Case Report, Clin Oncol Case Rep Vol: 5 Issue: 3
Advanced Alveolar Soft Part Sarcoma with CNS Metastases Response after Treatment with Atezolizumab: A Case Report
Denaly Chen1*, Shefali Chopra2, Philip Cheng3, and James S Hu1*
1Division of Oncology, Norris Comprehensive Cancer Center, University of Southern California, Los Angeles, CA
2Department of Pathology, Keck Hospital of University of Southern California, Los Angeles, CA, USA
3Department of Radiology, Keck Hospital of University of Southern California, Los Angeles, CA, USA
*Corresponding Author:
Denaly Chen
Division of Oncology,
Norris Cancer Center, University of Southern California,
USA
E-mail: Denaly.chen@med.usc.edu
Received: February 09, 2022, Manuscript No: COCR-22-53959;
Editor assigned: February 14, 2022, PreQC No: P-53959;
Reviewed: March 25, 2022; QC No: Q-53959;
Revised: March 29, 2022, Manuscript No: R-53959;
Published: April 03, 2022, DOI: 10.4172/cocr.5(3).220
Citation: Chen D, Chopra S, Cheng P, Hu JS (2022) Advanced Alveolar Soft Part Sarcoma with CNS Metastases Response after Treatment with Atezolizumab: A Case Report. Clin Oncol Case Rep 5:3
Abstract
Alveolar Soft-Part Sarcoma (ASPS) is a rare subtype of Soft Tissue Sarcoma (STS). The median age of ASPS patients is 25 years old. Conventional chemotherapy treatments are ineffective in ASPS, and thus, new treatments focus on targeted therapies and immunotherapies. Our patient is a 49-year-old male who presented with right buttock ASPS with diffuse pulmonary and brain metastases. His lesions showed a remarkable response to atezolizumab despite having advanced ASPS with metastatic disease to his lungs and brain. Further study into atezolizumab acting as a first line treatment option for metastatic ASPS is ongoing.
Keywords: Alveolar soft part sarcoma; Atezolizumab; CNS metastases; PD-1 inhibitors
Introduction
Alveolar Soft-Part Sarcoma (ASPS) is a rare subtype of Soft Tissue Sarcoma (STS), representing less than 1% of all diagnosed cases of STS. ASPS patients have a median age of 25 years old and there is a slight female predominance [1]. Clinically, cases typically involve an intramuscular or perimuscular thigh mass though cases have also been reported in other organs including the mediastinum, stomach, breast, uterine cervix, head and neck and other organ systems [2-10]. Although the tumor is characterized by indolence, there is a paradoxically high metastatic rate that include the central nervous system. Symptoms from metastatic disease, are often the first signs leading to diagnosis [11]. Conventional chemotherapy agents, such as anthracyclines or platinum based therapies, were found to have no response in ASPS [11,12]. Thus, treatment for ASPS has centered around surgical resection and/or Vascular Endothelial Growth Factor Receptor (VEGFR)-directed Tyrosine Kinase Inhibitors (TKIs) and immunotherapies [13]. In this case, we discuss a patient in which atezolizumab, a monoclonal antibody of IgG1 isotype against the protein Programmed Cell Death-Ligand 1 (PD-L1), resulted in a pronounced response in ASPS metastatic to the central nervous system with significant regression of brain and lung metastases.
Case Presentation
A 49-year-old white male presented for evaluation after a recent diagnosis of alveolar soft part sarcoma of the right buttock with extensive pulmonary nodular metastatic disease. A Positron Emission Tomography/Computed Tomography (PET/CT) scan done from skull base to mid-thigh showed multiple bilateral hypermetabolic pulmonary nodules with a right upper lobe nodule measuring 1.8 cm, a right upper lobe 2.2 cm lesion as well as pulmonary nodule in the left upper lobe measuring 2.8 cm, and a pulmonary nodule in the lingular segment measuring 3.8 cm with an SUV of up to 7. There was also a 4.9 cm × 3.2 cm soft tissue tumor mass in the right gluteus medius muscle with an SUV of 4.55 (Figure PET Axial Pelvis). Magnetic Resonance Imaging (MRI) of the brain noted numerous enhancing supratentorial anxd infratentorial nodules with surrounding vasogenic edema consistent with metastatic disease. The largest infratentorial lesion within the right cerebellar hemisphere measured 1.1 cm × 1.0 cm × 1.2 cm (Figure MR Brain). Diagnosis of alveolar soft part sarcoma was confirmed by biopsy of a left lung nodule as well as a CT guided needle biopsy of the right gluteal mass showed polygonal cells with eosinophilic granular cytoplasm, vesicular nucleus arranged in nest like pattern with a rich capillary vasculature (Figure 1a and Figure 1b). There was diffuse nuclear positivity for TFE3 supporting the diagnosis. (Figure 1c.)
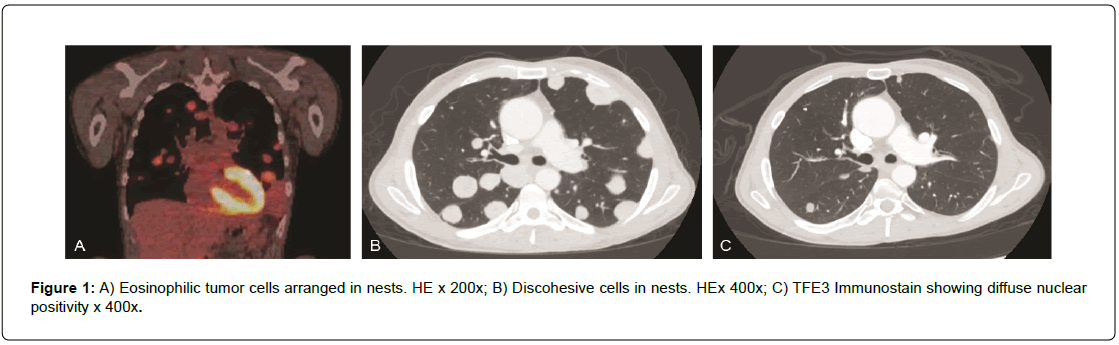
Figure 1: A) Eosinophilic tumor cells arranged in nests. HE x 200x; B) Discohesive cells in nests. HEx 400x; C) TFE3 Immunostain showing diffuse nuclear positivity x 400x.
The patient was started on atezolizumab and also underwent 10 fractions of whole brain radiation therapy. At 3 months, the patient’s restaging scans showed a mixed response with some lung nodules increasing in size while others showing stable or decreased size. Repeat MRI of the brain also demonstrated a mixed response as a 4 mm left frontal intraparenchymal metastasis increased in size to 9 mm with surrounding vasogenic edema, while significant treatment effect was noted in bilateral centrum semiovale and corona radiata with resolution of previously seen punctate enhancing diffuse metastatic disease. The patient then received stereotactic radiation to the growing brain mass (Figure 2).
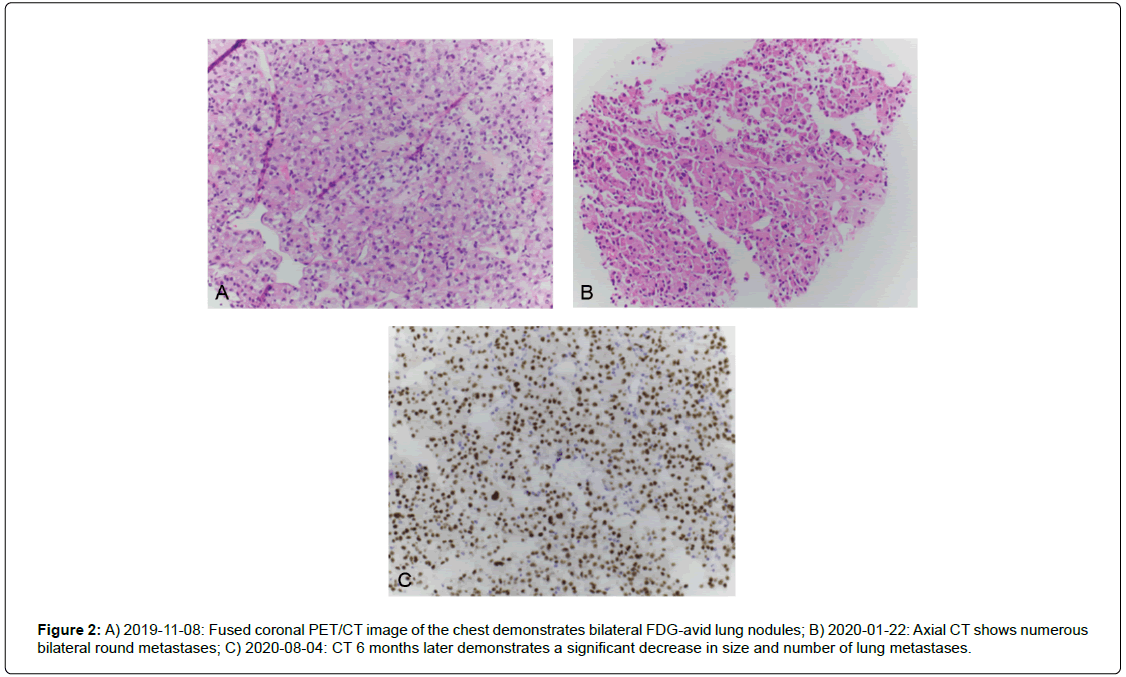
Figure 2: A) 2019-11-08: Fused coronal PET/CT image of the chest demonstrates bilateral FDG-avid lung nodules; B) 2020-01-22: Axial CT shows numerous bilateral round metastases; C) 2020-08-04: CT 6 months later demonstrates a significant decrease in size and number of lung metastases.
The patient continued to improve clinically and by 6 months, restaging signs showed significant interval decrease in size and number of bilateral metastatic pulmonary nodules and masses when compared to prior studies with a significant interval decrease in size of metastatic mediastinal lymph nodes as well as the right buttock mass (Figure 3) The patient is currently receiving atezolizumab and has experienced a continued remission now 8 months after starting atezolizumab.
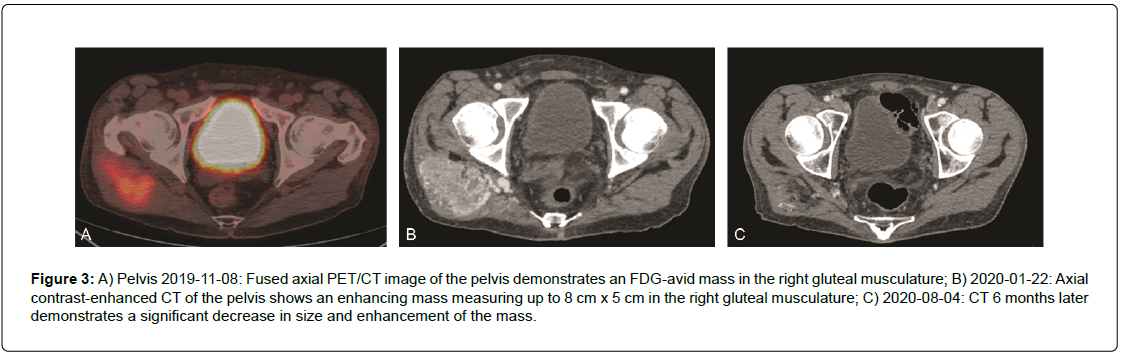
Figure 3: A) Pelvis 2019-11-08: Fused axial PET/CT image of the pelvis demonstrates an FDG-avid mass in the right gluteal musculature; B) 2020-01-22: Axial contrast-enhanced CT of the pelvis shows an enhancing mass measuring up to 8 cm x 5 cm in the right gluteal musculature; C) 2020-08-04: CT 6 months later demonstrates a significant decrease in size and enhancement of the mass.
Discussion
ASPS is a very rare subtype of soft tissue sarcoma first described in 1952 by Christopherson et al. [14]. The incidence of ASPS is one diagnosis per 10 million population per year, and it makes up less than 1% of all sarcomas [15]. Clinically, cases typically involve an intramuscular or perimuscular thigh mass in a young woman, though rare cases have also been reported in other organs including the mediastinum, stomach, breast, uterine cervix, and other organ systems [2-10]. Although the tumor is characterized by indolence, there is a paradoxically high metastatic rate. Symptoms related to organ involvement from metastatic disease are often the first signs leading to diagnosis [11]. At presentation, 35% of ASPS patients had AJCC Stage II or III disease, while 65% of patients had Stage IV disease. Brain metastases were noted in 19% of patients who presented with Stage IV (M1) disease, and the median survival of patients with M1 disease was 40 months [16].
The pathogenesis of this tumor is related to a non-reciprocal t(X;17) (p11;q25) translocation, forming the ASPL-TFE3a fusion gene that is characteristic of ASPS. The 17q25 gene locus and the TFE3 transcription factor gene from Xp11 form a fusion protein that drives hepatocyte growth factor receptor (MET) tyrosine kinase signaling [17,18] resulting in aberrant down stream signaling. MET’s role in cell survival and angiogenesis leads to ASPL-TFE3’s prooncogenic pathway and the pathogenesis of ASPS. Gene expression analysis has also shown that ASPS is associated with elevated expression of transcripts related to angiogenesis, such as MET and vascular endothelial growth factor (VEGF), as well as others involved in cell proliferation, metastasis, and steroid biosynthesis [15].
Treatment of ASPS involves surgical resection, but the relapse rate after surgery was 20%-30% at 5 years [19]. Since conventional chemotherapy does not achieve responses in ASPS, targeted therapies have been used to treat ASPS patients. Pazopanib, a multitargeted tyrosine kinase receptor inhibitor with activity against VEGFR 1, 2, and 3 and the platelet derived growth factor receptor (PDGFR), has US Food and Drug Administration approval for use in STSs refractory to other therapy. In a retrospective study of 30 patients who received pazopanib for treatment of advanced ASPS, there was 1 complete response, 7 partial responses, 17 stable disease, and 4 progressive diseases with median progression-free survival of 13.6 months (range 1.6-32.2) with 59% of patients progression-free at 1 year [20]. In a phase II trial of cediranib, an oral small molecule inhibitor of VEGF-1, VEGF-2, and VEGF-3, 43 patients were evaluated and the objective response rate was 35% with 15 patients (35%) having a partial response and 26 patients (60%) demonstrating stable disease [21]. Sunitinib, another multi-targeted TKI against VEGFR-1, VEGFR-2, fetal liver tyrosine kinase receptor 3 (FLT3), KIT, PDGFRα, and PDGFRβ, was also studied in a retrospective study and showed 6 partial responses in 9 patients and median progression free survival on treatment of 17 months [22]. Other more limited data exists for the efficacy of other TKIs in ASPS including sorafenib, tivantinib, imatinib, dasatinib, bevacizumab, cabozantinib S-malate, and crizotinib [13].
Immune Checkpoint Inhibitors (ICIs) are promising treatment options for ASPS. A recent study explored treatment of advanced or metastatic sarcomas by combining axitinib, a VEGF receptor tyrosine-kinase inhibitor, with pembrolizumab, an anti-PD-1 immune checkpoint inhibitor. In the 33 patients enrolled, 12 patients had ASPS, and the 3-month progression-free survival for these ASPS patients was 72.7% (95% CI 27.1-90.3) [23]. In a 50 patient study on patients with advanced sarcoma who received immunotherapy, best response was seen in two of the four ASPS patients who achieved partial response after anti-PD-L1 based therapy. The other two ASPS patients achieved stable disease [24]. At the Connective Tissue Oncology Society (CTOS) 2018 meeting in Rome, Italy, Coyne et al presented a study of 19 ASPS patients who qualified for inclusion and received atezolizumab. The Objective Response Rate (ORR) for these patients was 42% [25]. In our case, the patient demonstrated spread of ASPS to his lungs and numerous lesions in his brain including the supratentorial and infratentorial regions that necessitated immediate systemic treatment. Despite clinical improvement in the first three months of treatment and the addition of whole brain radiation, a mixed response was noted on CT of the chest and progressive disease was noted in the brain that required stereotactic radiation therapy. The patient was nonetheless continued on systemic treatment based on his subjective clinical improvement and by 6 months a definitive radiographic disease response was noted systemically and locally. This pattern of delayed radiographic response is not unusual in patients who respond to immunotherapy as the melanoma literature has noted responses taking up to one year with pseudo-progression occurring in some patients [25]. In ASPS, the median time to response was not reported and therefore this is the first reported study of this delayed time course of response in ASPS.
As our patient demonstrated significant response to atezolizumab despite his extensive metastatic disease, it is clear that atezolizumab should be further studied in patients who have advanced ASPS including those with metastasis to the lungs and the brain. Given that the patient population of ASPS has a median age of 25 years old, treatment with atezolizumab could significantly improve symptoms and disease burden, especially as surgical resection has a high rate of relapse.
Conclusion
Alveolar soft part sarcoma is a rare subtype of soft tissue sarcoma that currently possesses few effective therapeutic options. This case demonstrates a remarkable response of a symptomatic ASPS patient to atezolizumab after approximately six months therapy that included brain irradiation. The results of a phase II study assessing atezolizumab in ASPS patients is ongoing and will lend further insight into the role of immunotherapy in the management of ASPS (NCT03141684).
References
- Ferrari A, Sultan I, Huang TT, Rodriguez-Galindo C, Shehadeh A, et al. (2011) Soft tissue sarcoma across the age spectrum: A population-based study from the surveillance epidemiology and end results database. Pediatr Blood Cancer 57: 943-949.
Google Scholar Cross Ref - Aisner SC, Beebe K, Blacksin M, Mirani N, Hameed M (2001) Primary alveolar soft part sarcoma of fibula demonstrating ASPL-TFE3 fusion: A case report and review of the literature. Skeletal Radiol 37: 1047-1051.
Google Scholar Cross Ref - Amin MB, Patel RM, Oliveira P, Cabrera R, Carneiro V, et al. (2006) Alveolar soft-part sarcoma of the urinary bladder with urethral recurrence: A unique case with emphasis on differential diagnoses and diagnostic utility of an immunohistochemical panel including TFE3. Am J Surg Pathol 30: 1322-1325.
Google Scholar Cross Ref - Fadare O (2006) Uncommon sarcomas of the uterine cervix: A review of selected entities. Diagn Pathol 1: 30.
Google Scholar Cross Ref - Flieder DB, Moran CA, Suster S (1997) Primary alveolar soft-part sarcoma of the mediastinum: a clinicopathological and immunohistochemical study of two cases. Histopathology 31: 469-473.
Google Scholar Cross Ref - Liu TT, Chou YH, Lai CR, Chen CM, Tsou MH, et al (1997) Breast mass due to alveolar soft part sarcoma of the pectoris major muscle. Eur J Radiol 24: 57-59.
Google Scholar Cross Ref - Luo J, Melnick S, Rossi A, Burke RP, Pfeifer JD, et al. (2008) Primary cardiac alveolar soft part sarcoma. A report of the first observed case with molecular diagnostics corroboration. Pediatr Dev Pathol 11: 142-147.
Google Scholar Cross Ref - Park YK (1999) Primary alveolar soft part sarcoma of bone. Histopathology 35: 411-417.
Google Scholar - Wu J, Brinker DA, Haas M, Montgomery EA, Argani P (2005) Primary alveolar soft part sarcoma (ASPS) of the breast: report of a deceptive case with xanthomatous features confirmed by TFE3 immunohistochemistry and electron microscopy. Int J Surg Pathol 13: 81-85.
Google Scholar Cross Ref - Yagihashi S, Yagihashi N, Hase Y, Nagai K, Alguacil-Garcia A, et al. (1991) Primary alveolar soft-part sarcoma of stomach. Am J Surg Pathol 15: 399-406.
Google Scholar Cross Ref - Pennacchioli E, Fiore M, Collini P, Radaelli S, Dileo P, et al. (2010) Alveolar soft part sarcoma: Clinical presentation, treatment, and outcome in a series of 33 patients at a single institution. Ann Surg Oncol 17: 3229-3233.
Google Scholar Cross Ref - Ogose, A., et al., Alveolar soft part sarcoma in Japan: multi-institutional study of 57 patients from the Japanese Musculoskeletal Oncology Group. Oncology 6: 7-13.
Google Scholar Cross Ref - Paoluzzi L, Maki RG (2019) Diagnosis, prognosis, and treatment of alveolar soft-part sarcoma: A review. JAMA Oncol 5: 254-260.
Google Scholar Cross Ref - Christopherson WM, Foote FW, Stewart FW (1952) Alveolar soft-part sarcomas; structurally characteristic tumors of uncertain histogenesis. Cancer 5: 100-11.
Google Scholar Cross Ref - Brahmi MH, Vanacker A (2020) Novel therapeutic options for alveolar soft part sarcoma: antiangiogenic therapy, immunotherapy and beyond. Curr Opin Oncol 32: 295-300.
Google Scholar Cross Ref - Portera CA, Ho V, Patel SR, Hunt KK, Feig BW, et al. (2001) Alveolar soft part sarcoma: clinical course and patterns of metastasis in 70 patients treated at a single institution. Cancer 91: 585-591.
Google Scholar Cross Ref - Ladanyi M, Lui MY, Antonescu CR, Krause-Boehm A, Meindl A, et al. (2001) The der(17)t(X;17)(p11;q25) of human alveolar soft part sarcoma fuses the TFE3 transcription factor gene to ASPL, a novel gene at 17q25. Oncogene 20: 48-57.
Google Scholar Cross Ref - Williams A, Bartle G, Sumathi VP, Meis JM, Mangham DC, et al. (2011) Detection of ASPL/TFE3 fusion transcripts and the TFE3 antigen in formalin-fixed, paraffin-embedded tissue in a series of 18 cases of alveolar soft part sarcoma: Useful diagnostic tools in cases with unusual histological features. Virchows Arch 458: 291-300.
Google Scholar Cross Ref - Flores RJ, Harrison DJ, Federman NC, Furman WL, Huh WW, et al. (2018) Alveolar soft part sarcoma in children and young adults: A report of 69 cases. Pediatr Blood Cancer 65: e26953.
Google Scholar Cross Ref - Stacchiotti S, Mir O, Le Cesne A, Vincenzi B, Fedenko A, et al. (2018) Activity of Pazopanib and Trabectedin in Advanced Alveolar Soft Part Sarcoma. Oncologist, 2018. 23: 62-70.
Google Scholar Cross Ref - Kummar S, Allen D, Monks A, Polley EC, Hose CD, et al. (2013) Cediranib for metastatic alveolar soft part sarcoma. J Clin Oncol 31: 2296-2302.
Google Scholar Cross Ref - Stacchiotti S, Negri T, Zaffaroni N, Palassini E, Morosi C, et al. (2011) Sunitinib in advanced alveolar soft part sarcoma: Evidence of a direct antitumor effect. Ann Oncol 22: 1682-1690.
Google Scholar Cross Ref - Wilky BA (2019) Axitinib plus pembrolizumab in patients with advanced sarcomas including alveolar soft-part sarcoma: A single-centre, single-arm, phase 2 trial. Lancet Oncol 20: 837-848.
Google Scholar Cross Ref - Groisberg R, Hong DS, Behrang A, Hess K, Janku F, et al. (2017) Characteristics and outcomes of patients with advanced sarcoma enrolled in early phase immunotherapy trials. J Immunother Cancer 5: 100.
Google Scholar Cross Ref - Ma Y, Wang Q, Dong Q, Zhan L, Zhang J (2019) How to differentiate pseudoprogression from true progression in cancer patients treated with immunotherapy. Am J Cancer Res 9: 1546-1553.
Google Scholar
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi 