Research Article, Clin Oncol Case Rep Vol: 1 Issue: 1
A Sole Cytogenetic Abnormality r (2) in Acute Lymphocytic Leukemia Patient: An Indian Experience
Patel Dharmesh M, Trivedi Pina J, Brahmbhatt Manisha M and Patel Prabhudas S*
Cytogenetic Lab, Department of Cancer Biology, Gujarat Cancer & Research Institute, Asarwa, Ahmedabad-380016, India
*Corresponding Author : Dr. Prabhudas S. Patel
Professor and Head, Department of Cancer Biology The Gujarat Cancer & Research Institute, Asarwa, Ahmedabad-380016, India
Tel: (O) 79-22688479
Fax: 79-22685490
E-mail: prabhudas_@hotmail.comp
Received: January 19, 2018 Accepted: April 26, 2018 Published: April 30, 2018
Citation: Dharmesh PM, Pina T, Manisha BM, Prabhudas PS (2018) A Sole Cytogenetic Abnormality r (2) in Acute Lymphocytic Leukemia Patient: An Indian Experience. Clin Oncol Case Rep 1:1.
Abstract
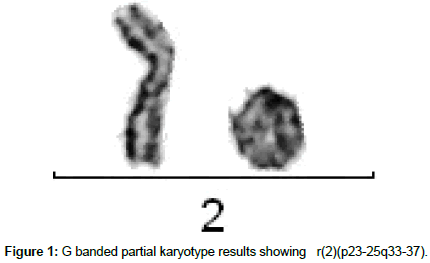
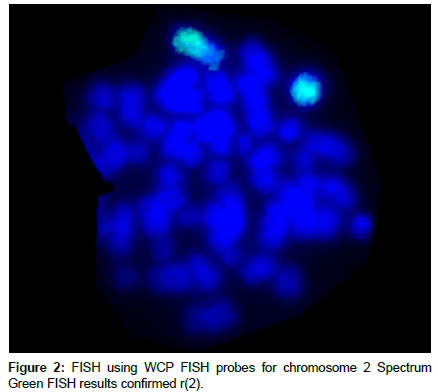
Cytogenetic abnormalities are established as important prognostic factor for Leukemia. After the first report of a ring chromosome in a case of human leukemia ring chromosomes have been infrequently (<10%) detected in hematopoietic neoplasias. However, ring chromosomes are part of complex karyotype associated with a poor prognosis. In this study we document a case of 21 years old Acute Lymphoblastic Leukemia (ALL) patient. Short term unstimulated bone marrow (BM) and peripheral blood lymphocyte cultures were performed. The karyotype report was 46, XY, r (2)(p23-25q33-37). Fluorescence in-situ hybridization using WCP for chromosome 2 confirmed presence of ring chromosome 2. In most cases, there are no confirmatory reports on an association between a specific leukemia subtypes with ring chromosome. This case report may constitute a distinctive subtype. Further analysis of such cases is required to define complete disease entity.
Keywords: Acute Lymphocytic Leukemia; Ring chromosome; Cytogenetics; Fluorescence in Situ Hybridization; Whole Chromosome paint Probe
Introduction
Acute lymphoblastic leukemia (ALL) is a neoplastic disease characterized by clonal expansion of leukemic cells in the bone marrow (BM), lymph nodes, thymus, or spleen. ALL is a genetic disease because essentially most patients harbor acquired genetic alterations (somatic mutations) that contribute to the increased proliferation, prolonged survival, and/or impaired differentiation of the lymphoid hematopoietic progenitors. In the majority, albeit not all, patients diagnosed with ALL, one or more of these genetic alterations are in the form of nonrandom numerical or structural chromosome aberrations that can be detected microscopically [1].
Identification and understanding the underlying genetic abnormalities in ALL play significant role, not only in the diagnosis but also in predicting the prognosis and, ultimately, understanding the leukemogenesis [2]. The cytogenetic abnormalities have been established to be very important for the prognosis of ALL patients [3]. Approximately 75% of childhood ALL cases harbor recurrent genetic abnormalities, including aneuploidy or structural chromosomal arrangements, detected by conventional karyotyping and fluorescence in situ hybridization (FISH) [4]. Translocations t(9;22)(q34;q11) [BCR-ABL1], t(12;21)(p13;q22) [ETV6-RUNX1 (TEL-AML1)], hyperdiploidy, and translocation t(4;11)(q21;q23) [MLL-AFF1(AF4)] in infants, are found at the highest frequency in childhood B-ALL. Other recurrent cytogenetic abnormalities include hypodiploidy and translocation t (1; 19)(q23;p13) [TCF3-PBX1 (E2A-PBX1)] [5]. The ring chromosome is a circular, structural abnormality composed of either multiple chromosomes or a single chromosome with loss of genetic material at telomeres one or both ends. This chromosomal rearrangement is often unstable with frequent recombination and may be accompanied by either loss or amplification of genetic material [6]. Considering that ring chromosomes are rare in ALL, it is difficult to risk stratify patient prognosis, particularly when the ring chromosome occurs as the sole abnormality.
We report here the first case of ring chromosome 2 as sole abnormality in ALL patient. The karyotype report was 46,XY,r(2) (p23-25q33-37) it shows presence of ring chromosome 2, which was confirmed using WCP FISH for chromosome 2 in all examined metaphase plates. Patient was expired within 6 months of diagnosis which indicates that there might be loss of some tumor suppressor genes from telomeric region of chromosome 2 which are responsible for leukemogenesis and shows poor prognosis.
Case History
A 21 years old male child with complaints of high grade fever was registered at our institute. Peripheral blood report revealed hemoglobin level 10.4 gm/dl, white blood cell count 18000/uL, blast cells 90%, and platelet count 33000/uL. Bone marrow aspiration showed marked proliferation of tumor blast cell population. Blast cells were medium to large in size, having high N:C ratio, round to oval nuclei, clumped nuclear chromatin, occasional 0-2 nucleoli with scanty cytoplasm. Sudan Black-B was negative, and periodic acid Schiff (PAS) was block positive, M:E ratio was altered, and megakaryocytes were not seen. Final diagnosis based on morphological findings was ALL-L2. Immunophenotype report was T-cell ALL. The patient received chemotherapy of standard protocol MCP 841 for ALL. The patient expired during 5th day of 1st induction period of treatment after 18 days of diagnosis.
Materials and Method
Conventional cytogenetic
Unstimulated culture of bone marrow aspirate was set up in RPMI-1640 medium supplemented with 20% Newborn Calf Serum, L-glutamine, and antibiotics (penicillin and streptomycin). The cells were cultured in incubator at 37°C followed by overnight incubation in presence of Colcemid (10 μl/8 ml of culture). The cultures were exposed to hypotonic solution (0.075 mol/L KCl) and fixed with Methanol:Acetic acid (3:1). The slides were prepared by air dry method and banding was carried out using Giemsa Trysin-EDTA as per standard protocol [7]. Twenty metaphases were analyzed and karyotyped. Karyotypes were described according to the International System for Human Cytogenetics Nomenclature 2013 [8].
FISH assay
FISH was performed on metaphase cells following the manufacturer’s guidelines (Abbott Molecular, Inc., Des Plaines, IL, USA). Whole Chromosome Painting for chromosome 2 (WCP 2) with Spectrum Green (SG) was used to confirm the karyotype results. The analysis of both conventional cytogenetics and WCP-FISH was carried out using BX-61 Olympus fluorescence microscope (Olympus, Japan) equipped with CCD camera.
Results
Conventional cytogenetics
Conventional cytogenetic results detected an abnormal male chromosome complement. The karyotype results revealed 46, XY, r(2)(p23-25q33-37). It shows presence of ring chromosome 2 in all analyzed metaphase plates (Figure 1).
FISH results with WCP 2 FISH probe
The metaphase FISH results of WCP FISH for chromosome 2 with SG showed formation of ring for chromosome 2 in all metaphases (Figure 2).
Discussion
Chromosomes are often regarded merely as static containers for genetic information. However, it is now becoming increasingly clear that chromosomes are highly dynamic structures with a tightly regulated organization. Normal DNA replication results in two linear sister chromatids organized in a parallel configuration so that symmetrical separation can occur at the metaphase-anaphase transition. However, a change in topology from linear to circular may totally disrupt this sequence of events. This is an attempt to delineate the consequences of such ring-shaped chromosomes in human cells. Previous studies have demonstrated that the initial formation of ring and dicentric chromosomes in malignant tumors can result from telomere shortening and subsequent fusion of chromosome ends [9].
The incidence of cases with ring chromosomes is generally low in human hematopoietic neoplasias, rather than in ALL it is about 3.4%. Among a consecutive series of 152 childhoods ALL, however, only one case with a ring chromosome was found [10]. Ring chromosome is often unstable, leading to concomitant genetic loss or amplification. This may explain why ring chromosome is extremely rare in hematopoietic malignancies [11].
In the present study out of 273 ALL patients; we have observed a case with ring chromosome 2. Literature reported that chromosome 2 was involved in less than 1% cases, of all rings formation cases in ALL [6]. In leukemias, for instance, the presence of ring chromosomes seems to be associated with poor prognosis. In some tumor sub entities, they are so frequent and typical that they can even serve as cytogenetic hallmarks for differential diagnosis [6]. In the present study, 0.6 month survival indicates shorter survival and poor prognosis. In Mitelman database, only one case described with r (2) as sole abnormality in 1996 by Martinue et al. [12]. Hence, the case is considered as rare finding [13].
Ring chromosomes come to clinical attentions either in association with developmental anomalies at the beginning of life or with telomere shortening in ageing and neoplastic cells. It is likely that a transition from a DNA-damage sensitive to a DNA-damage tolerant state explains the high instability of rings in some tumour cells compared to those in non-neoplastic cells. However, many tumours show a pattern similar to that of normal cells. Two main modes for chromosomal reorganization in tumours. In type 1 non-neoplastic cells, ring chromosome structure is relatively conserved: rings may be duplicated or lost, but structural rearrangements are rarely maintained in the cell population [14]. In type 2, rings show extensive structural variability and provide a means for gene amplification. Ring chromosomes are thus illustrative proofs that chromosomal behavior is not only a function of straightforward molecular interactions; chromosomal topology and the physiological context in which a certain chromosome aberration occurs must also be taken into consideration [14].
Conclusion
The case reported here together with conventional karyotype and WCP FISH (Molecular cytogenetics) finding highlights the importance of WCP FISH for patients with ring chromosomes. In accordance to literature our result exhibit that ring chromosomes are associated with a poor prognosis. It also showed a concordance between FISH results and G-banding analysis. Application of FISH on metaphases permitted to increase the potentials of conventional cytogenetics.
References
- Krzysztof M, David PH, Peter D (2009) Aplan Cytogenetics and Molecular Genetics of Acute Lymphoblastic Leukemia. Hematol Oncol Clin North Am 23: 991-1011.
- Downing JR, Harrison CJ (2009) Cytogenetics of pediatric and adolescent acute lymphoblastic leukemia. Br J Haematol 144: 147-156.
- Siddaiahgari SR, Awaghad MA, Latha MS (2015) Clinical, immunophenotype and cytogenetic profile of acute lymphoblastic leukemia in children at tertiary health care centre in India. Muller J Med Sci Res 6: 112-118.
- Mullighan CG (2012) The molecular genetic makeup of acute lymphoblastic leukemia. Hematol Am Soc Hematol Educ Program 2012: 389–396.
- Jennifer SW, Michael OA, Carlos AT (2014) Childhood B-acute lymphoblastic leukemia: a genetic update Exp Hematol Oncol 3: 16.
- Gebhart E (2008) Ring chromosomes in human neoplasias. Cytogenet Genome Res 121:149-173.
- Verma RS, Babu A (1995) (eds) Human Chromosomes, Manual of Basic Techniques. Pergamon Press. London, UK.
- Shaffer LG, McGowan-Jordan J, Schmid M, editors. ISCN (2013) an international system for human cytogenetic nomenclature Karger Medical and Scientific Publishers.
- Gisselsson D, Jonson T, Peterson A, Strmbeck B, Dal Cin P, et al. (2001) Telomere dysfunction triggers extensive DNA fragmentation and evolution of complex chromosome abnormalities in human malignant tumors. Proceedings of the National Academy of Sciences of the United States of America. 98: 12683-12688.
- Andreasson P, Höglund M, Bekassy AN, Garwicz S, Heldrup J, et al. (2000) Cytogenetic and FISH studies of a single center consecutive series of 152 childhood acute lymphoblastic leukemias. Eur J Haematol 65: 40–51.
- Adachi M (2012) Ring chromosome derived from BCR/ABL in an acute lymphocytic leukemia. Int J Hematol 1: 1-2.
- Martineau M, Clark R, Farrell DM, Hawkins JM, Moorman AV, et al. (1996) Isochromosomes in acute lymphoblastic leukaemia: i(21q) is a significant finding. Genes Chromosomes Cancer 17: 21-30.
- Mitelman Database of Chromosome Aberrations in Cancer (2000).
- Gisselsson D, Pettersson L, Hˆ glund M, Heidenblad M, Gorunova L, et al. (2000) Chromosomal breakage-fusion-bridge events cause genetic intratumor heterogeneity. Proc Natl Acad Sci U S A 97: 5357-5362.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi