Research Article, J Chem Appl Chem Eng Vol: 1 Issue: 1
Thermal Decomposition Studies on Iron(III) Complexes of 1,2-Bis(imino-4’-antipyrinyl) ethane with Varying Counter Ions
Nayathuparambil Thomas Madhu1* Esther Theresa Knittl2, Kibret Fekadu1, Hagos Feleke1, Kassa Abuyea1, Endalkachew Asefa1 and Wolfgang Linert2
1Department of Chemistry, University of Hawassa, Sidamo, Ethiopia
2Institute of Applied Synthetic Chemistry, Vienna University of Technology, Getreidemarkt, Vienna, Austria
*Corresponding Author : Nayathuparambil Thomas Madhu
Department of Chemistry, University of Hawassa, Sidamo, P.O.Box.05, Ethiopia
Tel: ++43-1-58801-15350
Fax: ++43-1-5816668-16299
E-mail: ntmadhu@hotmail.com
Received: August 28, 2017 Accepted: September 14, 2017 Published: September 21, 2017
Citation: Madhu NT, Knittl ET, Fekadu K, Feleke H, Abuyea K, et al. (2017) Thermal Decomposition Studies on Iron(III) Complexes of 1,2-Bis(imino-4’- antipyrinyl) ethane with Varying Counter Ions. J Chem Appl Chem Eng 1:1 doi: 10.4172/2576-3954.1000103
Abstract
Phenomenological and mechanistic characteristics of the thermal decomposition of perchlorate, nitrate, thiocyanate, chloride and bromide iron(III) complexes with the Schiff base 1,2-bis(imino-4’- antipyrinyl)ethane (GA) were studied by thermo gravimetric and derivative thermo gravimetric analyses. It was observed that the counter anions may have a major effect on the thermal stability of the present complexes.The decomposition reaction mechanism is in accordance with a ‘random nucleation with one nucleus on each particle, i.e. the Mampel model’.
Keywords: Iron(III) complexes; 1,2-Bis(imino-4’-antipyrinyl)ethane; Thermal decomposition; Mechanistic aspects
Introduction
A plethora of references describing metal complexes of Schiff bases have appeared in the literature during the past few decades [1-4] and some of them are very relevant as models for biological systems [5]. However, only little is known about the thermal behaviour of such complexes [6,7]. In relation to this and as the thermal studies of Schiff base-metal complexes is a part of our continuing interest [8-12], here, the phenomenological and mechanistic characteristics of five iron(III) complexes of an antipyrine Schiff base derivative, namely 1,2-bis(imino-4’-antipyrinyl)ethane (GA), having the empirical formula C24H24N6O2 with varying counter anions (perchlorate, nitrate, thiocyanate,chloride and bromide), are reported.
Experimental
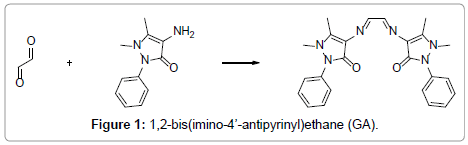
The ligand (GA) and the iron(III) complexes were prepared and analyzed as previously reported [2]. The ligand (GA) was prepared by a simple condensation reaction between glyoxal and 4-aminoantipyrine as starting materials (Figure 1). Thermogravimetric analysis was carried out on a Delta Series TGA 7 thermal analyzer under a nitrogen atmosphere using 10 mg of each sample. The heating rate was 10°C min-1 for each measurement. The mechanistic aspects of the thermal decomposition of the complexes were evaluated using a computer program in QBasic.
Results and Discussion
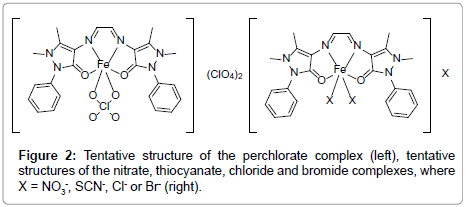
From the analytical and spectroscopic data [2], the five synthesised complexes can be formulated as [Fe(GA)(ClO4)](ClO4)2 and [Fe(GA) X2]X (where X = -NO3, -SCN, -Cl or -Br). In all complexes, GA acts as a neutral tetradentate ligand, which coordinates via both carbonyl oxygens and both azomethine nitrogen atoms. According to the molar conductance and the data from infrared spectra, it can be assumed that one of the perchlorate and two of the nitrate counter ions are coordinated in a bidentate and monodentate fashion in the respective complexes. In the thiocyanate complex, two of the counter anions are coordinated in a monodentate way, while in the halide complexes, two of the halides are coordinated. A spin free octahedral geometry can be assumed for all the synthesised complexes according to the electronic and magnetic moment studies (Figure 2). The TG and DTG curves of all five complexes are shown in the Figures 3-7.
Phenomenological aspects
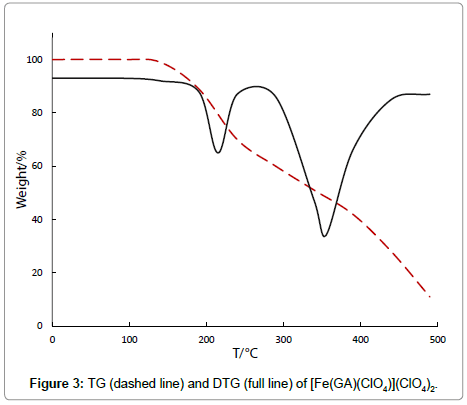
The phenomenological description of the TGA-curves of the present complexes is presented in Table 1. The perchlorate complex, [Fe(GA)(ClO4)](ClO4)2, has a two stage thermal decomposition process in the range of 146-482°C. It is stable without any mass loss up to 146°C, confirming the absence of little molecules like water or solvent in the complex. In the first stage of the decomposition (146°C-220°C) a mass loss (24.52%) is observed because of the conversion of three perchlorate anions into chlorides. The presence of chloride and the absence of perchlorate after the first stage are confirmed by the chemical analysis and the infrared spectrum, which were measured for the intermediate compound. The corresponding derivative thermo gravimetric peak is observed at 197°C for this stage. A mass loss of 54.72% can be observed in the range of 220°C-482°C, corresponds to the removal of the entire GA molecule with a DTG peak at 369°C. Qualitative analysis confirmed the final residue to be ferric chloride.
| Complex | Stage of decomposition | TG plateau (°C) | DTG peak (°C) | mass loss (%) |
|---|---|---|---|---|
| [Fe(GA)(ClO4)](ClO4)2 | I | 146-220 | 197 | 24.52 |
| II | 220-482 | 369 | 54.72 | |
| [Fe(GA)(NO3)2]NO3 | I | 142-264 | 192 | 27.00 |
| II | 264-497 | 325 | 31.00 | |
| III | 497-910 | 583 | 31.50 | |
| [Fe(GA)(SCN)2]SCN | I | 190-313 | 303 | 31.00 |
| II | 313-515 | 453 | 25.87 | |
| III | 515-801 | 636 | 32.02 | |
| [Fe(GA)Cl2]Cl | I | 211-400 | 307 | 36.01 |
| II | 400-874 | 637 | 36.17 | |
| [Fe(GA)Br2]Br | I | 240-408 | 350 | 29.10 |
| II | 408-856 | 620 | 29.00 |
Table 1: Phenomenological data of the thermal decomposition of the iron(III) complexes of GA.
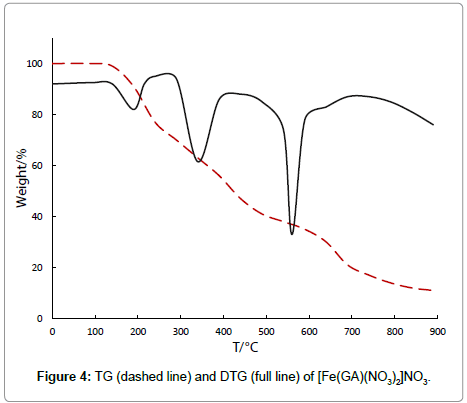
The nitrate complex, [Fe(GA)(NO3)2]NO3, decomposes in three stages. It is stable till 142°C revealing the absence of either water or solvent molecules in this complex. The thermal decomposition between 142°C-264°C in the first stage with a derivative thermogram at 192°C,which reveals a mass loss of 27%, can be assigned to the removal of three nitrate anions. The infrared spectrum of this intermediate residue after the first stage shows the absence of nitrate anions. The second stage (264°C-497°C), with a DTG peak at 325°C shows a mass loss of 31%, which is attributed to the removal of half of the ligand molecule [11], which is confirmed by an IR spectrum of the intermediate at 497°C. The third stage, which reveals a mass loss of 31.50%, between 497°C-910°C with a derivative thermo gravimetric peak at 583°C, can be assigned to the removal of the remaining ligand molecule, generating metal oxide as proved by qualitative analysis.
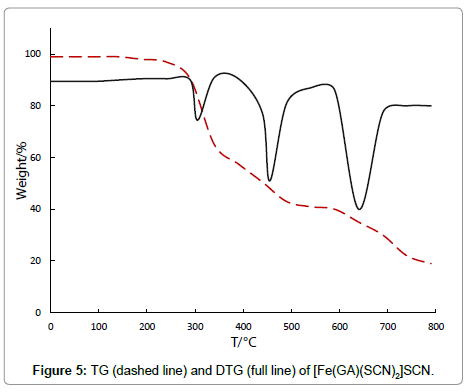
The thiocyanate complex, [Fe(GA)(SCN)2]SCN, undergoes a three stage decomposition pattern. The complex is stable up to 190°C indicating that there is neither water nor solvent molecules in the complex. The first decomposition stage (190°C-313°C) with a DTG peak at 303°C and a mass loss of 31%, which corresponds to the removal of half of the ligand molecule. The IR spectrum of the residue at 313°C shows the presence of parts of GA at this stage. The thermal decomposition between 313°C-515°C in the second stage with a derivative thermogram at 453°C reveals a mass loss of 25.87%, which happens to appear due to the decomposition of three thiocyanate anions. This observation is confirmed by the absence of thiocyanate ions in the intermediate compound obtained at 515°C, in IR spectra. The third stage (515-801°C) with the DTG peak at 636°C and a mass loss of 32.02% corresponds to the remaining half of GA. The decomposition gets completed at 801°C and the final residue is qualitatively proved to be anhydrous metal oxide.
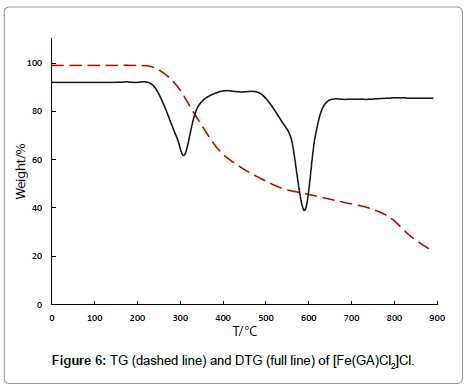
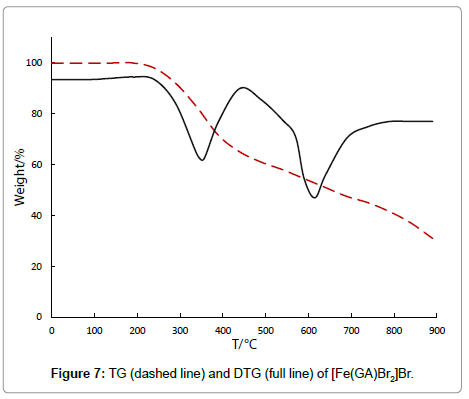
The chloride complex ester [Fe(GA)Cl2]Cl, decomposes in two stages and is stable till 211°C, revealing the absence of either water or solvent molecules in this complex. In the first decomposition process (211°C- 400°C), with a derivative thermogravimetric peak at 307°C, the observed mass loss of 36.01% corresponds to the removal of half of the ligand molecule. The IR spectrum of the residue after this stage shows the presence of parts of GA, indicating only partial removal of the ligand at this stage. The second stage (400°C-874°C) with a DTG peak at 637°C accounts to a mass loss of 36.17%, which can be attributed to the removal of the remaining half of the ligand molecule, yielding anhydrous ferric chloride as final product, which was confirmed by qualitative analysis. [Fe(GA)Br2]Br undergoes a two stage thermal decomposition. It is stable without any mass loss till 240°C, proving that there is neither water nor solvent in the complex. In the first stage of the decomposition (240°C-408°C), a DTG peak at 350°C appears with a mass loss of 29.10%, which happens to be due to the decomposition of half of the ligand molecule. The infrared spectrum of the residue after this stage shows the presence of GA, indicating only partial removal of the ligand at this stage. The thermal decomposition between 408°C-856°C in the second stage with a derivative thermogram at 620°C shows a mass loss of 29%. The decomposition of three thiocyanate anions happens in this stage. The second stage of the decomposition starts at 408°C and comes to an end at 856°C with the DTG peak at 620°C, regarding to the expulsion of the remaining half of the ligand molecule, yielding anhydrous ferric bromide as the final residue, proved by qualitative tests.
Structure and thermal stability correlation
From the phenomenological aspects, it has been clear that, even if the composition of all five complexes had been the same, there would have been a clear variation in their decomposition pattern. A three stage thermolytic pattern is observed for the thiocyanate and nitrate complexes, while the rest of the complexes decompose in two stages. Among the five complexes, the complex with nitrate as a counter ion is the least stable and the one with bromide as a counter ion is the most stable. The variation in the thermal stability proves that the counter anions play a major role on the thermal decomposition nature of the complexes [8-12], which can be brought in the following order: Bromide > Chloride > Thiocyanate > Perchlorate > Nitrate
Mechanistic aspects
All well-defined stages were selected to clearly state the thermal decomposition mechanism. The different g(α) forms are assumed to depend on the thermal decomposition mechanism as suggested by Šatava [13]. The correlation coefficient for all these nine forms were calculated (Table 2) and the form of g(α), for which the correlation has a maximum value, is chosen to be the mechanism of the reaction. Considering the above mentioned thermal decomposition reactions, the highest value of the correlation coefficient obtained for all welldefined stages corresponds to the “Mampel model” [7-12], which is
| No | Form of g(α) | Correlation coefficient (r) | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Perchlorate complex | Nitrate complex | Thiocyanate complex | Chloride complex | Bromide complex | |||||||||
| Stage I | Stage II | Stage I | Stage II | Stage III | Stage I | Stage II | Stage III | Stage I | Stage II | Stage I | Stage II | ||
| 1 | α2 | -0.9908 | -0.9843 | -0.9793 | -0.9852 | -0.9582 | -0.9696 | -0.9828 | -0.9823 | -0.9821 | -0.9733 | -0.9903 | -0.9950 |
| 2 | α+(1-α)ln(1-α) | -0.9954 | -0.9920 | -0.9884 | -0.9930 | -0.9756 | -0.9908 | -0.9959 | -0.9797 | -0.9906 | -0.9865 | -0.9955 | -0.9929 |
| 3 | [1-(1-α)1/3]2 | -0.9877 | -0.9563 | -0.9667 | -0.9620 | -0.9155 | -0.9175 | -0.9726 | -0.9765 | -0.9664 | -0.7830 | -0.9822 | -0.9087 |
| 4 | [1-(2/3)α]-(1-α)2/3 | -0.9968 | -0.9944 | -0.9917 | -0.9954 | -0.9828 | -0.9937 | -0.9891 | -0.9942 | -0.9935 | -0.9909 | -0.9971 | -0.9952 |
| 5 | -ln(1-α) | -0.9990 | -0.9990 | -0.9991 | -0.9993 | -0.9991 | -0.9993 | -0.9990 | -0.9992 | -0.9994 | -0.9990 | -0.9995 | -0.9991 |
| 6 | [-ln(1-α)]1/2 | -0.9988 | -0.9975 | -0.9985 | -0.9987 | -0.9982 | -0.9991 | -0.9984 | -0.9990 | -0.9988 | -0.9947 | -0.9994 | -0.9951 |
| 7 | [-ln(1-α)]1/3 | -0.9985 | -0.9924 | -0.9976 | -0.9967 | -0.9961 | -0.9990 | -0.9989 | -0.9984 | -0.9989 | -0.8654 | -0.9993 | -0.8582 |
| 8 | 1-(1-α)1/2 | -0.9970 | -0.9930 | -0.9917 | -0.9947 | -0.9815 | -0.9895 | -0.9911 | -0.9883 | -0.9932 | -0.9854 | -0.9971 | -0.9921 |
| 9 | 1-(1-α)1/3 | -0.9983 | -0.9962 | -0.9954 | -0.9976 | -0.9906 | -0.9935 | -0.9761 | -0.9947 | -0.9965 | -0.9927 | -0.9986 | -0.9929 |
Table 2: Correlation coefficients calculated using nine forms of g() for iron(III) complexes of GA.
g(α) = -ln(1–α)
Acknowledgement
We are grateful to Prof. E. Hallervorder for discussions and to the SRCE for financial support.
References
- Nayathuparambil TM, Pochayil KR (2000) Cobalt(II) complexes of 1,2-(diimino-4′-antipyrinyl)ethane and 4-N-(4′-antipyrylmethylidene)aminoantipyrine. Trans Met Chem 25: 287-292.
- Nayathuparambil TM, Pochayil KR (2001) Iron (III) complexes of 1,2-di(imino-4′-antipyrinyl)ethane. Synth React Inorg Met Org Chem 31: 1663-1673.
- Madhu NT, Tang JK, Hewitt IJ, Cle´rac R, Wernsdorfer W, et al. (2005) What makes a single molecule magnet? Polyhedron 24: 2864-2869.
- Lan Y, Novitchi G, Cle´racR, Tang JK, Madhu NT, et al. (2009) Di-, tetra- and hexanuclear iron(III), manganese(II/III) and copper(II) complexes of Schiff-base ligands derived from 6-substituted-2-formylphenols. Dalton Trans 38: 1721-1727.
- Hewitt IJ, Tang JK, Madhu NT, Cle´rac R, Buth G, et al. (2006) A series of new structural models for the OEC in photosystem II. Chem Comm 42: 2650-2652.
- Nath M (1991) Thermal studies on cobalt(II), nickel(II) and copper(II) complexes of Schiff base derived from salicylaldehyde and glycine. Themochim Acta 185: 11-24.
- Vinodkumar CR, Nair MKM, Radhakrishnan PK (2000) Thermal studies on lanthanide nitrate complexes of 4-n-(2′-furfurylidene)aminoantipyrine. J Therm Anal Cal 61: 143-149.
- Madhu NT, Radhakrishnan PK, Grunert M, Weinberger P, Linert W (2003) A thermal decomposition study on cobalt(II) complexes of 1,2-di(imino-4′-antipyrinyl)ethane. Thermochim Acta 400: 29.
- Madhu NT, Radhakrishnan PK, Grunert M, Weinberger P, Linert W (2003) Synthesis and thermal studies on iron(III) complexes of 4-N-(4′-antipyrylmethylidene)aminoantipyrine with varying counter ions. Thermochim Acta 407: 73.
- Madhu NT, Radhakrishnan PK, Williams E, Linert W (2005) Thermal decomposition studies on cobalt(II) complexes of 4-N-(4’-antipyrylmethylidene)aminoantipyrine with varying counter ions. J Therm Anal Calorim 79: 157-161.
- Madhu NT, Radhakrishnan PK, Linert W (2006) Thermal decomposition study on nickel(ii) complexes of 1,2-(diimino-4’-antipyrinyl)ethane with varying counter ions. J Therm Anal Calorim 84: 607-611.
- Madhu NT, Radhakrishnan PK, Linert W (2007) Thermolytic investigations on nickel(II) complexes of 4-N-(4′-antipyrylmethylidene)aminoantipyrine with various counterions. Int J Chem Kinet 39: 53.
- Satava V (1971) Mechanism and kinetics from non-isothermal TG traces. Thermochim Acta 2: 423.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi