Research Article, J Spine Neurosurg Vol: 6 Issue: 3
Use Of FIBERGRAFT BG Morsels Mixed With BMA In Anterior Cervical Discectomy And Fusion At 1, 2, 3 And 4 Levels: A Retrospective Analysis Of Fusion Results
Fortier L1*, Bauer L1 and Chung E2
1The Foundation for Spinal Restoration, 2811 Wilshire Blvd., Suite 850, Santa Monica, California, USA
2The Spine Institute of South Florida, 5210 Linton Blvd., Suite 103, Delray Beach, Florida, USA
*Corresponding Author : Dr. Luc Fortier
The Foundation for Spinal Restoration, 2811 Wilshire Blvd., Suite 850, Santa Monica, California 90403, USA
Tel: +1-418-656-5282
E-mail: luc.fortier@cshs.org
Received: March 28, 2017 Accepted: April 15, 2017 Published: April 22, 2017
Citation: Fortier L, Bauer L, Chung E (2017) Use of FIBERGRAFT BG Morsels Mixed with BMA in Anterior Cervical Discectomy and Fusion at 1, 2, 3 and 4 Levels: A Retrospective Analysis of Fusion Results. J Spine Neurosurg 6:3. doi: 10.4172/2325-9701.1000268
Abstract
Background: Fibergraft material is a synthetic bone graft substitute that has been proven in animal models to reveal 100% fusion in posterolateral spine fusion at 26 weeks. The purpose of this study is to analyze post-op CT scans to evaluate the fusion effectiveness of Fibergraft BG Morsels when combined with bone marrow aspirate (BMA) in one-, two-, three-, and four-level anterior cervical discectomy and fusion (ACDF).
Methods: In this retrospective, single-center medical record review, post-op cervical CT scans at approximately six months were evaluated in 27 patients by one senior orthopedic spine surgeon. The surgeon was blinded to the clinical outcomes, patient demographics and past medical history. Fusion results were given a grade of either I, II, III or IV based on the presence of trabecular bridging bone across both graft-vertebral body interfaces.
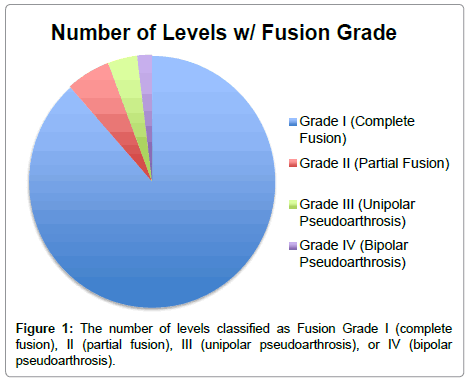
Results: A total of 52 levels were grafted in the 27 subjects who underwent the Smith-Robinson anterior cervical discectomy and fusion. 88.5% of the levels were classified as Grade I complete fusion, 5.8% of the levels classified as Grade II, 3.8% classified as Grade III, and 1.9% classified as Grade IV.
Conclusions: Fibergraft BG Morsels mixed with bone marrow aspirate (BA) is an effective bone graft option in one-, two-, three-, and four-level ACDF procedures. This synthetic bioactive glass bone graft substitute combined with BMA achieves high rates of fusion comparable to the biological performance of autologous bone graft.
Keywords: Cervical Discectomy; Spine fusion; Fibergraft
Introduction
Spinal fusion procedures are used to stabilize the spinal column for a number of disorders including, but not limited to, degenerative instability, traumatic instability, and abnormal curvature [1]. To assist in the body’s natural bone healing process, bone grafts are utilized during these procedures to bridge joints for arthrodesis [1].
Autologous bone graft has been considered the gold standard for obtaining successful spinal fusion [2]. The biological properties of autologous bone provide ideal osteoconductive, osteoinductive and ostogenic elements to aid in the bone healing response [3]. Unfortunately, autographs are also associated with many complications, such as infection at the donor site and donor site morbidity, increased blood loss and time under anesthesia, insufficient quality of donor bone, and residual pain and cosmetic flaws [3]. As a result, an industry of alternative bone grafts and synthetic bone graft substitutes has evolved to help alleviate morbidity traditionally associated with autologous bone grafting [3]. Today, an increasing number of synthetic bone graft formulations have been developed, leaving spine surgeons with a wide range of substitutes to choose from.
One bone graft substitute currently being utilized for spinal fusion includes FIBERGRAFT BG Morsels. FIBERGRAFT material is designed to have adequate structural integrity, while optimizing radiopacity and resorption. Each granule of FIBERGRAFT BG Morsels includes a porous outer shell encasing an intricate nest of 45S5 bioactive glass fibers and microspheres [4]. This unique design is responsible for leveraging the direct connectivity of fibers with exponentially increased surface area and optimized resorption rates, resulting in a bone graft substitute with an ultra-porous scaffold [4]. This osteoconductive scaffold is important because it maintains space within the fusion bed to allow de novo bone formation [1]. FIBERGRAFT’s increased surface area, continuous porosity, direct connectivity, and differential rate of resorption are thought to be responsible for its osteoconductive and osteoinductive biological performance, closely mimicking that of autologous bone grafts.
To date, FIBERGRAFT bioactive glass bone graft extender has been studied in various preclinical animal models that reveal 100% fusion in posterolateral spine fusion at 26 weeks [4]. However, no human studies have reviewed FIBERGRAFT’s fusion effectiveness as a bone graft extender in anterior cervical discectomy and fusion. The objective of this study was to examine the fusion effectiveness of FIBERGRAFT BG Morsels (bioactive glass) mixed with bone marrow aspirate (BMA) as an alternative fusion substrate to the “gold standard” bone graft during ACDF.
Methods
Study design
A retrospective, single-center, medical record review and analysis of cervical CT scans were performed in 27 patients by one surgeon to determine the fusion effectiveness of FIBERGRAFT BG Morsels mixed with BMA after 1-level, 2-level, 3-level, and 4-level anterior cervical discectomy and fusion (ACDF). CT images from each subject were selected by a senior orthopedic spine surgeon who was blinded to the clinical outcomes, patient demographics and past medical history. Fusion results were documented on all 27 patients approximately 6 months postoperatively using radiographic analysis evaluated for the presence of trabecular bridging bone across both graft-vertebral body interfaces [5].
Radiographic evaluation
The fusion results were graded by the senior orthopedic spine surgeon and based on a four-grade fusion system using the analysis of CT images. Grade I (complete fusion) implies cortical union of the graft at bone cranial and caudal ends and continuity of trabecular pattern between adjacent cranial and caudal vertebral bodies. Grade II (partial fusion) implies cortical union of the graft to the endplates at each end, however, with partial or absent trabecular continuity between the adjacent vertebral body bones at one or either end. Grade III (unipolar pseudoarthrosis) denotes cranial or caudal cortical nonunion with associated central trabecular discontinuity and Grade IV (bipolar pseudarthrosis) suggests both superior and inferior cortical non-union with a complete lack of central trabecular continuity [6].
Patient sample
The postoperative CT scans of 27 subjects who underwent grafting by the Smith-Robinson technique with FIBERGRAFT BG Morsels and BMA were assessed for fusion at one, two, three or four cervical spine levels. In all, a total of 52 levels were grafted. 12 subjects (44.4%) underwent a 1-level fusion, 8 subjects (29.6%) underwent a 2-level fusion, 5 subjects (18.5%) underwent a 3-level fusion, and 2 subjects (7.4%) underwent a 4-level fusion. Cervical CT scans were evaluated at a mean of 5.6 months postoperatively (range, 2 to 11 months).
The population consists of 14 females and 13 males, 51.9% and 48.1% respectively. The average age of the patient sample is 56 years old. 4 subjects were diagnosed with diabetes (14.8%), and 2 subjects self-reported being a smoker (7.4%). 5 subjects’ past surgical history was significant for spine surgery, including 1 subject who had 2 previous ACDF procedures and 1 subject who had 1 previous ACDF at the adjacent level.
*In addition to BMA, bone dust was also mixed with the Fibergraft BG Morsels in two of the ACDF cases analyzed. Local bone was also mixed with Fibergraft BG Morsels in one of the cases.
Results
27 anterior cervical discectomy and fusion (ACDF) procedures were performed on 27 individual patients between January 21 2015 and April 15 2016. A senior orthopedic spine surgeon reviewed postoperative cervical CT scans from the 27 subjects and 52 intervertebral levels grafted in total. The number of levels classified as Fusion Grade I (complete fusion), II (partial fusion), III (unipolar pseudoarthrosis), or IV (bipolar pseudoarthrosis) (Figure 1). Of the 52 levels that were grafted in total, 46 levels (88.5%) were classified as Grade I fusion, 3 levels (5.8%) were classified as Grade II fusion, 2 levels (3.8%) were classified as Grade III fusion, and 1 level (1.9%) was classified as Grade IV fusion. The CT images that revealed a fusion Grade II is on average 7.7 months status post-operation, Grade III is on average 6 months status post-operation, and Grade IV is 5-month status post-operation.
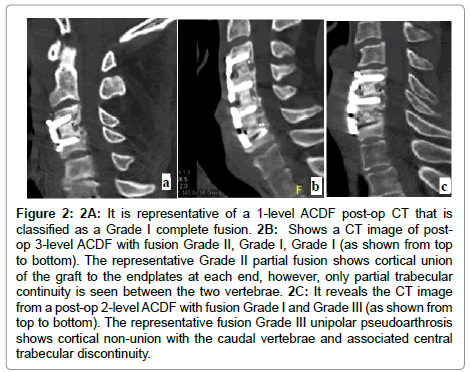
Representative of a 1-level ACDF post-op CT that is classified as a Grade I complete fusion. It shows a CT image of post-op 3-level ACDF with fusion Grade II, Grade I, Grade I (as shown from top to bottom). The representative Grade II partial fusion in Figure shows cortical union of the graft to the endplates at each end; however, only partial trabecular continuity is seen between the two vertebrae. This reveals the CT image from a post-op 2-level ACDF with fusion Grade I and Grade III (as shown from top to bottom). The representative fusion Grade III unipolar pseudoarthrosis in Image 3 shows cortical non-union with the caudal vertebrae and associated central trabecular discontinuity (Figure 2).
Figure 2: 2A: It is representative of a 1-level ACDF post-op CT that is classified as a Grade I complete fusion. 2B: Shows a CT image of postop 3-level ACDF with fusion Grade II, Grade I, Grade I (as shown from top to bottom). The representative Grade II partial fusion shows cortical union of the graft to the endplates at each end, however, only partial trabecular continuity is seen between the two vertebrae. 2C: It reveals the CT image from a post-op 2-level ACDF with fusion Grade I and Grade III (as shown from top to bottom). The representative fusion Grade III unipolar pseudoarthrosis shows cortical non-union with the caudal vertebrae and associated central trabecular discontinuity.
3 subjects were diagnosed with diabetes and had a total of 10 levels grafted. 1 subject was a self-reported smoker and had a total of 2 levels grafted. 1 subject was both diabetic and a smoker, and had 3 levels grafted shows the fusion grade that correlates to the levels grafted for diabetics, smokers, and both diabetic and smokers (Table 1).
| Grade I | Grade II | Grade III | Grade IV | |
|---|---|---|---|---|
| Diabetic | 8 | 0 | 1 | 1 |
| Smoker | 2 | 0 | 0 | 0 |
| Diabetic & Smoker | 2 | 1 | 0 | 0 |
Table 1: It shows the fusion grade that correlates to the levels grafted for diabetics, smokers, and both diabetic and smokers.
In all 27 cases, two 1-level ACDF procedures were grafted with bone dust in addition to Fibergraft BG Morsels and BMA. Both levels were classified as Grade I (complete fusion). One additional 1-level ACDF procedure was grafted with local bone in addition to Fibergraft BG Morsels, which was also classified as a Grade I (complete fusion).
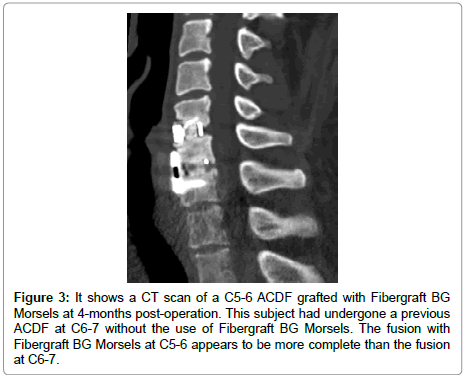
This shows a CT scan of a C5-6 ACDF grafted with Fibergraft BG Morsels at 4-months post-operation. This subject had undergone a previous ACDF at C6-7 without the use of Fibergraft BG Morsels. The fusion with Fibergraft BG Morsels at C5-6 appears to be more complete than the fusion at C6-7 (Figure 3).
Conclusions
The results of this retrospective study suggest that the use of Fibergraft BG Morsels mixed with bone marrow aspirate (BMA) is an effective osteoconductive bone graft option in 1-level, 2-level, 3-level and 4-level ACDF procedures. This synthetic bioactive glass bone graft substitute combined with BMA achieves high rates of fusion. Fibergraft material mimics the osteoconductive, osteoinductive and osteogenic biological performance of autologous bone graft, while simultaneously avoiding the complications and limitations associated with harvesting autograft. As an increasing number of bone graft substitute products become available, Fibergraft BG Morsels is evidenced to provide surgeons with an effective option with advantages to the “gold standard” autologous bone graft.
Declarations: Ethics approval and consent to participate. Because of the retrospective nature of this study, the trial was not registered.
Competing Interests: None
Funding: None
References
- Brecevich AT, Kiely PD, Yoon BV, Nguyen JT, Cammisa FP (2017) Effectiveness Comparison of Accell Evo3 and Grafton Demineralized Bone Matrix Putties against Autologous Bone in a Rat Posterolateral Spine Fusion Model. Spine J 17: 33-35.
- Boden SD, Schimandle JH (1995) Biologic Enhancement of Spinal Fusion. Spine 20: 113S-123S.
- Kadam A, Millhouse PW, Kepler CK, Radcliff KE, Fehlings MG (2016) Bone Substitutes and Expanders in Spine Surgery: A Review of Their Fusion Efficacies. International Journal of Spine Surgery 10: 33.
- FIBERGRAFT® BG Morsels Synthetic Bone Grafting. Prosidyan.
- Martin GJ, Haid RW, Macmillan M, Rodts GE, Berkman R (1999) Anterior Cervical Discectomy With Freeze-Dried Fibula Allograft. Spine 24: 852-859.
- Tan GH, Goss BG, Thorpe PJ, Williams RP (2007) CT-Based Classification of Long Spinal Allograft Fusion. Eur Spine J 16: 1875-1881.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi