Research Article, J Pharm Drug Deliv Res Vol: 14 Issue: 1
Sustained Drug Delivery in Pharmaceutical Dosage Forms for Novel Approaches of Formulation Design
Niladri Shekhar Dey*, Saumendu Deb Roy, Dibyendu Shil and Sushanta Kumar Das
Department of Pharmacy, Mata Gujri College of Pharmacy, Mata Gujri University, Bihar, India
*Corresponding Author: Niladri Shekhar Dey
Department of Pharmacy, Mata Gujri College of Pharmacy, Mata Gujri University, Bihar, India
E-mail: niladrisekhar111@gmail.com
Received date: 27 September, 2024, Manuscript No. JPDDR-24-148813;
Editor assigned date: 30 September, 2024, PreQC No. JPDDR-24-148813 (PQ);
Reviewed date: 15 October, 2024, QC No. JPDDR-24-148813;
Revised date: 13 February, 2025, Manuscript No. JPDDR-24-148813 (R);
Published date: 20 February, 2025, DOI: 10.4172/2325-9604.1000318.
Citation: Dey NS, Roy SD, Shil D, Das SK (2025) Sustained Drug Delivery in Pharmaceutical Dosage Forms for Novel Approaches of Formulation Design. J
Pharm Drug Deliv Res 14:1.
Abstract
Dosage form design involves the administration of API to achieve corrective or remedial effects on humans in the pharmaceutical field. Medication delivery technology consists of two main categories: Customary or standard medicament delivery devices and novel formulation technology for sustained medicament administration. Depending upon the formulation pattern, fluctuations in the API concentration may occur in serum level due to the direct release of medicament(s) from standard dosage forms. To maintain the steady-state concentration of API in serum level within the therapeutic window according to biopharmaceutics and pharmacokinetics, novel formulation delivery devices are advanced and modern technologies for controlled/sustained action for remedial or restorative response and these devices are also applicable for targeting at specific sites. Currently, sustained medicament administration is designed or framed to lessen or reduce undesired toxic effects as well as gastrointestinal irritation. The merit of this formulation design is that it is helpful for monitoring the dosing pattern and simultaneously lessens fluctuations in circulating the medicament concentrations in serum. Due to lower bulk density of gastro retentive devices lower than gastric fluid which float in gastric fluid and increase the residence time so that floating mechanism can be improved. Parenteral repository technique influences the sustainable delivery for the medicament via different nano formulation which also acts as a reservoir type of devices. Chrono-therapeutic is the novel technology for the administration of medicament at the right time, right dose for increase therapeutic efficacy with the design of microfabrication and 3D printing technology.
Keywords: Sustained medicament administration, Steadystate concentration, Parenteral repository technique, Chronotherapeutic, Microfabrication, 3D printing technology
Introduction
Medicament administration systems arethedevicesin which pharmaceutical formulations are designed or framed for the incorporation of Active Pharmaceutical Ingredient (API) along with nondrug components such as excipients or adjuvants to improve therapeutic responses or pharmacological actions in the human body [1]. The main objectives of drug delivery technologies or dosage form design are enhancing the safety and efficacy of API molecules subside the adverse side effects in patients which are known as therapeutic drug monitoring [2]. The formulation design is applicable for the application of API along with excipients and the release of therapeutic products at the site of action simultaneously reveals pharmacological action [3].
Compared with other oral solid and liquid medication, the goal of the easy administration of this formulation is to reach the medicament(s) in the blood stream and then therapeutic circle [4].
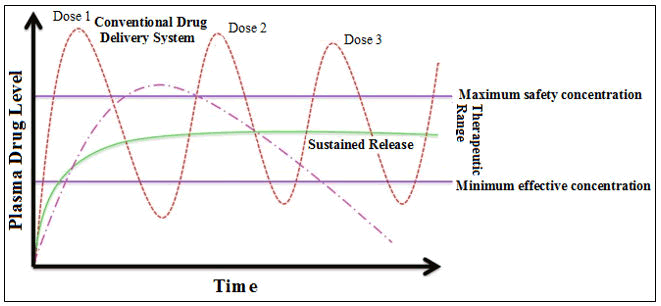
The immediate release of API after oral administration instantaneously increased medicament(s) concentration in systemic circulation possibly and may increase the toxicity [5]. This may subsequently create a rapid reduction in medicament(s) concentration to a sub therapeutic level due to frequent administration of the active medicament(s) [6] (Figure 1). To increase the API concentration within the therapeutic circle, sustainable technology is needed because of its longer duration of action in the substantial or plateau state and simultaneously lessens in the risk of adverse effects of medicament(s), maintenance dosage frequency and enhancement of patient compliance [7]. The modified release medicament(s) delivery devices can achieve optimum medicament entrapment capacity and safe action of the API molecule(s) via accidental discharge from the devices and easy to comfortable for administration to the patients [8].
Figure 1: Plasma drug concentrations used for conventional and controlled drug delivery.
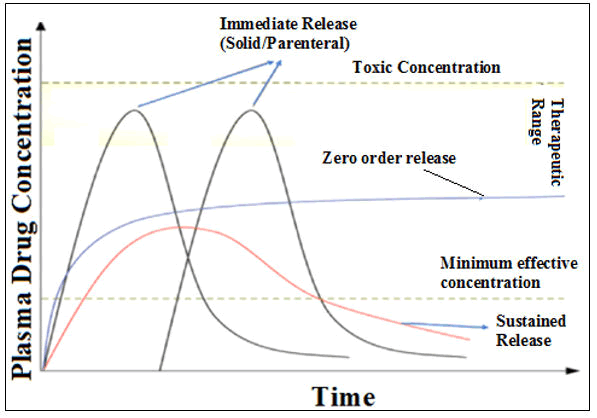
The maintenance of the release of medicament(s) from devices is necessary to achieve the desired therapeutic range that would yield the optimum plasma medicament concentration for longer period [9] (Figure 2). The main benefit of this sustainable design is improving the pharmacokinetic parameters by increasing the bioavailability, predictable gastrointestinal transit time, less localized gastrointestinal disturbances, optimum serum API concentration and greater therapeutic potency.
Figure 2: Drug plasma level and release profile.
Here, we focus on different technology of modified release dosage forms and their remedial response compared with that of customary dosage forms. Floating systems and micro balloons or hollow microspheres are also reported. The chrono-therapeutic treatment also influences the specific medicament(s) delivery according to time and improves the chrono-pharmacokinetic parameter profile at the serum level. We also highlight targeted action at the site along with the parenteral repository system in this novel medication technology.
Modified release API delivery design
- Extended release of medicaments.
- Sustained release pattern of medicament from different dosage forms.
- Delayed release of medicament from dosage forms.
- Targeted release technique with nano medicine.
- Design of prolonged action medication technology.
Different types of novel technology of formulation design are available as follows:
Extended release of the medicament: Two-fold reductions in dosing frequency revealed in the devices for the medicament in dosage form compared with the conventional medication. Novel and modified delivery design of active medicament(s) is under this type of formulation technology.
Sustained action of medicament from novel formulation: These types of devices are framed to discharge the API at an in a prearranged manner for fixed different time interval for regulation of plateau state medicament concentration in system circulation for longer time duration with few or no adverse drug reactions and toxic effects. This information has been reported through a variety of formulations, such as nanoparticles, liposomes, niosomes, carbon nanotubes, resealed erythrocytes and drug-polymer conjugates (examples of hydrogels).
Delayed release of medicament from dosage forms: A pharmaceutical dosage form that discharge the medicament(s) slowly for protracted period for better curative action. An initial portion from total API may be discharge immediately after the administration of the dosage and simultaneously the rest amount of API delivers slowly for longer period.
Targeted release dosage forms and nano medicine: Targeted action is also released the medicament at specific site of the effective organ. The medicaments used for the targeted discharge at various pharmaceutical doses may exhibit extended or immediate release characteristics as a novel medication strategy. The dosage form may be applied by passive targeting as well as active targeting by tagging with ligand molecules so that they can easily recognize the effector organ.
Mouth Dissolving Tablets (MDTs): MDTs which rapidly disintegrate in saliva after oral administration in the buccal cavity. MDTs loaded with API may be used with or without the addition of little water where the medicaments are dispersed in saliva and are swallowed with or without water [10].
Materials and Methods
Classification of extended-release technology
Based on the medicament discharge mechanism, sustained delivery technique is classified as follows:
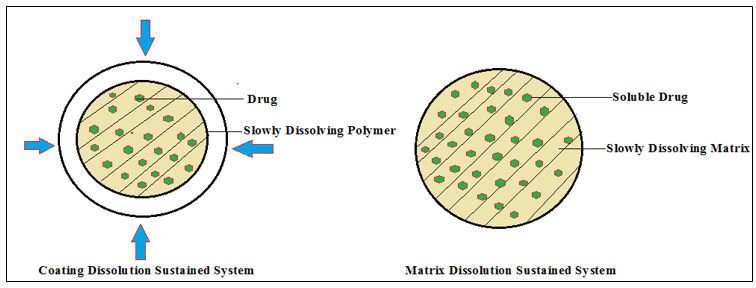
Dissolution-controlled drug delivery technology: Active pharmaceutical ingredients are either loaded with or uniformly embedded within polymeric membranes (reservoir systems) or matrices (monolithic systems), respectively which are slowly dissolving. API is incorporated with in the polymeric membrane for reservoir systems with low solubility.
Diffusion-controlled drug delivery technology: Modified API discharge devices via diffusion-based have been developed as polymeric matrix or reservoir systems, depending on their morphological structure. Reservoir systems are generally polymeric membranes (water insoluble) through which the medicament(s) diffuses, whereas in polymeric matrix systems, the medicament(s) are delivered by passing through the matrix itself. In diffusion-controlled release technology, API(s) is entrapped in and released via diffusion through polymeric membranes (reservoir systems) or polymeric matrices (monolithic systems) which are inertly insoluble in nature. These devices are mainly two categories such as control reservoirs and monolithic matrix device techniques. The rate-limiting step in diffusion-controlled systems is the diffusion of medicament(s).
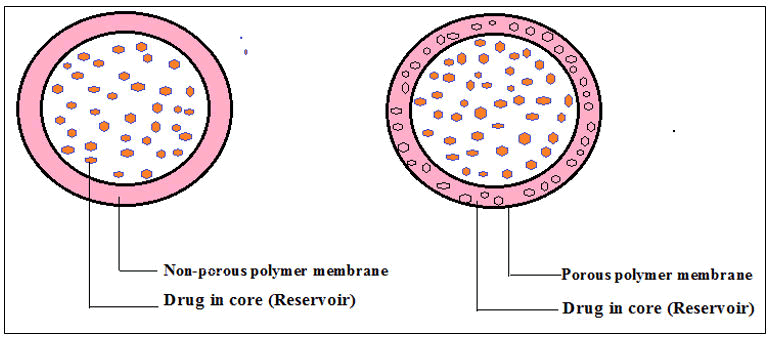
Monolithic and membrane-controlled systems are two types of diffusion-controlled systems. In membrane-controlled systems, the medicament is incorporated within the core which acts as a reservoir, and it is surrounded by a thin porous and nonporous polymeric core. The medicament released from the membrane by the diffusion process and the discharge rate depends upon the physicochemical characteristics of the API (molecular composition, formulation pattern, core and coating layer length) (Figure 3). Common technologies for fabricating membrane-controlled reservoir systems include press coating and encapsulation of tablets.
Figure 3: Membrane-controlled drug delivery systems.
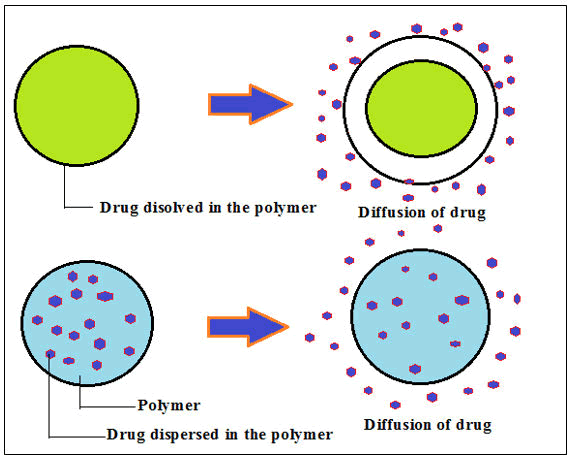
In these devices the API(s)are embedded throughout the polymer matrix devices homogenously. Medicament or API delivery occurs through diffusion when the boundary coating layer is eroded to an aqueous solution, allowing medicaments to slowly come out from the matrix devices for a specific time point (Figure 4). The API discharge pattern is generally proportional to the size of the matrix device i.e., when the size of the matrix lessens medicament(s) release falls off (Figure 5).
Figure 4: Diffusion monolithic or matrix-controlled medicament delivery technology.
Figure 5: Medicaments embedded in a polymer dissolving matrix.
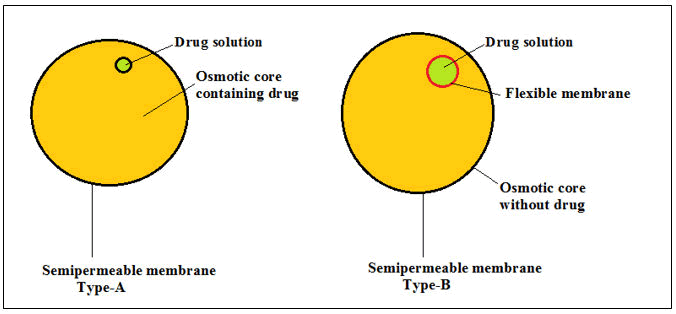
Modified formulation technology by osmotic pressure: This technology involves the use of osmotic pressure and osmogenes for controlled drug delivery. The procedure of solvent movement for solute molecules from a less concentrated media to more concentrated media known as osmosis occurs across as semipermeable penetrable screen (Figure 6). Osmotic pressure is formed by the movement of water within a semipermeable screen separated by the two various concentrations of solutes in solution. The basic ingredients used for osmotic delivery include medicament(s) which is known as osmogenes which may be incorporated into the formulation.
Figure 6: Schematic representation of type A and type B osmotic system.
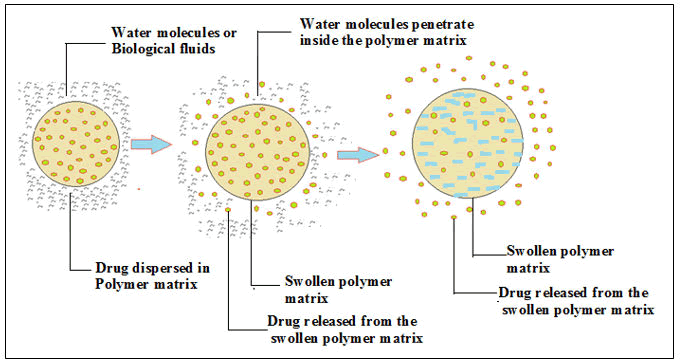
Swelling-controlled drug delivery devices: In this device, the APIs are uniformly embedded in the stringent or stubborn translucent polymer matrix (hydrophilic). In an aqueous solution, water penetrates through the matrix and below the ambient temperature; the glass transition temperature of the polymer reduces (Figure 7). This procedure forms the matrix in expand and flexible, and as a result slow medicament(s) diffuse out of from the inflate polymer matrix devices.
Figure 7: Schematic representation of swelling-controlled medicament delivery devices.
Gastro retentive technology
These devices are fabricated to increase the longer time of duration of API in the Gastrointestinal Tract (GIT) for sustained release medication for predetermined manner. These formulations are applicable for medicaments that are absorbed from the stomach in acidic media i.e., those poorly soluble in alkaline media such as furosemide, diazepam and albuterol and drugs which having narrow therapeutic index.
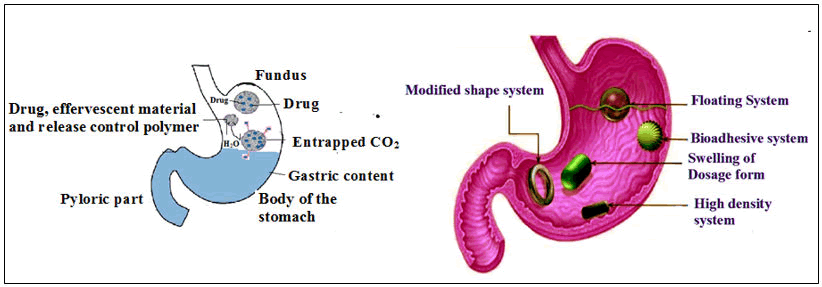
These drug delivery technologies consist of floating and nonfloating mechanisms. Based on the floating mechanism, there are two types such as fizzy or sparkling and not sparkling and depending on gastro retention function non-floating systems are again divided into four different patterns.
Floating system or low-density system: The bulk density of gastro retentive devices is lower than that of gastric fluids which remain buoyant i.e., able to float in the gastric lavage of the abdomen for longer time duration, without any alteration in the rate of gastric emptying. The medicament is discharged simultaneously in a preprogrammed technique from the devices. As a result, floating medicament(s)delivery devices increase the gastric residence time and control the fluctuation rate of serum API(s) concentrations.
The medication therapy is in a controlled manner at different time point from the device when the devices can float the gastric media. There are generally two types of floating drug delivery devices: Nonsparkling and gas-generating systems.
The inflatable devices consisted of a pilot device with a vacuum chamber loaded with volatile liquids. When the inflatable systems are administered in the gastric media (GI fluid), so that the vacuum spaces inflate, and it float in the GI fluid of stomach. The inflatable devices were constructed with a bio erodible and biodegradable polymer such as polyethylene and polyvinyl alcohol. The polymer starts to dissolve (due to polymer erosion) and gradually discharges the medicament when it floats in GI fluid. After discharge of the API, inflatable chamber will close (Table 1).
| Brand (Product) | Active medicament | Delivery system | Company |
| Xifaxan | Rifampicin | Bio adhesive tablets for gastro retention | Lupin, India |
| Cytotec | Misoprostal | Bilayer capsule devices based on floating | Pfizer, U.K. |
| Prazopress XL | Prazosin hydrochloride | Swelling based floating system on effervescent | Sun Pharma, Japan |
| Zanocin | Ofloxacin | Floating system based on effervescent | Ranbaxy, India |
| Coreg CR | Carvedilol | Osmotic system by gastro retention | Glaxo Smith Kline, UK |
| Cipro XR | Ciprofloxacin hydrochloride | Matrix based system based on erosion | Bayer, USA |
| Madopar HBS | Levodopa and benserzide | Controlled release capsule based on floating | Roche, UK |
| Gabapentin GR | Gabapentin | Swelling technology ACU-Form based on polymer | Depomed, USA |
Table 1: Marketed products for gastro-retention.
Floating intra gastric devices framed by a vacuum connected chamber and a microporous compartment serve as repository system for API. The method has been designed for intra gastric floating tablets of verapamil HCL via gel-forming agents such as Hydroxy Propyl Methyl Cellulose (HPMC), carbopol and xanthan gum. By incorporating via combination of anhydrous citric acid and sodium bicarbonate (sparkling mixture), buoyancy or floatability was achieved (Figure 8).
Figure 8: Schematic diagram of the floating design in GI fluid.
There are two types of matrix tablets such as single-layer and bilayer. Single-layer matrix tablets are designed or constructed by using API with hydrocolloid forming gel, and the bilayer matrix tablets are designed with one immediate release layer of medicament and another one is extended-release layer.
Micro balloons or hollow microspheres loaded with active medicament(s) with polymers were constructed via solvent diffusion or simple solvent evaporation processes to regulate the Gastric Residence Time (GRT). Upon evaporation of the solvent, gas is generated which disperses the polymer droplet and forms an interior orifice in the microsphere of the medicament with the polymer.
Parenteral repository formulation technology
Parenteral repository technology is gaining tremendous popularity among medical practitioners and scientists because of its significant clinical benefits of diseases. Parenteral repository formulations which act as medicament(s) reservoirs at the site of injection are combinations of oleaginous and aqueous suspensions that are administered through IM or SC route. From that reservoir the medicament(s) release simultaneously at a predetermined level and in a predictable manner according to the formulation design pattern. Long-acting formulations enhance or modify the therapeutic value of medicines so that the dosing frequency falls off or subsides. Parenteral repository technology has been designed to improve the efficacy and treatment of critical chronic diseases by decreasing adverse side effects in patients.
Several novel formulations of parenteral repository formulations as for e.g. solutions, emulsions, liposomes, micelles, implants, microparticles, nanoparticles and nano capsules are nowadays designed. Among them implants and micro-particles are utilized only as controlled release devices.
Several pharmaceutical formulations for controlled-release or sustained-release medication in parental dosage form have been developed.
Technology of parenteral depot formulations:
- Aqueous solution, viscous vehicle such as polyvinylpyrrolidine or gelatin.
- Colloidal suspensions have thixotropic nature.
- Immiscible water vehicles such as vegetable oil.
- Vasoconstrictor application.
- Administration of salt complexes and esters.
- Dispersions in microcapsules.
Depot formulation devices consist of several types as follows:
Dissolution of controlled depot devices
The absorption rate of API is controlled and governed by the dissolution of the medicament or depending on the biological fluid surrounding by the drug-loaded devices.
Complexation or salt formation: e.g. Benzathine penicillin G aqueous suspensions from alkali salt.
Microcrystalline suspensions: Macro crystals are larger size crystal molecules that dissolve and solubilize at a slower rate than smaller size crystal molecules and can be applied to regulate the rate of medicament dissolution.
Depot formulation (adsorption)
This formulation is framed by the conjugation of medicament with adsorbents. Thus, free species of the unbound active ingredient are available for adsorption. A fraction of the bound medicament is released to adjust the equilibrium stage when the unbound medicament is absorbed.
Depot formulations (encapsulation)
This depot type of formulation is designed by dispersing the medicament(s) in a diffusion matrix or encapsulating the medicament (solid) within a permeation barrier. Both of these two devices are manufactured from polymers that are either biodegradable or bioabsorbable in nature.
Depot formulation (esterification)
This formulation was constructed by synthesizing and manufacturing bio erodible esters of API(s) and then developing an injectable formulation. Medicament(s) loaded formulation forms drug reservoir devices at the specific area where injection is administered. The absorption rate is maintained by the fractionating or ramifying effect from the reservoirs to biological tissue fluid.
Excipients for designing the depot formulation technique use two major types of components to frame the formulation devices polymers and lipids.
Oil-based solutions
The medications were suspended or dissolved in oily solutions for the design of injectable repository systems that are traditionally used for hormones and antipsychotic drugs. Medicaments are generally used for dissolving or solubilizing pharmaceutical oils such assesame seed oil or triglycerides. Transformation of medicament from the oil phase into the biological tissue fluid via the intramuscular route injection which represents repository stage maintains sustained release. Spreading of the oil solution including muscle tissues influences the long acting or vibrating pattern via increasing of the surface area.
Liposomal systems
Liposomes are bilayer phospholipid vesicles (microscopic), and cholesterol may be used to improve structural rigidity for build-up liposomal lamellarity. The liposomal surface may be used for active targeting via the tagging of ligand molecules such as aptamers or antibodies with linker molecules so that they can easily recognize the malignant cells or sites of action. Liposomal formulation alone or the conjugation of linker molecules may provide uninterrupted or prolonged action of the encapsulated medicaments.
Both small and large conventional unilamellar lipid vesicles in combination with biocompatible and biodegradable ingredients to form liposomes or lipid vesicles in the gelact as repository system from where medicament(s) deliver slowly in uninterrupted manner.
In situ forming injectable gels
This technique is novel technology of repository system that is designed upon contact with biological fluid. Phase Separation Gels (PPSGs) which are phospholipids in nature can combine with the highly bio-resorbable solvent excipients. The addition of the medicament(s) in sterile filtration and dissolving or solubilizing the phospholipids in oil and ethanol mixtures can be applicable to produce suitable injectable formulations.
Microparticle repository injections are commercial drug products composed of which framed by bioresorbable polymersused in the field of oncology and CNS disorder. These may be delivered via different parenteral routes including the intramuscular, subcutaneous, and peritoneal routes.
Chrono therapeutic sustained drug delivery systems
Chrono therapeutic is modern technology of medication therapy in which the timing of medication administration is very important and specific drugs are utilized at the correct dose in particular diseases with synchrony of the human body in the day-night pattern and symptoms showing enhancing beneficial effects at the therapeutic level by avoiding the adverse effects.
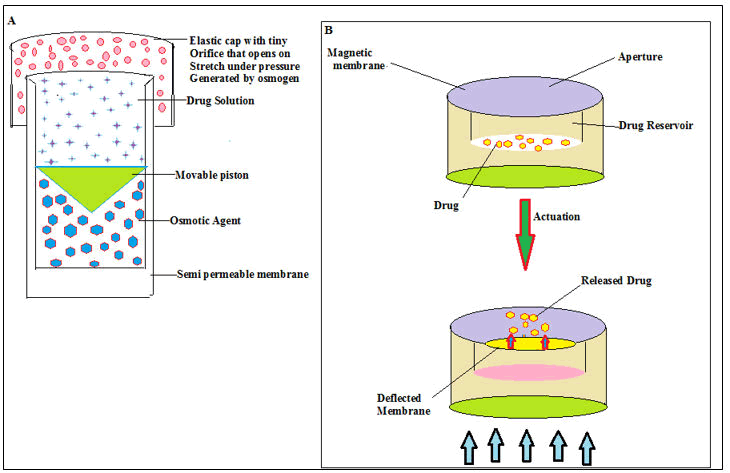
At present, chrono therapeutic is applied to the patients where deliver the active medicament(s) at the right time with the right dose at the target site of action. Depending upon regulation of chrono biology and circadian rhythms, after a lag time drug molecules are released completely (Figure 9). Diseases such as hypertension, ulcers, hypercholesterolemia, asthma, cancer, congestive heart failure, arthritis and diabetes are common. These diseases exhibit a significant night-time pattern of onset and symptom exacerbation.
Figure 9: (A) System based on an expandable orifice, (B) API(s) release from magnetically induced palpitating systems.
Results and Discussion
Novel technology applied to chrono therapeutic
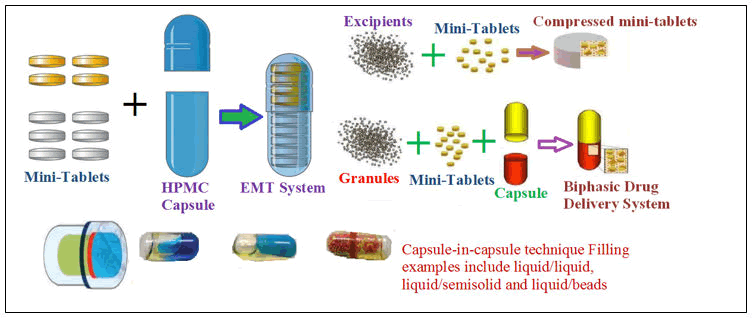
Capsule within capsule technology devices: Application of these technology devices mainly for chronic delayed medicament release in capsule dosage forms by orally. The inner layer of capsule is very small and contains a semisolid or liquid composition, and both capsules may be composed of either HPMC or gelatine. By using an appropriate coating layer, the outer and inner capsules can be targeted to the GI tract (Figure 10).
Figure 10: Schematic diagram of capsule within capsule technology.
In recent pharmaceutical fields time-dependent sustained or extended drug delivery system such as Vimovo TM and Combodart TM (GSK) amalgamation have been widely applied.
Merits or advantages
- Applicable for both zero-order and multiphase release.
- Two separate chambers in one single formulation unit.
- From an economic point of view, it enhances patient acceptability and compliance.
- Applicable for two separate parts of the GI system.
- Sustained, pulsed or delayed release characteristics are possible.
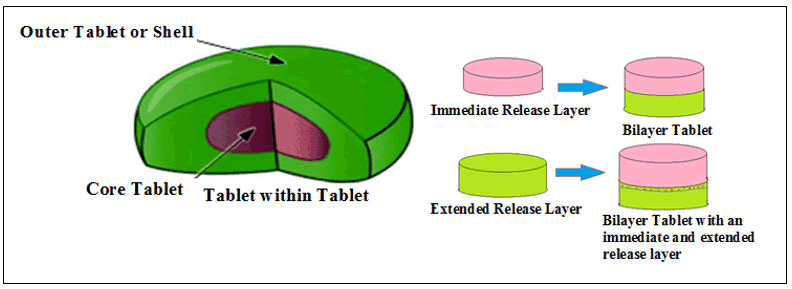
Tablet within tablet technology devices: In this technology granules are compressed by novel tableting machinery within tablet core which is prefabricated. This technique is a dry technique in which compression coating is necessary. The internal core is surrounded by covering ingredients to make up this type of tablet. During manufacturing of core of the tablet, is transported to a die cavity (larger) within center portion and entrapped with the coating granules.
The powder material is sprayed for coating on the upper portion of core scale to obtain tablet within tablet devices (Figure 11).
Figure 11: Schematic diagram of the immediate and extended release of drug(s) from bilayer tablets by tablet-in-tablet technology.
Capsule within tablet technology: These are recent novel combinations of modern technology that combine modified release capsules and controlled release tablets. API is incorporated within barrier material which is made of bio erodible and biologically acceptable polymeric materials.
Several thickness and lag times can be prepared by modification or alteration of the structure of the barrier material. The medicament is discharged in a predictable and palpitating manner when barrier material is eroded or may dissolve. During the processing of multiple and multifunctional units for oral administration flexible tablets were filled within a hard capsule shell. For chrono therapeutic medication different types of mini-tablets such as rapid-release, sustained-release, pulsatile and delayed-onset tablets are designed with several lag time periods for their discharge of the active medicament.
Technology for granules and tablets within capsule: In biphasic technology for delivery of the API at two different time points one is quick or immediate and second slow release at prearranged time intervals. When maximum API delivery is required rapidly and reduces the dosage frequency, biphasic techniques can be applied, where sharp initial release followed by a longer period of medicament(s) release phase (Table 2). Tablets and granules, which are designed for quick release of the medication, are used to incorporate in separate one layer and the second component layer where the medicament(s)is continuously released from the matrix, which keeps the plateau state concentration of medicament in serum level.
| Product (Trade Name) | Drug | Types |
| Desval ER tablets | Divalproex Sodium extended release tablets (250/500 mg/g) | Extended discharge API diffusion-controlled matrix tablets |
| Roliten OD | Tolterodine tartrate capsule (2/4 mg) for longer duration medicament discharge | Repository type control release pellets incorporated in empty capsule box |
| Entocost | Budenoside capsule (9 mg) | Novel formulation longer duration action capsule for colon targeted medicament discharge |
| Coamoxyclav ER tablets | Amoxicillin and potassium clavulanate tablets | Control release bilayer matrix tablets |
| Contifluo | Tamsolus in control release pellets | Diffusion and dissolution regulated control release pellets |
| Cifran OD | Ciprofloxacin tablets (500 mg/g) | Sparkling or fizzy floating matrix tablets |
Table 2: Commercially available novel formulation for longer duration.
Fabrication and design of polymeric delivery systems
Compression table methods involving biodegradable polymers which are new methods for the fabrication of controlled drug delivery devices are nowadays used for fabrication process.
Micro fabrication and 3D printing technologies are currently designed for the preparation of delayed release of API.
Micro fabrication technology: Microfabrication including photolithography and film deposition consists of repeated steps for creating delivery devices. Two-dimensional (2D) features are framed by CAD. The software was printed as a photolithography process with resolution of 0.5-1 μm.
Silicon which is a ring foundation may be used as master molds for inverse replicas of salt elastomers which are based upon the ultimate application of micron-sized features. In lithography technique poly (dimethyl siloxane) is used as the soft elastomer material.
Micro fluids are used for the manufacture of controlled-release via drug loaded carriers for tissue engineering applications, specifically in high-throughput field for medicament screening. Several Micromechanical System (MEMS) devices were established which could be effective for sustainably releasing medicaments. Based on their construction and mechanisms, MEMS are two types: multi reservoir and micro pump-based systems.
Multi-reservoir systems embedded with microchips which are used for delivering the medicaments with different predetermined release at time points. Micro pump-based systems are homogeneously distributed in micro fluidic networks to release API in a controlled manner via several actuation mechanisms such as piezoelectric pumping and battery-free magnetic pumping (Figure 12).
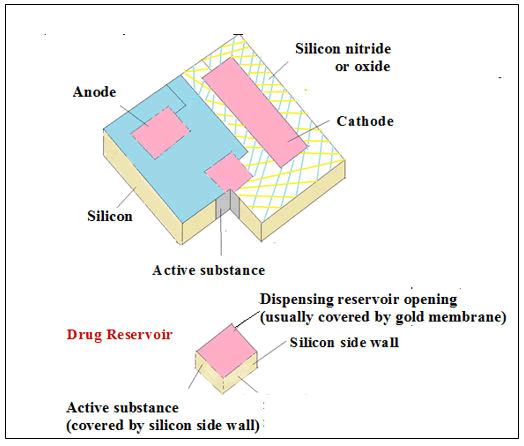
Figure 12: Implantable microchip (silicon) devices for adjusting the release rates of API via Electro chemical dissolution technique
Differential release of medicament(s) by microchip devices: Drug delivery devices that ensure the delivery of therapeutic medicament(s) at the site of the organ (right target) to improve therapeutic effectiveness bypass the presynaptic metabolism and eliminate toxic substances. Implantable medications are therefore designed to improve patient compliance and enhance local delivery.
Currently microchips have been developed for implantable systems with medicament discharge in palpitating patterns.
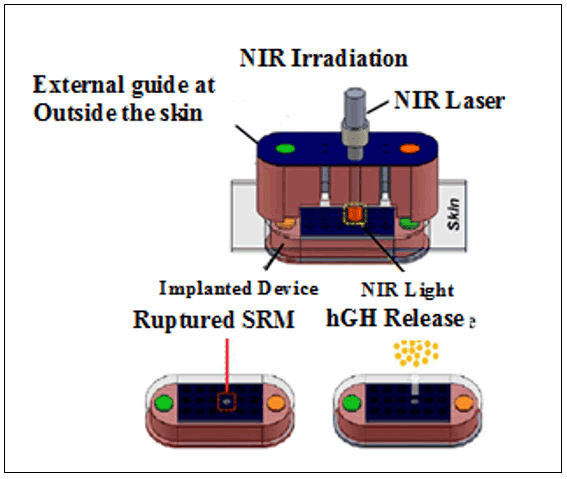
The development of the microchip was a sequential process that consisted of silicon wafers and microelectronic processing technology. Chemical substances (single or multiple) were loaded in the microchip in a ~25 nl reservoir coated with thin membranes such as gold which act as anodes (Figure 13).
Figure 13: Microchip device containing human Growth Hormone (hGH)incoporated in multiple reservoirs. The external guide and the device at the outer skin were magnetically attached (2, 3) A Stimuli- Responsive Membrane (SRM) was designed for generation of heat breaks it in 5 s upon NIR irradiation.
The combination of resonance (electron cyclotron) with photolithography which increases reactive ion may be applied on the upper side of the microchip layer made up of silicon nitride (pattern 17 mm × 17 mm).
Different erodible polymers with several molecular weights should be used for construction of the reservoir sealing chamber for controlling the degradation rates of the reservoirs to determine the time of release pattern of active medicaments from each reservoir.
3D printing technologies with tunable drug release
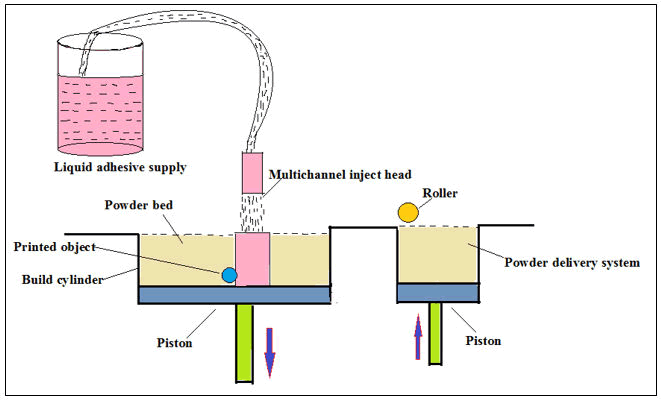
Three-dimensional (3D)-printed oral control drug delivery has reached the goal of providing a novel platform for drug delivery devices in the pharmaceutical industry and this type of technique is more beneficial for improving the effectiveness of personalized medicine (Figure 14). Fused deposition modeling, lithography-based techniques and power-bed inkjet techniques are generally used methods as are various types of 3D printed technologies that are designed or fabricated for polymeric modified sustained release oral dosage forms (Table 3).
Figure 14: Injection-based 3D printing technology.
| Fabrication methods | Merits | Restrictions |
| 3D printing of oral dosage form design by powder-bed inkjet | Relatively rapid and scalable producing Large entrapment capacity Multimaterial printing capability |
Brittle architecture with leakiness Low intention, high cost and not easy to use |
| Microfabrication technique of implantable microchip devices | Increase medicament bioavailability No burden and pain of repeated injection High magnification, instant and extensible production |
Invasive (requires surgeries) medicament outflow and biologically acceptable Issues. Relatively large and bulky devices need outside influence or external trigger and transfer unit/device. |
| 3D printing of oral dosage form design (Lithography) | High magnification, instant production. Multimaterials printing and producing vessels. |
Low medicament carrying capacity, risk of UV-induced scarring to medicament limited number of safe, biologically acceptable and printable photopolymers |
| FDM 3D printing of oral dosage form design | Inexpensive and accessible Producing complex. Multimaterials printing and vessels. |
Low medicament carrying capacity risk of UV-induced scarring to medicament limited number of safe, biologically acceptable, and printable photopolymers. |
Table 3: Fabrication methods of devices for adjustable medicament(s) release.
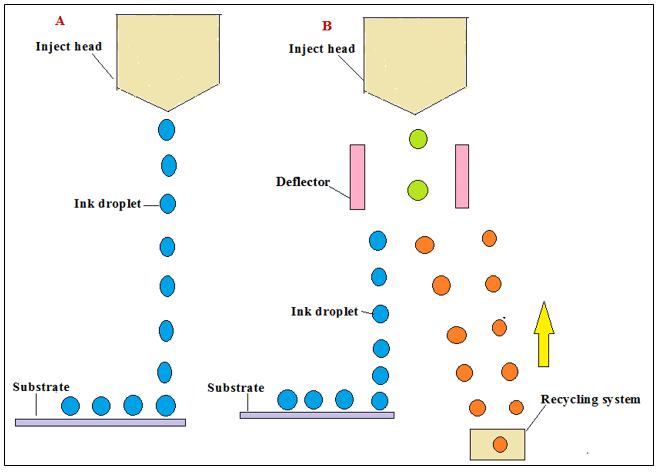
Jetting is another 3D part of novel technology where droplets are deposited on top and there are two types of jetting: material and binder jetting. The process where the building material droplets are deposited onto a foundation base is known as jetting of material. These material jetting methods are classified into two main categories of technology Dropping on Demand (DOD) and Continuously Injecting (CIJ).
The application of piezoelectric crystals (vapor bubbles) for increase in the voltage to large may be used for removing of input force upon forces the ink from the nozzle, the process known as DOD technique. In the CIJ technique upon the injection of charge droplets deflector plates deflect either away as waste products or onto the ground state and this process can be re-circulate (Figure 15).
Figure 15:(A) Drop on Demand (DoD); (B) Continuous Inkjet (CIJ).
Formulation of a sustained–release dosage via the use of natural herbal polymers
Numerous sustained release technologies such as water soluble or insoluble wax or polymer matrices, membrane-controlled systems and osmotic system matrices have been framed or designed. Drug delivery technology includes extended-release tablets with triple-layered, and a laser drilled-hole osmotic pump has been designed. For these novel formulations, sustained and modified control dosages of different excipients such as hydrophilic polymers must be incorporated during formulation which is very expensive. For this reason, herbal natural products normally serve as good alternatives for the fabrication of controlled or sustained release formulations. Because of their easy accessibility, low cost and eco-friendliness, the herbal materials are used as polymeric materials for designing matrix or layer tablets.
Researchers have been developing diclofenac sodium matrix tablets by using cashew gum and cross-linked cashew gum as controlled release polymers. Metformin hydrochloride matrix tablets were constructed by researchers by using hydrophilic synthetic and hydrophobic natural polymers for sustained–release tablets. Matrix tablets containing phytochemical constituents have been designed for a longer duration of action and beneficial for human healthcare.
Conclusion
Compared with customary dosage forms, controlled-release medication is widely accepted in the medical and pharmaceutical fields because of its modified dosage regimen and better bioavailability and therapeutic potency. Sustained action is beneficial for controlling the plateau stage concentration of medicament(s) in serum level followed by extending the duration of action. The modified release dose pattern is also influential and highly effective for palpitating and targeted action within the biological system. Medicament targeting at the desired location is much more effective in cancer chemotherapy and metastatic stage also.
The gastro retentive medicament delivery system is applicable for controlled release medication for patients suffering from chronic gastritis and ulceration as well as gut wall cancer. By minimizing the dosing frequency side effects and toxicity may subside during the treatment.
A wide range of medicament(s) can be used in GRDD with improved bioavailability and patient compliance. The parenteral repository system can influence extended release of the medicament at the serum level in prearranged technique via the use of biodegradable polymers, oily viscous solutions, multiple emulsions, liposomal and nanoparticle formulations and implantable medicament delivery technology.
Chrono therapeutic medication technology also influences medicament targeting by administration of active medicament(s) at specific sites in the disease state at appropriate times. Controlled API administration keeps the optimum medicament concentration within the range of minimum and maximum safety concentration which is essential according to the circadian rhythm of diseases. The use of pharmaceuticals coupled with nano medicines could improve medicament delivery technology which will be safer and more effective for therapeutic potency inhuman beings for future.
References
- Andrade C (2015) Sustained-release, extended-release, and other time-release formulations in neuropsychiatry. J Clin Psychiatry 76: e995-e999.
[Crossref] [Google Scholar] [PubMed]
- Sharma K, Thakur P, Agarwal S (2023) Formulation and evaluation of new sustained release floating microspheres of cilnidipine by solvent-diffusion evaporation technique. J Drug Deliv Ther 13: 102-111.
- Senapati S, Mahanta AK, Kumar S, Pralay Maiti P (2018) Controlled drug delivery vehicles for cancer treatment and their performance. Signal Transduct Target Ther 3: 1-19.
[Crossref] [Google Scholar] [PubMed]
- Adepu S, Ramakrishna S (2021) Controlled drug delivery systems: current status and future directions. Molecules 26: 5905.
[Crossref] [Google Scholar] [PubMed]
- Mankar SD, Shaikh SB (2020) Microencapsulation: Is an advance technique of drug formulation for novel drug delivery system. Res J Sci Tech 12: 201-210.
- Kumar D, Archana, Niranjan AK (2022) A comprehensive review on sustained release matrix drug delivery system. J Drug Deliv Ther 12: 249-253.
- Wang L, Liu X (2019) Sustained release technology and its application in environmental remediation: A review. Int J Environ Res Public Health 16: 2153.
[Crossref] [Google Scholar] [PubMed]
- Kottala N, Abebe A, Sprockel O, Bergum J (2012) Evaluation of the performance characteristics of bilayer tablets: part ii. impact of environmental conditions on the strength of bilayer tablets. AAPS Pharm Sci Tech 13: 1190-1196.
[Crossref] [Google Scholar] [PubMed]
- Devi D, Ghosh A, Mandal UK (2022) Sustained release matrix tablet of 100 mg losartan potassium: Formulation development and in vitro characterization. Braz J Pharm Sci 58: e20079.
- Wheless JW, Phelps SJ (2018) A clinician’s guide to oral extended-release drug delivery systems in epilepsy. J Pediatr Pharmacol Ther. 23: 277-292.
[Crossref] [Google Scholar] [PubMed]
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi