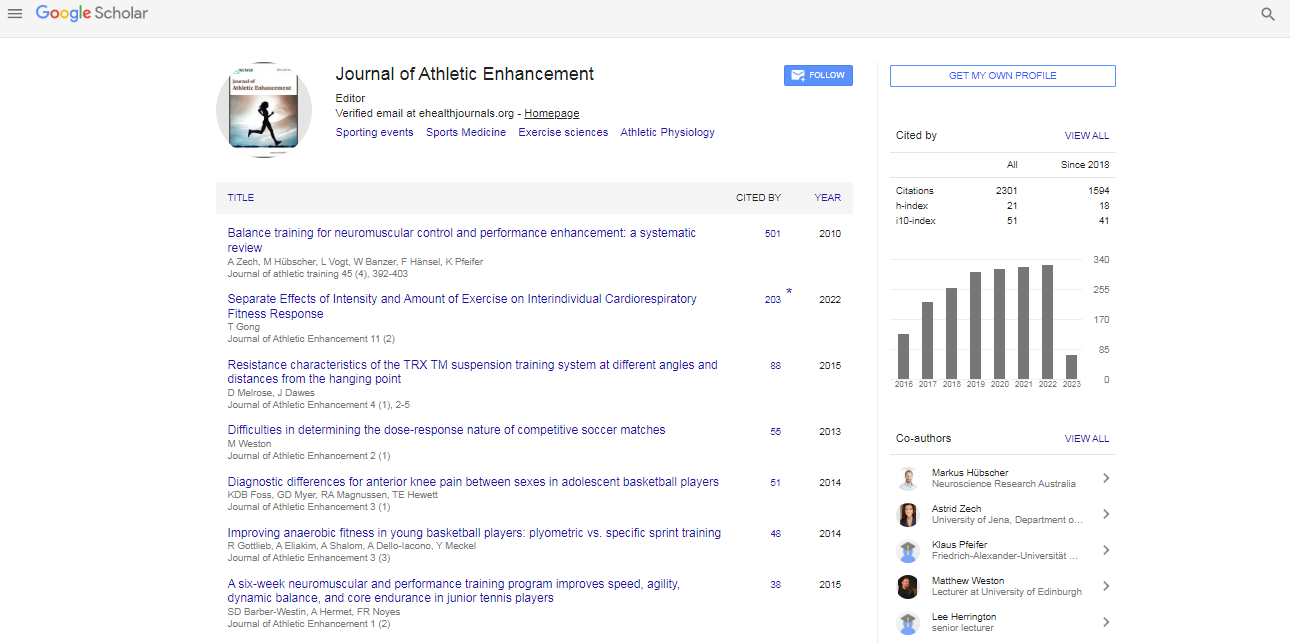
Research Article, J Athl Enhancement Vol: 3 Issue: 2
Multiple Sprint Exercise with a Short Deceleration Induces Muscle Damage and Performance Impairment in Young, Physically Active Males
| Brandon P Woolley1, John R Jakeman2 and James A Faulkner1* | |
| 1School of Sport and Exercise, Massey University, Wellington, New Zealand | |
| 2Department of Sport and Health Sciences, Oxford Brookes University, Oxford, UK | |
| Corresponding author : Dr. James Faulkner School of Sport and Exercise, Massey University, Private Bag 756, Wellington, 6140, New Zealand, Tel: +64 (0)4 801 5799 (Ext. 62104); Fax: +64 (0)4 801 4994 E-mail: J.Faulkner@massey.ac.nz |
|
| Received: October 16, 2013 Accepted: February 10, 2014 Published: February 15, 2014 | |
| Citation: Woolley BP, Jakeman JR, Faulkner JA (2014) Multiple Sprint Exercise with a Short Deceleration Induces Muscle Damage and Performance Impairment in Young, Physically Active Males. J Athl Enhancement 3:2. doi:10.4172/2324-9080.1000144 |
Abstract
Multiple Sprint Exercise with a Short Deceleration Induces Muscle Damage and Performance Impairment in Young, Physically Active Males
The purpose of the present study was to identify whether a bout of high-intensity multiple sprints elicits exercise-induced muscle damage (EIMD) of a similar magnitude to drop jumps. Methods: Ten physically active male volunteers (mean ± SD; 27 ± 3 y, 1.78 ± 0.06 m, 78.4 ± 7.5 kg) completed 10×10 drop jumps (DRP) and a bout of 40×15 m sprints with a 5 m deceleration zone (SPR) in a randomised repeated-measures crossover study. Muscle damage indices (creatine kinase (CK), lactate dehydrogenase (LDH), aspartate aminotransferase (AST), perceptual soreness and affective valence, range of motion and limb girth) and performance markers (vertical jump, agility and sprint performance) were gathered at baseline and 1, 24, 48 and 72 h post exercise. Results: The study findings revealed a significant Test by Time interaction for 15 m sprint performance (P<0.05), with a greater decrement in performance observed following SPR (~4%) compared to DRP (~1%). A significant main effect for Time for CK and AST activity, perceptual soreness, affective valence, limb girth, vertical jump and agility performance (P<0.05) was also revealed, although no Test by Time interaction was observed (P>0.05). Conclusion: This study indicates that a bout of multiple sprints with a short deceleration phase elicits greater reductions in sprint performance than drop jumps. This should be considered by individuals using similar sprint protocols in an applied context and by individuals looking to conduct research into EIMD using ecologically valid exercise protocols.
Keywords: Exercise-induced muscle damage; Drop jump; Delayed onset muscle soreness; Agility; Sprint
Keywords |
|
| Exercise-induced muscle damage; Drop jump; Delayed onset muscle soreness; Agility; Sprint | |
Abbreviations |
|
| ANOVA: Analysis of variance; AST: Aspartate Aminotransferase; BL: Baseline; CK: Creatine Kinase; DOMS: Delayed Onset Muscle Soreness; DRP: 10×10 drop jumps; EIMD: Exercise Induced Muscle Damage; FS: Feeling Scale; ICC: Intra-class Correlation; LDH: Lactate Dehydrogenase; CG: Mid-calf girth; MTG: Mid-thigh Girth; RBE: Repeated Bout Effect; ROM: Range of Motion; RPE: Rating of Perceived exertion; SPR: 40×15 m sprints with a 5 m deceleration zone; SSC: Stretch-Shortening Cycle; VAS: Visual Analogue Scale. | |
Introduction |
|
| Exercise-induced muscle damage (EIMD) occurs as a result of unaccustomed or eccentrically biased exercise and is predominantly characterised by impaired muscle function and muscle soreness [1-3]. It is well documented that EIMD also increases circulating plasma concentrations of myofibre proteins (e.g., creatine kinase (CK; [3,4]), causes swelling [5,6] and decreases an individual’s range of motion (ROM [7]). Perhaps of greatest concern to athletes and coaches is the negative influence of EIMD on sports performance, whereby an immediate and long lasting reduction in muscle strength and power is commonly observed [1,8]. However, the effects of EIMD on sprint and agility performance are not well established [1]. Semark et al. [9], for example, found no evidence to suggest that muscle damage negatively affected sprinting performance over 30 m. Conversely, reductions in 10 m sprint [1,10] and agility [1] performance have been reported 24 h and 48 h following muscle-damaging exercise. | |
| Isokinetic dynamometry [11,12], plyometric exercise [1,3] and ambulatory protocols such as downhill running [13,14] are typically used to elicit EIMD. However, to identify specific sporting or training implications following muscle damaging exercise, the exercise stimulus should involve muscle actions similar to those commonly employed during a particular activity [2,15]. Pre-season training for sports including rugby, field hockey and soccer typically involves high exercise loads in order to develop the necessary physical attributes required to compete at an optimal level throughout competitive matches [16]. Such sports require periods of maximal or near maximal, repeated sprint activity [6,17]. Depending on their positional play during a match, an individual may reach maximal speed over 30 times [18,19], covering distances between 14 and 22 m with each sprint [20,21]. Furthermore, deceleration plays an important role in the movement patterns of players as they rapidly decelerate to stop or change direction during games and training. As the quadriceps and hamstrings attempt to decelerate the body’s centre of mass, the mechanical stresses that are placed upon these muscle groups as they lengthen contribute to muscle damage [4,6]. This was demonstrated in a recent investigation in which a significant increase in CK activity, muscle soreness and limb girth, and a decrease in muscular strength, were exhibited in physically active participants after completing a single bout of sport specific multiple sprints (15×30 m, with a 10 m deceleration zone [6]). These effects, however, were observed in the absence of a control group. | |
| As an important component of athletic training, plyometric exercise has been identified as an appropriate method to induce muscle damage [22]. Bouts of drop jumps are commonly applied to induce and determine the negative effects of EIMD [1,3,10,22]. However, research is needed to examine muscle damage within the context of specific sporting tasks (i.e., field-based repeated sprints), to allow the magnitude of muscle damage and the required recovery duration to be appropriately established. This, in turn, may provide practical implications for coaches, strength and conditioning specialists and athletes participating in such activities [6]. | |
| The purpose of the present study was therefore to examine whether a bout of high-intensity multiple sprint exercise (experimental group), with a short deceleration zone, elicits a similar magnitude of exercise-induced muscle damage (EIMD) as compared to drop jumps (a common muscle damage procedure), when assessed by physiological, perceptual and performance indicators. It was hypothesised that a bout of high-intensity multiple sprints would elicit a similar magnitude of muscle damage to a bout of drop jumps. | |
Methods |
|
| Participants | |
| Ten physically active males (mean ± SD; 27 ± 3 years, 1.78 ± 0.06 m, 78.4 ± 7.5 kg) volunteered to take part in the present study. All participants were asymptomatic of injury and illness, regularly took part in team-based sports involving multiple sprint activity (3 sessions per week of greater than 1 h) and were in the competitive season of their respective sports. All participants had a similar history of weight training experience (>2 years) and had not engaged in specific lower limb eccentric or plyometric training for 6 months prior to taking part in this investigation. All participants provided written informed consent and completed a pre-exercise health-screening questionnaire and a risk stratification assessment [23]. Volunteers were asked to refrain from any exhausting physical activity or from using any form of recovery method (i.e., ice, massage, compression garments) in an attempt to attenuate the negative effects of EIMD during their participation in this study. The present study was conducted in agreement with the policies and guidelines of the institutional Human Ethics Committee. A priori calculations of statistical power indicated that this sample size was appropriate to satisfy power at or above 80% [3]. | |
| Procedures | |
| In a randomised repeated-measures counter-balance design, participants performed two bouts of EIMD exercise, separated by a period of 4 months to minimise the effect of a protective repeated bout effect (RBE [13,24]); a bout of multiple sprints (SPR; 40×15 m) and a bout of drop jumps (DRP; 10×10). Physiological, perceptual and performance characteristics were assessed in the following sequential order, prior-to (baseline [BL]) and following (1, 24, 48 and 72 h) the SPR and DRP damaging protocols: myofibre protein activity (plasma creatine kinase, lactate dehydrogenase, aspartate aminotransferase, perceived muscle soreness, knee- and hip-joint range of motion (ROM), thigh and calf girth, countermovement jump height, and agility and sprint performance. Data were collected 1 h after damaging exercise in order to provide an indicator of the magnitude of immediate muscle damage, without the confounding factor of acute fatigue resultant from either muscle damaging protocol. To compare the acute level of physical exhaustion from the damaging protocols (SPR, DRP), the rate of fatigue was calculated using the following equation: rate of fatigue=(25%first-25%last)/(25%first)×100, whereby 25%first and 25%last equated to the average sprint and jump height performance from the first 25% and last 25% of the damaging protocols (i.e., the first 10 and the last 10 15 m sprints; the first 25 and last 25 jump heights during DRP) [25]. | |
| Muscle-Damaging Exercise | |
| Multiple Sprints (SPR): Participants performed a single bout of 40×15 m maximal effort sprints, with a 5 m deceleration zone [17] in order to promote ecological validity, on a non-slip indoor playing surface. In accordance with previous research, a 30 s recovery period separated each sprint [17]. Infra-red timing gates (Multi-channel Timing System, Sportstec, NSW, Australia) were positioned at the start line (0 m) and at 5 and 15 m marks to allow sprint times to be recorded. Each sprint began from a stationary start, 1 m behind (–1 m) the first timing gate (0 m) to ensure that the timing gates were not prematurely triggered. A 3 s verbal countdown was provided before commencing each maximal sprint. Participants were instructed to sprint each 15 m trial as fast as possible. Upon crossing the final timing gate (15 m), each participant rapidly decelerated and came to a complete stop within the confines of the 5 m deceleration zone. Thereafter, the 30 s recovery period began. Each participant was given verbal encouragement to ensure maximal effort. Following the completion of each tenth sprint, participants reported their overall perception of exertion using the Borg 6-20 Ratings of Perceived Exertion (RPE) scale [26] and provided feelings of pleasure or displeasure (affective valence) using an 11-point bipolar Feeling scale (FS) ranging from +5 to -5 [27]. | |
| Drop jumps (DRP): Participants completed 10×10 repetitions of drop jumps, with 10 s separating each jump. A 60 s recovery period, which began after completion of the tenth jump of each set, was allowed between sets. Subjects were required to step from a 0.6 m high box, land (with both feet) in a squat position, with approximately 90o of hip and knee flexion, and then perform a maximal vertical jump in continuous movement, to elicit muscle damage. Participants were required to alternate between left and right legs as they stepped up onto the box. Following the completion of each set, participants were required to provide an RPE and FS score. Comparable protocols have been successfully applied to induce muscle damage to the knee extensor muscle group [1,3,10,22]. | |
| Assessment of sport performance | |
| Sprint performance: Sprint performance was determined over a 15 m distance in which participants performed three maximal effort sprints separated by a 2 min rest period [28]. The positioning of the timing gates, starting position and the instructions given to each participant were identical to those given during the multiple sprints mentioned above. | |
| Agility performance: Agility was assessed using a 3-cone shuttle drill test during four separate trials (Figure 1 [29]). Participants alternated between running to the left and to the right upon reaching the second cone. Participants had a 2 min recovery between trials to ensure adequate recovery [28]. An average of four trials was used for analysis. | |
| Figure 1: The 3-cone shuttle drill agility test. The athlete starts 1m behind cone 1 to ensure that the timing gates were not prematurely set off, and sprints and touches cone 2 directly in front, and then immediately returns to cone 1. Without stopping, the athlete changes direction, corners cone 2, and sprints directly to cone 3, on the athlete’s right-hand side. The athlete circles cone 3 to his left, then returns to the first cone (finish line) by cornering cone 2 and sprinting at full speed past cone 1. The test is repeated on both the right and left side. | |
| Vertical jump performance: Jump height was assessed using a vaned jump and reach apparatus, which provides the maximum height reached to the nearest centimetre (Yardstick, Swift Systems, Lismore, Australia [30]). Following verbal command and demonstration, participants were instructed to jump as high as possible, bending their knees on their downward countermovement (~90° of hip and knee flexion), and jumping vertically in the air with straight legs in order to achieve maximal height. The peak height of 3 jump trials was considered as optimal performance. | |
| Markers of muscle damage | |
| Myofibre protein activity: Plasma CK, LDH and AST activity was assessed from a 4 ml venous blood sample drawn from the antecubital vein. Each sample was then centrifuged (Medifuge, Heraeus Sepatech, Germany) for 10 min to obtain plasma samples [31]. Samples were frozen at -80°C until analysis. Automated spectrophotometry (Cobas 6000, Roche Diagnostics, Indianapolis, USA) was used to analyse each sample. | |
| Joint range of motion: Knee joint and hip joint ROM was determined using a plastic goniometer (E-Z Read Goniometer, Jamar, Sammons Preston Rolyan, USA) in accordance with procedures outlined by the ACSM [23]. To determine knee joint ROM the goniometer was placed upon the lateral epicondyle of the femur and aligned with the greater trochanter and the lateral malleolus. While lying prone, subjects actively flexed their knee joint through a full ROM from an extended position. To determine hip ROM, the goniometer was placed upon the greater trochanter and aligned with the femoral epicondyle and the axilla. While lying prone subjects were asked to extend their hip. Thereafter, hip flexion was determined with subjects lying in a supine position. Each of these reference points were marked with a permanent marker to ensure consistency on subsequent testing days. | |
| Perceived muscle soreness: Perceived soreness was assessed using a 10 cm visual analogue scale, with 0 (no pain) and 10 (worst pain ever) at the two extremes. Participants were instructed to assume an un-weighted squat of approximately 90o and mark their perceived soreness on a horizontal line between 0 and 10 [3]. | |
| Limb girth: Mid-thigh girth (MTG) and mid-calf girth (MCG) was assessed using a measuring tape following the method described by French et al. [5]. Measures were completed while participants were in a standing position. | |
| Statistical analysis | |
| A paired-samples t-test was used to compare the rate of fatigue and perceived exertion during and following DRP and SPR damaging protocols. Intra-class correlations (ICC) were used to assess testretest reliability prior-to and following the muscle damage. A series of two-way repeated-measures analysis of variance (ANOVA); Test (SPR, DRP)×Time (BL, 1 h, 24 h, 48 h and 72 h) assessed physiological and performance markers following DRP and SPR. Mauchly’s test of sphericity was used to test assumptions of equality of variance. Where violations of sphericity were evident, the Greenhouse-Geisser Epsilon value was used. Where statistical differences were observed, post-hoc analysis using a Bonferroni correction was used to reduce the risk of type I error. Alpha was set at P=0.05. All data were analysed using SPSS version 18. | |
Results |
|
| Muscle damaging exercise | |
| There were no significant differences in the rate of fatigue between the SPR (-1.24 ± 1.7%) and DRP (-0.13 ± 8.6%) damage protocols (t(9)=-0.38, P>0.05). Participants reported similar FS scores between damaging protocols at protocol completion (-0.22 ± 2.9 and 0.28 ± 1.7 for SPR and DRP, respectively; P>0.05). A greater RPE was however observed on completion of SPR (16.7 ± 1.8 and 14.6 ± 1.2 for SPR and DRP, respectively; t(8)=2.949, P<0.05). Test-retest reliability revealed high ICC’s prior-to and following the muscle damaging exercise for sprint (~0.95), agility (~0.88) and vertical jump (~0.94). | |
| Sprint performance | |
| A significant Test by Time interaction was only observed for 15 m (F(3,27)=3.00, P<0.05). Although sprint performance remained similar for SPR at 24, 48 and 72 h, a marked improvement was observed in DRP between 24 and 72 h (Figure 2). A significant Test main effect was revealed for 5 and 15 m sprint performance (all P<0.05). On average, a greater decrement in sprint performance was observed following SPR (4%) compared to DRP (1%) at 5 and 15 m, respectively. | |
| Figure 2: 15 m sprint performance expressed as a proportion of baseline following multiple sprints (SPR) and drop jump (DRP) induced muscle damage. Values are mean ± SD. | |
| Agility performance | |
| A significant main effect for Test (F(1,9)=4.56, P<0.05) and Time (F(3,27)=12.74, P<0.05) was observed for agility performance. Participants experienced a 3.2 ± 4.1% decrease in agility performance during SPR compared to a 1.5 ± 4.4% improvement following DRP (Figure 3). Regardless of the damaging protocol, agility performance significantly improved between 24 h and 72 h (P<0.05). There was no significant Test by Time interaction (F(3,27)=0.38, P>0.05). | |
| Figure 3: Agility performance expressed as a proportion of baseline following multiple sprints (SPR) and drop jump (DRP) induced muscle damage. Values are mean ± SD. | |
| Vertical jump performance | |
| Vertical jump height was significantly different across Time (F(3,27)=4.48, P<0.05) with a more pronounced recovery between 48 h and 72 h (3.2 ± 5.9%; Figure 4). No significant main effect for Test or a Test by Time interaction was observed (both P>0.05). | |
| Figure 4: Vertical jump performance expressed as a proportion of baseline following multiple sprints (SPR) and drop jump (DRP) induced muscle damage. Values are mean ± SD. | |
| Perceived muscle soreness | |
| Perceived muscle soreness significantly changed across Time following muscle-damaging exercise (F(4,36)=28.65, P<0.05), and followed a typical temporal pattern, increasing after insult from BL (1.6 ± 0.4) to 24 h (4.7 ± 1.6) before beginning to return to BL values between 24 h and 72 h (1.6 ± 0.9; Figure 5). | |
| Figure 5: Perceptual muscle soreness as determined using a visual analogue scale following multiple sprints (SPR) and drop jump (DRP) induced muscle damage. Values are mean ± SD. | |
| Myofibre protein activity | |
| Although approaching statistical significance, no main effect for Time was observed for CK (F(1.3,11.6)=3.27, P>0.05) or AST (F(1.33,11.93)=3.45, P>0.05) following damaging exercise (Table 1). Similarly, no Test, or Test by Time interaction was observed (P>0.05). No significant main effect for Time or Test, or a Test by Time interaction was observed for LDH (all P>0.05). | |
| Table 1: Creatine kinase (CK), lactated dehydrogenase (LDH) and aspartate aminotransferase (AST) activity following SPR and DRP induced muscle damage. | |
| Limb girth | |
| Relative to BL, only a Time main effect was observed for MTG (F(3,21)=4.82, P<0.05) and MCG (F(3,21)=2.78, P<0.05). Post-hoc analysis revealed an increase between 1 h and 24 h (0.6%) for MTG and an increase between 48 h and 72 h (0.3%) in MCG following muscle-damaging exercise. | |
| Range of motion | |
| Absolute ROM values for knee flexion, and hip extension/flexion revealed no significant main effect for Time or Test, or Test by Time interaction (all P>0.05). Similar findings were observed for knee extension, although a significant main effect for Time (F(1.9,17.06)=3.48, P<0.05) was observed. Post-hoc analysis demonstrated a significant reduction in knee extension observed between 1 h and 24 h (171.6 ± 1.5° to 169.0 ± 6.0°, respectively). | |
Discussion |
|
| The present study investigated whether a bout of high-intensity multiple sprints (SPR) elicits a similar magnitude of muscle damage when compared with a bout of drop jumps (DRP; a commonly used muscle damage procedure), by comparing the effect of SPR and DRP on performance markers, including sprint, agility and vertical jump. Although both SPR and DRP elicited muscle damage over the course of the study period (1, 24, 48 and 72 h), SPR had a more substantial impact on sports performance, impairing sprint performance to a greater degree than DRP. This may indicate that the sprinting protocol elicited a greater magnitude of strain on the muscle than the DRP protocol, most likely during the deceleration phase of the sprint, leading to greater reductions in muscular performance. The 4% (0.11 s) decrement in 5 and 15 m sprint performance following SPR was significantly greater than the 1% (0.05 s) reduction observed following DRP. This change in performance following muscle damage is consistent with previous research [1,10], thus demonstrating that repeated sprints with a short deceleration zone may impair sprint performance [1]. The greater decrement in performance for SPR was evident despite a statistically similar rate of fatigue and affective valence (pleasure-displeasure; FS) from the SPR and DRP muscle damaging protocols. It is however of particular interest to report that only DRP elicited observed improvements in sprint performance between 24 and 72 h (~3%; 0.08 s) post initial muscle insult (Figure 2). | |
| Similar findings were observed for agility performance. SPR elicited an average decrement of 3.2% (0.17 s) in agility performance compared to the 1.5% (0.05 s) improvement observed for DRP. Agility performance was impaired to the greatest degree 1 h after initial muscle insult (5.1 ± 3.4% vs. 0.7 ± 2.7% for SPR and DRP, respectively), suggesting that fatigue, as a result of the damaging exercise, was a pertinent reason for the increased time to complete the agility test. It is probable that improvements in agility times thereafter, evident in both groups, were due to participants becoming increasingly familiar with the agility test. However, the magnitude of the agility performance impairment is lower than that previously reported by Highton et al. [1] 48 h following initial muscle insult (8% decrement), and is most likely due to using a different agility protocol in the present study (3-cone shuttle drill) as compared to such previous research (agility-505 [1]). | |
| Decrements to sprint and agility performance may have occurred as a result of an inhibition of neural mechanisms in response to muscle soreness and a reduced reflex sensitivity during the stretchshortening cycle (SSC [1]). The activation of group III and IV free nerve endings, known to be sensitive to parameters associated with muscular damage (i.e., DOMS), are believed to potentially lead to an increased inhibitory drive to the α-motorneuron pool to protect the neuromuscular structures [1,10,32]. These changes occur concomitantly with a decrease in reflex sensitivity and are believed to negatively affect the force generating mechanisms of the muscle during the SSC [33]. As such, during the 15 m sprint test a reduced reflex sensitivity may have impaired the ability of damaged muscles to utilise ground impact forces, and thus less force would have been produced during the propulsive phase of leg movement [1]. This suggests that voluntary muscle actions are impaired via centrallymediated force-inhibiting neural mechanisms whereby, during dynamic muscle actions, these mechanisms attempt to prevent further damage to the muscle [1,10]. Based upon subjective scaling, research has demonstrated that a bout of repeated sprints leads to a greater level of pain to the hamstrings following muscle-damaging exercise [4,6]. This subjective score remained elevated for 72 h, which reflects the delayed recovery response evident in the present study between 48 h and 72 h. It may be proposed that in response to muscle soreness, reductions in neural drive affected a larger muscle mass influencing sprint performance to a greater degree than DRP damaging exercise. Unfortunately, the research team did not have access to an isokinetic dynamometer and as such were unable to monitor changes in muscular strength following the SPR and DRP muscle damaging protocols. This would have been useful with regards to providing additional support to the main outcome of this investigation as this marker is considered one of the most valid and reliable measures of muscle damage [34]. | |
| Reactive strength, which depends upon the contribution of both reflex and voluntary muscle actions, is the ability to reverse a movement from an eccentric contraction to a concentric contraction at a high speed [33]. Reduced mechanical efficiency, as represented by a decrease in muscular strength and reflex sensitivity, is believed to lead to an inability to effectively utilise ground impact forces during phases of breaking and push-off [1]. The decrement in vertical jump performance, consistent with previous research [3,33], may reflect a loss in reactive strength. | |
| Despite observed differences in sprint and agility performance between SPR and DRP, a similar perception of muscle soreness was observed between protocols at each follow-up assessment (1, 24, 48 and 72 h). Analogous with past research, VAS scores for perceived muscle soreness peaked 24 h after initial muscle insult before returning to pre-exercise levels at 72 h for both SPR and DRP [3,4,6,9]. Although the magnitude of soreness was not significantly different between protocols, anecdotal evidence indicated greater perceptual pain localised to the hamstring muscle group following SPR induced damage [4,6]. Future multiple sprint EIMD studies should consider the use of anatomical diagram of the body’s musculature, to provide a more accurate depiction of the locations of muscle soreness [4]. | |
| Localised swelling has been associated with muscle soreness [35]. As observed in the present study, an indicative marker of muscle swelling is an increase in MTG [6]. The influx of inflammatory mediators leads to the release of noxious chemicals, which activates muscle nociceptors causing a hyperalgesic state and increases sensitivity to pain [36,37]. The threshold for pain is ultimately lowered whereby stimuli may act upon the activated muscle nociceptors causing an excessive pain response. However, according to Howell et al. [37] an increase in pressure within the muscle compartment, most likely initiated by the muscle actions associated with performing a static squat, seemingly leads to a rise in muscle soreness. The present investigation did not observe a significant change in joint stiffness of the knee and hip following SPR and DRP induced muscle damage, although a significant reduction in knee extension between 1 h and 24 h is evident. French et al. [5] have demonstrated similar findings for knee flexion/extension and hip flexion/extension whereby changes were evident between BL and post-damaging exercise values, although these were not significant. | |
| Consistent with previous research CK activity [3,6,33], and AST activity [4] increased following a bout of muscle-damaging exercise. However, due to a substantial inter-individual variability this increase was not statistically significant. In this study, CK and AST activity remained elevated 48 h and 72 h following SPR induced damage, yet returned to baseline values between 48 h and 72 h for DRP induced muscle damage [22,33]. Howatson and Milak [6] reported a similar trend in CK activity after participants completed a single bout of sport-specific multiple sprints (15×30 m). | |
| The 4 month washout period between bouts of damaging exercise potentially limits this investigation as research has suggested that the RBE lasts up to, and including 6 months [38]. It is worth noting, however, that the duration reported by Nosaka and colleagues [39] was variant between subjects, and that the underlying mechanisms which may lead to the adaptations associated with the RBE are, to date, inconclusive and thus difficult to determine over time [39]. Moreover, this has been demonstrated on non-dominant elbow flexors and as such may not be a true reflection of the RBE during lower body exercise. | |
| To stimulate the necessary adaptations to the cytoskeletal framework, future training programmes may require the inclusion of activities that demand the generation of large forces [17]. Training programmes should involve repeated high-intensity efforts, with rapid breaking (deceleration) so as to condition athletes to the demands of their specific game [16,17]. As demonstrated in the present study, subsequent training sessions may be compromised for 72 h after exercise of this nature. Repair and remodelling of the damaged muscle may take several days or even weeks depending on the severity of the damage. Muscle damage may be attenuated or possibly prevented by training the affected muscle groups using the same muscle actions and exercising at the same intensities as the damaging exercise [31]. It may be of practical value to gradually introduce players to these stresses during pre-season training, increasing workloads over time in preparation for competitive matches. | |
Conclusion |
|
| In conclusion, the present study has shown that a single bout of high-intensity multiple sprints elicit a similar amount of muscle damage to a single bout of drop jumps. More importantly, however, the results suggest that high-intensity multiple sprints has a greater impact on sprint performance. Athletes, trainers and strength and conditioning coaches should be aware of the prolonged impairment in specific performance characteristics (up to 72 hours) that may be evident following a single bout of high-intensity multiple sprints, and as such modify subsequent training sessions in line with such information. Future research should consider whether a repeated bout of multiple sprints leads to the attenuation of the indices of muscle damage and whether specific recovery methods should be utilised for athletes who have completed bouts of high-intensity multiple sprints. | |
Acknowledgments |
|
| The authors wish to acknowledge the contributions of all those who participated in this investigation. | |
References |
|
|
|
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi