Research Article, J Nephrol Ren Dis Vol: 1 Issue: 2
The Dynamics of Carnitine, γ-butyrobetaine and Trimethylamine N-oxide in Diabetics and the Effects of Changes in Renal Function
Susumu Ogawa*, Manami Shimizu, Kazuhiro Nako, Masashi Okamura and Sadayoshi Ito
Division of Nephrology, Endocrinology and Vascular Medicine, Tohoku University Hospital, Japan
*Corresponding Author : Susumu Ogawa MD, PhD
Division of Nephrology, Endocrinology and Vascular Medicine, Tohoku University Hospital, 1-1 Seiryomachi, Aoba-ku, Sendai, Japan
Tel: 81-22-717-7163
Fax: 81-22-717-7168
E-mail: ogawa-s@hosp.tohoku.ac.jp
Received: March 23, 2017 Accepted: March 25, 2017 Published: April 02, 2017
Citation: Ogawa S, Shimizu M, Nako K, Okamura M, Ito S (2017) The Dynamics of Carnitine, γ-butyrobetaine and Trimethylamine N-oxide in Diabetics and the Effects of Changes in Renal Function. J Nephrol Ren Dis 1:2.
Abstract
Objectives: In diabetic kidney disease, vascular disorders progress alongside loss of renal function, possibly due to higher blood concentrations of angiopathic substances, such as gamma-butyrobetaine (γBB), produced by gut microbiota that feed on carnitine in dietary red meat. Therefore, we established the hypothesis that urinary γBB excretion declines with decreasing renal function, causing a rise in blood γBB concentration, leading to aggravated vascular injury.
Methods: Our subjects were non-diabetics with normal renal function (N group) and diabetics with reduced renal function (D group). We measured blood concentration and urinary γBB excretion to compare the groups. We also compared blood γBB levels and urinary γBB excretions in diabetics with diabetic kidney disease (eGFR<30 mL/min/1.73m2, DKD group) with diabetics without DKD (eGFR>30 mL/min/m2, NDKD group).
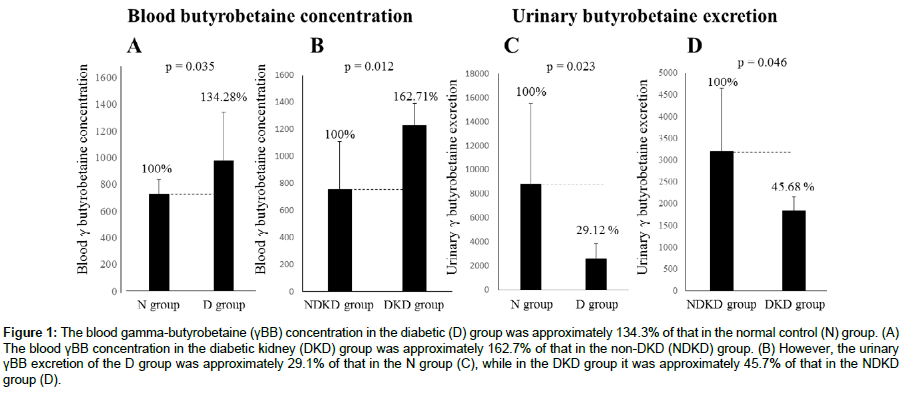
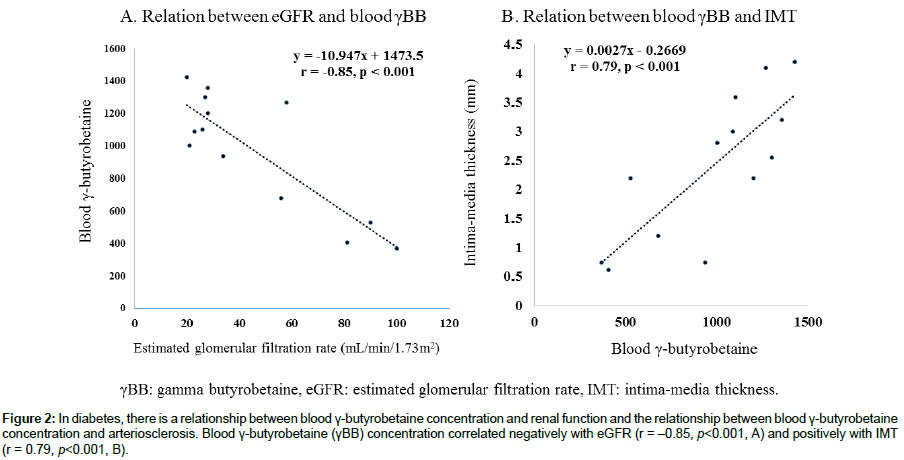
Results: Blood γBB concentration in the D group was approximately 134.3% compared with the N group. It correlated negatively with eGFR (r = –0.85, p<0.001) and positively with intima-media thickness (IMT, r = 0.79, p<0.001). Blood γBB concentration in the DKD group was approximately 162.7% compared with the NDKD group. However, urinary γBB excretion in the D group was approximately 29.1% compared with the N group, while the DKD group showed approximately 45.7% compared with the NDKD group.
Conclusions: Blood γBB concentration is higher in diabetics, which closely correlates with lower urinary excretion caused by renal hypo function. γBB, which damages blood vessels and aggravates vascular damage, may be implicated in the pathology of cardio renal correlation.
Keywords: γ-butyrobetaine; Trimethylamine N-oxide; Carnitine; Estimated glomerular filtration rate; Diabetic kidney disease; Intima-media thickness
Introduction
Vascular disorders progress alongside loss of renal function (cardio renal correlation), possibly due to higher blood concentrations of angiopathic substances. Gamma-butyrobetaine (γBB) is an angiopathic substance [1] produced by gut microbiota that feed on the carnitine present in dietary red meat. It is implicated in arteriosclerosis and long-term cardiovascular death [2]. Trimethylamine N-oxide (TMAO), its hepatic metabolite, is also linked to vascular disorders [3]. Our hypothesis is that urinary γBB excretion declines with decreasing renal function, causing a rise in its blood concentration and leading to the aggravation of vascular disorders. The dynamics of these substances in diabetes and the effects of changes in renal function have not yet been studied in detail. We investigated the dynamics of carnitine, γBB, and TMAO in the blood and urine of patients with diabetic kidney disease (DKD) and studied their relationships with renal function (eGFR) and intimamedia thickness (IMT).
Materials and Methods
Our subjects were non-diabetics with normal renal function (N group; n=9, eGFR=97.8 ± 15.6) and diabetics including individuals with reduced renal function (D group, n=13, eGFR=45.5 ± 28.4).
We measured their blood concentration and urinary excretion of carnitine, γBB, and TMAO for comparison between diabetics and non-diabetics. Carnitine, γBB and TMAO were measured using ultra performance liquid chromatography-tandem mass spectrometry based metabolome analysis [4]. The relative ratio of the detected peak area to that of the internal standard was used to eliminate systematic bias derived from injection volume variance and MS sensitivity.
We also divided the D group into individuals with reduced renal function (eGFR<30, DKD group; n=6, eGFR=24.7 ± 3.4) and without (eGFR>30, NDKD group; n=7, eGFR=69.3 ± 24.7). In the diabetic group, we examined the correlations between eGFR and IMT, as well as the concentration of γBB in the blood. The IMT was measured IMT, by the ATL Ultramark HDI 5000 Ultrasound System (Bothell).
A Kruskal–Wallis test and Dunn’s post-hoc test were used to assess the statistical significance of differences between D and N group samples. The Spearman’s rank correlation test was used to calculate correlations among IMT, eGFR, and the relative ratios of peak areas of the metabolites.
Results
No differences in blood carnitine concentration were seen between the D and N groups or between the DKD and NDKD groups. Urinary carnitine excretion was lower in the D group, but no differences were seen between the DKD and the NDKD groups. Blood TMAO concentration and urinary TMAO excretion were higher in the D group than the N group (137.5% and 239.3%, respectively), but no differences in urinary excretion were seen between the DKD and NDKD groups. Production of TMAO is higher in diabetics, but is not linked to low renal function. The blood γBB concentration in the D group was approximately 134.3% of that in the N group (Figure 1A). It correlated negatively with eGFR (r= –0.85, p<0.001) and positively with IMT (r=0.79, p<0.001) (Figure 2). The blood γBB concentration in the DKD group was approximately 162.7% of that in the NDKD group. However, the urinary γBB excretion of the D group was approximately 29.1% of that of the N group, while in the DKD group, it was approximately 45.7% of that in the NDKD group (Figure 1B-1D).
Figure 1: The blood gamma-butyrobetaine (γBB) concentration in the diabetic (D) group was approximately 134.3% of that in the normal control (N) group. (A) The blood γBB concentration in the diabetic kidney (DKD) group was approximately 162.7% of that in the non-DKD (NDKD) group. (B) However, the urinary γBB excretion of the D group was approximately 29.1% of that in the N group (C), while in the DKD group it was approximately 45.7% of that in the NDKD group (D).
Figure 2: In diabetes, there is a relationship between blood γ-butyrobetaine concentration and renal function and the relationship between blood γ-butyrobetaine concentration and arteriosclerosis. Blood γ-butyrobetaine (γBB) concentration correlated negatively with eGFR (r = –0.85, p<0.001, A) and positively with IMT (r = 0.79, p<0.001, B).
Conclusions
Blood γBB concentration is higher in diabetics. In addition to the factor of increased γBB synthesis from carnitine or of decreased carnitine synthesis from γBB (catalyzed by γBB deoxygenase), this rise is closely associated with lower urinary excretion caused by renal hypo function. However, urinary γBB excretion did not correlate to blood γBB concentration. This shows that metabolizing and excreting γBB are complex, although a small sample size is one potential factor.
Urinary γBB excretion decreases with falling renal function, so its blood concentration rises. It is possible that γBB, which damages blood vessels and aggravates vascular damage, may be implicated in the pathology of cardio renal correlation. The increase of blood TMAO concentration in the D group was not due to a decrease in urinary excretion. The generation of TMAO seemed to increase in diabetes. Although both γBB and TMAO are angiopathic substances, the mechanisms causing their increases are different and these dynamics in diabetics are very complex. All of the "uremic toxins" - small molecules that accumulate with impairment in renal function, similarly will correlate tightly with eGFR, and with IMT measures. The γBB might be one of such small molecules.
This research is preliminary and has a very small sample size. So we cannot confirm even the independence of γBB and IMT vs eGFR. Therefore, a larger-scale clinical study is necessary.
Highlights
The blood concentrations of gamma-butyrobetaine (γBB) and Trimethylamine N-oxide (TMAO) in a diabetic group are higher than those in a non-diabetic group.
Urinary γBB excretion declines with decreasing renal function, causing a rise in blood γBB concentration, leading to aggravation of vascular injury.
An increase of blood TMAO concentration in the diabetic group was not due to a decrease in urinary excretion.
Acknowledgement
We wish to thank the nurses of Tohoku University Hospital for their assistance with gathering the blood and urine samples for this study and for their efforts in gathering patient’s data. Our deep appreciation goes to all staff members who helped us in this study.
Sources of Funding
This work was supported by Tohoku University's Center for the Advancement of Higher Education President's Research Fund.
References
- Ussher JR, Lopaschuk GD, Arduini A (2013) Gut microbiota metabolism of L-carnitine and cardiovascular risk. Atherosclerosis 231: 456-461.
- Skagen K, Trøseid M, Ueland T, Holm S, Abbas A, et al. (2016) The Carnitine-butyrobetaine-trimethylamine-N-oxide pathway and its association with cardiovascular mortality in patients with carotid atherosclerosis. Atherosclerosis 247: 64-69.
- Koeth RA, Wang Z, Levison BS, Buffa JA, Org E, et al. (2013) Intestinal microbiota metabolism of L-carnitine, a nutrient in red meat, promotes atherosclerosis. Nat Med 19: 576-585.
- Hirayama A, Nakashima E, Sugimoto M, Akiyama S, Sato W, et al. (2012) Metabolic profiling reveals new serum biomarkers for differentiating diabetic nephropathy. Anal Bioanal Chem 404: 3101-3109.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi