Research Article, Endocrinol Diabetes Res Vol: 4 Issue: 2
The Influence of Cortisol and Age Differences on the Development of Insulin Resistance and Dyslipidemia in Euthyroid Patients
Abdelgayoum A Abdel-Gayoum*
Department of Clinical Laboratory Sciences, Faculty of Applied Medical Sciences, University of Hail, Hail, Saudi Arabia
*Corresponding Author : AA Abdel-Gayoum
Department of Clinical Laboratory sciences, Faculty of Applied Medical Sciences, University of Hail, Hail, Saudi Arabia
Tel: +966565714884
E-mail: aabdelgayoum@hotmail.com
Received: January 29, 2018 Accepted: July 25, 2018 Published: July 27, 2018
Citation: Abdel-Gayoum AA (2018) The Influence of Cortisol and Age Differences on the Development of Insulin Resistance and Dyslipidemia in Euthyroid Patients. Endocrinol Diabetes Res 4:2. doi: 10.4172/2470-7570.1000130
Abstract
Objectives: The present study was planned to investigate the influence of serum cortisol and age differences on insulin resistance and the associated lipid disturbances in euthyroid adults.
Methods: Hyperglycemic, hyperinsulinemic patients were recruited, and classified according to their insulin resistance (HOMA-IR) into: Normal insulin sensitivity (NIS), moderate- (MIR), high insulin resistance (HIR) with HOMA-IR<4, 4.1 to 6, and >6, respectively. The patients were also classified into three age-range groups: young age (YA), middle age (MA), Old age (OA); with age ranges <30, 31-40 and >40 yrs., respectively. Fasting serum was used for analysis.
Results: The serum cortisol of HIR was higher than NIS by 17.64%, and insulin higher than NIS and MIR by 272% and 121%, respectively. On the other hand, serum glucose of OA was higher than YA by 28.87%, whereas, insulin was lower than YA and MA by 52.08%, and 31.10%. Similarly, HOMA-IR was lower than YA and MA by 45.97% and 24.11%, respectively. There was significant correlation between serum cortisol and HOMA-IR (R=0.59, P=0.02) at ages> 45 years. Serum triglyceride in HIR was higher than NIS and MIR by 38.59% and 18.79%, and VLDL by 116% and 20.37%, respectively, whereas, HDL was lower than NIS by 11.38%.
Conclusion: Insulin resistant patients had elevated serum cortisol and insulin. There was significant correlation between cortisol and insulin resistance at older ages. The serum cortisol increased with age whereas insulin level decreased. Patients with high insulin resistance had dyslipidemia characterized by high serum TG, high VLDL-c and low HDL-c.
Keywords: Serum insulin; Cortisol; Insulin resistance; Age difference; Euthyroid; Dyslipidemia
Introduction
Insulin resistance is a metabolic disorder believed to play a major role in the pathogenesis of the metabolic syndrome, obesity, Type 2 diabetes and cardiovascular disease [1]. It is characterized with fasting hyperglycemia and hyperinsulinemia. Several factors are known to enhance the development of insulin resistance including thyroid disorder and excess serum cortisol levels. Cortisol is a steroid hormone that regulates glucose metabolism. It promotes gluconeogenesis [2]; inhibits pancreatic insulin secretion [3] and reduces insulin sensitivity in the adipose tissue and muscle [4]. Thus, the consequence of cortisol excess is the development of insulin resistance [5], characterized by disturbance of glucose and lipid metabolism and development of hypertension, all believed to be independent risk factors of cardiovascular diseases [6,7]. The prevalence of insulin resistance is known to be higher among patients with Cushing’s syndrome, and even repeated treatment with glucocorticoids may cause impairment of insulin sensitivity [8]. It was reported that insulin resistance may be influenced by age, degree of obesity and serum cortisol levels [9]. Despite the inverse relationship between age and insulin sensitivity [10]. It is debatable whether age is a primary determinant of the insulin resistance or a consequence of the aging related disorders [11,12]. In view of the factors influencing the pathogenesis of the insulin resistance, the present study was aimed to investigate the interrelated influences of fasting serum cortisol levels and age differences on the fasting serum insulin level and the status of insulin resistance and the associated changes in the lipid profiles in euthyroid adults.
Patients and Methods
Protocol of study: The study was conducted on patients with hyperglycemia and elevated fasting insulin levels visiting the Diabetic and Endocrinology Out-Patient Unit of King Khalid Hospital, Hail, Saudi Arabia between April 2016 and Sept. 2016. The patients clinical data entered in the record book of the laboratory were used in this study. A total of 78 newly diagnosed hyperglycemic and hyperinsulinemic patients were recruited for the study. Depending on their fasting serum glucose and insulin levels, the homeostasis model assessment –Insulin resistance (HOMA-IR) index was calculated by the method described by Mathew et al. [13]. The patients with values higher than 4.0 were classified as insulin resistant. All patients had an age range of 14 -77 years (mean age was 34.47 ± 11.20 years). A group of 38 subjects (M=9, F=29) with normal glycaemia and insulin sensitivity (HOMA-IR ≤ 4.0) were used as control (NIS) group. According to their HOMA-IR values the insulin resistant patients were classified into: Moderate Insulin Resistance (MIR) group: n= 35 patients (M=10, F=25) with HOMA-IR between 4.1 to 6; and High Insulin Resistance (HIR) group: n= 43, (M=20, F=23) with HOMA-IR > 6.0. The patients with thyroid disorders and patients under insulin or other hypoglycemic medications were excluded. 5 ml venous blood was collected from each patient after an overnight fast and serum was separated and used for the biochemical analysis. The protocol of experiment was explained and the consent was obtained from the participants. The study was approved by the ethical committee, Faculty of Applied Medical Sciences, University of Hail, Hail, Saudi Arabia. To study the influence of age on insulin resistance and cortisol secretion the patients were classified depending on age into three agerange groups: young age (YA) (n=52; M=21, F= 31), middle age (MA) (n=41; M=12, F= 29) and Old age (OA) (n= 23; M=4, F=19); with age ranges < 30, 31-40 and > 40 years, respectively.
Biochemical assays
The serum freeT3, freeT4, TSH, insulin and cortisol levels were assayed by Auto analyzer (ELecsys 2010, Cobas E 411-Mannheim Germany. The serum glucose, total cholesterol (TC), triglyceride (TG), high density lipoprotein-cholesterol (HDL-c), were measured by the automated spectrophotometer, Hitachi-717, utilizing commercial kits supplied by Roche Diagnostic, United Kingdom. The low density lipoprotein-cholesterol (LDL-c) and very low density lipoprotein (VLDL) were calculated by the Friedman equation. The estimated insulin sensitivity (QUICK-I) was calculated as described by Katz et al, [14].
Statistical analysis
The presented data are means ± SD. The significance of differences between the means was computed by one way analysis of variance, followed by Multiple Comparison Analysis. Spearman’s regression analysis was used to study the significance of correlation among the serum cortisol, HOMA-IR, serum insulin and glucose. P value less than 0.05 was considered.
Results
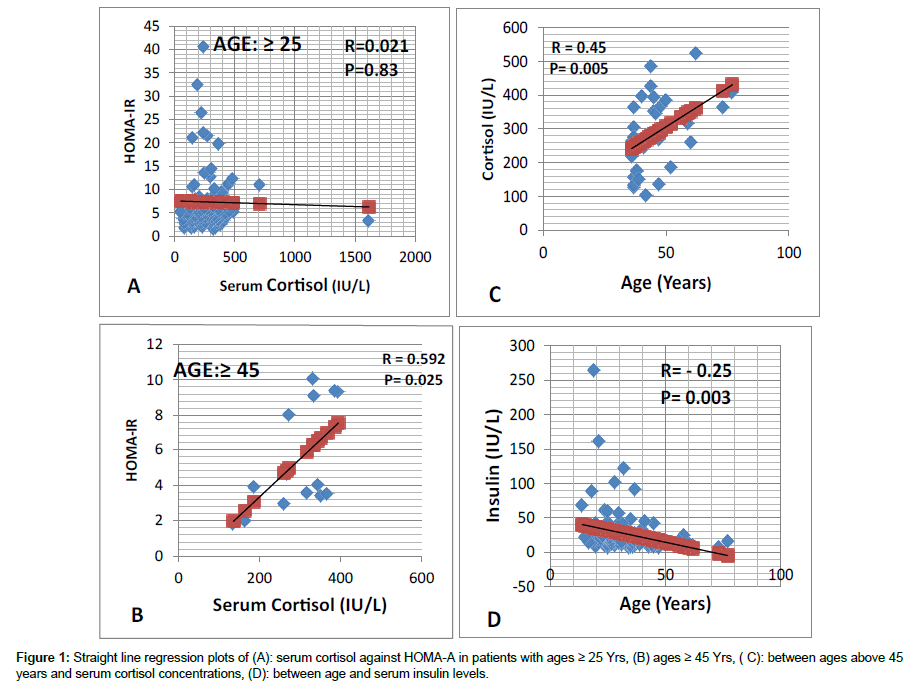
Table 1 summarizes the serum values of glucose, insulin, cortisol, thyroid hormones and the estimated insulin resistance in the three experimental groups. The mean ages in the three groups were not significantly different from each other. The fasting serum glucose concentration in the MIR group was slightly raised compared with NIS, whereas, that of the HIR group was significantly (P<0.001) higher compared with that of NIS and MIR by 63.90% and 53.80%, respectively. On the other hand, the serum insulin concentration in the MIR group was significantly higher than that of the NIS by 68.08%, whereas, that of HIR was significantly higher than NIS and MIR groups by 272% and 121%, respectively. Similarly, the mean HOMAIR of the MIR group was higher than NIS by 73.41%, whereas, that of HIR was higher than that of NIS and MIR by 430%, and 209%, respectively. On the other hand, the IR-beta showed a significant elevation in the MIR by 53.86 % compared with NIS, whereas, that of HIR was higher than that of NIS and MIR by 1.06-fold and 34.12 %, respectively. However, the serum cortisol level of the MIR was slightly elevated, whereas, that of the HIR was significantly higher than that of NIS by 17.64 %. On the other hand, the thyroid function parameters did not show significant differences in any of the groups and all parameters were within normal reference ranges. Table 2 depicts the serum glucose, insulin, cortisol, HOMA-IR in patients classified into three age (young, middle and old) groups. The fasting serum glucose of the old-age group was significantly higher than that of young -age group by 28.87% and not significantly different from the mid-age group. Whereas, the serum insulin concentration of the old group was significantly lower than that of the Young-age and Mid-age groups by 52.08%, and by 31.10 %, respectively. On the other hand, HOMA-IR of the old-age group was significantly lower than the young- and middle-age groups by 45.97% and 24.11%, respectively. Whereas, that of Middle-age group was lower than that of the younggroup by 18.80%. However, the serum cortisol level of the old group was significantly higher than that of the Young-and Mid-age groups by 14.61%, and 27.41%, respectively. Table 3 summarizes the lipid profile in the sera of the three insulin resistance levels groups. No significant differences were observed in serum total cholesterol levels in any of the three groups. However, the serum triglyceride level in the HIR group was significantly elevated by 38.59% and 18.79% compared with the NIS and MIR groups, respectively, whereas, that of the MIR group was higher than that of the NIS by 16.66%. On the other hand, the HDL-cholesterol level of the HIR group was significantly (P<0.05) lower than that of the NIS group by 11.38%. However, the serum LDL-cholesterol levels did not exhibit significant differences in any of the three groups, whereas, the VLDL level of the HIR group was significantly elevated compared to NIS and MIR groups by 116 % and 20.37%, respectively. On the other hand, that of MIR was higher than NIS by 80.00%. As shown in Figure 1 (A, B, C, D) there was no significant correlation between serum cortisol and HOMA-IR (R=0.02, p= 0.83) when all patients with ages 25 years and above were included. However, exclusion of younger ages (below 30, 35, 40, or 45 years) rendered the correlation between serum cortisol and HOMA concomitantly higher (R=0.14, 0.35, 0.49 and 0.59, respectively). Similarly, exclusion of young ages (below 45) resulted in a significant positive correlation between age and cortisol (R=0.45, p=0.005) whereas, the correlation between age and serum insulin levels exhibited a significant negative correlation, and did not require exclusion of the young ages (R=- 0.25, p=0.003).
| NIS (IR< 4) | MIR (IR= 4.1- 6) | HIR (IR> 6) | |
|---|---|---|---|
| Age (Years) | 39.25 ± 5.36 | 30.65 ± 4.72 | 31.00 ± 4.07 |
| Glucose(mmol/l) | 5.79 ± 0.64 | 6.17 ± 0.70 | 9.49 ± 1.98 a¥b¥ |
| Insulin(mIU/L) | 11.25 ± 2.44 | 18.91 ± 3.60 a¥ | 41.89 ± 3.31 a¥b¥ |
| HOMA-IR | 2.85 ± 0.45 | 5.00 ± 0.54 a** | 16.02 ± 1.02 a¥b¥ |
| Cortisol (IU/L) | 269.25 ± 13.36 | 291.97 ± 15.85 | 316.76 ± 17.46 a** |
| TSH( mU/L) | 2.16 ± 0.31 | 2.17 ± 0.30 | 2.23 ± 0.33 |
| T4 ( pmol/L) | 13.81 ± 1.02 | 15.02 ± 2.33 | 14.36 ± 2.06 |
| T3( pmol/L) | 7.32 ± 1.02 | 8.55 ± 1.52 | 10.12 ± 1.33 |
Table 1: The fasting serum glucose, insulin, cortisol, insulin resistance (HOMA-IR) and thyroid function parameters in subjects with normal insulin sensitivity (NIS), patients with moderate (MIR) and high insulin resistance (HIR). Presented data are mean ± SD. * P< 0.05, ** P< 0.01, ¥ P< 0.001. (a): significantly different from NIS; (b): significantly different from MIR.
| YA (≤ 30Yrs) | MA (31 - 40Yrs) | OA (> 40 Yrs) | |
|---|---|---|---|
| Age (Yrs) | 24.51 ± 3.21 | 34.89± 5.11 | 52.27± 5.61 |
| Serum Glucose (mmol/L) | 6.51 ± 1.44 | 7.77 ± 2.04 | 8.39 ± 1.83 a** |
| Serum Insulin (mIU/L) | 31.43 ± 3.64 | 21.86 ± 2.71 a** | 15.06 ± 2.24 a¥b¥ |
| HOMA-IR | 10.31 ± 2.35 | 7.34 ± 1.21 a* | 5.57 ± 1.05 a¥b* |
| Serum Cortisol (IU/L) | 288.70 ± 13.32 | 259.70± 10.52 | 330.90 ± 12.22 a¥b¥ |
Table 2: The fasting serum glucose, insulin, cortisol, insulin resistance in euthyroid subjects classified into different age groups.
| NIS | MIR | HIR | |
|---|---|---|---|
| Serum TC (mmol/L) | 4.86 ± 1.02 | 4.83 ± 0.98 | 4.81 ± 1.12 |
| Serum TG(mmol/L) | 1.14 ± 0.12 | 1.33 ± 0.09a* | 1.58 ± 0.11 a¥b* |
| HDL-cholesterol (mmol/L) | 1.23 ± 0.05 | 1.16 ± 0.06 | 1.09 ± 0.08a* |
| LDL- cholesterol (mmol/L) | 3.33 ± 0.21 | 3.13 ±0.36 | 3.07 ± 0.26 |
| VLDL-cholesterol (mmol/L) | 0.30 ± 0.04 | 0.54 ± 0.06 a** | 0.65 ± 0.04 a¥b* |
Table 3: Serum total cholesterol (TC), triglyceride (TG)and lipoprotein profiles in subjects with normal insulin sensitivity (NIS), moderate insulin resistance (MIR) and high insulin resistance (HIR).
Discussion
Insulin resistance is a metabolic disorder believed to play a major role in the pathogenesis of the metabolic syndrome, obesity, Type 2 diabetes and cardiovascular disease [1]. Excess glucocorticoids are known to be among various factors involved in the development of diabetes [15]. In the present study the group with high fasting insulin and glucose levels and with highest estimated insulin resistance exhibited the highest serum cortisol levels. This finding was in congruence with the reports that revealed elevated serum cortisol levels in hypertensive patients with high fasting glucose and insulin resistance [16,17]. Glucocorticoids are known to promote gluconeogenesis by inducing the expression of the hepatic gluconeogenic genes and suppress the cellular uptake of glucose by inhibiting the membrane glucose transporter-4 [16]. Moreover, investigators have found an inverse relationship between the rate cortisol clearance from circulation and the body insulin sensitivity [18] and repeated treatment with glucocorticoids was shown to induce hyperinsulinemia and hyperglycemia suggesting a role for cortisol in the pathogenesis of insulin resistance [19]. Interestingly however, the present results exhibited that the correlation between serum cortisol and insulin resistance was significant only when the younger patients with ages below 25 years were excluded and became highly significant when only patients with ages older than 45 years were considered. In a study conducted on Chinese elderly population it was indicated that the morning serum cortisol levels in the elderly individuals were significantly higher than that of their younger counterparts [20]. This suggests that the influence of cortisol as an underlying factor for insulin resistance becomes more prominent at older ages. Moreover, the increase in age was accompanied with increase in the serum cortisol concentrations and a concomitant decrease in the insulin levels. This finding suggests that as the individuals grew older the pancreatic insulin secretion dropped gradually with concomitant increase in the cortisol levels playing the major role in the development of hyperglycemia. This was also found by Kamba et al. [21]. who showed that higher serum cortisol levels were significantly associated with decreased insulin secretion in the physiological cortisol range of Japanese population. However, the estimated insulin resistance also gradually dropped with age due to the reduction in insulin levels, but it did not fall below 5.0. Some authors reported that postprandial glucose remained elevated for longer time in nondiabetic elderly adults than in younger ones, suggesting an age-related decline in insulin sensitivity and glucose tolerance [22]. In response to cortisol-induced peripheral insulin resistance and in an attempt to maintain euglycaemia, the pancreatic β-cells undergo several morphological and functional adaptations that result in hyperinsulinaemia, whereas, failure of the β-cells to compensate leads to disruption of glucose homeostasis ending up in hyperglycemia. In experiments with cultured ß-cells of pancreas it was shown that glucocorticoids suppressed the insulin secretion [23]. Moreover, it was shown that the glucose-induced insulin secretion was decreased in animals with over expressed glucocorticoid receptors in the ß-cells of pancreas [24,25]. Suggesting that glucocorticoids may directly suppress the ß-cells function independent of their induction of insulin resistance. It was reported that several abnormalities including amyloid deposition occurred in the ß-cells and insulin secretion dropped in elderly people beside their decreased insulin sensitivity towards insulin tropic factors [26]. Glucocorticoids were also shown to stimulate adipocytes lipolysis through activation of hormone sensitive lipase enzyme [27]. Causing elevation of the circulating free fatty acids that enhances the development of insulin resistance [28,29].
The present results also indicated that patients with high insulin resistance had high levels of serum TG levels with unaltered serum cholesterol concentrations. This was in accordance with the reports of Miccoli et al., [30]. who observed elevation of serum TG, reduction of HDL-cholesterol, and increased small dense LDL with normal LDLcholesterol in patients with impaired insulin activity. It is believed that insulin stimulates the uptake of free fatty acids and TG by liver and adipose tissue and enhances lipogenesis. Thus, when the adipose tissue loses insulin sensitivity, the circulating TG levels increase concomitantly [31]. The elevated serum TG levels were suggested to be due to increased hepatic secretion of VLDL and impaired clearance of chylomicrons and VLDL with increased fatty acid flux from adipose tissue [32]. In the presence of increased level of circulating VLDL the cholesterol-ester transfer protein (CETP) catalyzes the exchange of the VLDL- TG for LDL- and HDL-cholesterol ending up with TG-enriched LDL. These modified LDL particles are gradually catabolized by the hepatic lipase converting them into the atherogenic small dens LDL particles [30]. Moreover, these insulin resistant patients have reduced post heparin lipoprotein lipase activity due to increased apoprotein CIII /CII ratio that results in increased circulating chylomicrons and VLDL with reduced expression of LDL receptors that delays removal of chylomicrons remnant and LDL from circulation [33,34]. This probably explains our findings of unaltered LDL-cholesterol levels with significantly elevated TG concentrations. The Hypertriglyceridemia seems to be the effects of both impaired insulin action and the high serum cortisol levels. In support to this assumption, Prodam and coworkers [17]. observed a strong positive correlation between the serum cortisol levels and serum triglycerides, fasting glucose and insulin resistance. However, in contrast to the impairment of insulin resistance to the lipoprotein lipase activity, elevated serum cortisol was shown to enhance its activity that may enhance the catabolism lipoprotein TG and release of free fatty acids into circulation [35,36]. These two opposing effects of cortisol and the insulin resistance on the lipoprotein lipase activity seem to determine the extent of hypertriglyceridemia observed in these patients. Moreover, insulin resistance was shown to enhance the secretion of apoprotein B48 causing increased synthesis of chylomicrons [36]. Indeed, all these events lead to the accumulation of postprandial TGrich lipoproteins in circulation.
Conclusion
Euthyroid patients with insulin resistance had elevated serum cortisol levels. The serum cortisol was showed significant association with insulin resistance only at older ages. The serum cortisol levels increased with age whereas the serum insulin level decreased. Patients with high insulin resistance had dyslipidemia characterized by high serum TG, high VLDL and low HDL-cholesterol levels with unaltered total and LDL-cholesterol concentrations.
Acknowledgement
The author is grateful to Mr. Naif Falah and Zaid AlDosari in the Clinical Laboratory at King Khaled Hospital for their assistance in biochemical analysis and data collection. This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Conflict of interest
The author declares that no conflict of interest exists.
References
- Farag A, Karam J, Nicasio J, McFarlane SI (2007) Prevention of Type 2 diabetes: an update. Curr.Diab.Rep 7: 200-207.
- Vander Kooi BT, Onuma H, Oeser JK, Svitek CA, Allen SR et al. (2005) The glucose-6-phosphatase catalytic subunit gene promoter contains both positive and negative glucocorticoid response elements. Mol Endocrinol 19: 3001-3022.
- Davani B, Khan A, Hult M, Martensson E, Okret S, et al. (2000) Type 1 11beta-hydroxysteroid dehydrogenase mediates glucocorticoid activation and insulinrelease in pancreatic islets. J Biol Chem 275: 34841-34844.
- Qi D, Rodrigues B (2007) Glucocorticoids produce whole body insulin resistancewith changes in cardiac metabolism. Am J PhysiolEndocrinolMetab 292: 654-667.
- Pyorala M, Miettinen H, Halonen P, Laakso M, Pyorala K (2000) Insulin resistance syndrome predicts the risk of coronary heart disease and stroke in healthy middle-aged men: the 22-year follow-up results of the Helsinki Policemen Study. Arterioscler ThrombVasc Biol 20: 538-544.
- De Onis M, Blossner M, Borghi E (2010) Global prevalence and trends of overweight and obesity among preschool children.ract Am J ClinNutr 92: 1257-1264.
- Nadeau KJ, MaahsDM, Daniels SR, Eckel RH (2011) Childhood obesity and cardiovascular disease: links and prevention strategies. Nat Rev Cardiol 8: 513-525.
- Isidori AM, Graziadio C, Paragliola RM, Cozzolino A, Ambrogio AG, et al. (2015) The hypertension of Cushing's syndrome: controversies in the pathophysiology and focus on cardiovascular complications. J Hypertens 33: 44-60.
- Sinha R, Fisch G, Teague B, TamborlaneWV, Banyas B (2002) Prevalence of impaired glucose tolerance among children and adolescents with marked obesity. N Eng J Med 346: 802-810.
- Facchini FS, Hua N, Abbasi F, Reaven GM (2001) Insulin resistance as a predictor of age-related diseases. J Clin Endocrinol Metab 86: 3574-3578.
- Basu R, Breda E, Oberg AL, Powell CC, Dalla Man C, et al. (2003) Mechanisms of the age-associated deterioration in glucose tolerance: contribution of alterations in insulin secretion, action, and clearance. Diabetes 52: 1738-1748.
- Matthews DR, Hosker JP, Rudenski AS, Naylor BA, Treacher DF, et al. (1985) Homeostasis model assessment: insulin resistance and β-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 28:412-419.
- Katz A, Nambi SS, Mather K, Baron AD, Follmann DA, et al. (2000) Quantitative insulin sensitivity check index: a simple, accurate method for assessing insulin sensitivity in humans. J Clin Endocrinol Metab 85: 2402-2410.
- De Guia RM, Rose AJ, Herzig S (2014) Glucocorticoid hormones and energy homeostasis. Horm Mol Biol Clin Investig 19:117-128.
- Rafacho A, Ortsater H, Nadal A, Quesada I (2014) Glucocorticoid treatment and endocrine pancreas function: implications for glucose homeostasis, insulin resistance and diabetes. J Endocrinol 223: 49-62.
- Prodam F, Ricotti R, Agarla V, Parlamento S, Genoni G, et al. (2013) High-end normal adrenocorticotropic hormone and cortisol levels are associated with specific cardiovascular risk factors in pediatric obesity: a cross-sectional study. BMC Medicine 11: 44.
- Holt HB, Wild SH, Postle AD, Zhang J, Koster G, et al (2007) Cortisol clearance and associations with insulin sensitivity, body fat and fatty liver inmiddle-aged men. Diabetologia 50: 1024-1032.
- Bahr V, Pfeiffer AF, Diederich S. (2002) The metabolic syndrome X and peripheral cortisol synthesis. Exp Clin Endocrinol Diabetes 110: 313-318.
- Zhao Z-Y, Lu F-H, Xie Y, Fu Y-R, Bogdan A, et al. (2003) Cortisol secretion in the elderly. Influence of age, sex and cardiovascular disease in a Chinese population. Steroids. 68: 551-555.
- Kamba A, Daimon M, Murakami H, OtakaH, Matsuki K, et al. (2016) Associationbetween Higher Serum Cortisol Levels andDecreased Insulin Secretion in a GeneralPopulation. PLoS ONE.
- Short KR, Bigelow ML, Kahl J, Singh R, Coenen-Schimke J, et al. (2005) Decline in skeletal muscle mitochondrial function with aging in humans. Proc Natl Acad Sci USA 102: 5618-5623.
- Lambillotte C, Gilon P, Henquin JC. (1997) Direct glucocorticoid inhibition of insulin secretion. An in vitro study of dexamethasone effects in mouse islets. J Clin Invest 99: 414-423.
- Delaunay F, Khan A, Cintra A, Davani B, Ling ZC, et al. (1997) Pancreatic beta cells are important targets for the diabetogenic effects of glucocorticoids. J Clin Invest 100: 2094-2098.
- Blondeau B, Sahly I, Massouridès E, Singh-Estivalet A, Valtat B, et al. (2012) Novel transgenic mice for inducible gene overexpression in pancreatic cells define glucocorticoid receptor-mediated regulations of beta cells. PLoS One.
- Scheen AJ (2005) Diabetes mellitus in the elderly: insulin resistance and/or impaired insulin secretion? Diabetes Metab 31: 5S27-5S34.
- Djurhuus CB, Gravholt CH, Nielsen S, Mengel A, Christiansen JS, et al. (2002) Effects of cortisol on lipolysis and regional interstitial glycerol levels in humans. Am J Physiol Endocrinol Metab 283: E172-E177.
- Gao Z, Zhang X, Zuberi A, Hwang D, Quon MJ, et al. (2004) Inhibition of insulin sensitivity by free fatty acids requires activation of multiple serine kinases in 3T3-L1 adipocytes. Mol Endocrinol. 18: 2024-2034.
- Geer EB, Islam J, Buettner C (2014) Mechanisms of Glucocorticoid-Induced Insulin Resistance. Focus on Adipose Tissue Function and Lipid Metabolism. Endocrinol Metab Clin North Am 43: 75-102.
- Miccoli R, Bianchi C, Penno G, Del Prato S (2008) Insulin Resistance and Lipid Disorders. Future Lipidology 3: 651-664.
- Jianping Ye (2013) Mechanisms of insulin resistance in obesity. Front Med 7: 14-24.
- Ginsberg HN, Zhang YL, Hernandez-Ono A (2005) Regulation of plasma triglycerides in insulin resistance and diabetes. Arch Med.Res 36: 232-240.
- Ginsberg HN, Zhang YL, Hernandez-Ono A (2006) Metabolic syndrome: focus on dyslipidemia. Obesity 14: 41-49.
- Cohn JS, Patterson BW, Uffelman KD, Davignon J, Steiner G (2004) Rate of production of plasma and very-low-density lipoprotein (VLDL) apolipoprotein C-III is strongly related to the concentration and level of production of VLDL triglyceride in male subjects with different body weights and levels of insulin sensitivity. J. Clin. Endocrinol. Metab 89: 3949-3955.
- Krsek M, Rosicka M, Nedvidkova J, Kvasnickova H, Hana V, et al. (2006) Increased lipolysis of subcutaneous abdominal adipose tissue and altered noradrenergic activity in patients with Cushing’s syndrome: an in-vivo microdialysis study. Physiol Res 55: 421-428.
- Shulman GI (2014) Ectopic fat in insulin resistance, dyslipidemia, and cardiometabolic disease. N Engl J Med 371: 1131-1141.
- Haidari M, Leung N, Mahbub F, Uffelman KD, Kohen-Avramoglu R, et al. (2002) Fasting and postprandial overproduction of intestinally derived lipoproteins in an animal model of insulin resistance. Evidence that chronic fructose feeding in the hamster is accompanied by enhanced intestinal de novo lipogenesis and ApoB48-containing lipoprotein overproduction. J Biol Chem 277: 31646-31655.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi