies by inhibiting the production of 7-DHC and GGPP, respectively, as mentioned above. Therefore, statins are contraindicated for the treatment of osteoporosis. We emphasize that physicians should be cautious when prescribing statins to treat women with osteoporosis.
Figure 7: Effect of VK2 supplementation on lumbar bone mineral density (LBMD; A) and incidence of new bone fracture (B), respectively, after 2 years of treatment in 241 osteoporosis patients
The Figure in panel (A) was reproduced, and the data for (B) were taken from Shiraki et al. [68] with permission from the publisher. The control group (without treatment; n = 121) and the VK2-treated group (n = 120), who received 45 mg/day oral VK2, were followed for LBMD, and occurrence of new clinical fractures. Data are means ± SEs.. Gla-osteocalcin (cOC, %), Glu-osteocalcin (ucOC, %), and VK2 are serum levels at the end of the follow-up period. VK2 treatment increased the ratio of cOC to ucOC, and reduced bone fracture frequency (%) as compared to that in controls (p = 0.0273). P-values indicate significant differences compared with the control group.
Statins increase the risk of cancers by causing VD3 and VK2 deficiencies
VD3 suppresses the proliferation of mouse myeloid leukemia cells and induces their differentiation into macrophages [69,70]. In 1993, Corder et al. [71] reported that, in their study using stored serum, the mean serum VD3 level in 181 men was slightly but significantly lower in cancer patients than in healthy age- and sexmatched volunteers (p = 0.002). Higher levels of VD3 reduce the risk of prostate cancer. In men over the age of 57, a reduced VD3 level was found to be an important risk factor for palpable and anaplastic tumors. The anti-carcinogenic effects of VD are exerted by differentiation, induction of apoptosis, and cell cycle arrest of cancer cells [69,70,72-74]. VD3 reduces the risk of various cancers such as breast, colon, prostate, and blood-based neoplastic cell cancers. VD and calcium supplementations reduce the risk of all types of cancers in postmenopausal women [75].
VK2 regulates the transcription of the enzyme that phosphorylates PKA via the SXR/PXR- or GGCX-independent pathway. Protein kinase-mediated VK action is associated with anti-tumor effects as well as steroidogenesis [76]. VK2 induces apoptosis in several types of tumor cells. Therefore, the VD3 and VK2 insufficiencies induced by statin use can increase the risk of several types of cancer.
Conclusions
Statins induce VD3 and VK2 deficiencies by inhibiting 7-DHC and GGPP production, respectively, besides inhibiting cholesterol production. Both VD3 and VK2, which up regulate OC and MGP mRNA levels by activating their respective nuclear receptors, play crucial roles in maintaining bone health, glucose homeostasis, and steroidogenesis. Moreover, insufficient levels of GGPP induced by statins cause disturb the secretion of peptide hormones, such as insulin and LH. Deficiencies of these intermediate metabolites of the mevalonate pathway induce type 2 diabetes, arteriosclerosis, cardiovascular disease, osteoporosis, and CKD, and disrupt endocrine function. The biochemical mechanisms of the actions of statins and warfarin reviewed here must be considered when these medicines are prescribed for long-term use.
Acknowledgements
We thank A. Ito, H. Shirakawa, N. Takumi, Y. Minegishi, A. Ohashi, Z.H. Howlader, Y. Ohsaki, T. Goto, and M. Komai from Tohoku University, and T. Sato from J-Oil mills Inc., Japan, for permission to reproduce the data for Figure 5, and the American Society for Nutrition and John Wiley and Sons, for permission to reproduce a part of the data for Figure 3 and 7, respectively.
References
- Shavelle DM, Takasu J, Budoff MJ, Mao S, Zhao XQ, et al. (2002) HMG CoA reductase inhibitor (statin) and aortic valve calcium. Lancet 359: 1125-1126 .
- Son BK, Kozaki K, Iijima K, Eto M, Kojima T, et al. (2006) Statins protect human aortic smooth muscle cells from inorganic phosphate-induced calcification by restoring Gas6-Axl survival pathway. Circ Res 98: 1024-1031 .
- Liu JC, Hao WR, Hsu YP, Sung LC, Kao PF, et al. (2016) Statins dose-dependently exert a significant chemopreventive effect on colon cancer in patients with chronic obstructive pulmonary disease: A population-based cohort study. Oncotarget 7: 65270-65283 .
- Ganda OP (2016) Statin-induced diabetes: incidence, mechanisms, and implications. F1000 Res .
- Culver AL, Ockene IS, Balasubramanian R, Olendzki BC, Sepavich DM, et al. (2012) Statin use and risk of diabetes mellitus in postmenopausal women in the Women's Health Initiative. Arch Intern Med 172: 144-152 .
- Cederberg H, Stancakova A, Yaluri N, Modi S, Kuusisto J, Laakso M (2015) Increased risk of diabetes with statin treatment is associated with impaired insulin sensitivity and insulin secretion: a 6 year follow-up study of the METSIM cohort. Diabetologia 58: 1109-1117 .
- Okuyama H, Langsjoen PH, Ohara N, Hashimoto Y, Hamazaki T, et al. (2016) Medicines and Vegetable Oils as Hidden Causes of Cardiovascular Disease and Diabetes. Pharmacology 98: 134-170 .
- Okano T, Shimomura Y, Yamane M, Suhara Y, Kamao M, et al. (2008) Conversion of phylloquinone (Vitamin K1) into menaquinone-4 (Vitamin K2) in mice: two possible routes for menaquinone-4 accumulation in cerebra of mice. J Biol Chem 283: 11270-11279 .
- Nakagawa K, Hirota Y, Sawada N, Yuge N, Watanabe M, et al. (2010) Identification of UBIAD1 as a novel human menaquinone-4 biosynthetic enzyme. Nature 468: 117-121 .
- Yi Z, Yokota H, Torii S, Aoki T, Hosaka M, Zhao S, et al. (2002) The Rab27a/granuphilin complex regulates the exocytosis of insulin-containing dense-core granules. Mol Cell Biol 22: 1858-1867 .
- Davidson JS, Eales A, Roeske RW, Millar RP (1993) Inhibition of pituitary hormone exocytosis by a synthetic peptide related to the rab effector domain. FEBS Lett 326: 219-221.
- Kanzaki M (2006) Insulin receptor signals regulating GLUT4 translocation and actin dynamics. Endocr J 53: 267-293 .
- Chiu TT, Jensen TE, Sylow L, Richter EA, Klip A (2011) Rac1 signalling towards GLUT4/glucose uptake in skeletal muscle. Cell Signal 23: 1546-1554 .
- Sylow L, Moller LL, Kleinert M, Richter EA, Jensen TE (2014) Rac1--a novel regulator of contraction-stimulated glucose uptake in skeletal muscle. Exp Physiol 99: 1574-1580.
- Suhara Y, Wada A, Okano T (2009) Elucidation of the mechanism producing menaquinone-4 in osteoblastic cells. Bioorg Med Chem Lett 19: 1054-1057 .
- Haussler MR, Haussler CA, Jurutka PW, Thompson PD, Hsieh JC, et al. (1997) The vitamin D hormone and its nuclear receptor: molecular actions and disease states. J Endocrinol 154 Suppl : S57-73 .
- Carvallo L, Henriquez B, Paredes R, Olate J, Onate S, et al. (2007) 1alpha,25-dihydroxy vitamin D3-enhanced expression of the osteocalcin gene involves increased promoter occupancy of basal transcription regulators and gradual recruitment of the 1alpha,25-dihydroxy vitamin D3 receptor-SRC-1 coactivator complex. J Cell Physiol 214: 740-749 .
- Tabb MM, Sun A, Zhou C, Grun F, Errandi J, et al. (2003) Vitamin K2 regulation of bone homeostasis is mediated by the steroid and xenobiotic receptor SXR. J Biol Chem 278: 43919-43927 .
- Ichikawa T, Horie-Inoue K, Ikeda K, Blumberg B, Inoue S (2006) Steroid and xenobiotic receptor SXR mediates vitamin K2-activated transcription of extracellular matrix-related genes and collagen accumulation in osteoblastic cells. J Biol Chem 281: 16927-16934 .
- Oury F, Sumara G, Sumara O, Ferron M, Chang H, et al. (2011) Endocrine regulation of male fertility by the skeleton. Cell 144: 796-809 .
- Oury F, Ferron M, Huizhen W, Confavreux C, Xu L, et al. (2013) Osteocalcin regulates murine and human fertility through a pancreas-bone-testis axis. J Clin Invest 123: 2421-2433 .
- Zhao L, Chen XJ, Zhu J, Xi YB, Yang X, et al. (2015) Lanosterol reverses protein aggregation in cataracts. Nature 523: 607-611 .
- Holick MF (2000) Sunlight and vitamin D: The bone and cancer connections. Radiation Protection Dosimetry 65-71 .
- Hirota Y, Tsugawa N, Nakagawa K, Suhara Y, Tanaka K, et al. (2013) Menadione (vitamin K3) is a catabolic product of oral phylloquinone (vitamin K1) in the intestine and a circulating precursor of tissue menaquinone-4 (vitamin K2) in rats. J Biol Chem 288: 33071-33080 .
- Darwish H, DeLuca HF (1993) Vitamin D-regulated gene expression. Crit Rev Eukaryot Gene Expr 3: 89-116.
- Kitazawa S, Kajimoto K, Kondo T, Kitazawa R (2003) Vitamin D3 supports osteoclastogenesis via functional vitamin D response element of human RANKL gene promoter. J Cell Biochem 89: 771-777 .
- Ichikawa T, Horie-Inoue K, Ikeda K, Blumberg B, Inoue S (2007) Vitamin K2 induces phosphorylation of protein kinase A and expression of novel target genes in osteoblastic cells. J Mol Endocrinol 39: 239-247 .
- Baron RA, Seabra MC (2008) Rab geranylgeranylation occurs preferentially via the pre-formed REP-RGGT complex and is regulated by geranylgeranyl pyrophosphate. Biochem J 415: 67-75 .
- Ren JC, Zhu Q, Lapaglia N, Emanuele NV, Emanuele MA (2005) Ethanol-induced alterations in Rab proteins: possible implications for pituitary dysfunction. Alcohol 35: 103-112 .
- Gomes AQ, Ali BR, Ramalho JS, Godfrey RF, Barral DC, et al. (2003) Membrane targeting of Rab GTPases is influenced by the prenylation motif. Mol Biol Cell 14: 1882-1899 .
- Leung KF, Baron R, Seabra MC (2006) Thematic review series: lipid posttranslational modifications. geranylgeranylation of Rab GTPases. J Lipid Res 47: 467-475 .
- Yu S, Yehia G, Wang J, Stypulkowski E, Sakamori R, et al. (2014) Global ablation of the mouse Rab11a gene impairs early embryogenesis and matrix metalloproteinase secretion. J Biol Chem 289: 32030-32043 .
- Barbarin A, Frade R (2011) Procathepsin L secretion, which triggers tumour progression, is regulated by Rab4a in human melanoma cells. Biochem J 437: 97-107 .
- Steffan JJ, Dykes SS, Coleman DT, Adams LK, Rogers D, et al. (2014) Supporting a role for the GTPase Rab7 in prostate cancer progression. PloS one 9: e87882 .
- Meng Z, Edman MC, Hsueh PY, Chen CY, Klinngam W, et al. (2016) Imbalanced Rab3D versus Rab27 increases cathepsin S secretion from lacrimal acini in a mouse model of Sjogren's Syndrome. American journal of physiology Cell physiology 310: 942-954 .
- Ohnishi H, Samuelson LC, Yule DI, Ernst SA, Williams JA (1997) Overexpression of Rab3D enhances regulated amylase secretion from pancreatic acini of transgenic mice. J Clin Invest 100: 3044-3052.
- Nashida T, Imai A, Shimomura H (2006) Relation of Rab26 to the amylase release from rat parotid acinar cells. Arch Oral Biol 51: 89-95 .
- Lee NK, Sowa H, Hinoi E, Ferron M, Ahn JD, et al. (2007) Endocrine regulation of energy metabolism by the skeleton. Cell 130: 456-469 .
- Drucker DJ (2006) The biology of incretin hormones. Cell Metab 3: 153-165 .
- Mizokami A, Yasutake Y, Higashi S, Kawakubo-YT, Chishaki S, et al. (2014) Oral administration of osteocalcin improves glucose utilization by stimulating glucagon-like peptide-1 secretion. Bone 69: 68-79 .
- Zhou M, Ma X, Li H, Pan X, Tang J, et al. (2009) Serum osteocalcin concentrations in relation to glucose and lipid metabolism in Chinese individuals. Eur J Endocrinol 161: 723-729 .
- Yeap BB, Chubb SA, Flicker L, McCaul KA, Ebeling PR, et al. (2010) Reduced serum total osteocalcin is associated with metabolic syndrome in older men via waist circumference, hyperglycemia, and triglyceride levels. Eur J Endocrinol 163: 265-272 .
- Yasuma T, Yano Y, D'Alessandro-Gabazza CN, Toda M, Gil-Bernabe P, et al. (2016) Amelioration of diabetes by protein S. Diabetes 65: 1940-1951 .
- Benzakour O, Gely A, Lara R, Coronas V (2007) Gas-6 and protein S: vitamin K-dependent factors and ligands for the TAM tyrosine kinase receptors family. Med Sci 23: 826-833 .
- Lamson DW, Plaza SM (2003) The anticancer effects of vitamin K. Altern Med Rev 8: 303-318 .
- Wimmel A, Rohner I, Ramaswamy A, Heidtmann HH, Seitz R, et al. (1999) Synthesis and secretion of the anticoagulant protein S and coexpression of the Tyro3 receptor in human lung carcinoma cells. Cancer 86: 43-49.
- Dormady SP, Zhang XM, Basch RS (2000) Hematopoietic progenitor cells grow on 3T3 fibroblast monolayers that overexpress growth arrest-specific gene-6 (GAS6). Proc Natl Acad Sci U S A 97: 12260-12265 .
- Beulens JW, van der AD, Grobbee DE, Sluijs I, Spijkerman AM, et al. (2010) Dietary phylloquinone and menaquinones intakes and risk of type 2 diabetes. Diabetes Care 33: 1699-1705 .
- Ferron M, Wei J, Yoshizawa T, Del Fattore A, DePinho RA, et al. (2010) Insulin signaling in osteoblasts integrates bone remodeling and energy metabolism. Cell 142: 296-308 .
- Luo G, Ducy P, McKee MD, Pinero GJ, Loyer E, Behringer RR, Karsenty G (1997) Spontaneous calcification of arteries and cartilage in mice lacking matrix GLA protein. Nature 386: 78-81 .
- Jono S, Ikari Y, Vermeer C, Dissel P, Hasegawa K, et al. (2004) Matrix Gla protein is associated with coronary artery calcification as assessed by electron-beam computed tomography. Thromb Haemost 91: 790-794 .
- Hashimoto Y, Mori M, Kobayashi S, Hanya A, Watanabe S, et al. (2014) Canola and hydrogenated soybean oils accelerate ectopic bone formation induced by implantation of bone morphogenetic protein in mice. Toxicology Reports 1: 955-962 .
- Geleijnse JM, Vermeer C, Grobbee DE, Schurgers LJ, Knapen MH, vet al. (2004) Dietary intake of menaquinone is associated with a reduced risk of coronary heart disease: the Rotterdam Study. J Nutr 134: 3100-3105 .
- Trion A, Schutte-Bart C, Bax WH, Jukema JW, van der Laarse A (2008) Modulation of calcification of vascular smooth muscle cells in culture by calcium antagonists, statins, and their combination. Mol Cell Biochem 308: 25-33 .
- Kupcsik L, Meurya T, Flury M, Stoddart M, Alini M (2009) Statin-induced calcification in human mesenchymal stem cells is cell death related. J Cell Mol Med 13: 4465-4473.
- Saremi A, Bahn G, Reaven PD (2012) Progression of vascular calcification is increased with statin use in the Veterans Affairs Diabetes Trial (VADT). Diabetes Care 35: 2390-2392 .
- Lieben L, Masuyama R, Torrekens S, Van Looveren R, Schrooten J, et al. (2012) Normocalcemia is maintained in mice under conditions of calcium malabsorption by vitamin D-induced inhibition of bone mineralization. J Clin Invest 122: 1803-1815 .
- Fusaro M, Gallieni M, Rebora P, Rizzo MA, Luise MC, et al. (2016) Atrial fibrillation and low vitamin D levels are associated with severe vascular calcifications in hemodialysis patients. J Nephrol 29: 419-426 .
- Ito A, Shirakawa H, Takumi N, Minegishi Y, Ohashi A, et al. (2011) Menaquinone-4 enhances testosterone production in rats and testis-derived tumor cells. Lipids Health Dis 10: 158 .
- Shirakawa H, Ohsaki Y, Minegishi Y, Takumi N, Ohinata K, et al. (2006) Vitamin K deficiency reduces testosterone production in the testis through down-regulation of the Cyp11a a cholesterol side chain cleavage enzyme in rats. Biochim Biophys Acta 1760: 1482-1488 .
- Fusaro M, Crepaldi G, Maggi S, Galli F, D'Angelo A, Calo L, et al. (2011) Vitamin K, bone fractures, and vascular calcifications in chronic kidney disease: an important but poorly studied relationship. J Endocrinol Invest 34: 317-323 .
- Krueger T, Westenfeld R, Ketteler M, Schurgers LJ, Floege J (2009) Vitamin K deficiency in CKD patients: a modifiable risk factor for vascular calcification? Kidney Int 76: 18-22 .
- Mundy G, Garrett R, Harris S, Chan J, Chen D, et al. (1999) Stimulation of bone formation in vitro and in rodents by statins. Science 286: 1946-1949.
- Edwards CJ, Hart DJ, Spector TD (2000) Oral statins and increased bone-mineral density in postmenopausal women. Lancet 355: 2218-2219 .
- Chan KA, Andrade SE, Boles M, Buist DS, Chase GA, et al. (2000) Inhibitors of hydroxymethylglutaryl-coenzyme A reductase and risk of fracture among older women. Lancet 355: 2185-2188 .
- Wada Y, Nakamura Y, Koshiyama H (2000) Lack of positive correlation between statin use and bone mineral density in Japanese subjects with type 2 diabetes. Arch Intern Med 160: 2865 .
- Booth SL, Tucker KL, Chen H, Hannan MT, Gagnon DR, et al. (2000) Dietary vitamin K intakes are associated with hip fracture but not with bone mineral density in elderly men and women. Am J Clin Nutr 71: 1201-1208 .
- Shiraki M, Shiraki Y, Aoki C, Miura M (2000) Vitamin K2 (menatetrenone) effectively prevents fractures and sustains lumbar bone mineral density in osteoporosis. J Bone Miner Res 15: 515-521.
- Abe E, Miyaura C, Sakagami H, Takeda M, Konno K, et al. (1981) Differentiation of mouse myeloid leukemia cells induced by 1 alpha,25-dihydroxyvitamin D3. Proc Natl Acad Sci U S A 78: 4990-4994.
- Miyaura C, Abe E, Kuribayashi T, Tanaka H, Konno K, et al. (1981) 1 alpha,25-Dihydroxyvitamin D3 induces differentiation of human myeloid leukemia cells. Biochem Biophys Res Commun 102: 937-943.
- Corder EH, Guess HA, Hulka BS, Friedman GD, Sadler M, et al. (1993) Vitamin D and prostate cancer: a prediagnostic study with stored sera. Cancer Epidemiol Biomarkers Prev 2: 467-472 .
- Wyllie AH, Morris RG, Smith AL, Dunlop D (1984) Chromatin cleavage in apoptosis: association with condensed chromatin morphology and dependence on macromolecular synthesis. J Pathol 142: 67-77 .
- Wu G, Fan RS, Li W, Ko TC, Brattain MG (1997) Modulation of cell cycle control by vitamin D3 and its analogue, EB1089, in human breast cancer cells. Oncogene 15: 1555-1563 .
- Pilon C, Urbanet R, Williams TA, Maekawa T, Vettore S, et al. (2014) 1alpha, 25-Dihydroxyvitamin D(3) inhibits the human H295R cell proliferation by cell cycle arrest: a model for a protective role of vitamin D receptor against adrenocortical cancer. J Steroid Biochem Mol Biol 140: 26-33 .
- Lappe JM, Travers-Gustafson D, Davies KM, Recker RR, Heaney RP (2007) Vitamin D and calcium supplementation reduces cancer risk: results of a randomized trial. Am J Clin Nutr 85: 1586-1591 .
- Shibayama-Imazu T, Aiuchi T, Nakaya K (2008) Vitamin K2-mediated apoptosis in cancer cells: role of mitochondrial transmembrane potential. Vitam Horm 78: 211-226.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi