Case Report, Cocr Vol: 7 Issue: 12
A Rare Case of Peritoneal Well-Differentiated Papillary Mesothelioma Coincidentally Associated with Spermatic Cord Mesothelial Hyperplasia
Hiroshi Sonobe1*, Rika Omote1, Ryosuke Hamano2, Hiroyuki Yanai3, Kazuki Nabeshima4
1Department of Diagnostic Pathology, National Hospital Organization (NHO) Fukuyama Medical Center, Hiroshima, Japan
2Department of Surgery, National Hospital Organization (NHO) Fukuyama Medical Center, Hiroshima, Japan
3Department of Diagnostic Pathology, Okayama University Hospital, Okayama, Japan
4Department of Diagnostic Pathology, Pathological Diagnosis Center, Fukuoka Tokushukai Hospital, Fukuoka, Japan
- *Corresponding Author:
- Dr. Hiroshi Sonobe
Department of Diagnostic Pathology, National Hospital Organization (NHO) Fukuyama Medical Center,4-14-17 Okinogami-cho, Fukuyama, Hiroshima, Japan
E-mail: hnsonobe@gmai.com
Received: Dec 01, 2024; Manuscript No: COCR-24-158885
Editor Assigned: Dec 09, 2024; PreQC Id: COCR-24-158885(PQ)
Reviewed: Dec 20, 2024; QC No: COCR-24-158885(Q)
Revised: Dec 24, 2024; Manuscript No: COCR-24-158885(R)
Published: Jan 30, 2024; DOI: 10.4173/cocr.7(12).385
Citation: Sonobe H.. et al., (2024) A Rare Case of Peritoneal Well-Differentiated Papillary Mesothelial Tumor Coincidentally Associated with Spermatic Cord Mesothelial Hyperplasia. Clin Oncol Case Rep 7:12
Abstract
Background: We report a case of incidental association between Well-Differentiated Papillary Mesothelial Tumor (WDPMT) of the peritoneum and Nodular Histiocytic/Mesothelial Hyperplasia (NHMH) of the spermatic cord.
Case Presentation: The male patient was in his 40s with no history of asbestos exposure. A computed tomography scan of a gradually enlarging inguinal hernia revealed seminal hydrocele and ascites. Laparoscopy revealed multiple small nodules in the peritoneum. Histologically, the seminal hydrocele contained a nodule within the hernia sac. The nodule was mainly composed of histiocytes positive for CD68 (Cluster of Differentiation 68), with a few scattered reactive mesothelial cells positive for calretinin and epidermal growth factor-like domain 1 (HEG1). The spermatic cord lesion was diagnosed as NHMH. The resected peritoneal nodule had an edematous stroma with a highly branching papillary pattern, and the surface was covered with a single layer of non-dysplastic mesothelial cells positive for calretinin and HEG1. No nuclear deletion of BRCA1-Associated Protein 1 (BAP1), Methyl-Thioadenosine Phosphorylase (MTAP), or Merlin/Neurofibromatosis 2 (NF2) was found in the superficial mesothelial cells, and the diagnosis was WDPMT.
Conclusion: In some cases, WDPMT may not be differentiated from Mesothelioma In Situ (MIS), or from diffuse or localized mesothelioma. In cases of MIS as well as diffuse or localized mesothelioma with a conspicuous papillary pattern, these tumors are difficult to differentiate from WDPMT when only the superficial components are obtained by biopsy, particularly owing to the lack of confirmation of invasion. In such cases, the findings of nuclear deletion of BAP1 (BRCA1-associated protein 1), MTAP, and Merlin/NF2 using immunohistochemistry and homozygous deletion of p16 cyclin-dependent kinase 1 inhibitor 2A using fluorescent in situ hybridization help to exclude the possibility of WDPMT or diagnose MIS, and diffuse or localized mesothelioma. Given the rarity of WDPMT, molecular studies are still underway, and further research is needed.
Keywords: Well-Differentiated Papillary Mesothelial Tumor (WDPMT); Mesothelial tumor; Nodular Histiocytic/Mesothelial Hyperplasia(NHMP) ; Inguinal hernia; Peritoneum; Mesothelioma In Situ (MIS)
Keywords
Well-Differentiated Papillary Mesothelial Tumor (WDPMT); Mesothelial tumor; Nodular Histiocytic/Mesothelial Hyperplasia (NHMH); Inguinal hernia, Peritoneum, Mesothelioma In Situ (MIS)
Introduction
In the previous WHO classification (2015), the category of "mesothelial tumors" included the following subcategories: Adenomatoid Tumor (AT), Well-Differentiated Papillary Mesothelioma (WDPM), Multi-Cystic Peritoneal Mesothelioma (MCPM), malignant mesothelioma in situ, diffuse malignant mesothelioma, and localized malignant mesothelioma. However, in the new WHO classification (2021), the category has been changed to "pleural and pericardial tumors," and the subcategory of MCPM, which is a peritoneal lesion, has disappeared. Additionally, the word "malignant" has been omitted from “malignant mesothelioma”, and it has become “mesothelioma.” Hence, malignant mesothelioma in situ became Mesothelioma In Situ (MIS), while diffuse malignant mesothelioma and localized malignant mesothelioma became diffuse mesothelioma and localized mesothelioma, respectively. Furthermore, since the name “mesothelioma” now has a malignant connotation, well-differentiated papillary mesothelioma with a good prognosis was renamed Well-Differentiated Papillary Mesothelial Tumor (WDPMT) [1-3]. Nodular Histiocytic/Mesothelial Hyperplasia (NHMH) is a rare unique lesion that develops in various sites, including the peritoneum, lung, cardiac muscle, inguinal hernia, and spermatic cord, and can mimics a malignant tumor [4].
WDPMT of the peritoneum and NHMH of the spermatic cord are rare individual mesothelial diseases [4,5]. Here in we report a case in which typical WDPMT of the peritoneum and NHMH of the spermatic cord developed simultaneously, along with the clinical and immunohistochemical findings of them. Additionally, regarding WDPMT of the peritoneum, we reviewed the literature and discussed the laparoscopic, clinicopathological, and molecular pathological findings.
Case Presentation
The patient was a man in his 40s with no history of asbestos exposure. He visited our hospital for a detailed examination because his right inguinal hernia had gradually been enlarged over approximately 2 months. A computed tomography scan revealed a hydrocele of the spermatic cord and ascites. The right spermatic cord lesion was extirpated using an anterior approach (Figure 1). Laparoscopy revealed more than 10 scattered small nodules in the peritoneum. A visceral peritoneal lesion in the right hepatic lobe (green arrow) was resected for pathological examination (Figure 2).
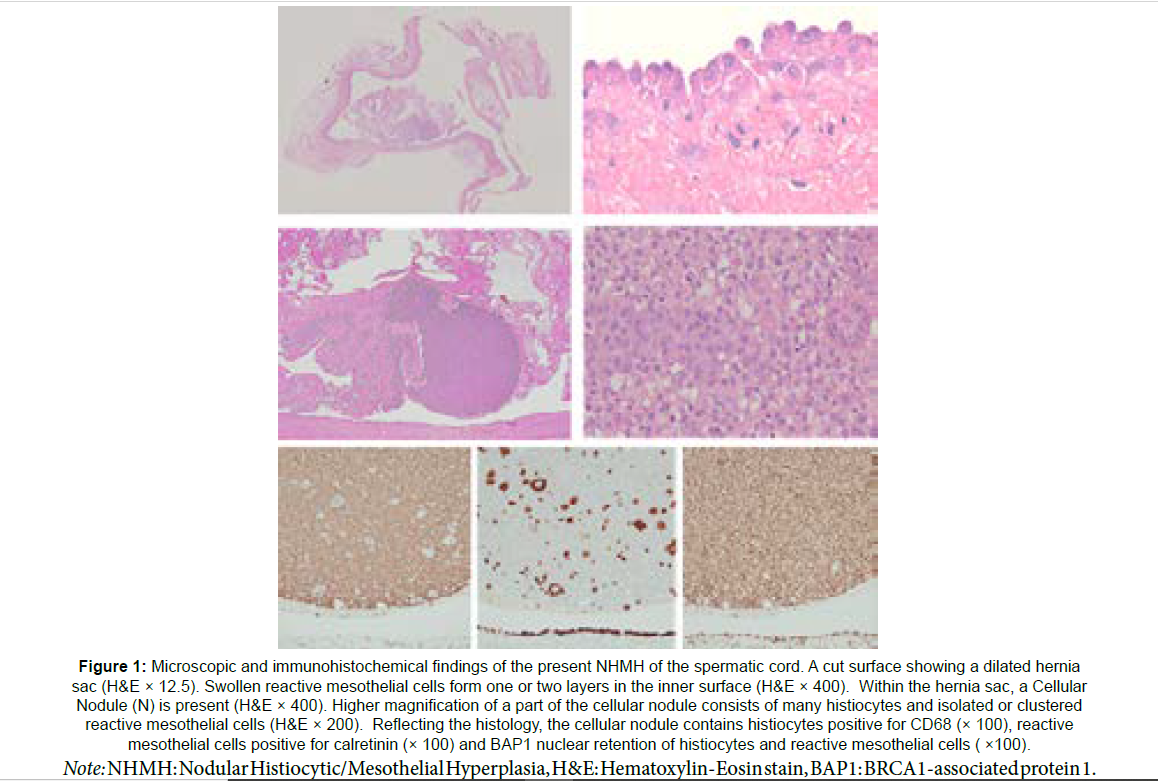
Figure 1: Microscopic and immunohistochemical findings of the present NHMH of the spermatic cord. A cut surface showing a dilated hernia sac (H&E × 12.5). Swollen reactive mesothelial cells form one or two layers in the inner surface (H&E × 400). Within the hernia sac, a Cellular Nodule (N) is present (H&E × 400). Higher magnification of a part of the cellular nodule consists of many histiocytes and isolated or clustered reactive mesothelial cells (H&E × 200). Reflecting the histology, the cellular nodule contains histiocytes positive for CD68 (× 100), reactive mesothelial cells positive for calretinin (× 100) and BAP1 nuclear retention of histiocytes and reactive mesothelial cells ( ×100).
Note: NHMH: Nodular Histiocytic/Mesothelial Hyperplasia, H&E: Hematoxylin-Eosin stain, BAP1: BRCA1-associated protein 1.
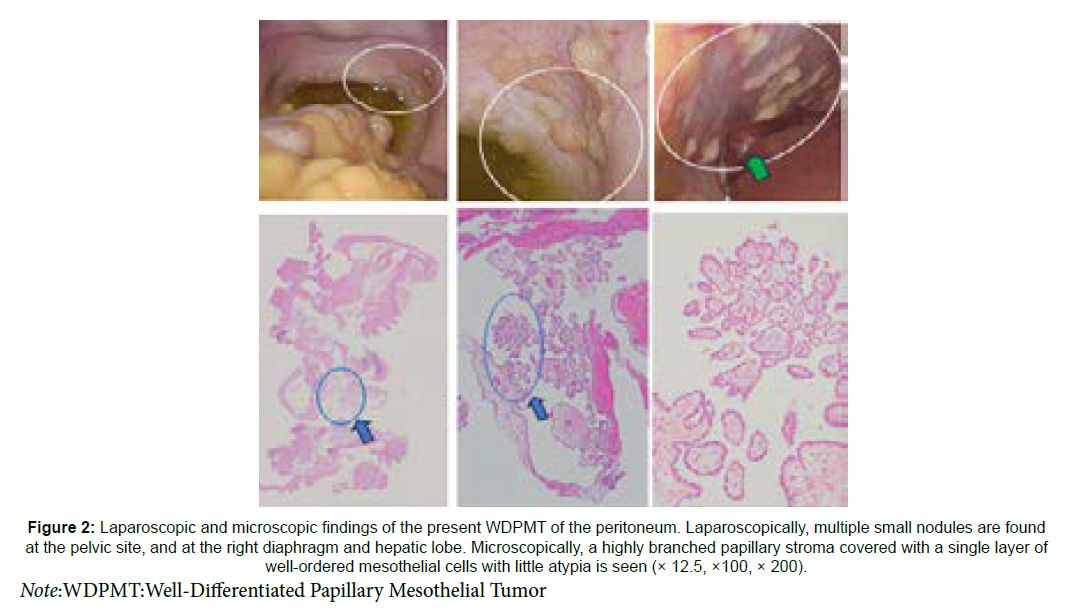
Figure 2: Laparoscopic and microscopic findings of the present WDPMT of the peritoneum. Laparoscopically, multiple small nodules are found at the pelvic site, and at the right diaphragm and hepatic lobe. Microscopically, a highly branched papillary stroma covered with a single layer of well-ordered mesothelial cells with little atypia is seen (× 12.5, ×100, × 200).
Note:WDPMT:Well-Differentiated Papillary Mesothelial Tumor
Histological examination of the extirpated spermatic cord lesion revealed a dilated hernia sac, with the inner surface was covered by one or two layers of swollen reactive mesothelial cells. Within the sac, reactive mesothelial cells proliferated in an irregular reticular pattern with fibrin deposition and protruded into the cavity, and a cellular spherical Nodule (N) was formed with dense proliferation of histocytes and a small number of reactive mesothelial cells found in clustered or isolated patterns (Figure 1). Reflecting such histology, the histiocytes were positive for CD68 and the mesothelial cells were for calretinin and heated development protein with epidermal growth factor like domain 1 (HEG1). Additionally, both the histiocytes and mesothelial cells showed nuclear retention in immunohistochemistry for BRACA1-Associated Protein 1 (BAP1) (Figure 1). Methylthioadenosine Phosphorylase (MTAP), and Merlin/Neurofibromatosis 2 (NF2) (Table 1). Based on these findings, this lesion was diagnosed as NHMH.
| Anti-Body | Lesion Tissue |
|---|---|
| Caletinin | Positive for a few mesothelial cells |
| HEG1 | Positive for a few mesothelial cells |
| CD68 | Positive for many histiocytes |
| CEA | Negative for histiocytes abd mesothelial cells |
| BAP1 | Retained in nuclei of mesothelial cells |
| MTAP | Retained in nuclei of mesothelial cells |
| Merlin/NF2 | Retained in nuclei of mesothelial cells |
Table 1: Immunohistochemistry results in present NHMH of the spermatic cord.
HEG1: Heart development protein with EGF like domains 1 CEA: Carcinoembryonic Antigen, BAP1: BRCA1-associated protein 1, MTAP: Methyl-Thioadenosine Phosphorylase Merlin/NF2: Merlin/Neurofibromatosis 2.
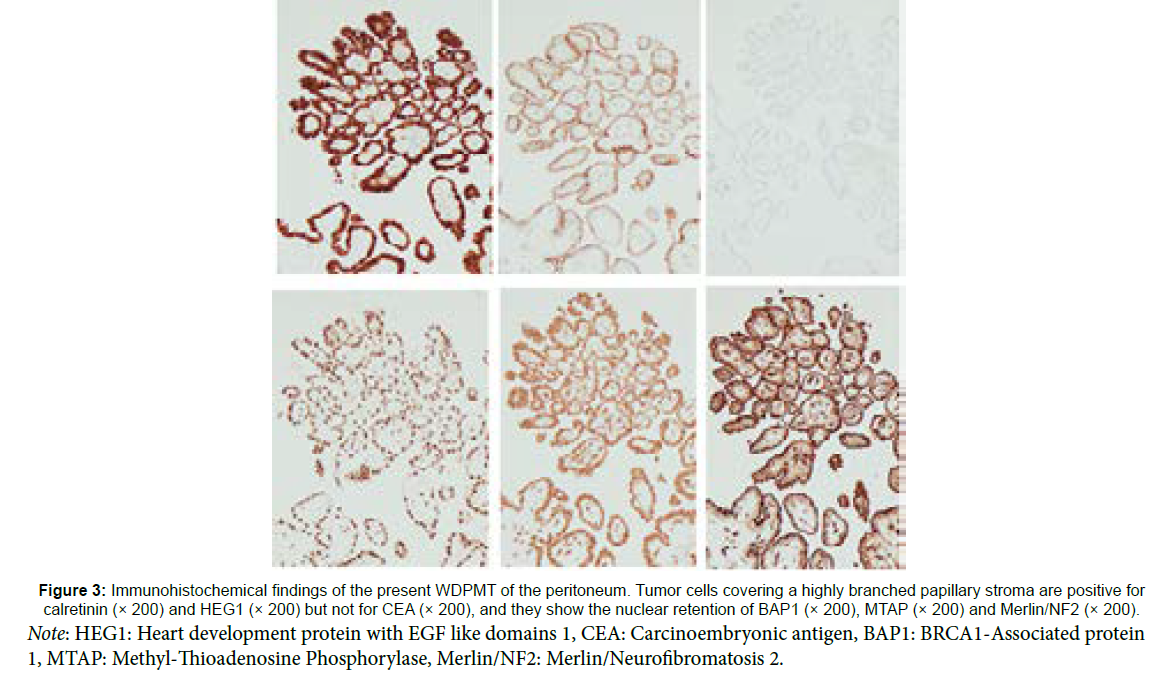
The resected peritoneal lesion was histologically (green arrow) characterized by edematous stroma with a highly branched papillary structure and was covered by a single layer of well-ordered mesothelial cells with little atypia. These mesothelial cells did not exhibit any mitotic (Figure 2). Mesothelial cells were immunohistochemically positive for Calretinin (Figure 3). But negative for carcinoembryonic antigen and Claudin-4. Additionally, Nuclear retention of the tumor cells by BAP1, MTAP, and Merlin/NF2 was observed (Figure 2) (Table 2). Based on these findings, the lesion was diagnosed as WDPMT.
Figure 3: Immunohistochemical findings of the present WDPMT of the peritoneum. Tumor cells covering a highly branched papillary stroma are positive for calretinin (× 200) and HEG1 (× 200) but not for CEA (× 200), and they show the nuclear retention of BAP1 (× 200), MTAP (× 200) and Merlin/NF2 (× 200).
Note: HEG1: Heart development protein with EGF like domains 1, CEA: Carcinoembryonic antigen, BAP1: BRCA1-Associated protein 1, MTAP: Methyl-Thioadenosine Phosphorylase, Merlin/NF2: Merlin/Neurofibromatosis 2.
| Anti-Body | Tumor Tissue |
|---|---|
| Caletinin | Positive |
| HEG1 | Positive |
| CEA | Negative |
| Claudin 4 | Negative |
| BAP 1 | Retained in nuclei of tumor cells |
| MTAP | Retained in nuclei of tumor cells |
| Merlin/NF2 | Retained in nuclei of tumor cells |
Table 2: Immunohistochemistry results in present WDPMT of the peritoneum.
HEG1: Heart development protein with EGF like domains 1 CEA: Carcinoembryonic antigen, BAP1: BRCA1-Associated protein 1, MTAP: Methyl-Thioadenosine Phosphorylase Merlin/NF2: Merlin/neurofibromatosis 2.
A cut surface showing a dilated hernia sac (H&E × 12.5). Swollen reactive mesothelial cells form one or two layers in the inner surface (H&E × 400). Within the hernia sac, a Cellular Nodule (N) is present (H&E × 400). Higher magnification of a part of the cellular nodule consists of many histiocytes and isolated or clustered reactive mesothelial cells (H&E × 200). Reflecting the histology, the cellular nodule contains histiocytes positive for CD68 (× 100), reactive mesothelial cells positive for calretinin (× 100) and BAP1 nuclear retention of histiocytes and reactive mesothelial cells ( ×100).
NHMH: Nodular Histiocytic/Mesothelial Hyperplasia, H&E: Hematoxylin-Eosin stain, BAP1: BRCA1-associated protein 1
Laparoscopically, multiple small nodules are found at the pelvic site, and at the right diaphragm and hepatic lobe. Microscopically, a highly branched papillary stroma covered with a single layer of well-ordered mesothelial cells with little atypia is seen (× 12.5, ×100, × 200).
WDPMT: Well-Differentiated Papillary Mesothelial Tumor
Tumor cells covering a highly branched papillary stroma are positive for calretinin (× 200) and HEG1 (× 200) but not for CEA (× 200), and they show the nuclear retention of BAP1 (× 200), MTAP (× 200) and Merlin/NF2 (× 200).
Discussion
As mentioned above, the patient had NHMH associated with a spermatocele. A similar case has been reported, so far [6,7]. NHMHs are rare reactive lesions consisting of a mixture of histiocytes and reactive mesothelial cells. This disease concept was first established in 1975 [6]. As benign mesothelial hyperplasia named this disease NHMH in 1997, and the name has now become common [8,9]. Used the name nodular histiocytic hyperplasia because the disease was primarily composed of histiocyte proliferation. Regarding localization, rare NHMH showing reactive changes occurs at various sites, including the peritoneum, lungs, and cardiac muscle, although the lungs and cardiac muscle have no mesothelial cells [8, 10-12]. Concerning its etiology or mechanism, NHMH is related to various conditions, including inflammations, infections, and tumors and it has been speculated that adhesion molecules and ligands after mesothelial cell damage and cytokine stimulation occur [5,13]. Reviewed 74 cases of NHMH in 2022 [5]. Accordingly, the age range was wide (1.5 years to 87 years), and the male/female ratio was 32/39, with an unknown 3. The lesions ranged in size from 2 mm to 6.0 cm and were either single or multiple. They occurred in various locations, with lung lesions being associated with rickettsial pneumonia, lung cancer, and lung adenocarcinoma; groin lesions associated with inguinal hernia and spermatocele; pericardium lesions associated with rheumatic pericarditis, coronary artery disease, and pericardial cyst; and pelvic cavity lesions associated with abortion and endometriosis. Histologically, 72 cases showed a predominance of large histiocytes, 1 case showed a predominance of mesothelial cells, and 1 case showed an equal distribution of histiocytes and mesothelial cells, with histiocyte predominance in most case. In the case, which occurred in a man in his 40s, histiocytes predominantly proliferated to a greater extent than mesothelial cells. Although it is only a speculation, it is believed that chronic physical irritation persisted in the inguinal hernia with the spermatic cord tissue, which protruded from the patient’s body surface, causing histiocytes to accumulate around the detached mesothelial cells, resulting in the development of NHMH.
In the present case, the peritoneal lesion was diagnosed as WDPMT according to the new WHO classification (2021). The patient is alive and doing well 10 months after the biopsy. Representative peritoneal mesothelial tumors include AT, WDPMT, MCPM, MIS, diffuse mesothelioma, and localized mesothelioma [1-3]. Their main characteristics are as follows.: AT occurs more frequently in the uterus and the retroperitoneum than in the peritoneum. Histologically, enlarged mesothelial tumor cells grow in a cord-like arrangement between fibrous and hyalinized tissues. WDPMT occurs as single or multiple small nodular lesions in the peritoneum. Histologically, it shows a highly branched papillary pattern of edematous stroma covered by aligned mesothelial cells with little atypia. MCPM forms multiple cysts of various sizes, which are composed of mesothelial cells with little atypia. In MIS, mesothelial cells with nuclear atypia spread on the peritoneal surface with no invasion, and MIS slowly progresses to diffuse mesothelioma [14,15]. However, when MIS grows in a papillary pattern, it may be difficult to distinguish it from WDPMT [16]. In diffuse and localized mesothelioma, mesothelial cells with nuclear atypia proliferate in various patterns [1,3]. When both the tumors grow in a papillary fashion, it can be difficult to determine the presence or absence of invasion if only the superficial components are biopsied, making distinguishing them from WDPMT challenging [1,2]. On the other hand, immunohistochemically, MIS essentially shows nuclear deletion of BAP1 [2], and diffuse mesothelioma and localized mesothelioma demonstrate not only nuclear deletions of BAP1, but also MTAP and NF2, as well as homozygous deletion of p16 (CDK1N2A) using fluorescent in situ hybridization but WDPMT does not. These findings are useful in distinguishing WDPMT from the other three diseases [1,2].
Few reports have been published on the clinicopathological findings of WDPMT reviewed 75 cases [17]. The ages of the patients ranged from 18 years to 69 years (median, 42 years), and the male-female ratio was 58:17. The lesions occurred as a single nodule or multiple miliary lesions in the visceral or parietal peritoneum. Histologically, 67 cases showed typical findings of WDPMT, 6 cases indicated microinvasion, and 1 case revealed malignant transformation. Four cases were associated with AT, 2 cases with MCM, and 2 cases with both AT and MCM. Malpica A et al. reviewed 26 cases of peritoneal WDPMT in women [18]. The ages of the patients ranged from 23 yeras to 75 years (median 47 years). None of the patients had a history of asbestos exposure. In 24 of the 26 cases, the tumor was discovered incidentally during surgery for benign or malignant lesions. In 13 cases, the tumor was found as single or multiple lesions, measuring 0.1cm to 2 cm, and was found anywhere in the peritoneum of the abdominal cavity. Regarding the prognoses, 22 cases were disease-free, 1 recurred after 4 years, and 3 died of other causes in the follow-up period of 5 months to 112 months. Furthermore, an interesting study has been reported on the prognosis. In the hyperthermia registration cases in Sweden, the 5-year disease-free survival period was compared between the peritoneal benign mesothelioma group (WDPMT and MCPM) and the peritoneal malignant mesothelioma group treated with hyperthermia-chemotherapy. The former had a disease-free survival rate of approximately 60%, whereas the latter had a disease-free survival rate of approximately 30% [19]. Regarding malignant transformation of WDPMT, Costanzo L et al in 2014 reported a case as malignant transformation of WDPMT. In the case, a 66-years-old patient developed mediastinal lymph node metastasis and pulmonary infiltration 13 years after the diagnosis of pleural WDPMT, and malignancy was confirmed by biopsy. Hower, because BAP1 immunohistochemical staining was not performed for the pleural lesion and pleural WDPMT was exceptional, whether it was really a WDPTM case is questionable [20].
As already mentioned, the present MDPMT showed immunohistochemical nuclear retention of BAP1, MTAP, and Merlin/NF2, but detailed molecular studies were not performed. Molecular or genetical studies on diffuse mesothelioma and MIS have included detailed analyses using whole exome sequencing [21,22]. Such studies have been rarely performed [23]. Advocated that BAP1 nuclear retention was require to diagnose WDPMT or differentiate it from MI [24]. Analyzed NF2 at 22q12.3 in a female patient who was diagnosed with WDPMT through laparoscopic biopsy and survived for 4.5 years and found LOH. Thus, the authors speculated that this abnormality was an early change in WDPMT. NF2 abnormalities are related not only to malignant mesothelioma but also to its development. However, abnormalities in BAP1 are considered early events in tumor development in diffuse mesothelioma, while abnormalities in NF2 are late events [25]. Based on these findings, it is reasonable to conclude that WDPMT and diffuse mesothelioma have different molecular mechanisms of action. However, research on genetic abnormalities in WDPMT is limited, and further data should be accumulated.
Conclusion
Herein, we reported a case of typical peritoneal WDPMT occurring simultaneously with NHMH of the spermatic cord. The association should be coincidental, as the co-occurrence of the two lesions has not been reported previously. Peritoneal WDPMT is classified as a benign tumor in the new WHO classification. It is important to recognize that peritoneal WDPMT is classified as a benign tumor in the new WHO classification, and WDPMT develops more frequently in women with no history of asbestos exposure, and unlike MIS and mesothelioma, when growth in a distinct papillary pattern does not show nuclear deletions on BAP1, MTAP, or Merlin/NF2 immunostaining.
Data availability
The datasets generated or analyzed during the present study are available from the corresponding author on reasonable request.
Declarations
Conflicts of interest. The authors declare no conflicts of interest.
Funding
None
Ethics approval and consent to participate
All procedures, including the handling of personal information, were conducted in accordance with the ethical standards of the Helsinki Declaration of 1964 and its later versions by the responsible committee at the NHO Fukuyama Medical Center.
Author contribution
HS concerned the conceptualization of the study, study design, data acquisition, data analysis, data interpretation, drafting of the manuscript, critical revision of the manuscript for important intellectual content, and final approval of the version to be published. HS, RO, HY, KN discussed the pathological characterization of the tumor and subsequently arrived at a diagnosis of nodular histiocytic/mesothelial hyperplasia of the spermatic cord and well differentiated papillary mesothelial tumor of the peritoneum. RH made the clinical diagnosis based on the patient’s history, symptoms, and images, and performed the surgery and follow-up.
Acknowledgment
We thank the staff of the Department of Diagnostic Pathology, NHO Fukuyama Medical Center for their support. In addition, the authors thank Editage for proofreading this manuscript.
References
- Sauter JL, Dacic S, Galateau-Salle F, Attanoos RL, Butnor KJ. et al. (2022). The 2021 WHO classification of tumors of the pleura: advances since the 2015 classification. J Thorac Oncol. 17:608–622.
[Google Scholar], [Crossref]
- Churg A, Galateau-Salle F. (2022). Well differentiated papillary mesothelial tumor: a new name and new problems. Mod Pathol. 35:1327-1333.
[Google Scholar], [Crossref]
- Churg A. (2024). New developments in mesothelial pathology. Histopathology 84:136-152..
[Google Scholar], [Crossref]
- Vogin G, Hettal L, Vignaud JM, Dartigues P, Goere D. et al. (2019). Well-developed papillary mesothelioma of the peritoneum: a retrospective study from the RENAPE Observational Registry. Ann Surg Oncol 26:852–860.
[Google Scholar], [Crossref]
- Grech J, Lilley CM, Martinbianco EM, Ding X, Mirza KM. et al. (2022). Nodular histiocytic/mesothelial hyperplasia mimicking mesenteric metastasis. 14:e24971.
[Google Scholar], [Crossref]
- Rosai J, Dehner LP. (1975). Nodular mesothelial hyperplasia in hernia sacs: a benign reactive condition simulating a neoplastic process. 35:165-175.
[Google Scholar], [Crossref]
- Chen HJ, Li DH, Zhang J. (2017). A case of spermatic cord cyst with nodular histiocytic/mesothelial hyperplasia. Asian J Androl 19:505–506.
[Google Scholar], [Crossref]
- Chan JK, Loo KT, Yau BK, Lam SY. (1997). Nodular histiocytic/mesothelial hyperplasia: a lesion potentially mistaken for a neoplasm in transbronchial biopsy. Am J Surg Pathol. 21:658–663.
[Google Scholar], [Crossref]
- Ordonez N, Ro JY, Ayala AG. (1998). Lesions described as nodular mesothelial hyperplasia are primarily composed of histiocytes. Am J Surg Pathol. 22:285-292.
[Google Scholar], [Crossref]
- Chung Y, Abdul R, Jang SM, Choi JS, Jang K. et (2016). Pelvic nodular histiocytic and mesothelial hyperplasia in a patient with endometriosis and uterine leiomyoma. J Pathol Transl Med. 50:397-400.
[Google Scholar], [Crossref]
- Cabibi D, Lo Iacono G, Raffaele F, Dioguardi S, Ingrao S. et al. (2015). Nodular histiocytic/mesothelial hyperplasia as consequence of chronic mesothelium irritation by subphrenic abscess. Future Oncol 11(24 Suppl):51-55.
[Google Scholar], [Crossref]
- Chikkamuniyappa S, Herrick J, Jagirdar JS. (2004). Nodular histiocytic/mesothelial hyperplasia: a potential pitfall Ann Diagn Pathol. 8:115-120.
[Google Scholar], [Crossref]
- Suarez-Vilela D, Lzquierdo-Garcia FM. (2002) Nodular histiocytic/mesothelial hyperplasia: a process mediated by adhesion molecules? 40:299–300.
[Google Scholar], [Crossref]
- Vitlarov N, Burtscher E, Pfeiffenberger E, Wahl A, Hodja V, et al. (2023) Peritoneal papillary mesothelioma in situ: BAP1 mutation with indolent behavior for 15 years. Virchows Arch. 483:873-878.
[Google Scholar], [Crossref]
- Churg A, Galateau-Salle F, Roden AC, Attanoos R, von der Thusen JH, et al. (2020) Malignant mesothelioma in situ: morphologic features and clinical outcome. Mod Pathol 33:297-302.
[Google Scholar], [Crossref]
- Galateau-Salle F, Hamilton T, MacNeill A, Hofman V, Sequeiros R, et al. (2023) Mesothelioma in situ mimicking well-differentiated papillary mesothelial tumor. Am J Surg Pathol 47:611-617.
[Google Scholar], [Crossref]
- Sun M, Zhao L, Lao W, Yu L, Wang J, et al. (2019) Well-differentiated papillary mesothelioma: a 17-year single institution experience with a series of 75 cases. Ann Diagn Pathol 38:43-50.
[Google Scholar], [Crossref]
- Malpica A, Sant'Ambrogio S, Deavers MT, Silva EG. (2012) Well-differentiated papillary mesothelioma of the female peritoneum: a clinicopathologic study of 26 cases. Am J Surg Pathol 36: 117–127.
[Google Scholar], [Crossref]
- Cashin PH, Jansson Palmer G, Asplund D, Graf W, Syk I, et al. (2019) Peritoneal mesothelioma in Sweden: a population-based study. Cancer Med 8:6468-6475.
[Google Scholar], [Crossref]
- Costanzo L, Scarlata S, Perrone G, Rossi L, Papa A, et al. (2014) Malignant transformation of well-differentiated papillary mesothelioma 13 years after the diagnosis: a case report. Case Rep Clin Respir J. 8:124-129
[Google Scholar], [Crossref]
- Guo G, Chmielecki J, Goparaju C, Heguy A, Dolgalev I, et al. (2015) Whole-exome sequencing reveals frequent genetic alterations in BAP1, NF2, CDKN2A, and CUL1 in malignant pleural mesothelioma. Cancer Res. 75:264-269.
[Google Scholar], [Crossref]
- Dacic S, Roy S, Lyons MA, von der Thusen JH, Galateau-Salle F, et al. (2020) Whole exome sequencing reveals BAP1 somatic abnormalities in mesothelioma in situ. Lung Cancer 149:1-4.
[Google Scholar], [Crossref]
- Hassan A, Prabhakaran S, Pulford E, Hocking AJ, Godbolt D, et al. (2024) The significance of BAP1 and MTAP/CDKN2A expression in well-differentiated papillary mesothelial tumour: a series of 21 cases and a review of the literature. Pathology. 56:662-670.
[Google Scholar], [Crossref]
- Nemoto H, Tate G, Kishimoto K, Saito M, Shirahata A, et al. (2012) Heterozygous loss of NF2 is an early molecular alteration in well-differentiated papillary mesothelioma of the peritoneum. Cancer Genet. 205:59.
[Google Scholar], [Crossref]
- Zhang M, Luo JL, Sun Q, Harber J, Dawson AG, et al. (2021) Fennell DA. Clonal architecture in mesothelioma is prognostic and shapes the tumour microenvironment. Nat Commun. 12:1751.
[Google Scholar], [Crossref]
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi