Case Report, Clin Oncol Case Rep Vol: 5 Issue: 4
Bilateral Primary Cerebellopontine Angle Lymphoma Mimicking NF-2: A Rare Case Report with Review of Literature
Hardik Siroya*
Department of Neurosurgery, National Institute of Mental Health and Neuro Sciences, Bengaluru, Karnataka, India
*Corresponding Author: Hardik Siroya
Department of Neurosurgery, National Institute of Mental Health and Neuro Sciences, Bengaluru, Karnataka, India
E-mail: siroyahardik@hotmail.com
Received: April 04, 2022; Manuscript No: COCR-22-59499;
Editor Assigned: April 06, 2022; PreQC Id: COCR-22-59499(PQ);
Reviewed: April 21, 2022; QC No: COCR-22-59499(Q);
Revised: April 23, 2022; Manuscript No: COCR-22- 59499(R);
Published: April 29, 2022; DOI: 10.4172/cocr.5(4).227
Citation: Siroya H (2022) Bilateral Primary Cerebellopontine Angle Lymphoma Mimicking NF-2: A Rare Case Report with Review of Literature. Clin Oncol Case Rep 5:4
Abstract
Background and Importance: Primary CNS lymphomas (PCNSL) are a rare entity. Even rarer is CNS lymphomas of bilateral Cerebellopontine Angle (CPA) mimicking NF-2. The rarest form is bilateral cerebellopontine lymphomas extending into Internal auditory meatus along with multiple other CNS lesions. Management in this regard can only be done after definitive diagnosis. Symptomatic lesions leading to surgery hardly find description in literature.
Clinical Presentation: We present a 55 year old male with the rarest form PCNSL presenting with multiple cranial nerve deficits and involving bilateral CPA. Surgery confirmed the diagnosis in otherwise presumed case of NF-2. Management pearls along with histological analysis are highlighted.
Conclusion: We assume CNS lymphomas are a great mimicker and biopsy proven diagnosis and change the course of management with extremely good outcomes.
Keywords: Cerebellopontine angle lymphoma; NF-2 mimic; Primary CNS lymphoma
Background and Importance
Diffuse large cell lymphoma of B-cell origin (DLBCL) is the most common subtype of Primary CNS lymphoma to involve the brain. PCNSL constitutes 0.7%-1% of NHL and less than 1% of all intracranial tumours [1]. It can mimic myriad features ranging from stroke, mastoiditis, cranial nerve deficits, seizures(8%-10%), focal deficits (50%-75%), neuropsychiatric manifestation (25%- 35%), encephalitis, raised ICT (Intracranial Tension)-(1%-3%) etc. [2]. Multiple intracranial lesions can be present in 20%-50% of the patients [3]. The usual sites of involvement is lobar followed by basal ganglia, thalamus, periventricular white matter, corpus callosum, cerebellar vermis etc. [4,5]. Cerebellopontine angle involvement is rare accounting for less than 0.5% [6]. Bilateral involvement of Cerebellopontine Angle (CPA) is even rarer. Most CPA do not extend into internal auditory meatus. Involvement of bilateral internal auditory meatus presenting as Neurofibromatosis type 2 has been reported only four times in literature [2,7-9] . Ours is the fifth case in this regard. After doing systematic search and review of indexed data on PUBMED, CROSSREF and GOOGLE scholar from 1956 till date, we could find only 41 cases of CPA lymphomas among which 35 are primary, 6 secondary. Amongst the primary, one was associated with HIV. We present here a case report of the rarest bilateral CPA lymphoma mimicking NF-2. A review of the literature revealed four case reports of PCNSL presenting with bilateral symmetric cerebellopontine angle tumours.
This case report has been reported in line with the SCARE Criteria.
Clinical Presentation
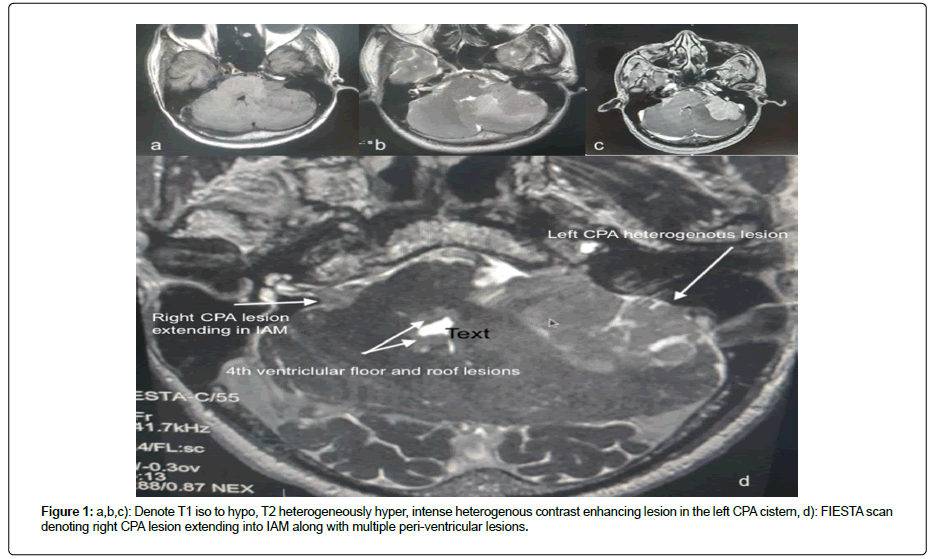
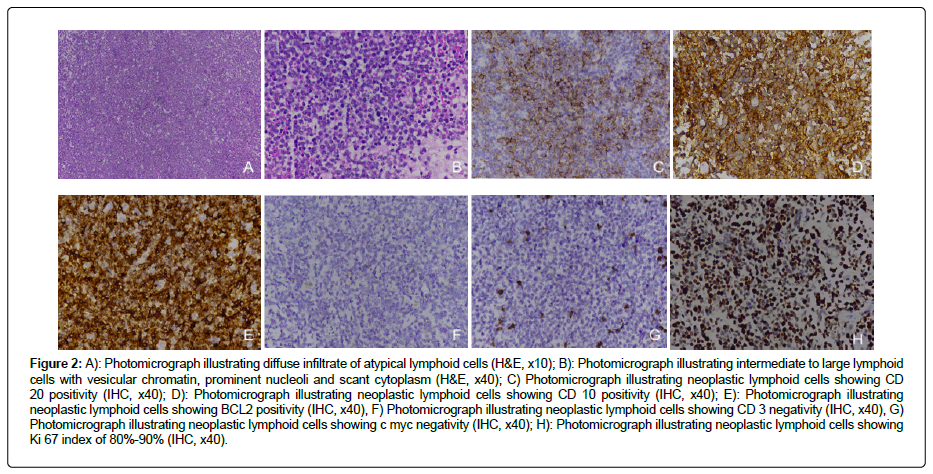
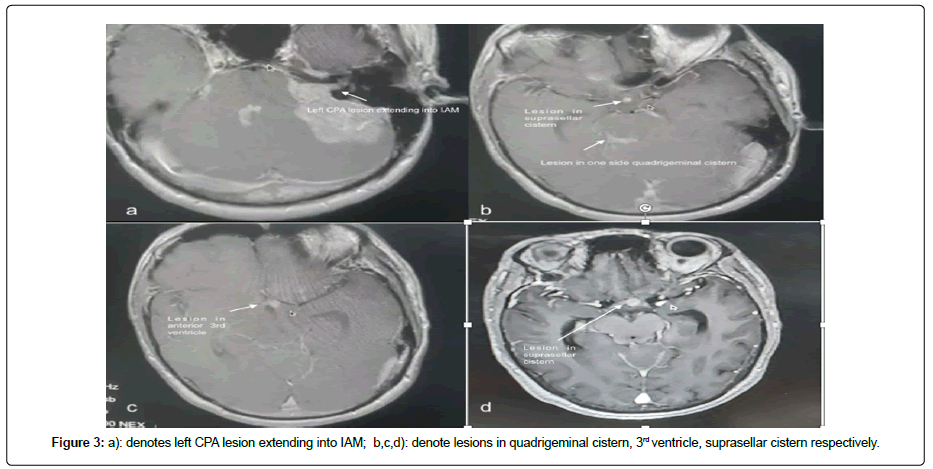
55-year-old male patient with nil comorbidities came with history of localised left temporal headache since 3 months which progressively became holocranial and throbbing type since 2 months. Since 3 months there was progressive hearing loss in the left ear with early morning vomiting episodes for 2 months. He also complained of drunken gait, swaying to the left side with intermittent dizziness for 1 month. On examination he was fully conscious, obeying with left grade II LMN facial palsy. Left ear demonstrated moderately severe hearing loss. CT brain was suggestive of an extra-axial well defined hyperdense lesion in the left CPA with extension and dilation of IAM. It was broad based towards petrous dura with. Mass effect on adjacent cerebellar folia and oedema. Ill-defined hypodensities were noted in the cerebellar parenchyma and along roof of 4th ventricle. There was partial effacement of 4th ventricle and resultant dilation of 3rd and bilateral lateral ventricles suggestive of Koos grade 4. The right side tumour was Koos grade 2. In view of raised ICT features and obstructive hydrocephalus, right side ventriculo-peritoneal shunt was done. MRI was suggestive of T1 hypo, T2 heterogeneously hyper lesion (Figure 1) with intense heterogenous contrast uptake (Figure 2). Lesion measured 3.8 cm × 3.7 cm × 3.5 cm placed in left CPA cistern. It showed central necrosis with perilesional oedema extending posteriorly indenting over left middle cerebellar peduncle and mass effect on brainstem s/o Koos grade 4. There were T2/FLAIR hyperintensities with intense contrast enhancing small lesions noted in roof of 4th ventricle, suprasellar cistern extending into 3rd ventricular floor (Figure 1 and Figure 3). There was another small lesion in the right CPA cistern extending into ipsilateral IAM with no mass effect. The 4th and 3rd ventricular lesion were deemed as ependymomas whereas the CPA lesions were thought to be schwannomas making a presumptive diagnosis of NF-2 spectra. Patient underwent Left Retrosigmoid approach, lateral suboccipital craniotomy and excision of lesion under cranial nerve monitoring. Intraoperative findings: Yellowish grey, soft suckable, solid cystic moderately vascular lesion with an ill-defined plane with Pons and superiorly with the Cerebellar surface. Intracapsular decompression was done. Part of the capsule adjoining the facial nerve was left behind. The IAM was dilated and involved, part extending into IAM was also excised. Post-operative scans were suggestive a small residue along 7th nerve. His postoperative course was uneventful with persistent grade II facial palsy. Histopathology revealed tumour cells arranged in sheets. There was a perivascular distribution of large monomorphic lymphoid cells predominantly centroblastic type having 1-3 prominent nucleoli. Small mature lymphocytes were also see. Immunohistochemistry revealed tumour cells positive for CD20, CD10, BCL2 (Figure 2) and negative for CD3, cMyc, Tdt (Figure 2). Ki 67 index was 70%-80%. After this report patient underwent metastasis workup which did not reveal any primary lesion. No lymph node enlargement was noted in the body. HIV test was negative. Currently patient is receiving combination radio chemotherapy. Follow up of 6 weeks showed complete resolution of facial weakness.
Figure 2: A): Photomicrograph illustrating diffuse infiltrate of atypical lymphoid cells (H&E, x10); B): Photomicrograph illustrating intermediate to large lymphoid cells with vesicular chromatin, prominent nucleoli and scant cytoplasm (H&E, x40); C) Photomicrograph illustrating neoplastic lymphoid cells showing CD 20 positivity (IHC, x40); D): Photomicrograph illustrating neoplastic lymphoid cells showing CD 10 positivity (IHC, x40); E): Photomicrograph illustrating neoplastic lymphoid cells showing BCL2 positivity (IHC, x40), F) Photomicrograph illustrating neoplastic lymphoid cells showing CD 3 negativity (IHC, x40), G) Photomicrograph illustrating neoplastic lymphoid cells showing c myc negativity (IHC, x40); H): Photomicrograph illustrating neoplastic lymphoid cells showing Ki 67 index of 80%-90% (IHC, x40).
Discussion
Primary Non-Hodgkin’s Lymphoma (NHL) of the Central Nervous System (CNS) of Diffuse Large Cell B-Cell Lymphoma (DLBCL) is malignant and is the most common lymphoma (>90%) affecting the CNS [10-13]. Other lesser variants include mantle cell lymphoma. Burkitt’s lymphoma, marginal variant, T-cell lymphoma, intermediate DLBCL type, Hodgkin’s disease, Lymphoplasmacytic Lymphoma (LPL) accounting for remaining less than 10% cases [12,13]. These tumours are highly radio-chemo sensitive due to their high mitotic index (Ki-67) ranging from 10% to 90% [10]. PCNSL usually have a mitotic index (Ki-67) in very high range from 50%- 90% [10].
CPA tumours usually constitute following tumours likewise Vestibular schwannoma (85%-90%), Meningioma (2%-7%) [14,15], cholesteatomas, 7th and 5th nerve schwannoma, epidermoids, arachnoid cysts together constitute the rest 5% [14]. Other rare tumours include haemangiomas, hemangioblastomas, gliomas, lipomas, chondrosarcomas, teratomas, metastasis, abscess mimicking tumour etc [14]. Primary CPA lymphomas are the rarest (≤ 0.5%) with only few countable case reports described in literature [1]. They are usually supratentorial and occur singly [3]. Infratentorial lesion tend to occur mainly in cerebellum and in periventricular areas. Multiple lesions , infratentorial location is quite rare and CPA location is even rarer [3]. Among these bilateral occurrence mimicking NF-2 have been reported only four times. Secondary CNS lymphomas are reported even lesser accounting only 1/10th of PCNSL and usually involve multiple cranial nerves. They account for approximately 5%- 15% of systemic lymphomatous malignancies [16,17]. Lymphomas in general are associated with immune deficiency syndromes in most cases. The mean age at diagnosis for immunosuppressed is approximately 30 years whereas for immunocompetent is just above 55 years [10, 18, 16]. Most case reports report female patients above 45 years, whereas multi-centre studies have revealed male predilection [16]. Presumptive diagnosis is usually done by clinical presentation which is mainly a picture of inflammatory process reduced by steroids, whereas; confirmatory diagnosis is done either by biopsy, autopsy of resectional surgery. The latter is beneficial only in cases of progressive deficits(debulking), large lesion or if the presentation is like any other CPA lesion.
MRI with T1,T2/FLAIR are suggestive whereas T1 with gadolinium along with hyper density on CT is confirmatory. The usual picture involves T1 and T2/FLAIR iso to hypo with homogenous intense contrast enhancement and homogenous hyper density on CT. Such picture is also present in meningiomas albeit dural tail which may assist in diagnosis. MR spectroscopy is additive in this regard. Any deviation from the above, can obscure the diagnosis more in favour of schwannoma. Our case showed T2/ FLAIR heterogeneity with heterogenous contrast uptake extending and invading IAM (internal acoustic meatus), which led to a diagnosis of CPA schwannoma. Furthermore IAM erosion or involvement in very rare in lymphomas seen mainly in neurolymphomatosis which itself is extremely rare (≤ 0.2%).The only feature favouring lymphoma was multiple periventricular small lesions which were unusual for NF-2. Nonetheless the Wishart-Phenotypic variant may present with periventricular ependymomas and gliomas. Jaiswal et al (Table 1) had reported a similar case with multiple intracranial lesions along with bilateral CPA lymphomas in which the right CPA mass was excised as preoperative diagnosis was schwannoma [7]. Intraoperatively there was no adhesion with the nerves and IAM was not involved. Frozen section revealed lymphoma after which only partial decompression was done followed by Radio-chemotherapy. Garth et al (Table 1) in 1993 were the first to report bilateral CPA lymphoma with a non-conclusive pre-surgical diagnosis [9]. Patient underwent left side craniotomy in view of compressive symptoms and signs of cerebellopontine angle. In their case no IAM involvement or dilation was noted intraoperatively. Larner et al (Table 1) reported a case with large bilateral CPA masses [8]. Based on CSF cytology and patients response to steroids, a presumptive diagnosis of inflammatory vs metastatic pathology was kept. After 2 months of steroids patient again developed increased symptoms which prompted biopsy of one of the tumours revealing lymphomatous pathology. Patient had a rapid deteriorating course with death ensuing with 3 months of 1st symptoms. In 2007, Lenarz et al (Table 1) reported a similar case that was previously treated for colon carcinoma [2]. In this regard, the presumptive diagnosis was kept as metastasis. But CSF cytology revealed lymphoma. Hence chemotherapy was initiated. Patient’s scan revealed bilateral involvement of IAM. Present case illustrates a PCNSL with multiple infratentorial and supratentorial lesions in an immunocompetent male with bilateral CPA lesions involving IAM.
Table 1. Reprents camparison between prior 4 studies and the present study.
| Studies | Year | No. of Cases | Age | Sex | CT | MRI | Internal auditory meat us extension | Other extension | Other lesions | Cranial nerve defects | Primary or secondary | Histopathology | Surgery | HIV |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Jaiswal et al | 2004 | 1 | 36 | M | b/l T1-iso, T2-hyper, homogenous enhancement | Yes | Nil | Small masses in riaght lateral ventricle and anterior third ventricle region | VII, VIII, cerebellar, headache | Primary | DLBCL,L26,CD20 Positive | Left side craniotomy and excision + Combination radiochemotherapy | Neg | |
| Lenarz et al | 2007 | 1 | 68 | F | b/l intense homogenous enhancement | Yes bilateral | Nil | Meningeal Metastasis t/c/o Colon cancer | VI, VII, VIII, Headache, Diplopia | Primary | DLBCL | Chemotherapy (HD-MTX)+ Steriods | Neg | |
| Garth et al | 1993 | 1 | 68 | F | b/l homogenous enhancement | b/l homogenous enhancement | Nil | Nil | NIL | VIII cerebellar dysarthria | Primary | DLBCL, L26 Positive | Left side craniotomy and excision + radiochemotherapy | Neg |
| Larner et al | 1999 | 1 | 73 | M | Bilateral symmetrical CPA intense homogenous enhancement | Nil | Nil | History of Guillain-Barre 14 years back.No. h/o immuno suppression | V, cerebellar double vision, Progressive speech disturbance, Right facial numbness | Primary | DLBCL-CD20 positive CD3 negative | Biopsy and Corticosteriods | Neg | |
| This study | 2021 | 1 | 55 | M | Hyperdense lesion | Bilateral Heterogenous intense enhancement | Yes, Bilateral | Nil | Suprasellar 3rd, quadrig eminal cisternal and 4th ventricular small lesions | VII, VIII, cerebellar, headache | Primary | DLBCL CD20, CD10, BCL2 positive, CD3 Negative | Left side excision+Combination radiochemotherapy | Neg |
The treatment after diagnosis is induction chemotherapy that aims for a complete radiographic response (CR), followed by consolidative therapy [19]. The median survival was approximately 12 to 18 months after best treatment during pre-chemotherapy era [20,17]. Current data suggests an improved median disease free survival of approximately 40 months after combined modality treatment with focused high dose chemotherapy (HD-MTX), Cranial RT, high dose cytarabine, temozolomide, cytarabine, etoposide, vincristine, carmustine, ifosfamide, thiotepa, and cyclophosphamide [10,19]. Consolidation includes high-dose radiation (45 Gy), low dose radiation (23.4 Gy), chemotherapy with carmustine, thiotepa, cyclophosphamide, busulfan, cytarabine, and etoposide. The maximum Overall Survival (OS) achieved with above regimen at 3 years was 87% with CR [19]. The role of surgery is to provide diagnosis in absence of confirmatory CSF cytology; so also decompression in impending herniation.
Conclusion
Bilateral CPA PCNSL in immunocompetent males is very rare to find. Treatment protocols for such lesions are standardized and chemotherapy remains corner stone in management. Nonetheless, overall prognosis which was poor in spite of best treatment has significantly improved with combination modality treatment in present era.
References
- Tadmor R, Davis KR, Roberson GH, Kleinman GM (1978) Computed tomography in primary malignant lymphoma of the brain. J Comput Assist Tomogr 2: 135-140. [Google Scholar] [Cross Ref]
- Lenarz M, Durisin M, Becker H, Lenarz T, Nejadkazem M (2007) Primary central nervous system lymphoma presenting as bilateral tumors of the internal auditory canal. Skull Base Off J North Am Skull Base Soc Al 17: 409-412. [Google Scholar] [Cross Ref]
- Tomlinson FH, Kurtin PJ, Suman VJ, Scheithauer BW, O'Fallon JR, et al. (1995) Primary intracerebral malignant lymphoma: A clinicopathological study of 89 patients. J Neurosurg 82: 558-566. [Google Scholar]
- Cellerier P, Chiras J, Gray F, Metzger J, Bories J (1984) Computed tomography in primary lymphoma of the brain. Neuroradiol 26: 485-492. [Google Scholar] [Cross Ref]
- Enzmann DR, Krikorian J, Norman D, Kramer R, Pollock J, et al (1979) Computed tomography in primary reticulum cell sarcoma of the brain. Radiology 130:165-170. [Google Scholar] [Cross Ref]
- Wang YT, Su HH, Hou Y, Chu ST, Lai PH, et al. (2007) Diffuse large B-cell lymphoma of the cerebellopontine angle in a patient with sudden hearing loss and facial palsy. J Chinese Med Ass 70: 294-297. [Google Scholar] [Cross Ref]
- Jaiswal AK, Mahapatra AK, Sharma MC (2004) Primary central nervous lymphoma presenting as bilateral cerebellopontine angle lesions: A rare case report. J Clin Neurosci Off J Neurosurg Soc Australas 11: 328-331. [Google Scholar] [Cross Ref]
- Larner AJ, D’Arrigo C, Scaravilli F, Howard RS (1999) Bilateral symmetrical enhancing brainstem lesions: An unusual presentation of primary CNS lymphoma. Eur J Neurol 6: 721-723. [Google Scholar] [Cross Ref]
- Garth RJ, Coddington R, Brightwell AP (1993) Primary cerebral lymphoma presenting with bilateral cerebellopontine angle lesions. J Laryngol Otol 107: 937-939. [Google Scholar] [Cross Ref]
- Abrey LE, DeAngelis LM, Yahalom J (1998) Long-term survival in primary CNS lymphoma. J Clin Oncol Off J Am Soc Clin Oncol 16: 859-863. [Google Scholar]
- Shenkier TN, Blay JY, O’Neill BP, Poortmans P, Thiel E, et al. (2005) Primary CNS lymphoma of T-cell origin: A descriptive analysis from the international primary CNS lymphoma collaborative group. J Clin Oncol 23: 2233-2239. [Google Scholar]
- Lim T, Kim SJ, Kim K, Lee JI, Lim DH, et al. (2011) Primary CNS lymphoma other than DLBCL: A descriptive analysis of clinical features and treatment outcomes. Annals Hematol 90: 1391-1398. [Google Scholar] [Cross Ref]
- Yang XL, Liu YB (2017) Advances in Pathobiology of Primary Central Nervous System Lymphoma. Chin Med J Engl 130: 1973-1979. [Google Scholar] [Cross Ref]
- Brackmann DE, Bartels LJ (1980) Rare tumors of the cerebellopontine angle. Otolaryngol Head Neck Surg 88: 555-559. [Google Scholar] [Cross Ref]
- Kendall B, Symon L (1977) Investigation of patients presenting with cerebellopontine angle syndromes. Neuroradiol 13: 65-84. [Google Scholar] [Cross Ref]
- Bathla G, Hegde A (2016) Lymphomatous involvement of the central nervous system. Clin Radiol 71: 602-609. [Google Scholar] [Cross Ref]
- Wang CC, Carnevale J, Rubenstein JL (2014) Progress in central nervous system lymphomas. Br J Haematol 166: 311-325. [Google Scholar] [Cross Ref]
- Ferreri AJM, Reni M, Pasini F, Calderoni A, Tirelli U, et al. (2002) A multicenter study of treatment of primary CNS lymphoma. Neurology 58: 1513-1520. [Google Scholar] [Cross Ref]
- Han CH, Batchelor TT. (2017) Diagnosis and management of primary central nervous system lymphoma. Cancer 123: 4314-4324. [Google Scholar] [Cross Ref]
- Hochberg FH., Miller DC. (1988) Primary central nervous system lymphoma. J Neurosurg 68: 835-853. [Google Scholar]
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi