Research Article, J Fashion Technol Textile Eng S Vol: 0 Issue: 5
A Study of Electrospinning and Characterization of Poly (Æ-caprolactone) Nanofibers
Riaz T1,2*, Delaite C1, Khenoussi N2, Adolphe D2 and Schacher L2
1LPIM, University of Haute Alsace, 3 bis rue Alfred Werner 68093 Mulhouse, France
2LPMT, University of Haute Alsace, 11 rue Alfred Werner 68093 Mulhouse, France
*Corresponding Author : Tabinda Riaz
Doctorant LPIM/LPMT, University of Haute Alsace, 3 bis rue Alfred Werner 68093 Mulhouse, France
E-mail: Tabinda.riaz@uha.fr
Received: September 19, 2017 Accepted: October 08, 2018 Published: October 13, 2018
Citation: Riaz T, Delaite C, Khenoussi N, Adolphe D, Schacher L (2018) A Study of Electrospinning and Characterization of Poly (ÆÂ-caprolactone) Nanofibers. J Fashion Technol Textile Eng S5:005. doi:10.4172/2329-9568.S5-005
Abstract
Nanofibers of poly (Ɛ-caprolactone) (PCL) were produced by electrospinning a 10 wt% solution of PCL/Chloroform and ethanol at room temperature. For electrospinning the voltage was varied from 15-30 kV in seven regular intervals while keeping the feed rate (0.5 ml/h) and needle tip to collector distance (25 cm) constant at ambient atmospheric conditions (22 ± 2�?C and 40% R.H). This led to the study that how voltage variations effect the final morphology and diameter of nanofibers. SEM micrographs of the samples elaborated their morphology as heterogeneous and homogeneous mixing of nanofibers and their diameters ranging from 90-200 nm. Moreover, the crystallinity ratio (C.R) and thermal behavior of pure PCL and its electrospun nanofibers were studied using Differential Scanning Calorimeter (DSC). Results showed that the Tg of pure PCL and of nanofibers was same as -63.8�?C and the crystallinity ratio of pure PCL was 40% that increased to 50% after electrospinning. A viscosity analysis was done for different solvents of PCL taking 10 wt% PCL for each. Chloroform, THF, acetone, o-xylene and a combination of chloroform with ethanol that showed chloroform with the highest viscosity as 3645 mPa.s and o-xylene with the lowest as 331 mPa.s. The rheological study of these solvents helped in determining the appropriate solvent for PCL electrospinning. So, a combination of chloroform and ethanol was selected as the best choice as it has the optimum viscosity of 425 mPa.s with 10 wt% PCL allowing having higher concentrations of PCL with good solubility for electrospinning.
Keywords: Electrospinning; Morphology; Nanofibers; Scanning electron microscopy
Introduction
The idea of using an electric field to spin fibers from a charged polymer melt or solution dates back to 1930s. This process was termed as electrospinning which was a simple method to manufacture the nanofibers from polymer solutions to a wide submicron range of diameters around 100 nm [1-5]. The operating principle of the process involves the application of electric field (10-30 kV) to a polymer solution jet coming from a needle hole and a grounded collector surface. As the polymer jet travels under applied electric field, it is stretched and polymer chains are shaped thus producing the fine nanofibers having versatile properties. On the way solvent evaporates and the charged fibers are collected on the collector surface. Charged polymer solution forms a conical shape at the tip of needle, called Taylor Cone. Nanofibers are formed when electric charge at the needle tip overcomes the surface tension and viscosity. Due to their unique surface area per volume ratio these nanofibers is of high interest in their biomedical applications like drug delivery systems, wound dressings, scaffolds and tissue engineering.
The structure of nanofibers is governed by different variable that are interrelated including the electrospinning conditions (applied voltage, needle to collector distance, feed rate), molecular chain length of polymer, attributes of solution (concentration, solvent nature, viscosity of solution). All these factors are to be studied and optimized in order to get the desired diameters, morphology and mechanical properties of resultant fibers.
Among all the parameters applied voltage is one of the most important factors that control the molecular structure of nanofibers. As for the applied voltage, the Coulombic forces, at low voltages are not enough to overcome surface tension of the polymer solution which results in droplets or bead formation instead of nanofibers. When voltage increases, the surface tension and viscolelastic forces begin to be balanced thus forming a straight stable charged jet that produces fine fiber diameter distribution. But if the applied voltage keeps on increasing sometimes Coulombic forces overcome the viscoelastic forces, breaking the charged jet on its way thus yielding coarser diameters [6].
So, not only the suitable solvent and solution viscosity influences the final structure of nanofibers but also the suitable applied voltage plays a vital role in shaping the morphology of spun fibers as well as their crystallinity and tensile properties [7].
In past few decades, biodegradable and biocompatible polymers have become of significant attentions because they are environmentally benign so are used extensively in biomedical applications. This is the reason that poly (ÆÂ-caprolactone) (PCL) has got major score as a promising biopolymer and it can be used in many medical applications. Its long degradation time, bioactivity and stable nature have been documented by many researchers. PCL is semi-crystalline and hydrophobic polymer that is soluble in most of the organic solvents and is FDA approved with no cytotoxicity. It has good mechanical behavior and tensile properties that make it a good candidate for electrospinning material [8].
The aim of this study was to determine a good solvent for the PCL to make a suitable solution for electrospinning with an appreciable viscosity and to investigate the effect of applied voltage on the nanofiber diameter and morphology. Many different solvents like acetone, THF, chloroform, a combination of chloroform and ethanol and o-xylene were tried and tested for their rheological behavior. Thus on the basis of results a combination of chloroform and ethanol was selected for a 10 wt% concentration of PCL. At the electrospinning part, applied voltage was selected as variable and other parameters like concentration, needle to collector distance and feed rate were kept constant. A set of seven samples was prepared by applying seven different voltages (15 kV, 18 kV, 20 kV, 23 kV, 25 kV, 28 kV and 30 kV) with a needle to collector distance from 25 cm and fed rate was 0.5 ml/h.
Experimental Procedures
Materials and methods
Poly (ÆÂ-caprolactone) (PCL) with molecular weight of 80,000 g/ mol was purchased from Sigma-Aldrich and was used as received. Chloroform stabilized with ethanol (Carlo Erba with purity>99.0%), THF stabilized with BHT (Carlo Erba with purity>99.5%), Acetone and Ethanol as reagent grade (Honeywell with purity>99.5%), o-xylene reagent grade (Sigma-Aldrich with purity>98.0%) were used without any further purification. A 30 ml sample was prepared for electrospinning by taking 10 wt% PCL dissolved in Chloroform with further addition of 3 ml of ethanol to this solution at room temperature. To get a homogeneous solution it was left on an agitator overnight at moderate agitation. For viscosity analysis 10 ml samples were prepared with 10 wt% PCL concentration and in case of acetone, THF and o-xylene solutions were agitated and heated at a temperature range of 40-50˚C for almost 3-4 h to get a clear solution.
Electrospinning process
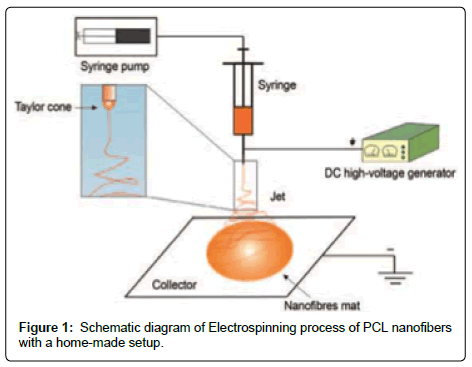
A home-made vertical electrospinning apparatus was used for the experiments in this research. 20 ml solution was loaded to a plastic syringe with a metallic needle of 14 gauge. A syringe pump was employed to maintain a uniform polymer solution feed rate and a high voltage source (0-30 kV) was connected to the enclosed electrospinning chamber to supply a controlled electric field between the needle tip to the grounded collector made of 20×20 cm Teflon plate covered with aluminum foil. All the experiments were carried out at room temperature (22 ± 2˚C) and relative humidity was almost 40%. The needle to collector distance (D) was set at 25 cm and solution feed rate was 0.5 ml/h. seven different voltages as 15, 18, 20, 23, 25, 28 and 30 kV were applied to have seven different samples of electrospun nanofibers. The electrospinning time for each sample was 15 min. A schematic diagram of electrospinning process is shown below in Figure 1.
Characterization
The diameter and morphology of electrospun fibers was explained by using SEM micrographs. Image J software was used to measure the diameters of nanofibers. For the viscosity measurements the Anton Par MCR302 rheometer was used with a constant shear rate of 100 s-1 for 20 points. In order to analyze the melting point and crystallinity of pure PCL as well as produced nanofibers DSC-Q200 equipment was employed.
Results and Discussion
Viscosity analysis
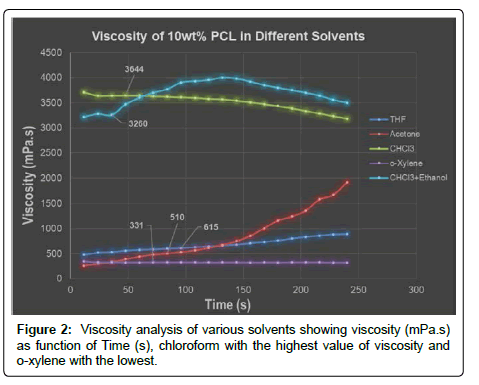
All of the PCL samples (PCL/Chloroform, PCL/THF, PCL/ Acetone, PCL/o-xylene and PCL/Chloroform+ethanol) with 10 wt% concentration showed clear and transparent behavior. Plate-plate rheological measurements showed that all solutions showed a non-Newtonian behavior under shear rate of 100 s-1 and indicates viscosity as a function of time and a gradual increase in viscosity after some time that is due to the evaporation of solvent [9].
It is clear from the rheograms in Figure 2 that PCl with only chloroform and a combination of chloroform and ethanol showed the highest viscosity at 3645 mPa.s and 3200 respectively. Whereas, o-xylene has the lowest value at 331 mPa.s. This shows that chloroform is a good solvent for PCL as polymer chains occupy more hydrodynamic volume and vice versa. But at the same time, this high viscosity at only 10 wt% of PCL also limits the possibility of electrospinning with higher concentration as 12-20 wt%. Acetone and THF exhibit a good viscosity behavior with low viscosity values. It showed a possibility that with these solvents more concentrations could be tried for electrospinning for different morphologies and fiber diameters. So, chloroform with a little amount of ethanol was selected for the electrospinning of PCL as it gives the required honey like viscosity needed for the aid process.
SEM and effect of voltage on nanofibers diameters
The nanofibers in the electrospinning are formed under the impact on applied voltage on the polymer solution. Thus, voltage eventually affects the morphology of nanofibers in terms of chain alignments and polymer chain stretching. As the voltage changes, it affects the shape of Taylor Cone at the tip of spinneret and the velocity of polymer jet. In literature researchers observed an irregular morphology and larger fiber diameters with increasing voltage [10].
In this study, the applied voltage seemed to have an influence on both nanofiber diameter and morphology. In Figure 3, it can be clearly seen from the SEM images that voltage change has shaped the nanofiber morphology and depicts a change in diameters. For a fixed needle to collector distance of 25 cm and feed rate 0.5 ml/h, 10 wt% PCL nanofibers have a diverse set of diameters at voltages 18,20,23 and 25 kV with a heterogeneous network of nanofibers from very fine thread like appearance to average normal diameters. For these samples diameters ranges from 90-200 nm. This can be proved from their coefficients of variation (CV%) that showed the maximum value of 30 at 18 kV voltage which is obvious in the SEM image in Figure 3. On the other hand, images of higher voltages 28 and 30 kV showed a uniform morphology with diameters 114107 nm with lower CV% as 11 and 18 respectively. This fact is directly related to their visual presentation in SEM micrographs. The largest diameter value was observed as 204.4 nm at 20 kV with CV% as 15. All these results verify that applied voltage has a notable impact on electrospun nanofibers in terms of polymer solution conductivity and molecular chain alignment (Table 1).
| Voltage (kV) | Diameters (nm) | CV% |
|---|---|---|
| 15 | 157.5 | 14% |
| 18 | 91.8 | 30% |
| 20 | 204.4 | 15% |
| 23 | 172.2 | 19% |
| 25 | 153 | 23% |
| 28 | 114 | 11% |
| 30 | 107.9 | 18% |
Table 1: Nanofiber diameters and applied voltage.
Thermal properties and crystallinity ratio
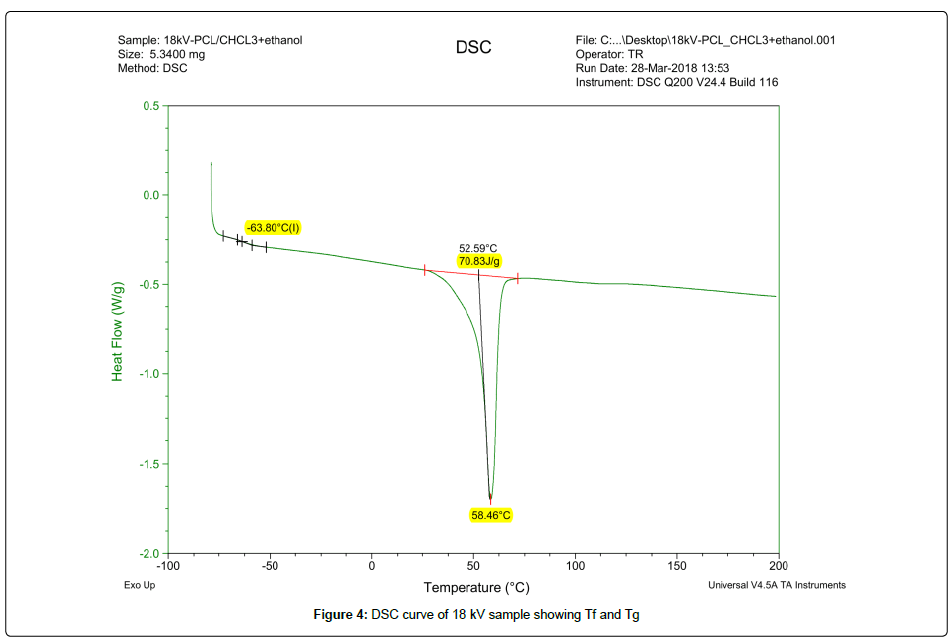
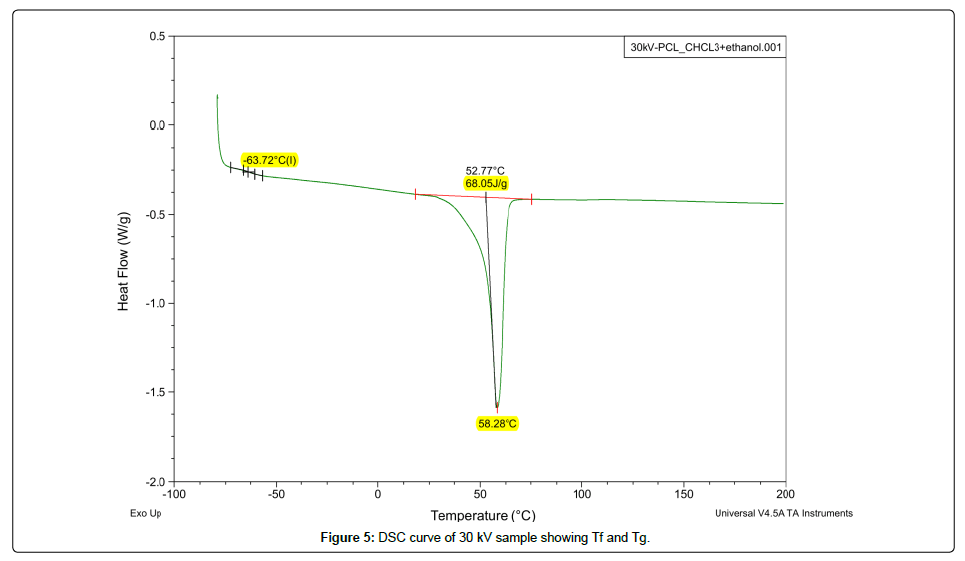
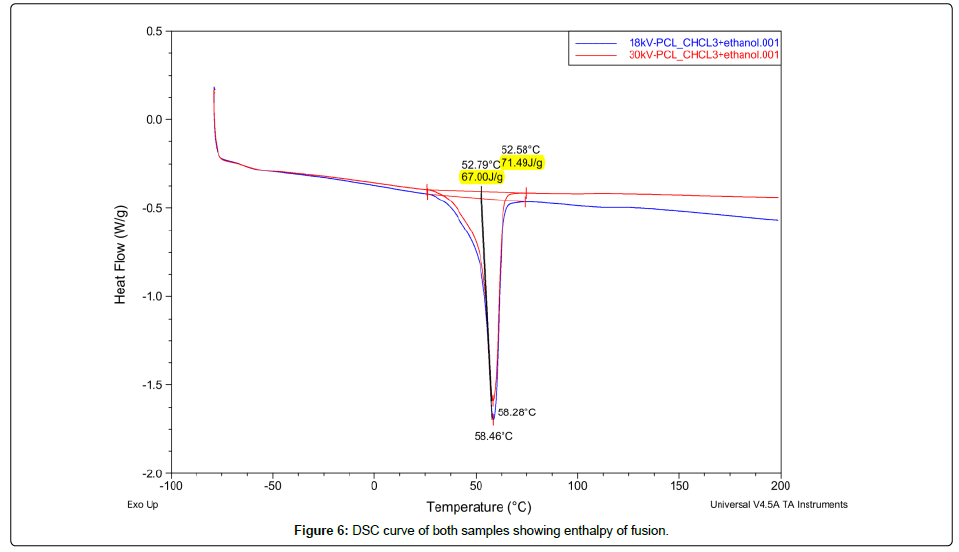
Thermal behavior of pure PCL and its nanofibers was analyzed by using DSC-Q200 for a temperature range of -50˚C to 200˚C. Two electrospun samples of voltage 18 kV and 30 kV were selected to study their thermal properties. These samples represented both morphologies of nanofibers and their thermograms can be observed in Figures 4-6. DSC analysis of pure PCL showed a glass transition temperature (Tg) of -63.80˚C and crystallinity ratio 40%. Both the samples at 18 kV and 30 kV had the same Tg like pure PCL and their melting points (Tf) were almost same at 58.28˚C and 58.46˚C but their crystallinity ration was increased to 48% and 50% respectively. Crystallinity ration was calculated by dividing the enthalpy of fusion of nanofibers by the enthalpy of 100% crystalline PCL that is almost 142 J/g. A rise in crystallinity ratio of electrospun PCL as compared to pure is an indication to more aligned polymer chains after facing voltage during electrospinning that leads to higher crystallinity [11].
Conclusion
PCL electrospun nanofibers were successfully achieved with diameters in range of 90-200 nm by applying different voltages keeping other parameters constant. After analyzing this effect, it can be stated that applied voltage plays an important role in shaping the morphology and diameter of nanofibers. But it also depends upon the polymer solution viscosity and obviously the correct solvent selection.
Chloroform and ethanol with 10 wt% PCL worked as a good polymer solution with viscosity of 3200 mPa.s that worked well to produce uniform jets of nanofibers. Thermal properties like Tf and Tg showed the similar trend as of pure PCL for both samples of nanofibers. But the crystallinity ratio was increased by 10% after electrospinning of PCL. To conclude it is clear about this study that solvent nature and voltage has significant effect on morphology of nanofibers and physical properties like crystallinity. This could be very interesting in finalizing the application of such nonwoven nano structures of different fields of science as smart drug delivery systems, wound dressings or tissue engineering.
References
- Reneker DH, Yarin AL, Fong H, Koombhongse S (2000) Bending instability of electrically charged liquid jets of polymer solutions in electrospinning. J Appl Phys 87: 4531–4548.
- Ma QA, Cebe P (2010) Phase structure of electrospun poly (trimethylene terephthalate) composite nanofibers containing carbon nanotubes. J Therm Anal Calorim 102: 425–434.
- Reneker DH, Chun I (1996) Nanometre diameter fibres of polymer, produced by electrospinning. Nanotechnology 7: 216–223.
- Qin XH (2010) Structure and property of electrospinning PAN nanofibers by different preoxidation temperature. J Therm Anal Calorim 99: 571-75.
- Stephens JS, Frisk S, Megelski S, Rabolt JF, Chase DB (2001) Real time Raman studies of electrospun fibers. Appl Spectrosc 55: 1287-1290.
- Wannatong L, Sirivat A, Supaphol P (2004) Effects of solvents on electrospun polymeric fibers: preliminary study on polystyrene. Polym Int 53: 1851–1859.
- Luo CJ, Stride E, Edirisinghe M (2012) Mapping the influence of solubility and dielectric constant on electrospinning polycaprolactone solutions. Macromolecules 45: 4669-4680.
- Woodruff MA, Hutmacher DW (2010) The return of a forgotten polymer-Polycaprolactone in the 21st century. Prog Polym Sci 35: 1217-1256.
- Nezarati RM, Eifert MB, Cosgriff-Hernandez E (2013) Effects of humidity and solution viscosity on electrospun fiber morphology. Tissue Eng Part C Methods 19: 810–819.
- Haider A, Haider S, Kang IK (2015) A comprehensive review summarizing the effect of electrospinning parameters and potential applications of nanofibers in biomedical and biotechnology.
- Zong X, Kim K, Fang D, Ran S, Hsiao BJ, et al. (2002) Structure and process relationship of electrospun bioabsorbable nanofiber membranes. Polymer 43: 4403–4412.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi