Case Report, Clin Oncol Case Rep Vol: 5 Issue: 1
Cetuximab Responsiveness in Patients with Non-Small Cell Lung Cancer and EGFR Exon 20 Insertion Alterations
Mina Nikanjam1, Shumei Kato1*, Jacob J Adashek2, and Razelle Kurzrock1
1Center for Personalized Cancer Therapy and Division of Hematology and Oncology, UC San Diego Moores Cancer Center, San Diego, USA
2Department of Internal Medicine, University of South Florida, Tampa, FL, USA
*Corresponding Author:
Shumei Kato
Center for Personalized Cancer Therapy and Division of Hematology and Oncology
UC San Diego Moores Cancer Center, San Diego, CA, USA
E-mail: smkato@health.ucsd.edu
Received: September 16, 2021, Manuscript No: COCR-21-42180;
Editor assigned: September 18, 2021, PreQC No: P-42180;
Reviewed: November 22, 2021; QC No: Q-42180;
Revised: December 05, 2021, Manuscript No: R-42180;
Published: January 10, 2022, DOI: 10.4172/cocr.5(1).210
Citation: Nikanjam M, Kato S, Adashek JJ, Kurzrock R (2022) Cetuximab in Patients with Non-Small Cell Lung Cancer and EGFR Exon 20 Insertion Alterations. Clin Oncol Case Rep 5:1
Abstract
Epidermal Growth Factor Receptor (EGFR) exon 20 insertion alterations represent 4%-10% of all EGFR mutations in Non-Small Cell Lung Cancer (NSCLC) and result in resistance to standard EGFR-directed therapies. EGFR exon 20 insertions restrict the size of the kinase pocket, prohibiting the entry of approved EGFR kinase inhibitor drugs. Structural In Silico modeling also predicts that EGFR exon 20 insertion anomalies increase attractive electrostatic dimerization, hence stabilizing the activating dimer configuration. EGFR antibodies such as cetuximab that interfere with dimerization may lead to responses. We identified three non-smoking patients with NSCLC and EGFR exon 20 insertions treated with cetuximab-based therapy. All three patients demonstrated clinical benefit. A 58-year-old woman achieved prolonged stable disease lasting 9 months, while a 76-year-old woman and 38-year-old man maintained a partial response with progression-free survivals of 13 months and 32 months, respectively. In conclusion, cetuximab merits further investigation as it appears to be an additional promising therapy for overcoming EGFR exon 20 insertion-related resistance.
Keywords: Cetuximab; EGFR; Exon 20 insertion; NSCLC
Introduction
Erlotinib is an effective small molecule EGFR inhibitor that is approved for NSCLC harboring an exon 19 deletion or exon 21 (L858R) substitution mutation [1,2]. However, resistance can develop through additional mutations, such as the EGFR T790M, which can be overcome by osimertinib, now often used as front-line treatment in EGFR-mutated NSCLC [3]. EGFR exon 20 insertions represent 4%-10% of all EGFR alterations in NSCLC, and mediate resistance to several approved EGFR inhibitors, with response rates of only 3%-9% for erlotinib, gefitinib, and the second-generation EGFR inhibitor afatinib [4-6]. Recently, however, two drugs have been approved for this indication, including a bispecific EGFR/MET antibody amivantamab, and a small molecule inhibitor mobocertinib, based on response rate of ~40% and 28%, respectively.
In silico modeling demonstrated that the EGFR exon 20 insertions result in an activated kinase with a small pocket into which most kinase inhibitors do not fit, therefore necessitating a kinase inhibitor of smaller size, such as poziotinib [4-8]. Computer simulations also show that the EGFR exon 20 insertion stabilizes the active dimer configuration, hence possibly sensitizing to an agent such as an EGFR antibody that prevents dimerization [7]. Anecdotal cases further suggest that the EGFR antibody cetuximab may be able to overcome exon 20 insertion resistance in the clinic [5,7,9].
We present a case series of three patients with EGFR exon 20 insertions who showed benefit from cetuximab-based treatment.
Methods
Molecular Tumor Board
Patients with EGFR exon 20 insertion alterations were identified from the PREDICT and the Molecular Tumor Board database (NCT02478931). We followed Institutional Review Board guidelines for data analysis and any investigational studies for which patients provided written informed consent.
Genotyping results and interpretation of the molecular results
In each of the three cases, an EGFR exon 20 insertion was identified via next generation sequencing (NGS) of tumor tissue: EGFR exon 20 insertion (H773_V774lnsPH) (case 1); EGFR exon 20 insertion (H773_V774insPH) (ase 2); EGFR exon 20 (D770_P772delsinKG) (Case 3). These EGFR exon 20 insertions represent <10% of EGFR alterations in lung cancer [5].
Results
Case 1
A76-year-old non-smoking woman (history of chronic lymphocytic leukemia) was noted to have increasing lung ground-glass opacities by computerized tomographic (CT) scan. Transbronchial biopsy showed adenocarcinoma. Next generation sequencing (NGS) of lung cancer tissue (Foundation Medicine, 324 genes, https://www.foundationmedicine.com/genomic-testing) showed EGFR exon 20 insertion (H773_V774lnsPH) without other alterations; NGS of blood-derived circulating tumor DNA (ctDNA) (Guardant Health, https://guardanthealth.com/, 73 genes), NRAS G13V, NF1 R69fs, NF1 S340fs, and BRCA2 E1335fs mutations. The patient declined therapy for eight months, when repeat CT scan showed a growing left lung mass; repeat biopsy demonstrated adenocarcinoma. The patient was started on cetuximab for EGFR exon 20 insertion and trametinib for NRAS and NF1 mutations. She was on therapy for 13 months with a partial response (Figure 1). The patient died due to septic shock from colon perforation, which was felt to be unrelated to therapy or her cancer. Side effects were limited to rash.
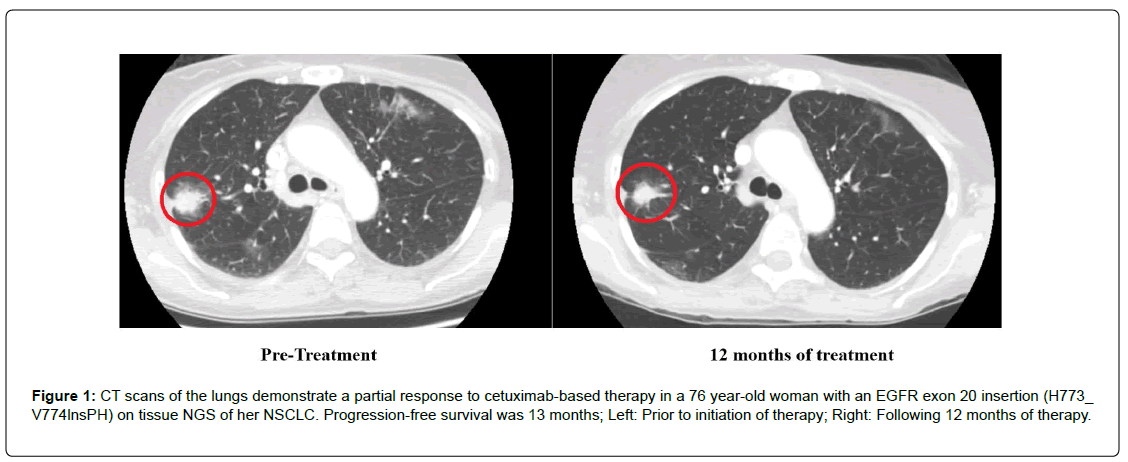
Figure 1: CT scans of the lungs demonstrate a partial response to cetuximab-based therapy in a 76 year-old woman with an EGFR exon 20 insertion (H773_ V774lnsPH) on tissue NGS of her NSCLC. Progression-free survival was 13 months; Left: Prior to initiation of therapy; Right: Following 12 months of therapy.
Case 2
A 58-year-old non-smoking woman was diagnosed with metastatic NSCLC after presenting with cough. Pleural nodule biopsy showed an EGFR exon 19 deletion (L747_P753>S). She was treated with erlotinib (>2 years) followed by osimertinib (16 months) until progression. Repeat biopsy showed a new EGFR exon 20 insertion (H773_V774insPH) (Foundation Medicine, 324 genes NGS) (https://www.foundationmedicine.com/genomic-testing) along with TP53 G244A and SPTA1 alterations. The patient was started on afatinib and cetuximab, with stable disease lasting nine months. Side effects included only low-grade diarrhea and abdominal pain.
Case 3
A 38-year-old non-smoking man presented with back pain due to cord compression and bone lesions. Left iliac biopsy showed metastatic lung adenocarcinoma harboring an EGFR exon 20 alteration (D770_P772delsinKG) (Caris Life Sciences (https://www.carislifesciences.com/cmi-overview/). He received thoracic spine and sacrum radiation. He was started on carboplatin, paclitaxel, bevacizumab, and cetuximab. Following three months of therapy, the chemotherapy was discontinued and he was transitioned to maintenance cetuximab and bevacizumab, which he received for a total of 29 months with a partial response. Side effects were limited to skin toxicity, which necessitated a two-month drug holiday (after eight months of cetuximab and bevacizumab); he resumed these therapeutic antibodies without further significant adverse events. Treatment was discontinued after a total of 32 months from its start, due to progression. There were no other serious side effects.
Patient Updates
As noted in the patient histories, all three patients had non-smoking NSCLC and all benefitted from cetuximab-based therapy. The first patient (age 76) achieved a PR lasting 13 months (Figure 1) and then died of unrelated causes. The second patient (age 58) had stable disease that lasted 9 months. The third patient (38-year-old) attained a PR that lasted 32 months. The most common side effect was rash.
Discussion
EGFR exon 20 insertions are resistant to classic EGFR small molecule inhibitors and are associated with poor NSCLC outcomes. Structurally, this alteration results in a small kinase domain that is not accessible to conventional kinase inhibitors [4-8]. Combination therapy with erlotinib and cetuximab has previously been studied in a clinical trial as a means to improve EGFR targeting, and a partial response ongoing at 42 months was reported in a NSCLC with an EGFR exon 20 insertion alteration [7]. A separate study of EGFR exon 20 insertion NSCLC treated with afatinib with cetuximab noted objective responses in three of four patients, though afatinib alone is ineffective [5,9]. These observations are consistent with in silico modeling observations showing that the EGFR exon 20 insertion activates EGFR by bringing the dimerization domains together, while EGFR antibody attenuates dimerization (Figure 2) [7]. In our case series, the three cetuximab-treated patients with EGFR exon 20 insertions had a PFS of 9, 13 and 32 months, respectively. The main side effect was rash.
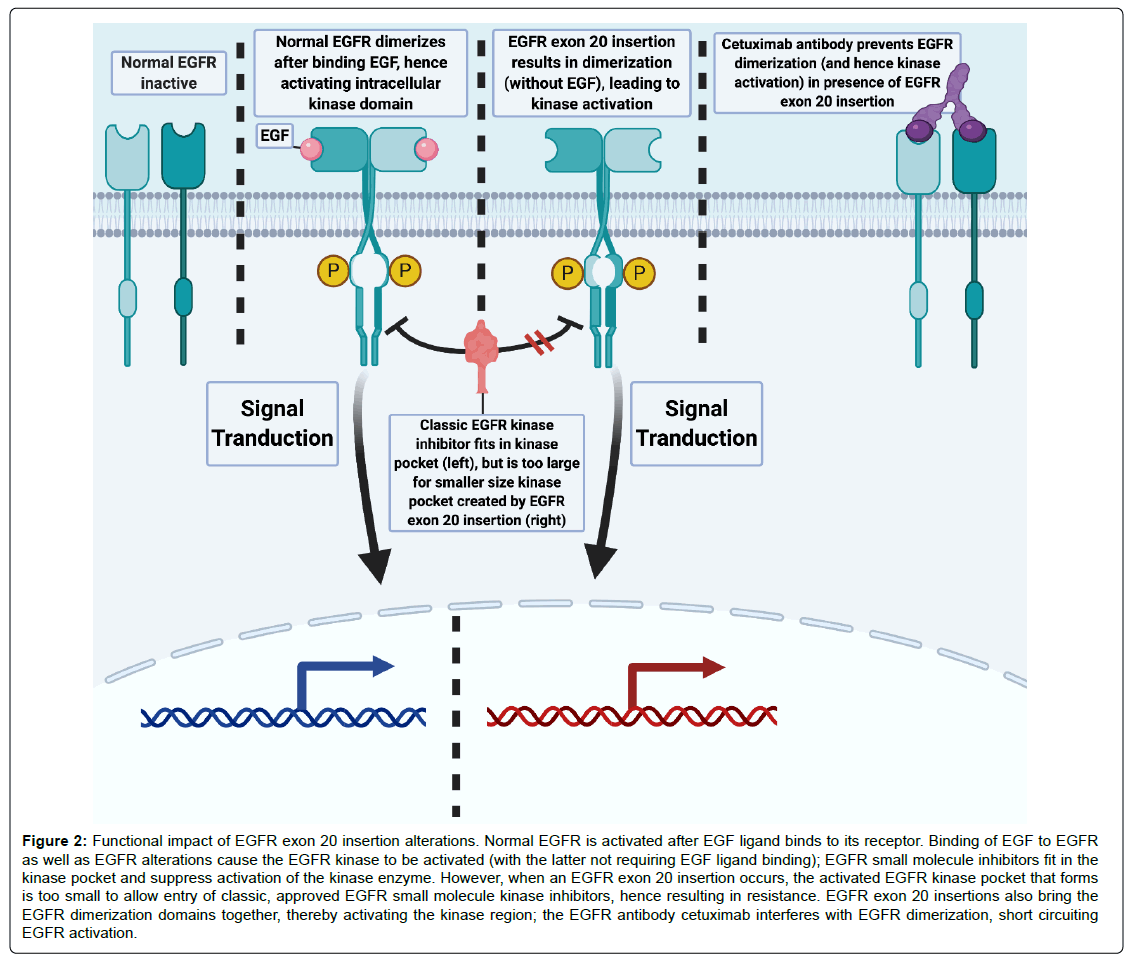
Figure 2: Functional impact of EGFR exon 20 insertion alterations. Normal EGFR is activated after EGF ligand binds to its receptor. Binding of EGF to EGFR as well as EGFR alterations cause the EGFR kinase to be activated (with the latter not requiring EGF ligand binding); EGFR small molecule inhibitors fit in the kinase pocket and suppress activation of the kinase enzyme. However, when an EGFR exon 20 insertion occurs, the activated EGFR kinase pocket that forms is too small to allow entry of classic, approved EGFR small molecule kinase inhibitors, hence resulting in resistance. EGFR exon 20 insertions also bring the EGFR dimerization domains together, thereby activating the kinase region; the EGFR antibody cetuximab interferes with EGFR dimerization, short circuiting EGFR activation.
Poziotinib is a small size novel kinase inhibitor designed to overcome EGFR exon 20 insertion resistance by fitting into the small kinase pocket created by EGFR exon 20 insertions [6,8]. In the clinic, it has activity, but reported median PFS is 5.6 months in EGFR exon 20 insertion NSCLC [5,6,8]. Reviews of other molecules show the following 5: luminespib, a heat shock protein 90 inhibitor, 17% response rate (2.9 month median PFS); and TAK-788 (mobocertinib), a novel small molecule inhibitor, 28% response rate (https://www.targetedonc.com/view/fda-approves-mobocertinib-for-egfr-exon-20-positive-mnsclc). Amivantamab is of special interest. It is a bispecific EGFR-MET antibody that has shown promising preclinical and clinical activity in patients with EGFR exon 20 insertions [10] and was recently granted for accelerated approval by the Food and Drug Administration with a 40% response rate (median duration=11 months) (https://www.fda.gov/drugs/resources-information-approved-drugs/fda-grants-accelerated-approval-amivantamab-vmjw-metastatic-non-small-cell-lung-cancer).
In conclusion, while our sample size is small, it is of interest that all three patients treated had clinical benefit from cetuximab, including two partial responses, and that the benefit duration was between 9 and 32 months. Furthermore, responses, albeit anecdotal, to cetuximab in other patients with NSCLC and EGFR exon 20 insertions have also been reported, including patients with partial responses ongoing at 3.5 years [5,7]. While afatinib itself has a response rate of ~9% in EGFR exon 20 insertion NSCLC, the combination of cetuximab and afatinib resulted in objective responses in 3 of 4 such patients [5,9]. Interestingly, an exceptional response to a trastuzumab-based (ERBB2 antibody) regimen has been documented in a NSCLC with an analogous exon 20 alteration (insertion 774-775 AYVM) in a different Erb receptor family member-ERBB2 [10]. These observations demonstrate the value of molecular tumor board discussions, in silico modeling, and of individualized therapy [7,11-17]. Prospective cetuximab evaluation in EGFR exon 20 insertion NSCLC is warranted.
Disclaimer
Ethics approval and consent to participate: This study was performed in accordance with the UCSD IRB guidelines for data analysis and for any investigational treatments for which patients gave consent.
Consent for Publication: Not applicable
Funding: This study was funded in part by the Joan and Irwin Jacobs philanthropic fund and by National Cancer Institute grant P30 CA023100.
Acknowledgements: Figure was partially created with biorender.com. 6.5 Disclosures/Competing Interests Razelle Kurzrock has research funding from Incyte, Genentech, Merck Serono, Pfizer, Sequenom, Foundation Medicine, Guardant Health, Grifols, and Konica Minolta, as well as consultant fees from LOXO, X-Biotech, Actuate Therapeutics, Genentech and NeoMed. She receives speaker fees from Roche, and has an equity interest in ID by DNA and Curematch, Inc.
Availability of Data and Material: The data that support the findings of this study are available from the corresponding author upon reasonable request.
Authors’ Contributions: MN performed the chart review and wrote the manuscript. RK conceptualized the study and wrote the manuscript. SK reviewed the manuscript. Disclosures Shumei Kato serves as a consultant for Foundation Medicine. Speaker’s fee: Roche. Research grant: ACT Genomics, Sysmex, Konica Minolta, OmniSeq. Razelle Kurzrock has research funding from Incyte, Genentech, Merck Serono, Pfizer, Sequenom, Foundation Medicine, Guardant Health, Grifols, and Konica Minolta, as well as consultant fees from LOXO, X-Biotech, Actuate Therapeutics, Genentech and NeoMed. She receives speaker fees from Roche, and has an equity interest in IDbyDNA and Curematch, Inc. Mina Nikanjam received salary support from Genentech and owns Genentech stock.
References
- Midha A, Dearden S, McCormack R (2015) EGFR mutation incidence in non-small-cell lung cancer of adenocarcinoma histology: A systematic review and global map by ethnicity (mutMapII). Am J Cancer Res 5: 2892-2911.
- Janku F, Garrido-Laguna I, Petruzelka LB, Stewart DJ, Kurzrock R (2011) Novel therapeutic targets in non-small cell lung cancer. J Thorac Oncol 6: 1601-1612.
- Soria JC, Ohe Y, Vansteenkiste J (2018) Osimertinib in untreated egfr-mutated advanced non-small-cell lung cancer. N Engl J Med 378: 113-125.
- Vyse S, Huang PH (2019) Targeting EGFR exon 20 insertion mutations in non-small cell lung cancer. Signal Transduct Target Ther 4: 5.
- Li AM, Boichard A, Felip E, Kurzrock R (2020) New therapeutic approaches to overcoming resistant EGFR exon 20 alterations. Crit Rev Oncol Hematol 151: 102990.
- Robichaux JP, Elamin YY, Tan Z (2018) Mechanisms and clinical activity of an EGFR and HER2 exon 20-selective kinase inhibitor in non-small cell lung cancer. Nat Med 24: 638-646.
- Tsigelny IF, Wheler JJ, Greenberg JP (2015) Molecular determinants of drug-specific sensitivity for epidermal growth factor receptor (EGFR) exon 19 and 20 mutants in non-small cell lung cancer. Oncotarget 6: 6029-6039.
- Heymach J, Negrao M, Robichaux J (2018) OA02. 06 a phase II trial of poziotinib in EGFR and HER2 exon 20 mutant non-small cell lung cancer (NSCLC). J Thoracic Oncol 13: S323-S324.
- Van Veggel B, de Langen AJ, Hashemi SMS (2018) Afatinib and cetuximab in four patients with egfr exon 20 insertion-positive advanced nsclc. J Thorac Oncol 13: 1222-1226.
- Yun J, Lee SH, Kim SY (2020) Antitumor activity of amivantamab (JNJ-61186372), an EGFR-MET bispecific antibody, in diverse models of egfr exon 20 insertion-driven nsclc. Cancer Discov 10: 1194-1209.
- Falchook GS, Janku F, Tsao AS, Bastida CC, Stewart DJ, et al. (2013) Non-small-cell lung cancer with HER2 exon 20 mutation: Regression with dual HER2 inhibition and anti-VEGF combination treatment. J Thorac Oncol 8: e19-e20.
- Kato S, Kim KH, Lim HJ (2020) Real-world data from a molecular tumor board demonstrates improved outcomes with a precision N-of-one strategy. Nat Commun 11: 4965.
- Patel M, Kato SM, Kurzrock R (2018) Molecular tumor boards: Realizing precision oncology therapy. Clin Pharmacol Ther 103: 206-209.
- Schwaederle M, Parker BA, Schwab RB (2014) Molecular tumor board: The University of California-San Diego moores cancer center experience. Oncologist 19: 631-636.
- Itchins M, Lau B, Hudson AL (2020) ALK-rearranged non-small cell lung cancer in 2020: Real-world triumphs in an era of multigeneration alk-inhibitor sequencing informed by drug resistance profiling. Oncologist 25: 641-649.
- Zhao P, Peng L, Wu W (2019) Carcinoma of unknown primary with EML4-ALK fusion response to ALK inhibitors. Oncologist 24: 449-454.
- Sicklick JK, Kato S, Okamura R (2019) Molecular profiling of cancer patients enables personalized combination therapy: the I-PREDICT study. Nat Med 25: 744-750.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi 