Short Communication, Biomater Med Appl Vol: 1 Issue: 1
Thermal Analysis of Two Niobo-Phosphate Glasses
TJS Oliveira, EPL Junior, AS Paula, MO Domingos, D Navarro da Rocha and MH Prado da Silva*
Military Institute of Engineering-IME, Materials Science Post Grade Programme, Pça. Gen. Tiburcio, 80, Praia Vermelha, Urca, Rio de Janeiro, Brazil
*Corresponding Author : MH Prado da Silva
Military Institute of Engineering-IME, Materials Science Post Grade Programme, Brazil
E-mail: marceloprado@ime.eb.br
Received: September 13, 2017 Accepted: October 10, 2017 Published: October 16, 2017
Citation: Oliveira TJS, Junior EPL, Paula AS, Domingos OM, Rocha DN, et al. (2017) Thermal Analysis of Two Niobo-Phosphate Glasses. Biomater Med Appl 1:1.
Abstract
In the present study, two new biocompatible glasses, with the same components, but in different proportions, were produced.The thermal behavior of both glasses was analyzed and the glass transition temperatures (Tg) were determined by differential scanning calorimetry (DSC) for both glasses. The DSC analyses showed that the bioactive glass with Ca/P = 1.68 showed higher Tg than the bioactive glass with Ca/P = 1.33.
Keywords: Niobo-phosphate; Glass; Glass transition; Thermal analysis
Introduction
Studies on the synthesis of phosphate glasses have been reported from different authors, mainly due to their effect on chemical stability [1-3]. Different applications such as in electronic components [4-6], as well as on sealing systems [7], lasers [8] and biomedical materials [9-11] have been reported.
Niobo-phosphate glasses designed to be biocompatible have been reported by several authors [11-13]. In previous studies, a CaO-P2O5-Nb2O5-CaF2 glass was developed and tested in vitro [12,13]. However, transition temperature of this new glass remained undetermined. In the present study, a new biocompatible glass, with the same components, but in different proportions, was produced. The thermal behavior of both bioactive glasses was analyzed and the glass transition temperatures (Tg) were determined by differential scanning calorimetry (DSC) for both glasses.
Experimental
Two niobo-phosphate glasses were produced by mixing different amounts of Nb2O5, H3PO4, CaCO3 and CaF2. The mixtures were melted at 1350°C and cooled into ultrapure water. The designed glasses compositions are shown on Table 1.
| Sample | CaO (mol%) | CaF2(mol%) | P2O5(mol%) | Nb2O5(mol%) |
|---|---|---|---|---|
| BG1 | 50.1 | 12.5 | 18.6 | 18.8 |
| BG2 | 20.0 | 20.0 | 30.0 | 30.0 |
Table 1: Glasses compositions.
The produced glasses were milled in a ball mill Marconi MA350/E, using alumina balls. The obtained powder was analyzed by DSC and X-ray Diffraction (XRD). The DSC analyses were performed in a differential scanning calorimeter DSC 404F1 Pegasus (Netzsch) in a heating rate of 20ºC/min from 30ºC to 500ºC, and above 500ºC up to 1000ºC with 5 and 10ºC/min. The analyses were realized with Pt-Rh crucibles and under argon atmosphere. The results were treated with Netzsch Proteus software.
Structural characterization was performed by X-ray diffractometry in a Panalytical X’Pert Pro diffractometer with CoKα at 40 kV, 45 mA with an iron (Fe) filter. The scan was made in 0.02° step from 10° to 100°. The results were treated with the software X’Pert HighScore Plus.
Results and Discussion
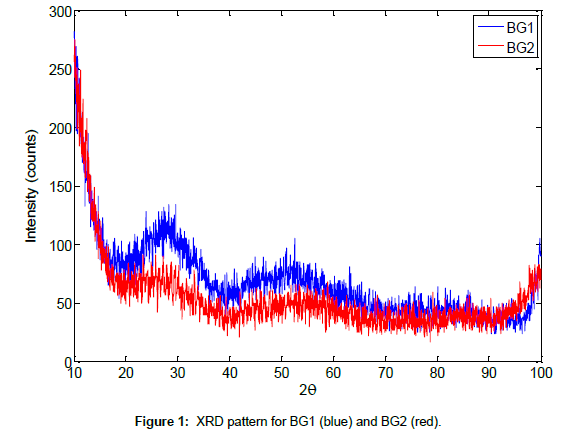
The XRD patterns of both glasses are shown in Figure 1. An amorphous band was observed between 20º < 2θ < 40º, confirming that both samples are strongly amorphous. Another band was observed between 40º < 2θ < 60º, indicating the presence of low-range crystallinity.
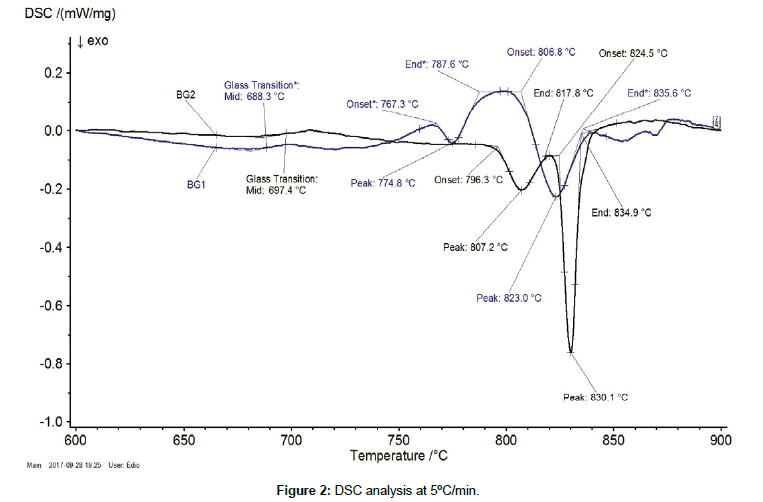
Figures 2 and 3 show the DSC analysis, for both glasses, with distinct heating rates (5 a 10ºC/min) in interest (5 and 10°C/min). Theses runs were performed in order to confirm if the variation in heat flow between 650ºC and 750ºC was a Tg and exothermic peaks associated to phase transformation at high temperature range. Figure 2 and Table 2 show that Tg = 688.3ºC for BG1 and Tg = 697.4ºC for BG2 at 5°C/min.
| TG | Peak1 | Peak2 | |||||
|---|---|---|---|---|---|---|---|
| Sample | start | Peak | finish | start | Peak | finish | |
| Heating rate: 5ºC/min | |||||||
| BG1 | 688.3 | 767.3 | 774.8 | 787.6 | 806.8 | 823.0 | 835.6 |
| BG2 | 697.4 | 796.3 | 807.2 | 817.8 | 824.5 | 830.1 | 834.9 |
| Heating rate: 10ºC/min | |||||||
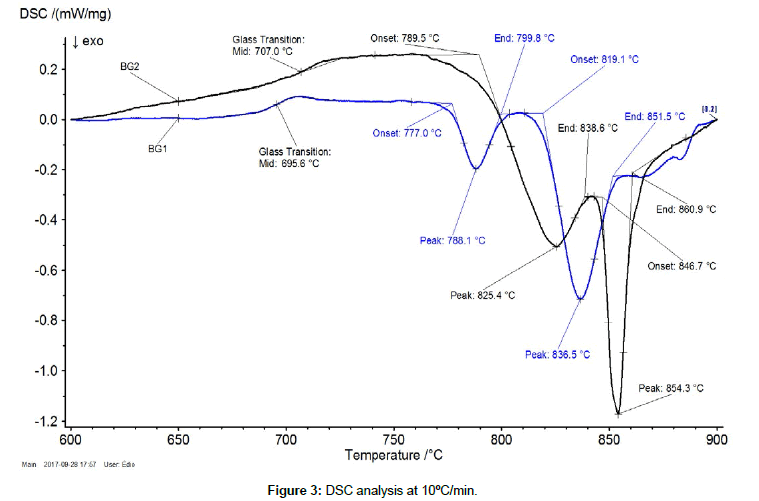
| BG1 | 695.6 | 777.0 | 788.1 | 799.8 | 819.1 | 836.5 | 851.5 |
| BG2 | 707.0 | 789.5 | 825.4 | 838.6 | 846.7 | 854.3 | 860.9 |
Table 2: Summary of DSC results for Glasses 1 and 2.
It is conventional to adopt the Tg values from DSC analyses with heating rate of 10°C/min. Figure 3 and Table 2 show the DSC results for both glasses under heating rate of 10°C/min. The glass transition temperature for BG1 was found to be Tg = 695.6°C and, for BG2, Tg = 707.0ºC. Glass 1 has higher glass modifier content than glass 2, what explains the Tg reduction. In addition, BG2 has higher Nb2O5 content than BG1. Sene et al. reported a rise in the Tg of niobophsophate glasses with increasing Nb2O5 content. This was explained in the basis of Nb-O-Nb and Nb-O-P being stronger than P-O-Nb bondings, and thus requiring higher relaxation temperatures.
Above Tg temperature, both glasses exhibit crystalline structure and revealed distinct peaks and phase transformation temperature. Glass 1 DSC curve exhibit four exothermic peaks, where the first two peaks are related with more significative energy associated the peak area. However, Glass 2 DSC curve exhibits only the first two exothermic peaks associated with highest temperature transformation and apparently higher peak area for the first peak, when compared to Glass 1 curve. The probable phases, according to glasses compositions, are calcium phosphates and calcium niobates.
Conclusion
Glass 1 showed lower Tg than glass 2. Additionally, glass 1 shows Ca/P = 1.68. This ratio is very close to that of hydroxyapatite (Ca/P = 1.67), and this is a promising feature regarding bioactivity. Both glasses can be converted to glass ceramics after heat treatments between 750ºC and 900ºC.
References
- Sales BC, Boatner LA (1986) Physical and chemical characteristics of lead-iron phosphate nuclear waste glasses. J Non-Cryst Solids 79: 83-116.
- Peng YB, Day DE (1991) High thermal expansion phosphate glasses. Glass Technol 32:166-173.
- Metwalli E, Karabulut M, Sidebottom DL, Morsi MM, Brow RK (2004) Properties and structure of copper ultraphosphate glasses. J Non Cryst Solids 344: 128-134.
- Fuxi G (1990) New glass-forming systems and their practical application. J Non-Cryst Solids 123:385-399.
- Zhong HJ, Chen GH, Cui SC, Chen JS, Yang Y, et al. (2015) Luminescence and energy transfer of Tm/Tb/Mn tri-doped phosphate glass for white light-emitting diodes. J Mater Sci: Mater Electron 26: 8130-8135.
- AEL KJ, Rabardel L, Couzi M, Mansouri I, Flem GLE (1993) Glass Formation in the Na2O-TiO2-P2O5 System. J Solid State Chem 102: 400-407.
- Brown RK, Tallant DR (1997) Structural Design of Sealing Glasses. J Non Cryst Solids 222: 396-406.
- Sardar DK, Gruber JB, Zandi B, Hutchinson JA, Trussell CW (2003) Judd-Ofelt analysis of the Er3+ (4f11) absorption intensities in phosphate glass: Er3+, Yb3. J Appl Phys 93: 2041-2046.
- Franks K, Salih V, Knowles JC, Olsen I (2002) The effect of MgO on the solubility behavior and cell proliferation in a quaternary soluble phosphate based glass system. J Mater Sci: Mater Med 13: 549-556.
- Prado da Silva MH, Lemos AF, Ferreira JM, Lopes MA, Santos JD (2002) Production of porous biomaterials based on glass-reinforced hydroxyapatite composites. Key Eng Mater 230-232: 483-486.
- Carvalho CN, Martinelli JR, Bauer J, Haapasalo M, Shen Y, et al. (2015) Micropush-out dentine bond strength of a new gutta-percha and niobium phosphate glass composite. Int Endod J 48: 451-459.
- Prado da Silva MH, Ramirez CM, Granjeiro JM, Rossi AM (2008) In vitro assessment of new Niobium Phosphate Glasses and Glass Ceramics. Key Eng Mat 361-363: 229-232.
- Fernandes GVO, Alves GG, Linhares ABR, Prado da Silva MH (2012) Evaluation of Cytocompatibility of Bioglass-Niobium Granules with Human Primary Osteoblasts: A Multiparametric Approach. Key Engineering Materials 493-494: 37-42.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi