Short Communication, J Womens Health Issues Care Vol: 10 Issue: 8
The Corpus Luteum, A Source and Target of Androgen
Agata M. Parsons1*, Jason E. Bruemmer2, Fiona K. Hollinshead1, Astrid M. Iturbe Franzius1 and Gerrit J. Bouma1
1College of Veterinary Medicine and Biomedical Sciences, Colorado State University, Fort Collins, Colorado, 80523, USA
2USDA APHIS WS, National Wildlife Research Center, Fort Collins, Colorado 80523, USA
*Corresponding Author: Parsons AM College of Veterinary Medicine and Biomedical Sciences, Colorado State University, Fort Collins, Colorado, 80523, USA, E-mail: Agata.Parsons_Aubone@colostate.edu
Received date: July 22, 2021; Accepted date: August 05, 2021; Published date: August 12, 2021
Citation: Parsons AM, Bruemmer JE, Hollinshead FK, Franzius AMI, Bouma GJ (2021) The Corpus Luteum, A Source and Target of Androgen.J Womens Health, Issues Care 10:8.
Abstract
Normal physiological levels of androgens are important for female reproductive health. However, high levels of androgens, as seen in women with Polycystic Ovarian Syndrome (PCOS) (8%-13% of reproductive age women) [1] or gestational diabetes (incidence in US of 10%) [2] are associated with reproductive complications. Pathologies accompanying significantly elevated testosterone levels involve impaired ovulation and early pregnancy maintenance. Formation of the Corpus Luteum (CL) requires a normal ovulation process to occur. The CL plays a central role in the establishment and early maintenance of a pregnancy. Here we present a brief report on the function of the CL and our current understanding on the potential role and function of testosterone and the activated androgen receptor.
Keywords: Corpus luteum; Testosterone; Androgen receptor; Female fertility
Introduction
Androgens and fertility in women
Androgens are a necessary component of female reproductive physiology acting directly through their receptors or indirectly after aromatization into estrogens. Normal levels of androgens are necessary for optimal ovarian function, whereas an excess of androgen results in follicle arrest and anovulation [3]. The importance of proper levels of androgens is highlighted in women affected by Polycystic Ovarian Syndrome (PCOS), where an excess of androgen from ovaries is a key feature. Hyperandrogenism leads to anovulation, abnormal follicular development and reduced fertility [4]. Moreover, if pregnancy does occur, there is an increased risk of pregnancy complications, including intrauterine growth restriction and premature delivery. Alternatively, Female Androgen Insufficiency Syndrome [5], which is caused by a decline in testosterone as a result of ovarian, adrenal, or central dysfunction, not only decreases libido in women, but also is accompanied by a decrease in follicular development and fertility.
Androgens are synthesized and secreted by the ovaries and adrenal glands in women. In addition, testosterone precursors (e.g., DHEA) from these endocrine glands are converted into testosterone and other androgens by adipose tissue. Testosterone is the major androgen in women, and its action in target tissues are mediated through the androgen receptor (AR) or, after its aromatization to estradiol, through estrogen receptors. Androgen signaling is critical for normal female reproduction function. AR knockout mice exhibit impaired ovarian folliculogenesis, as androgens promote follicle growth and reduce granulosa cell apoptosis via their ARs [6]. Moreover, androgens control ovarian steroidogenesis downstream of FSH signaling by regulating the expression of key steroidogenic enzymes StAR, P450scc and 3β-HSD [7]. Finally, an increase in testosterone in women can suppress normal menstruation and ovulation [8], and thus potentially CL formation and adequate production of steroidal hormones such as progesterone, estrogen and androgens, which are essential for the establishment and maintenance of early pregnancy.
The CL as a source of steroids, including androgens
The CL is a temporary endocrine gland within the ovary and develops from the luteinization of theca and granulosa cells from the mature follicle following breakdown of the basal lamina immediately prior to rupture of the follicle during ovulation [9]. The process of luteinization is driven by luteinizing hormone (LH) which is secreted from the anterior pituitary gland. Theca and granulosa cells become small and large steroidogenic luteal cells, respectively. The main function of the CL is hormone synthesis, primarily progesterone, followed by estrogen and androgens, for establishment and maintenance of early pregnancy [10]. LH stimulates the synthesis of these hormones by acting through cyclic adenosine monophosphate (cAMP) regulating genes essential for hormone synthesis and luteal development [11]. During early human pregnancy, hCG, secreted by embryonic trophoblast cells, prevents the regression and stimulates the CL. Human luteal regression in the menstrual cycle is caused by a large reduction in the responsiveness of the aging CL to LH, which can be overcome by elevated concentrations of hCG during maternal recognition of pregnancy [12].
CL is essential for an adequate menstrual cycle and for the establishment and maintenance of early pregnancy. Defects in CL formation or function leads to an inability to produce and maintain adequate levels of progesterone and is clinically manifested by short menstrual cycles and infertility. CL pathologies associated with high levels of testosterone as seen in women with PCOS are associated with impaired angiogenesis in the ovary, including the CL. Compromised CL function may cause miscarriage by its inability to produce adequate levels of progesterone which has been reported in women with PCOS [13].
Steroid hormones play a local role in the formation, maintenance and regression of the CL [14,15]. Progesterone exerts its function on the CL through its nuclear progesterone receptor (PGR), membrane progestin receptors, and progesterone receptor membrane components (PGRMC) 1 and 2 [16]. Progesterone localizes to small and large luteal cells and promotes luteal development and sustains luteal structure-function during the menstrual cycle and early pregnancy [17]. Estrogen acts through its nuclear estrogen receptor (ESRs) 1 and 2, or a membrane G Protein–Coupled Estrogen Receptor 1 (GPER1) [18] and is also detected in small and large luteal cells. Studies in rats and rabbits showed that estradiol has a luteotropic effect, protecting the CL and promoting progesterone production [19].
The CL as a target of androgen
AR has been identified in pig and rat CL [20,21], as well as human CL [22]. In human CL, AR is localized to both large and small luteal cells. In addition, AR is present in CL from sheep, horse and dog (Figure 1), suggesting AR has a fundamental role in mammalian CL formation and/or function, and indicates that androgens target the CL.
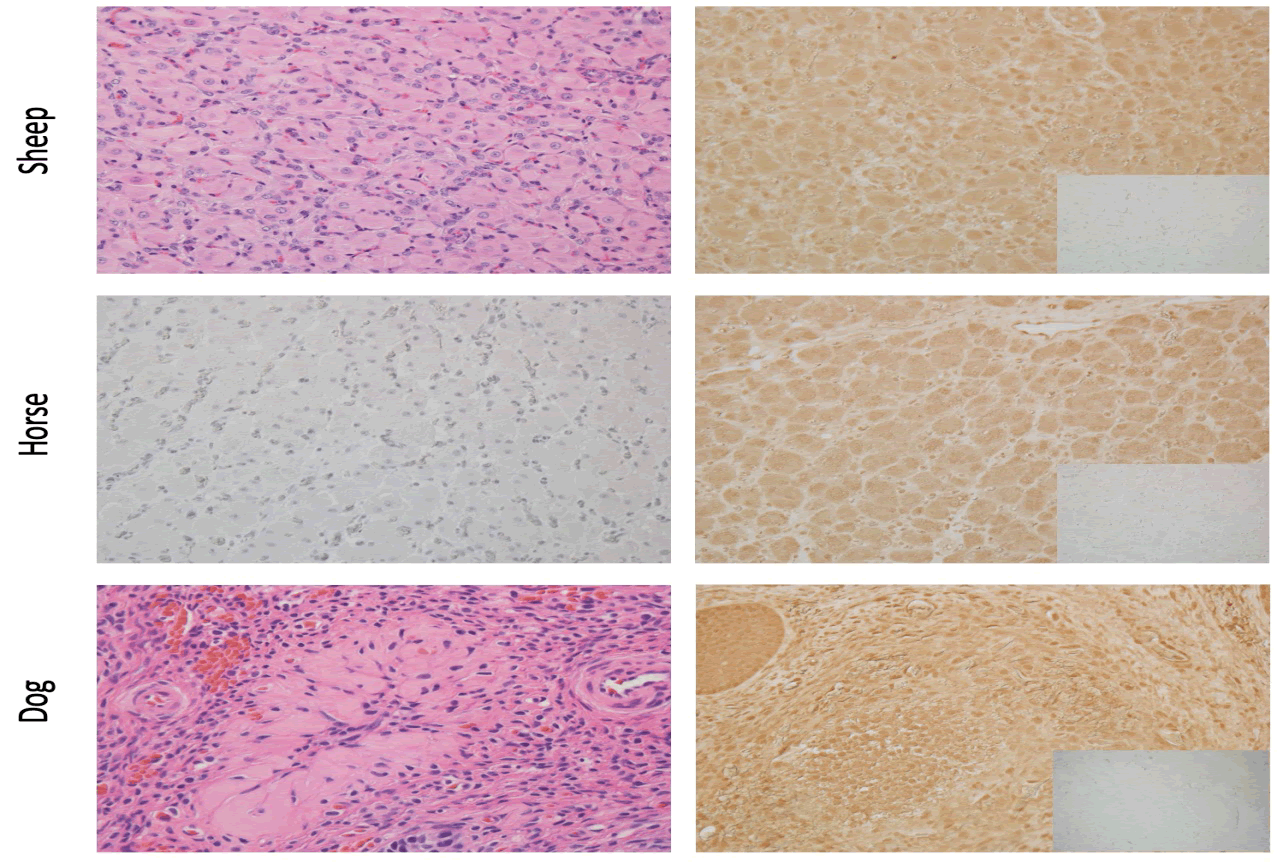
Figure 1: Androgen Receptor (AR) localization reveals AR in corpora lutea (CL) from sheep, horse and dog ovaries. Left panel indicates hematoxylin and eosin (sheep and dog), and hematoxylin (horse) staining of CL. Right panel indicates AR localization (brown staining) in CL by immunohistochemistry. Bottom right insert is a “no-primary antibody” negative control. All images were taken at 20 X.
AR signaling potentially stimulates progesterone production in the CL as androgen signaling through the AR is known to regulate steroidogenesis by regulating the expression of key steroidogenic enzymes [7]. Studies in human, ovine and bovine luteal cells revealed that dihydrotestosterone (DHT), inhibits progesterone secretion. The inhibitory effect of DHT on progesterone secretion possibly is due to decreased expression of StAR, P450scc, and/or 3βâ?HSD, through AR activation.
Discussion
Finally, women with PCOS present with CL insufficiency primarily due to defects in vascularization. Currently very little is known about the potential impact of AR regulation on vascularization in CL development or function. Vascularization is finely regulated by expression of angiogenic factors, including vascular endothelial growth factor, angiopoietins, fibroblast growth factor, platelet-derived growth factors and tumor necrosis factor. Vascular endothelial growth factor was consistently reported to be elevated in women with PCOS, and is associated with altered ovarian blood flow and vascularization [23]. Angiogenic factors like VEGFA are regulated by androgens binding to AR [24]. Therefore, regulation of angiogenesis by androgens and their receptors may explain the CL pathologies in cases of hyperandrogenism, and impaired development and function of the CL resulting in a shortening of the luteal phase, and subfertility due to early embryonic loss.
Conclusion
A greater understanding of androgens mechanism of action in/on the CL will further elucidate the pathophysiology of luteal function and failure in reproductive pathologies in women that are associated with altered levels of androgens, and potentially provide a way for novel therapies to be developed.
References
- Witchel SF, Teede HJ, Peña AS (2020) Curtailing PCOS. Pediatr Res 87(2):353-361.
- Lende M, Rijhsinghani A (2020) Gestational Diabetes: Overview with Emphasis on Medical Management. Int J Environ Res Public Health 17(24):9573.
- Astapova O, Minor BMN, Hammes SR (2019) Physiological and Pathological Androgen Actions in the Ovary. Endocrinology 160(5):1166-1174.
- Hart R (2008) PCOS and infertility. Panminerva Med 50(4):305-314.
- Bachmann G, Bancroft J, Braunstein G (2002) Female androgen insufficiency: The Princeton consensus statement on definition, classification, and assessment. Fertil Steril 77(4):660-665.
- Walters KA, Allan CM, Handelsman DJ (2008) Androgen actions and the ovary. Biol Reprod 78(3):380-389.
- Hasegawa T, Kamada Y, Hosoya T, Fujita S, Nishiyama Y, et al. (2017) A regulatory role of androgen in ovarian steroidogenesis by rat granulosa cells. J Steroid Biochem Mol Biol 172:160–165.
- Goodman NF, Cobin RH, Futterweit W (2015) American Association of Clinical Endocrinologists, American College of Endocrinology, and Androgen Excess and Pcos Society Disease State Clinical Review: Guide to the Best Practices in the Evaluation and Treatment of Polycystic Ovary Syndrome-Part 1. Endocr Pract 21(11):1291-1300.
- Stouffer R (1996) Corpus Luteum Formation and Demise. In: Adashi EY, Rock RA, Rosenwaks Z, editors. Reproductive Endocrinology, Surgery, and Technology. Ultrasound Obstet Gynecol 25(5): 498–507.
- Tavaniotou A, Albano C, Smitz J, Devroey P (2002) Impact of ovarian stimulation on corpus luteum function and embryonic implantation. J Reprod Immunol 55(2):123-130.
- Retamales I, Carrasco I, Troncoso JL, Las Heras J, Devoto L, et al. (1994) Morpho-functional study of human luteal cell subpopulations. Hum Reprod 9(4):591-596.
- Chen SU, Chen RJ, Shieh JY (2010) Human chorionic gonadotropin up-regulates expression of myeloid cell leukemia-1 protein in human granulosa-lutein cells: Implication of corpus luteum rescue and ovarian hyperstimulation syndrome. J Clin Endocrinol Metab 95(8):3982-3992.
- Patil K, Hinduja I, Mukherjee S (2021) Alteration in angiogenic potential of granulosa-lutein cells and follicular fluid contributes to luteal defects in polycystic ovary syndrome. Hum Reprod 36(4):1052-1064.
- Rosenfeld CS, Wagner JS, Roberts RM, Lubahn DB (2001) Intraovarian actions of estrogen. Reproduction 122:215–26.
- Peluso JJ (2006) Multiplicity of progesterone’s actions and receptors in the mammalian ovary. Biol Reprod 75:2–8.
- Kowalik MK, Rekawiecki R, Kotwica J (2013) The putative roles of nuclear and membrane-bound progesterone receptors in the female reproductive tract. Reprod Biol 13:279–89.
- Stouffer RL (2003) Progesterone as a mediator of gonadotrophin action in the corpus luteum: beyond steroidogenesis. Hum Reprod Update 9(2):99-117.
- Rosenfeld CS, Wagner JS, Roberts RM, Lubahn DB (2001) Intraovarian actions of oestrogen. Reproduction 122:215–226.
- Yuh KC, Keyes PL (1982) Relationships between estrogen receptor and estradiol-stimulated progesterone synthesis in the rabbit corpus luteum. Biol Reprod 27(5):1049-1054.
- Somczynska M, Szotys M, Duda M, Tabarowski Z, Sakiewicz A (2006) Androgen receptor in the reproductive systems of pregnant pigs and rats. Reprod Biol 6(Suppl. 1):113–118.
- Szotys M, Somczynska M, Galas J, Duda M, Sakiewicz A (2007) Expression of androgen receptor and 3beta-hydroxysteroid dehydrogenase in corpora lutea during pregnancy in the rat. Reprod Fertil Dev 19:356–65.
- Sasano, H, Suzuki, T (1997) Localization of steroidogenesis and steroid receptors in human corpus luteum. Classification of human corpus luteum (CL) into estrogen-producing degenerating CL, and nonsteroid-producing degenerating CL. Reprod Endocrinol 15:345–351.
- di Pietro M, Pascuali N, Parborell F, Abramovich D (2018) Ovarian angiogenesis in polycystic ovary syndrome. Reproduction 155(5):R199-R209.
- Cleys ER, Halleran JL, Enriquez VA (2015) Androgen receptor and histone lysine demethylases in ovine placenta. PLoS One 10(2):e0117-472.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi